Antimicrobials to Prevent and Treat Infections Antimicrobials to

Antimicrobials to Prevent and Treat Infections: Antimicrobials to Prevent and Treat Infections August, 2018

Antimicrobials to Prevent and Treat Infections: § Non-Systemic § Non-Resistant § Highly Effective in ALL Key Pathogens § § -Bacterial & Antibiotic Resistant Infections -Viral Infections -Spore and Fungal Infections -Chemical/Biological Warfare Pathogens

Safe Harbor/Disclaimer This presentation contains forward-looking statements. These statements involve substantial known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking statements we make. The forward-looking statements in this presentation represent our views as of the date of this presentation. We anticipate that the subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we have no current intention of doing so except to the extent required by applicable law. You should, therefore, not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this presentation. This presentation also contains estimates and other statistical data made by independent parties and by us relating to the market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. 3

Antimicrobial Resistant Infections -One of our most serious health threats (US & Global) High Levels of antibiotic resistance found worldwide, new data shows – January, 2018 Antimicrobial resistance is one of our most serious health threats. Patients with resistant infections are much more likely to die, and survivors have significantly longer hospital stays, delayed recuperation, and long-term disability. – April, 2013 Global use of antibiotics soars as resistance crisis worsens – March, 2018 Antibiotic resistance: An old problem with new ramifications – November, 2017 Antibiotic-Resistant Bugs in the 21 st Century – A Clinical Super-Challenge Drug companies told to do more to tackle ‘superbug’ crisis. The rise of drug-resistant bacteria is a growing threat to modern medicine – January, 2018 It is more difficult than ever to eradicate infections caused by antibiotic-resistant “superbugs, ” and the problem is exacerbated by a dry pipeline for new antimicrobials – January, 2009 4
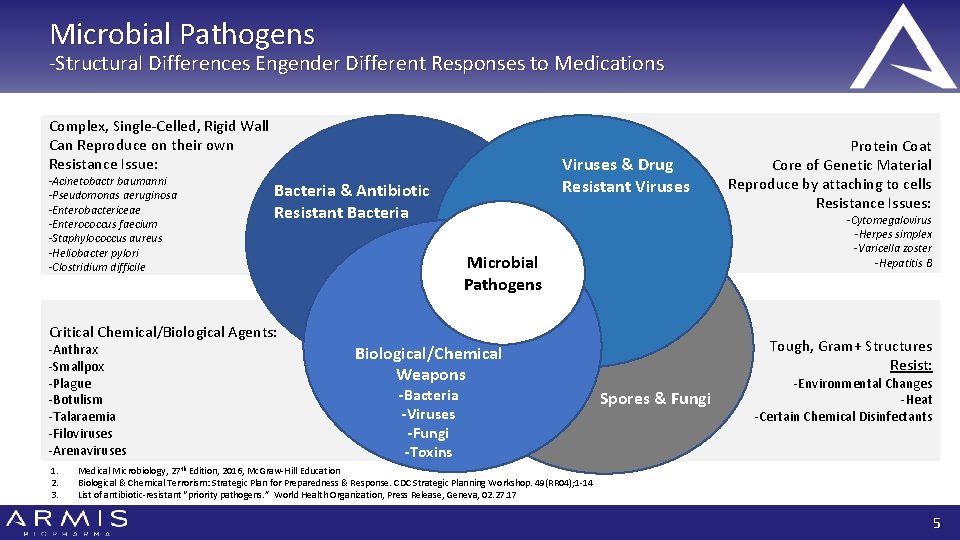
Microbial Pathogens -Structural Differences Engender Different Responses to Medications Complex, Single-Celled, Rigid Wall Can Reproduce on their own Resistance Issue: -Acinetobactr baumanni -Pseudomonas aeruginosa -Enterobactericeae -Enterococcus faecium -Staphylococcus aureus -Heliobacter pylori -Clostridium difficile Viruses & Drug Resistant Viruses Bacteria & Antibiotic Resistant Bacteria -Cytomegalovirus -Herpes simplex -Varicella zoster -Hepatitis B Microbial Pathogens Critical Chemical/Biological Agents: -Anthrax -Smallpox -Plague -Botulism -Talaraemia -Filoviruses -Arenaviruses 1. 2. 3. Tough, Gram+ Structures Resist: Biological/Chemical Weapons -Bacteria -Viruses -Fungi -Toxins Protein Coat Core of Genetic Material Reproduce by attaching to cells Resistance Issues: Spores & Fungi -Environmental Changes -Heat -Certain Chemical Disinfectants Medical Microbiology, 27 th Edition, 2016, Mc. Graw-Hill Education Biological & Chemical Terrorism: Strategic Plan for Preparedness & Response. CDC Strategic Planning Workshop. 49(RR 04); 1 -14 List of antibiotic-resistant “priority pathogens. ” World Health Organization, Press Release, Geneva, 02. 27. 17 5

Healthcare Acquired Infections (HAIs): Common and Costly Large Problem Made Worse by Antibiotic Resistance >100 M HAIs Globally 1 Additional costs of HAI estimated at $11 K - $38 K per patient in U. S. 2 Antibiotic Resistance in the U. S 4. : • Causes >2 Million illnesses and 23, 000 deaths • Costs $2. 2 B to treat 1. 2. 3. 4. 5. Additional length of stay ranges from 3 -10 days 3 • ~55% of US hospitals are unprofitable and 2, 600 will incur readmission penalties 5, 6 • Medicare no longer reimburses HAIs Report on Burden of Healthcare Associated Infections Worldwide. World Health Organization. 2011 The direct medical cost of HAI in US hospitals and the benefits of prevention. Centers for Disease Control and Prevention, March 2009 Joint Commission Center for Transforming Healthcare, Dec. 2014 Antibiotic Resistance Infections Treatment Costs Have Doubled Since 2002, Now Exceeding $2 B Annually. Health Affairs. 2018; 37(4): https: //doi. org/10. 1377/hlthaff. 2017. 1153 Kaiser Health News, June 05 2017; 6: Health Affairs Journal, https: //doi. org/10. 1377/hlthaff. 2015. 1193 6
Drug Resistance -Why we are facing a “post antibiotic” reality § Antibiotic resistance occurs when bacteria develop the ability to defeat drugs designed to kill them § Every time antibiotics are used, biological pressure on bacteria promotes development of resistance § The loss of effective antibiotics will undermine our ability to fight infectious diseases 1. Antibiotic Resistance Threats in the US, 2013, Centers for Disease Control and Prevention 7
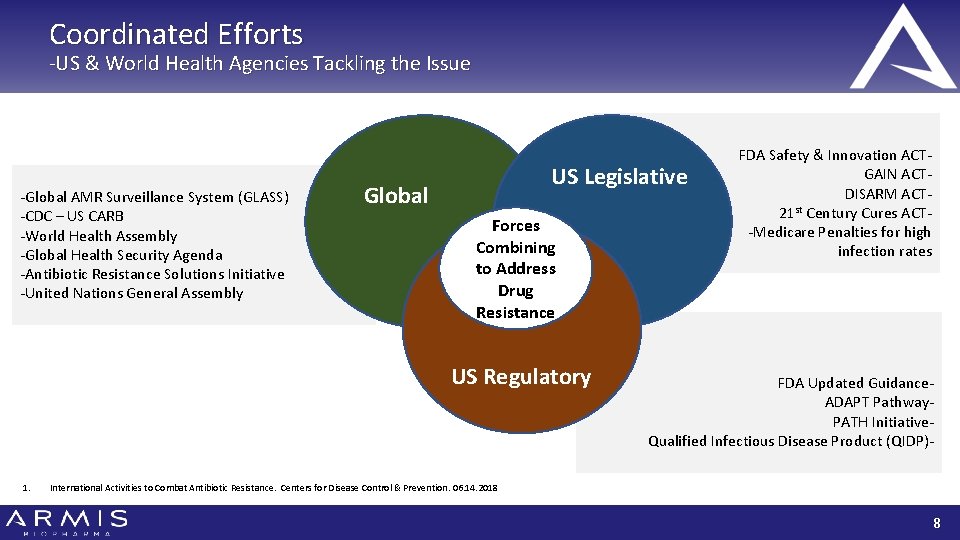
Coordinated Efforts -US & World Health Agencies Tackling the Issue -Global AMR Surveillance System (GLASS) -CDC – US CARB -World Health Assembly -Global Health Security Agenda -Antibiotic Resistance Solutions Initiative -United Nations General Assembly US Legislative Global Forces Combining to Address Drug Resistance US Regulatory 1. FDA Safety & Innovation ACTGAIN ACTDISARM ACT 21 st Century Cures ACT-Medicare Penalties for high infection rates FDA Updated Guidance. ADAPT Pathway. PATH Initiative. Qualified Infectious Disease Product (QIDP)- International Activities to Combat Antibiotic Resistance. Centers for Disease Control & Prevention. 06. 14. 2018 8

Veriox™: Breakthrough Technology § New & Undefeatable Mechanism of Action (M of A) - (Microbes cannot develop resistance to the M of A) § Breeches the cell wall (or protein coat) § Destroys the contents of the cell § Gram+, Gram-, Spores, Fungi, Virus § Non-Systemic = Maximum Efficacy § § Able to deliver higher, sustained concentrations of antimicrobial agents Reduced possibility of systemic toxicity Not reliant on the patients “physiology” to deliver the antimicrobial to the site of the pathogen Beneficial bacteria is preserved in the digestive tract § Multiple Healthcare Applications § § § Surface Disinfectant (Operating Rooms, Ventilators, Intensive Care Units) Impregnated into surgical devices (Catheters, Orthopedic Implants, Wound Dressings, Surgical Mesh) Emergency Wipes (Trauma Cases, Chemical/Biological Warfare) Topical Therapeutics (i. e. Uncomplicated Skin & Skin Structure Infections, Impetigo & Post Burn Fungal Infections) Foot and Toenail Fungus Acne 1. The role of topical antibiotics used as prophylaxis in surgical site infection prevention. J Antimicrob Chemother (2011), 66 (4): 693 -701 2. Topical antimicrobial therapy for treating chronic wounds. Clin Infect Dis, 2009, vol. 49 9
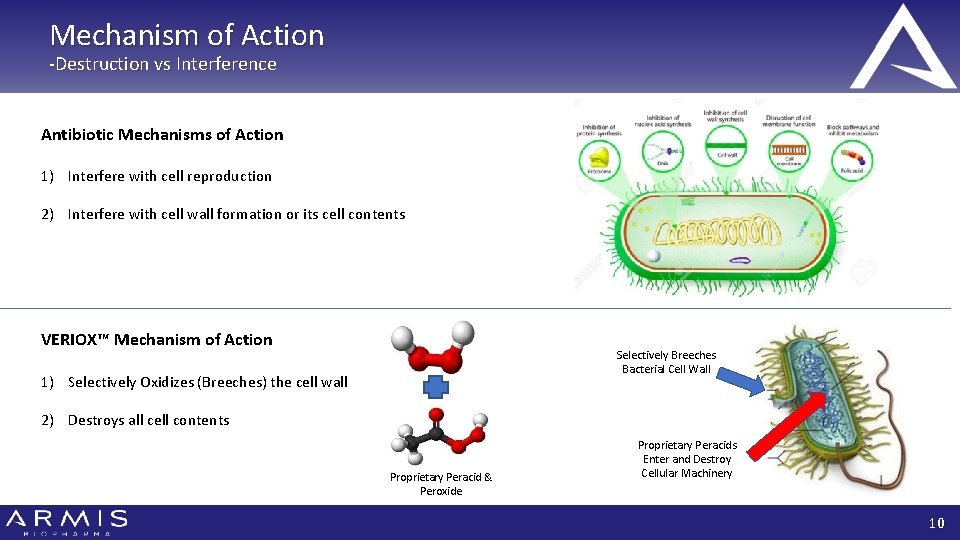
Mechanism of Action -Destruction vs Interference Antibiotic Mechanisms of Action 1) Interfere with cell reproduction 2) Interfere with cell wall formation or its cell contents VERIOX™ Mechanism of Action Selectively Breeches Bacterial Cell Wall 1) Selectively Oxidizes (Breeches) the cell wall 2) Destroys all cell contents Proprietary Peracid & Peroxide Proprietary Peracids Enter and Destroy Cellular Machinery 10
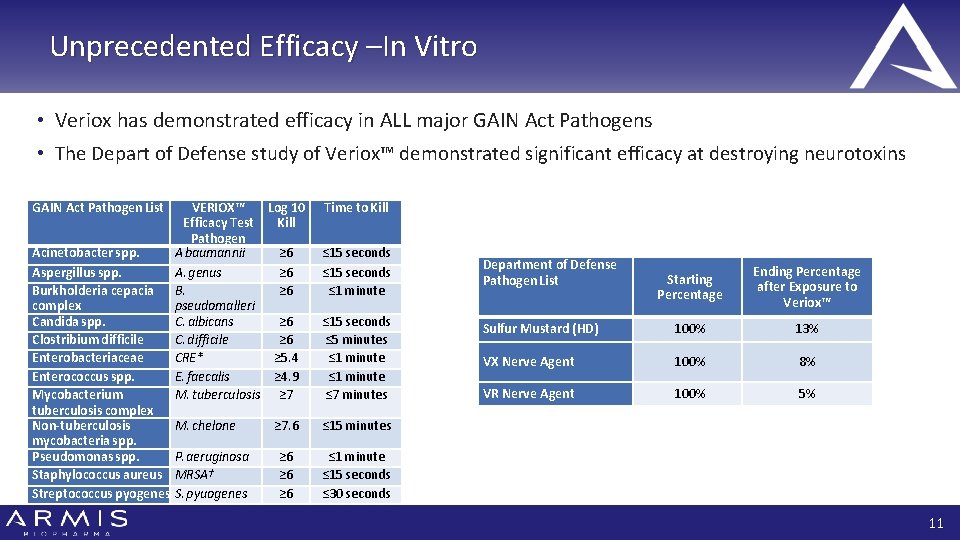
Unprecedented Efficacy –In Vitro • Veriox has demonstrated efficacy in ALL major GAIN Act Pathogens • The Depart of Defense study of Veriox™ demonstrated significant efficacy at destroying neurotoxins GAIN Act Pathogen List Acinetobacter spp. Aspergillus spp. Burkholderia cepacia complex Candida spp. Clostribium difficile Enterobacteriaceae Enterococcus spp. Mycobacterium tuberculosis complex Non-tuberculosis mycobacteria spp. Pseudomonas spp. Staphylococcus aureus Streptococcus pyogenes VERIOX™ Log 10 Efficacy Test Kill Pathogen A baumannii ≥ 6 A. genus ≥ 6 B. ≥ 6 pseudomalleri C. albicans ≥ 6 C. difficile ≥ 6 CRE* ≥ 5. 4 E. faecalis ≥ 4. 9 M. tuberculosis ≥ 7 M. chelone P. aeruginosa MRSA† S. pyuogenes Time to Kill ≤ 15 seconds ≤ 1 minute ≤ 15 seconds ≤ 5 minutes ≤ 1 minute ≤ 7 minutes ≥ 7. 6 ≤ 15 minutes ≥ 6 ≥ 6 ≤ 1 minute ≤ 15 seconds ≤ 30 seconds Department of Defense Pathogen List Starting Percentage Ending Percentage after Exposure to Veriox™ Sulfur Mustard (HD) 100% 13% VX Nerve Agent 100% 8% VR Nerve Agent 100% 5% 11
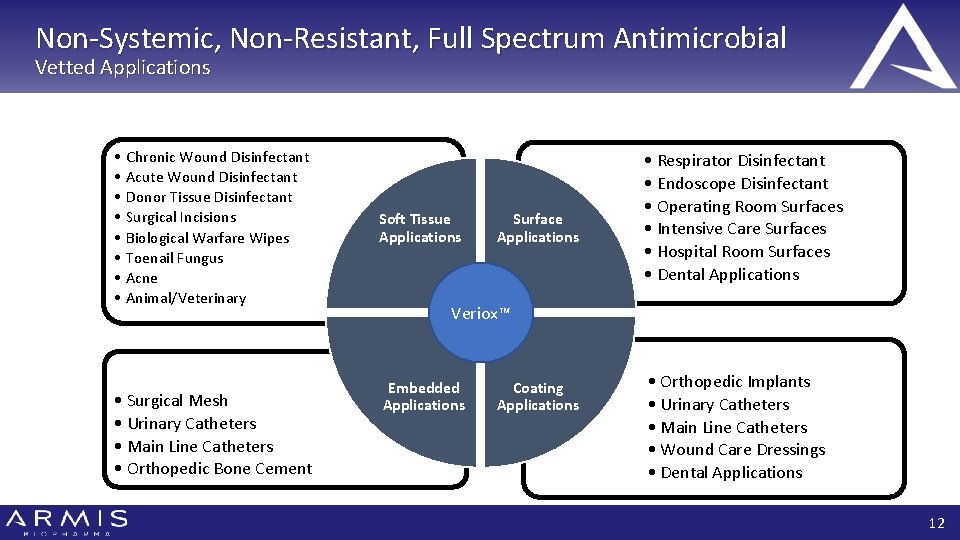
Non-Systemic, Non-Resistant, Full Spectrum Antimicrobial Vetted Applications • • Chronic Wound Disinfectant Acute Wound Disinfectant Donor Tissue Disinfectant Surgical Incisions Biological Warfare Wipes Toenail Fungus Acne Animal/Veterinary • Surgical Mesh • Urinary Catheters • Main Line Catheters • Orthopedic Bone Cement Soft Tissue Applications Surface Applications • Respirator Disinfectant • Endoscope Disinfectant • Operating Room Surfaces • Intensive Care Surfaces • Hospital Room Surfaces • Dental Applications Veriox™ Embedded Applications Coating Applications • Orthopedic Implants • Urinary Catheters • Main Line Catheters • Wound Care Dressings • Dental Applications 12
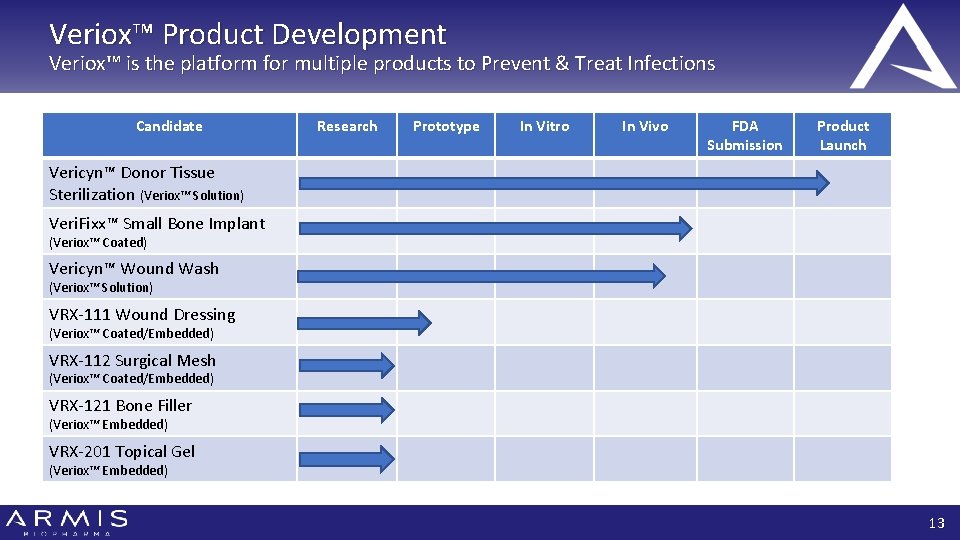
Veriox™ Product Development Veriox™ is the platform for multiple products to Prevent & Treat Infections Candidate Research Prototype In Vitro In Vivo FDA Submission Product Launch Vericyn™ Donor Tissue Sterilization (Veriox™ Solution) Veri. Fixx™ Small Bone Implant (Veriox™ Coated) Vericyn™ Wound Wash (Veriox™ Solution) VRX-111 Wound Dressing (Veriox™ Coated/Embedded) VRX-112 Surgical Mesh (Veriox™ Coated/Embedded) VRX-121 Bone Filler (Veriox™ Embedded) VRX-201 Topical Gel (Veriox™ Embedded) 13

LEAD PRODUCT CANDIDATES: VERICYN™ Veri. Cyn™: Veri. Cyn™ (stabilized aqueous solution of VERIOX) broad spectrum, non-resistant donor tissue sterilization Initial Indication: Veri. Cyn™ for donor tissue is intended for eradicating bacteria, viruses, and fungi from both hard and soft donor tissues and organs The Issue: Donor tissues carrying various bacteria and/or viruses have the potential to transfer to the recepient. 1 An unacceptable number of donations are rendered unusable due to the present of pathogens. Veri. Cyn™ is being tested for use in donor sterilization by an international tissue bank Administration: Integrated into donor tissue bank sterilization process 1. Recommendations for screening of donor and recipient prior to solid organ transplantation and to minimize transmission of donor–derived infections. Clinical Microbiology and Infection. 2014; 20(7): 10 -18. 14
LEAD PRODUCT CANDIDATES: VERIFIXX™ Veri. Fixx™: The CHD Small Bone Implant with Veri. Fixx™ coating (VERIOX™ plus a biodegradable polymer) on an orthopedic implant Indication: Veri. Fixx™ implant is indicated for the fixation of osteotomies/ reconstruction of the lesser toes after corrective procedures for hammertoe, claw and mallet toe. Revenue Potential: With 650 K procedures a year in the U. S. in 2017* estimated gross sales for toe implants could exceed $30 M/year. Shows Point-of-Care potential for other markets like spine, large bone, dental and trauma ($35 B addressable market*). Application: To be used in the distal phalanges of the toes or fingers to fuse bone defects and joints. * i. Data and Frost & Sullivan Market Research Reports 15

LEAD PRODUCT CANDIDATES: VERICYN™ Veri. Cyn™: Veri. Cyn™ (stabilized aqueous solution of VERIOX) broad spectrum, non-resistant antimicrobial wound wash Initial Indication: Veri. Cyn™ is intended for moistening and debriding acute and chronic dermal lesions, such as pressure ulcers, stasis ulcers, diabetic ulcers, post-surgical wounds, first and second degree burns, abrasions and minor skin irritations and intended to be used in conjunction with negative pressure wound therapy (NPWT) Revenue Potential: Over 1 M wounds treated per year. Revenue potential is +$100 M/yr. Addressable market is estimated for NPWT to be $100 M/yr. Administration: Irrigate the wound post debridement or during negative pressure wound therapy. 16

ADDITIONAL VERIOX™ PRODUCTS VERIOX™ Surgical Mesh: • VERIOX™ Coated Versions • Full integration into hernia area • Decreased internal scarring – reduction in pain associated with integration • Decreased infection rate • >$1 B Addressable Market* VERIOX™ Wound Dressing: • VERIOX™ Impregnated Versions • Fully cross-linked Type 1 Collagen – Sourcing FDA approved base matrix for removable wound dressing • >$1 B Addressable Market* * Frost & Sullivan Market Research Reports 17

ADDITIONAL VERIOXTM PRODUCTS: -TOPICAL THERAPEUTIC Market: • • • Uncomplicated Skin and Skin Structure Infections, Impetigo and Fungal Burn Antimicrobial/Antifungal - Will prevent infection during healing process >$4 B addressable market in U. S. Only competitive products are limited use (gram positive efficacy only) Low COGS will provide ability to gain rapid adoption Targeted Gel & Cream Attributes: • • • Short term application – Will wash off with water Antimicrobial/Antifungal - Will prevent infection during healing process Fast release of active ingredient Potential for quicker healing Used for topical infections Gel Formulation 18

INTELLECTUAL PROPERTY ESTATE • Our peracid technology platform is currently protected by 8 U. S. issued patents and multiple corresponding incountry international patents • In addition, we have 4 pending U. S. patent applications • These patents and applications relate to various aspects of the peracid technology and its application to our intended product portfolio 19
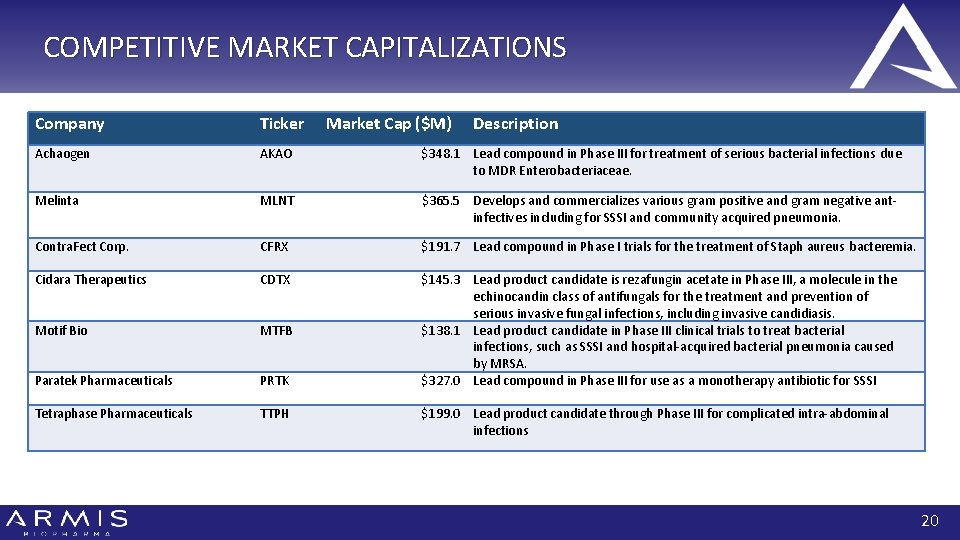
COMPETITIVE MARKET CAPITALIZATIONS Company Ticker Market Cap ($M) Description Achaogen AKAO $348. 1 Lead compound in Phase III for treatment of serious bacterial infections due to MDR Enterobacteriaceae. Melinta MLNT $365. 5 Develops and commercializes various gram positive and gram negative antinfectives including for SSSI and community acquired pneumonia. Contra. Fect Corp. CFRX $191. 7 Lead compound in Phase I trials for the treatment of Staph aureus bacteremia. Cidara Therapeutics CDTX Motif Bio MTFB Paratek Pharmaceuticals PRTK $145. 3 Lead product candidate is rezafungin acetate in Phase III, a molecule in the echinocandin class of antifungals for the treatment and prevention of serious invasive fungal infections, including invasive candidiasis. $138. 1 Lead product candidate in Phase III clinical trials to treat bacterial infections, such as SSSI and hospital-acquired bacterial pneumonia caused by MRSA. $327. 0 Lead compound in Phase III for use as a monotherapy antibiotic for SSSI Tetraphase Pharmaceuticals TTPH $199. 0 Lead product candidate through Phase III for complicated intra-abdominal infections 20
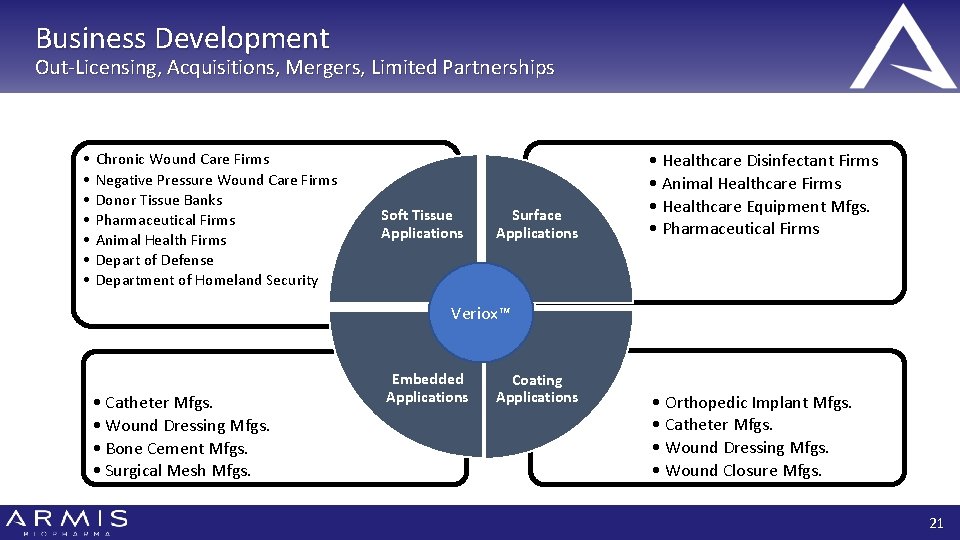
Business Development Out-Licensing, Acquisitions, Mergers, Limited Partnerships • • Chronic Wound Care Firms Negative Pressure Wound Care Firms Donor Tissue Banks Pharmaceutical Firms Animal Health Firms Depart of Defense Department of Homeland Security Soft Tissue Applications Surface Applications • Healthcare Disinfectant Firms • Animal Healthcare Firms • Healthcare Equipment Mfgs. • Pharmaceutical Firms Veriox™ • Catheter Mfgs. • Wound Dressing Mfgs. • Bone Cement Mfgs. • Surgical Mesh Mfgs. Embedded Applications Coating Applications • Orthopedic Implant Mfgs. • Catheter Mfgs. • Wound Dressing Mfgs. • Wound Closure Mfgs. 21

Leadership Team § Theodore E. Ziemann Chairman and CEO § Bob Kali Chief Financial Officer § Brian Doughty President and COO § Franklin Okumu VP Product Development & Manufacturing (Advisor) 22

Leadership Team/Collaborators Leadership Experience Collaborators Ted Ziemann Chairman and Chief Executive Officer Brian Doughty, MBA, MPA President and Chief Operating Officer Bob Kali Chief Financial Officer Franklin Okumu. Ph. D. VP Product Development & Manufacturing (Advisor) Scott Noblitt. Ph. D. Director of Product Development 23
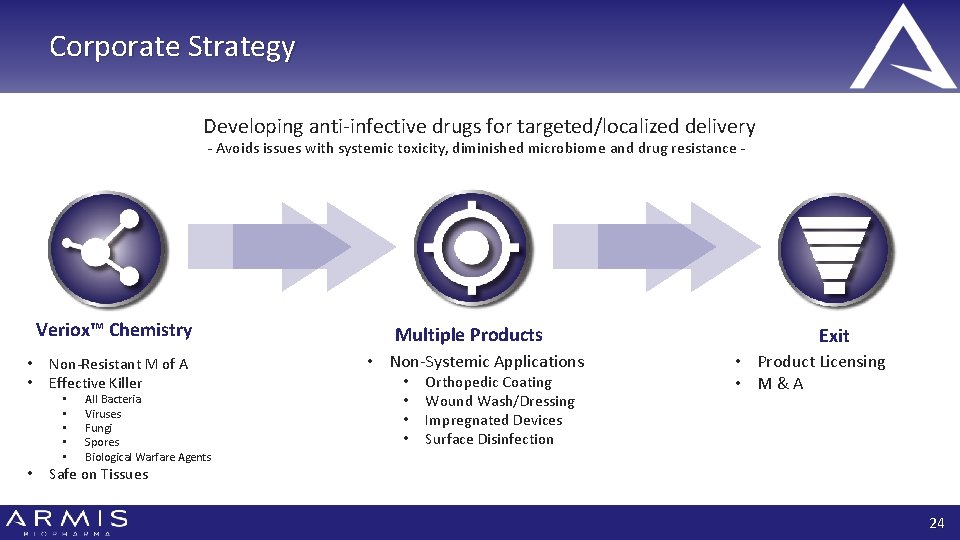
Corporate Strategy Developing anti-infective drugs for targeted/localized delivery - Avoids issues with systemic toxicity, diminished microbiome and drug resistance - Veriox™ Chemistry • • Non-Resistant M of A Effective Killer • • • All Bacteria Viruses Fungi Spores Biological Warfare Agents Multiple Products • Non-Systemic Applications • • Orthopedic Coating Wound Wash/Dressing Impregnated Devices Surface Disinfection Exit • Product Licensing • M&A Safe on Tissues 24

Contact CHD Bioscience research laboratories are located in the Research Innovation Center, Fort Collins, Colorado CHD Bioscience Corporate Headquarters are located at the Harmony Corporate Center in Fort Collins Colorado. Ted Ziemann Chairman & CEO ted. ziemann@armisbiopharma. com Brian Doughty President and COO Brian. doughty@armisbiophrma. com www. armisbiopharma. com
- Slides: 25