Antimicrobial Stewardship a Strategy to Combat Antimicrobial Resistant

Antimicrobial Stewardship a Strategy to Combat Antimicrobial Resistant Pathogens Trish M. Perl, MD, MSc Jay P. Sanford Professor of Medicine, Chief of Infectious Diseases UTSW Adjunct Professor of Medicine Johns Hopkins University

Objectives • Present the background and rationale for antimicrobial stewardship in healthcare • Demonstrate the value of antimicrobial stewardship programs for improved patient care and controlling costs • Review the role of technology and innovation • Discuss the GCC strategy to reduce resistance

Objectives • Present the background and rationale for antimicrobial stewardship in healthcare • Demonstrate the value of antimicrobial stewardship programs for improved patient care and controlling costs • Review the role of technology and innovation • Discuss the GCC strategy to reduce resistance
Antimicrobial Timeline 1928 1940 1941 1944 1972 2000 2010 Florey, Chain, Heatley, and Abrahamof demonstrated Widespread production penicillin Fleming discovers penicillin Human studies for penicillin therapeutic potential in mouse infectionbegin study from Penicillium (Oxford) chrysogenum

The Irony: Antimicrobial Timeline 1928 1940 1941 1944 1972 2000 2010 w Time, February 25, 1966 • “Nearly all experts agree that [by the year 2000] bacterial and viral diseases will have been wiped out. Probably atherosclerotic heart disease will also have been eliminated. Cells have only a few secrets still hidden from probers, who are confident that before the year 2000 they will have found the secret that causes cancer. ” w William H. Stewart, Surgeon General, 1969 • “It is time to close the book on infectious diseases” w Robert Petersdorf, President IDSA, 1978 • “Even with my great personal loyalty to infectious diseases, I cannot conceive of the need for 309 more infectious disease experts [the number taking the boards that year] unless they spend their time culturing each other”
The Reality: Antimicrobial Timeline 1928 1940 1941 1944 1972 Cephalosporins Penicillin 2000 2010 Linezolid Erythromycin Daptomycin Ampicillin Carbapenems Vancomycin Methicillin Sulfonamides

Principles of Antimicrobial Resistance 1. Given sufficient time and drug use, antibiotic 2. 3. 4. 5. resistance will emerge. Resistance is progressive, evolving from low levels through intermediate to high levels. Organisms resistant to one antibiotic are likely to become resistant to other antibiotics. Once resistance appears, it is likely to decline slowly, if at all. The use of antibiotics by any one person affects others in the extended as well as the immediate environment. Levy. NEJM 1998

The World of Antimicrobial Resistance https: //www. cgdev. org/files/1424207_file_CGD_DRWG_FINAL. p df
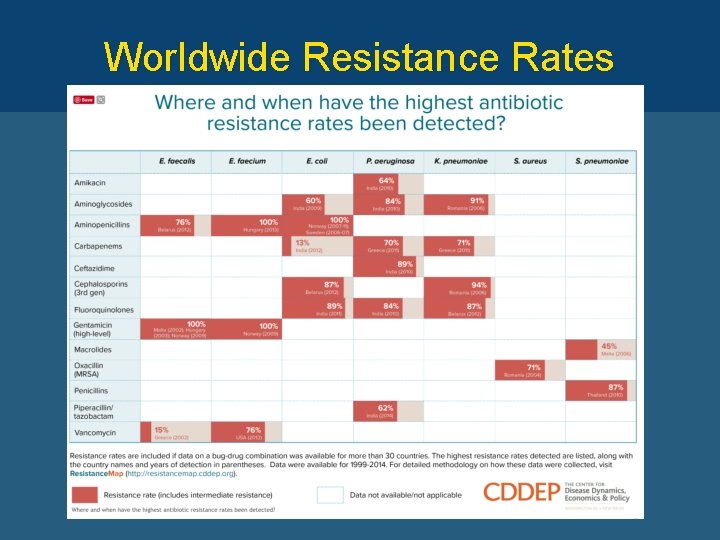
Worldwide Resistance Rates
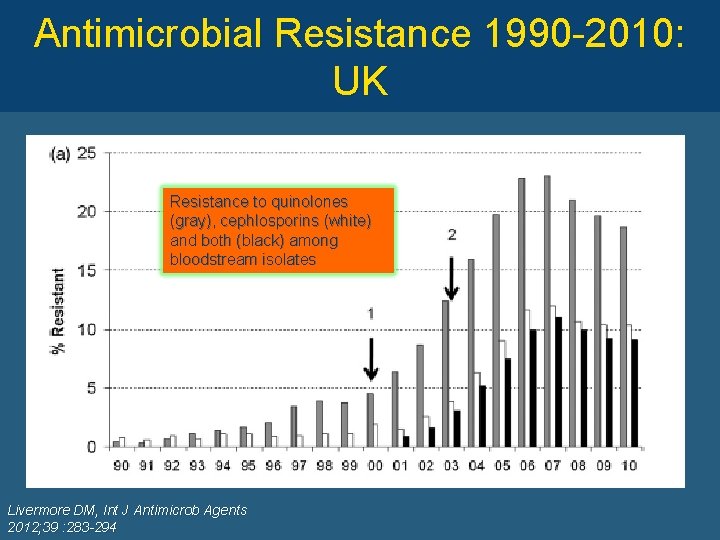
Antimicrobial Resistance 1990 -2010: UK Resistance to quinolones (gray), cephlosporins (white) and both (black) among bloodstream isolates 410 662 5758 Livermore DM, Int J Antimicrob Agents 2012; 39 : 283 -294
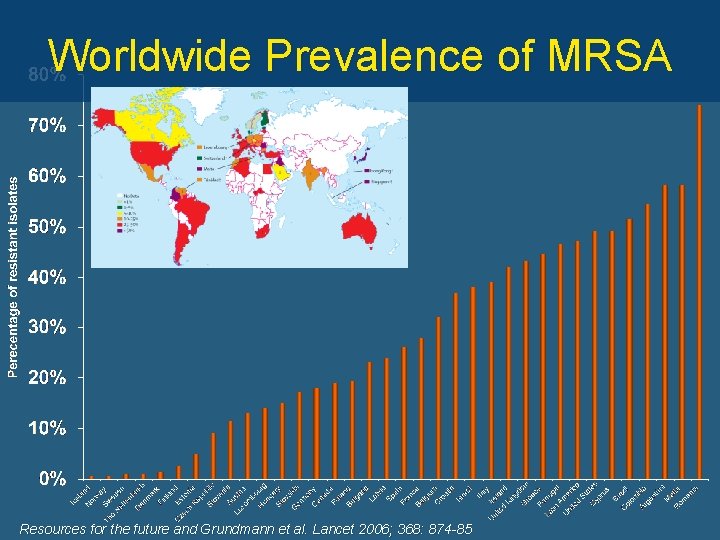
Worldwide Prevalence of MRSA Resources for the future and Grundmann et al. Lancet 2006; 368: 874 -85
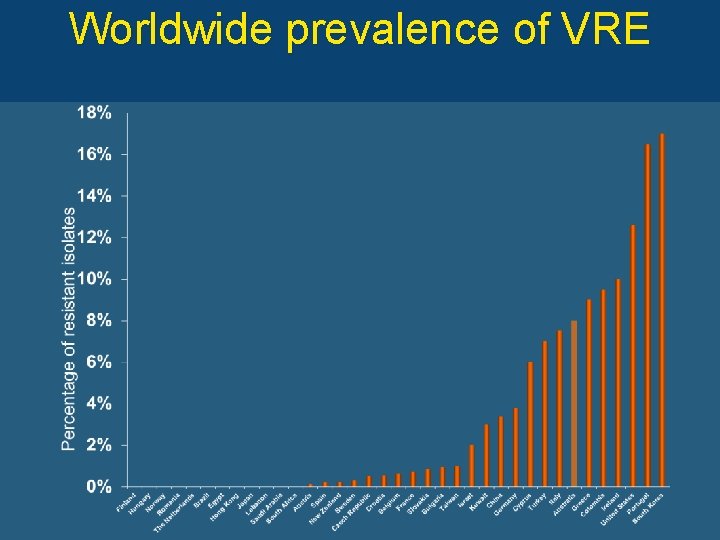
Worldwide prevalence of VRE

Global distribution of ESBL’s Gales et al Clinical Infectious Diseases 2001; 32: S 146 -S 155
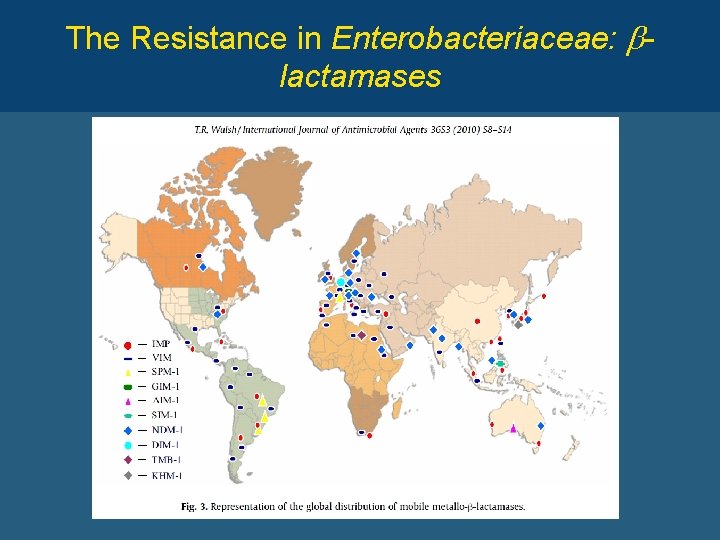
The Resistance in Enterobacteriaceae: blactamases

Worldwide Expansion • France: 2005. KPC 2 in a K. pneumoniae from a patient who has been in New York for medical treatment • Colombia: 2006 • Israel: 2007 • China: 2007 • Greece: 2008 • France: 2009

Carbapenemases • The emergence of ESBL-producing Enterobacteriaceae has increased carbapenem use. • Carbapenemases confer resistance to all b-lactams (penicillins / cephalosporins and carbapenems) and the bla gene is commonly associated with resistance genes • New York City--carbapenem-resistant K pneumoniae rose from 9% in 2002 to 18% in 2004, to 38% in 2008. • Widespread in Greece, Israel, Italy, Turkey, China and Brazil • KPC, the most important carbapenemase in Enterobacteriaceae, is endemic in USA [8% of Klebsiella in 2007 – 37 States] Southern Medical Journal 2011(104)
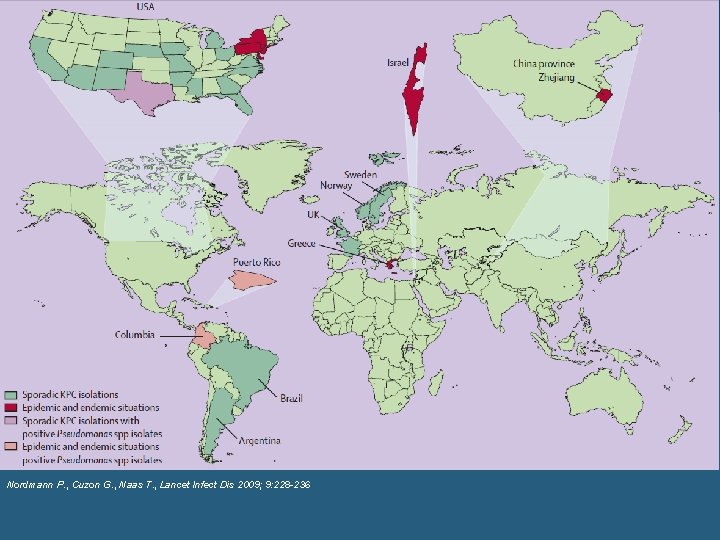
The resistance in Enterobacteriaceae: blactamases Southern Medical Journal • Volume 104, Number 1, January 2011 Nordmann P. , Cuzon G. , Naas T. , Lancet Infect Dis 2009; 9: 228 -236

Globe Trotting: Spread of KPC-containing Slide Klebsiella pneumoniae 1 1 1 2 1 4 Wernli D et al. PLo. S Medicine 2011 1
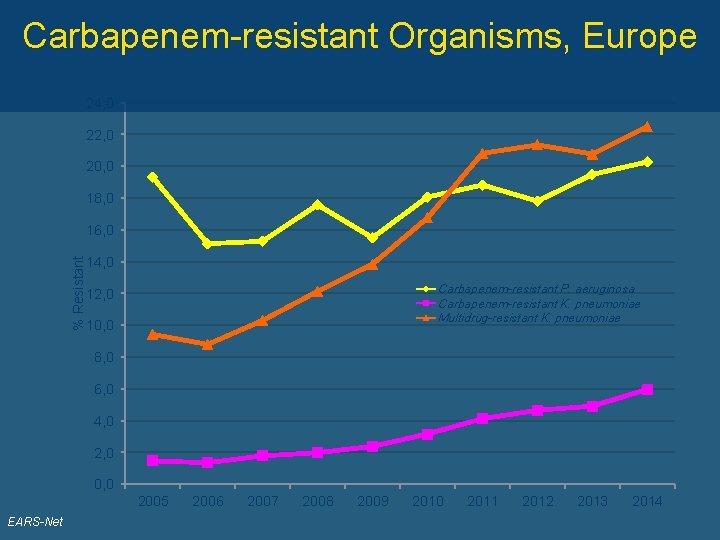
Carbapenem-resistant Organisms, Europe 24, 0 22, 0 20, 0 18, 0 % Resistant 16, 0 14, 0 Carbapenem-resistant P. aeruginosa Carbapenem-resistant K. pneumoniae Multidrug-resistant K. pneumoniae 12, 0 10, 0 8, 0 6, 0 4, 0 2, 0 0, 0 2005 EARS-Net 2006 2007 2008 2009 2010 2011 2012 2013 2014
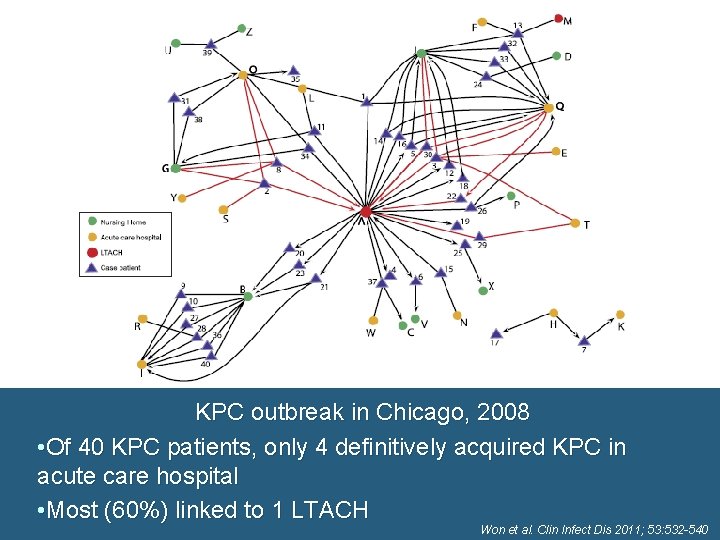
KPC outbreak in Chicago, 2008 • Of 40 KPC patients, only 4 definitively acquired KPC in acute care hospital • Most (60%) linked to 1 LTACH Won et al. Clin Infect Dis 2011; 53: 532 -540
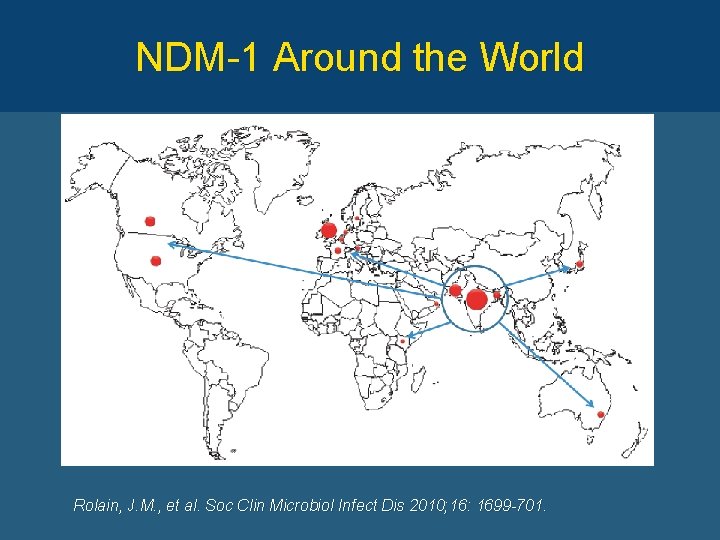
NDM-1 Around the World Rolain, J. M. , et al. Soc Clin Microbiol Infect Dis 2010; 16: 1699 -701.

The Newest Kid on the Block
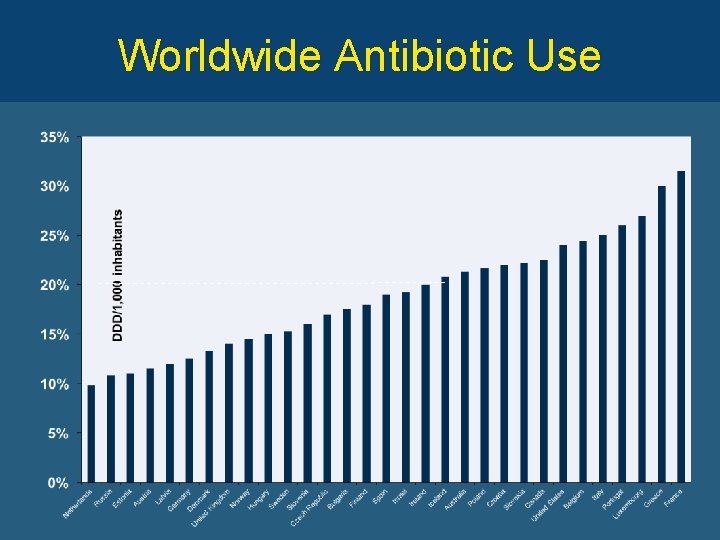
Worldwide Antibiotic Use
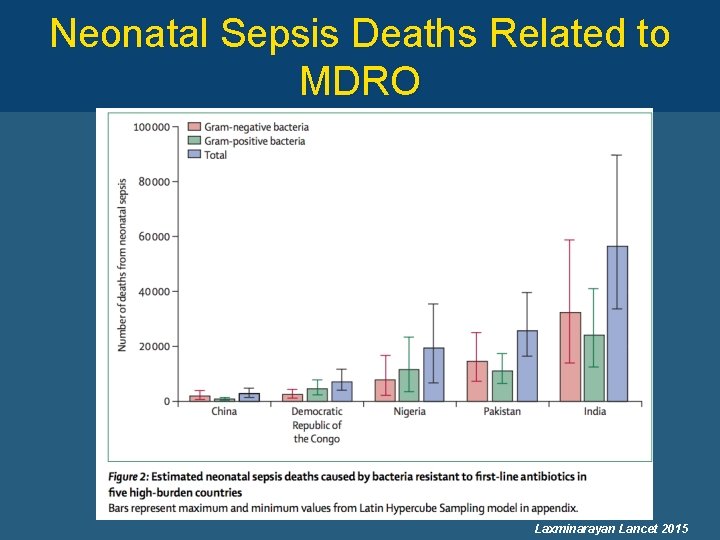
Neonatal Sepsis Deaths Related to MDRO Laxminarayan Lancet 2015
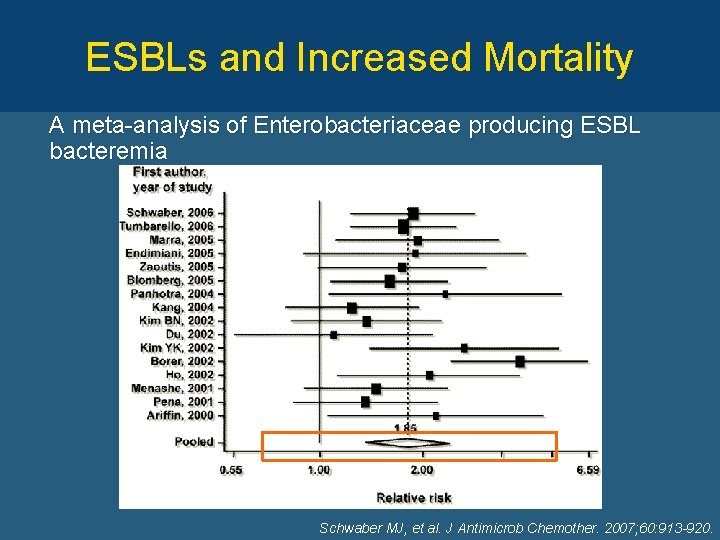
ESBLs and Increased Mortality A meta-analysis of Enterobacteriaceae producing ESBL bacteremia Schwaber MJ, et al. J Antimicrob Chemother. 2007; 60: 913 -920.
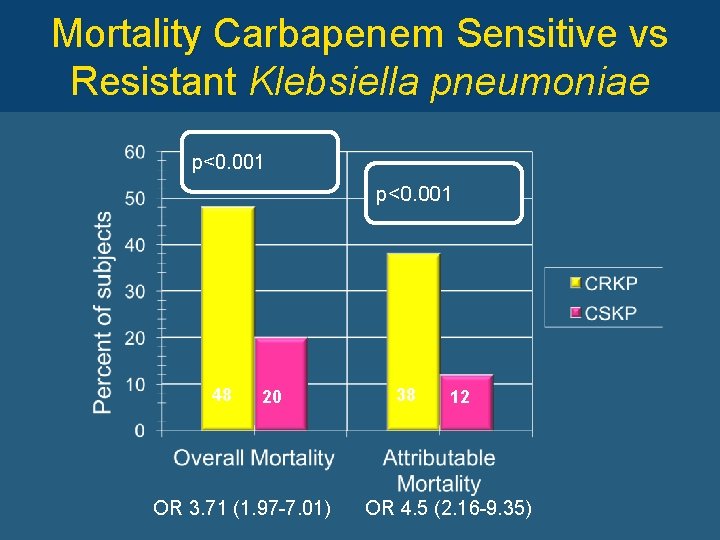
Mortality Carbapenem Sensitive vs Resistant Klebsiella pneumoniae p<0. 001 48 20 OR 3. 71 (1. 97 -7. 01) 38 12 OR 4. 5 (2. 16 -9. 35)

28 October 2021 27

The Numbers • 193 countries have signed on to the UN effort to tackle antimicrobial resistance • 700, 000 deaths a year are related to resistance in HIV, malaria and TB. This will increase to 10 million in 2050. • In 2016 490, 000 cases of MDR TB were reported; only 54% were successfully treated • ¼ antibiotics are unnecessary

Antimicrobial resistance is not a future threat looming on the horizon. It is here, right now and the consequences are devastating. Dr. Margaret Chan, The Hague 25 June 2014 28 October 2021 29

“In the Middle East, antimicrobial resistance is a crisis at the present time. It stems from a wide range of problems; however, there are few studies from this region about factors associated with proliferating resistance. ” Al Tawfiq et al Expert Review in Antimicrobial Therapy 2010

Why We Need to Do Something? • Prevalence of multidrug-resistant gram-negative, Gram positive and other bacteria increasing worldwide • Considerable morbidity and mortality from infections with MDR organisms • Few antibiotics in development to treat these infections – use of older antibiotics previously discontinued because of toxicity • Vulnerable patients at most risk • Costly to patients and the healthcare system
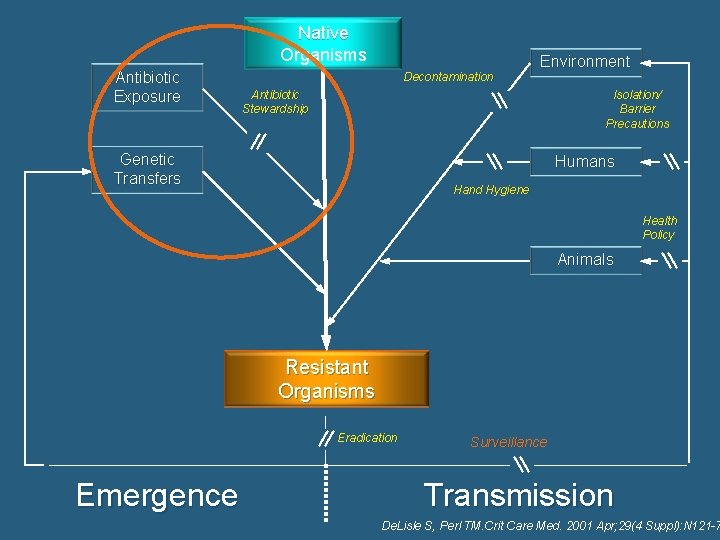
Native Organisms Antibiotic Exposure Decontamination Environment Antibiotic Stewardship Isolation/ Barrier Precautions Genetic Transfers Humans Hand Hygiene Health Policy Animals Resistant Organisms Eradication Emergence Surveillance Transmission De. Lisle S, Perl TM. Crit Care Med. 2001 Apr; 29(4 Suppl): N 121 -7
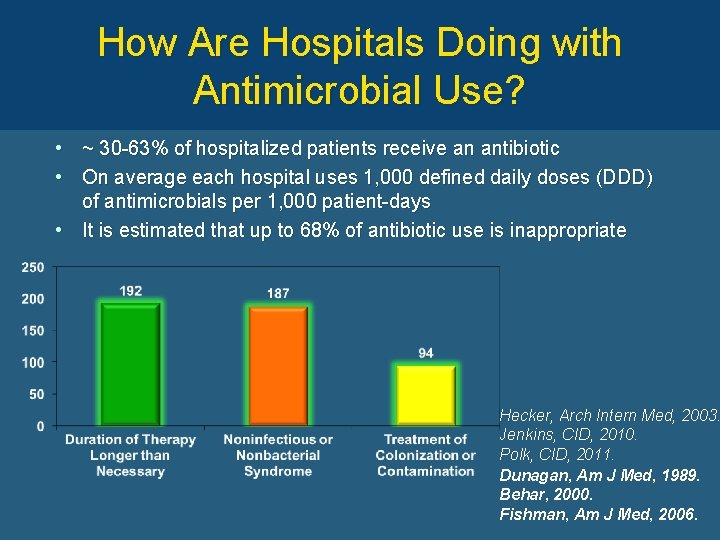
How Are Hospitals Doing with Antimicrobial Use? • ~ 30 -63% of hospitalized patients receive an antibiotic • On average each hospital uses 1, 000 defined daily doses (DDD) of antimicrobials per 1, 000 patient-days • It is estimated that up to 68% of antibiotic use is inappropriate Hecker, Arch Intern Med, 2003. Jenkins, CID, 2010. Polk, CID, 2011. Dunagan, Am J Med, 1989. Behar, 2000. Fishman, Am J Med, 2006.
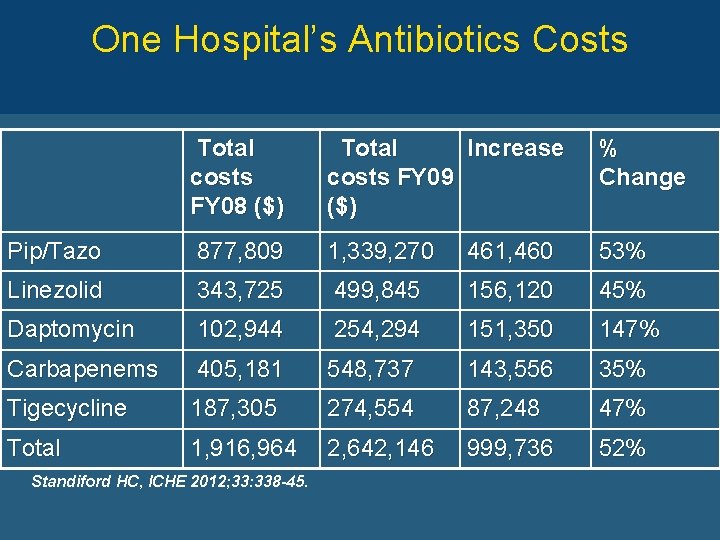
One Hospital’s Antibiotics Costs Total costs FY 08 ($) Total Increase costs FY 09 ($) % Change Pip/Tazo 877, 809 1, 339, 270 461, 460 53% Linezolid 343, 725 499, 845 156, 120 45% Daptomycin 102, 944 254, 294 151, 350 147% Carbapenems 405, 181 548, 737 143, 556 35% Tigecycline 187, 305 274, 554 87, 248 47% Total 1, 916, 964 2, 642, 146 999, 736 52% Standiford HC, ICHE 2012; 33: 338 -45.

Antimicrobial Stewardship The optimal selection, dosage, and duration of antimicrobial treatment that results in the best clinical outcome for the treatment or prevention of infection, with minimal toxicity to the patient and minimal impact on subsequent resistance. Owens RC et. al. Pharmacotherapy 2004; 24(7): 896– 908

Rationale for a Stewardship Program 1. Patient care: “The primary goal. . . is to optimize clinical outcomes while minimizing unintended consequences. . including toxicity, selection of pathogenic organisms (such as C. difficile), and the emergence of resistance. ” 2. Financial: “Effective antimicrobial stewardship programs can be financially self-supporting and improve patient care. … in both larger academic hospitals and smaller community hospitals. ” Guidelines for Developing an Institutional Program to Enhance Antimicrobial Stewardship: CID 2007; 44: 159 -77

Objectives • Describe the magnitude of the problem. • Present the background and rationale for antimicrobial stewardship in healthcare • Demonstrate the value of antimicrobial stewardship programs for improved patient care and controlling costs • Provide theoretical and practical considerations to make the case for implementing an antimicrobial stewardship program
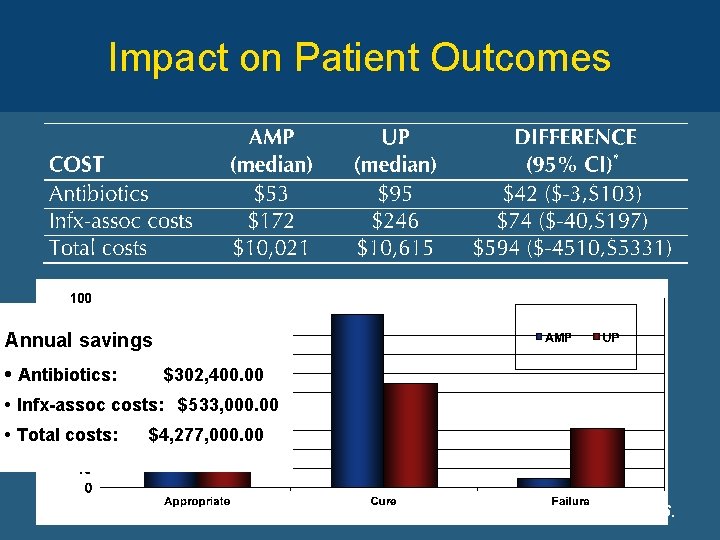
Impact on Patient Outcomes Annual savings • Antibiotics: $302, 400. 00 • Infx-assoc costs: $533, 000. 00 • Total costs: $4, 277, 000. 00 Fishman, Am J Med 2006.

Inappropriate Antimicrobial Use: Ambulatory Setting • Special considerations: – Antibiotic prescriptions unchecked – Frequently encounter conditions (pharyngitis, otitis media, sinusitis, and upper respiratory tract infections) that are >80% caused by viral infection – Impractical to change antibiotics after prescription • 72% of patients that present with an upper respiratory tract infection expect to receive a prescription for an antibiotic • Antibiotics are prescribed in 21% of acute pediatric ambulatory visits (>70% are for upper respiratory tract infections, representing 10 million antibiotic prescriptions per year) • Of the providers who gave a prescription for antibiotic, 20% felt the antibiotic was clinically indicated Cooper, Ann Intern Med, 2001. Bisno, CID, 2002. Hersh, Pediatrics, 2011. Macfarlane, BMJ, 2007.

Impact on Appropriate Dosing • Clinical decision support system to appropriately dose based on renal function implemented using academic detailing • Results – Appropriate dosing of gentamicin increased from 63% to 87% – Appropriate dosing of vancomycin increased from 47% to 77% – Appropriate use of gentamicin therapeutic monitoring increased from 70% to 90% Roberts GW et al. J Am Med Inform Assoc. 2010; 17: 308 -12.

Does Stewardship Decrease Antimicrobial Costs? Post-prescription review, tertiary centers Tertiary care ID fellow, critical Antimicrobial teaching hospital care pharmacist charges/patient significantly reduced ($1, 287 vs. $1, 873, p<0. 04) Oklahoma City VA ID pharmacist IV antibiotic costs savings $145, 942/annually • There are multiple examples of the impact of post prescription review in smaller hospitals and in international settings Fraiser GL et al. Arch Intern Med. 1997; 157: 1689 -94. Gentry CA et al. Am J Health Syst Pharm. 2000; 57: 268 -74.

Does Stewardship Decrease Antimicrobial Costs? Pre-prescription approval County Hospital Baylor ID faculty hold pager 24/7 U Pennsylvania Hospital Formal ASP with ID pharmacists White AC et al. Clin Infect Dis. 1997; 25: 230 -239. Fishman N. Am J Med. 2006; 119: S 53 -S 61. Antibiotic costs savings $803, 910 in 1 year Antibiotic costs savings $302, 400 in 1 year
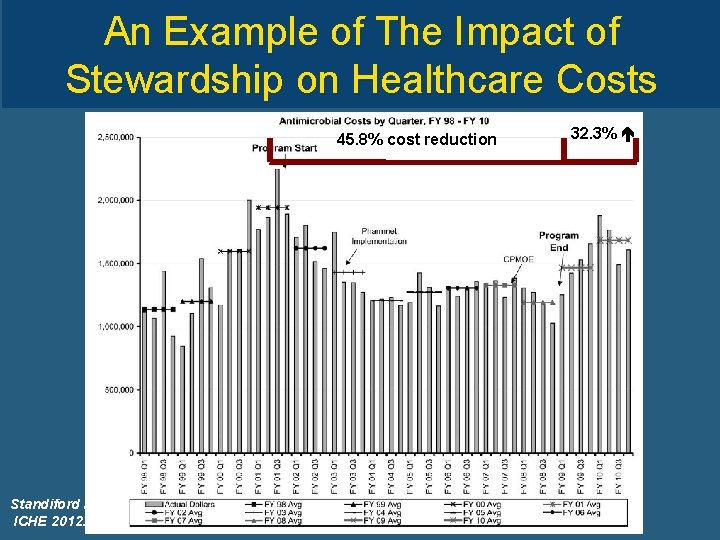
An Example of The Impact of Stewardship on Healthcare Costs 45. 8% cost reduction Standiford HC, ICHE 2012. 32. 3%

Stewardship Decreases LOS Hospital LOS Oklahoma City VA ID pharmacist Mean LOS baseline 13. 3 days Mean LOS with program 10. 8 days (p < 0. 01) ID / critical care MDs & algorithm Mean ICU stay 9 vs. 15 days (p = 0. 04) ICU LOS Pittsburg VA Gentry CA et al. Am J Health Syst Pharm. 2000; 57: 268 -74. Singh N et al. Am J Respir Crit Care Med. 2000; 162: 505 -11.

Stewardship Decreases Resistance • Ben Taub General Hospital, 1994 • ID attendings held approval pager 24/7 – All broad-spectrum antibiotic required approval (ticar/clav, imipenem, aztreonam, ceftazidime, ciprofloxacin, amikacin, IV fluconazole) • Increased susceptibility of GNR P. aeruginosa susceptibilities, 1/93 -12/93 vs. 7/94 -6/95 Ticar/clav 83 89 Imipenem Aztreonam Ceftazidime 83 95 White et al. CID. 1997; 25: 230 70 88 76 92 Cipro 83 87

Impact on Antimicrobial Resistance Cipro N=39 N=42 Standard Antibiotic duration 3 days 10 days LOS ICU 9 days 15 days Antibiotic 15% resistance/ superinfection 35% Singh N, Am J Respir Crit Care Med 2000.
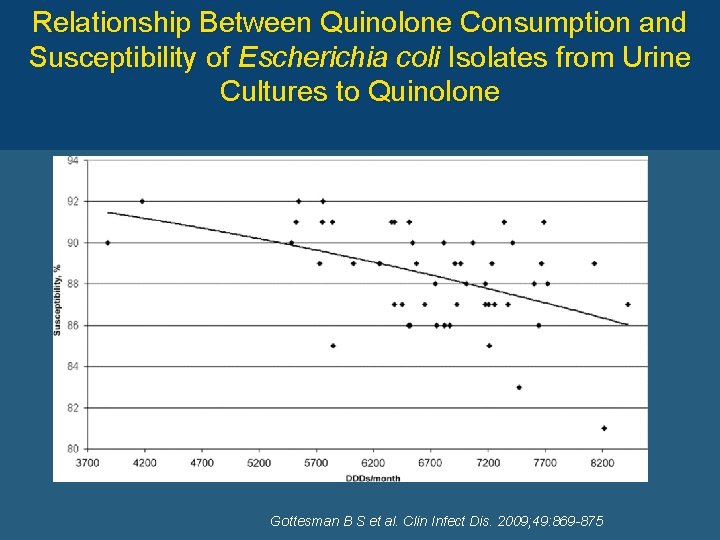
Relationship Between Quinolone Consumption and Susceptibility of Escherichia coli Isolates from Urine Cultures to Quinolone Gottesman B S et al. Clin Infect Dis. 2009; 49: 869 -875
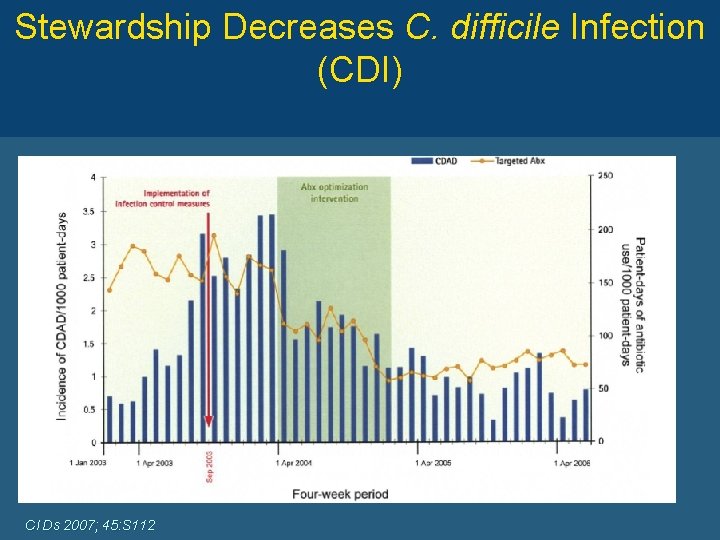
Stewardship Decreases C. difficile Infection (CDI) Cl Ds 2007; 45: S 112
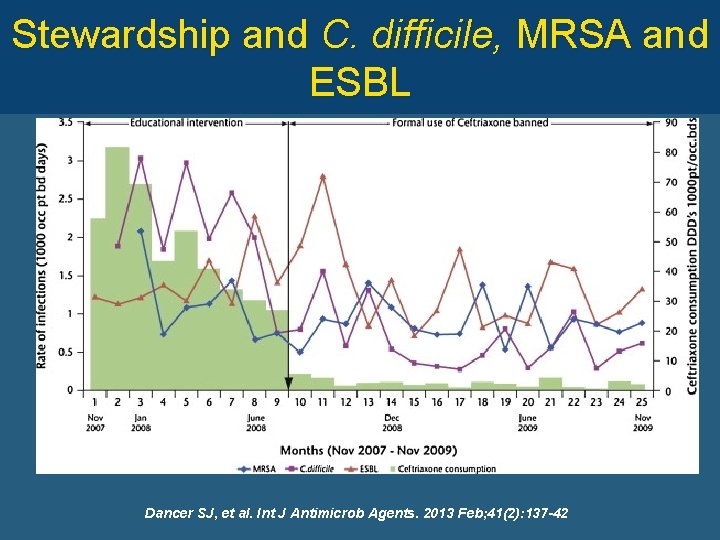
Stewardship and C. difficile, MRSA and ESBL Dancer SJ, et al. Int J Antimicrob Agents. 2013 Feb; 41(2): 137 -42
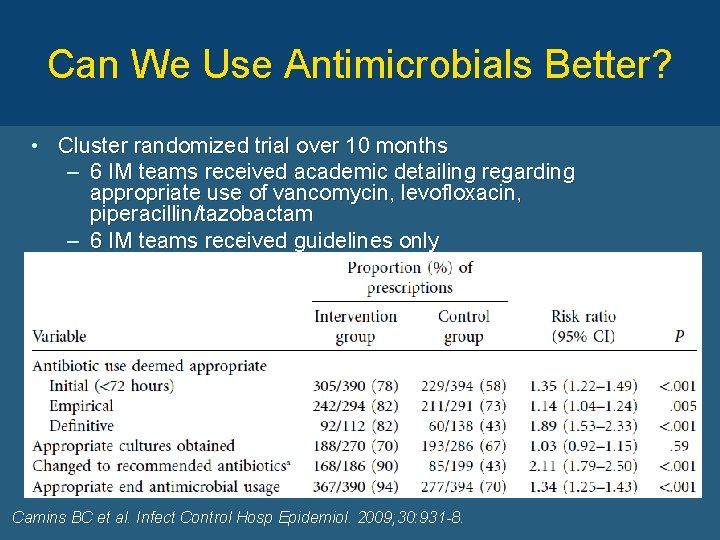
Can We Use Antimicrobials Better? • Cluster randomized trial over 10 months – 6 IM teams received academic detailing regarding appropriate use of vancomycin, levofloxacin, piperacillin/tazobactam – 6 IM teams received guidelines only Camins BC et al. Infect Control Hosp Epidemiol. 2009; 30: 931 -8.

Stewardship Optimizes Surgical Prophylaxis • Intervention to standardize surgical prophylaxis – – – Simplify drug options Standardize dosing Improve timing • Results – All doses correct – Reduction in dosing after incision (20% to 7%) – Annual cost savings $112, 000 Willemsen I et al. J Hosp Infect. 2007; 67: 156 -160.

Stewardship Improves Renal Dosing • Dosing of antimicrobials is key to assure adequate levels yet not produce renal insufficiency. Clinical decision support system was used to appropriately dose antimicrobials on renal function (academic detailing) • Results – in appropriate gentamicin dosing: 63% to 87% – in appropriate vancomycin dosing: 47% to 77% – Appropriate use of therapeutic monitoring: 70% to 90% Roberts GW et al. J Am Med Inform Assoc. 2010; 17: 308 -12.

Objectives • Present the background and rationale for antimicrobial stewardship in healthcare • Demonstrate the value of antimicrobial stewardship programs for improved patient care and controlling costs • • Review the role of technology and innovation Provide theoretical and practical considerations to make the case to hospital leadership for implementing an antimicrobial stewardship program

Urine Cultures • Asymptomatic bacteriuria is common – Post-menopausal females: 2. 8 -8. 6% – Long-term care residents: 15 -50% • Pyuria (in the absence of symptoms) is not an acceptable treatment indication • Treatment of asymptomatic bacteriuria is associated with no decreased risk of UTI and is associated with increased adverse events • Algorithms to promote appropriate interpretation of urine cultures have decreased antibiotic use by 50% Asscher, BMJ, 1969. Nicolle, Ann Intern Med, 1989. Loeb BMJ, 2005. Lin, Ann Intern Med. 2008.

Procalcitonin • Procalcitonin: biomarker increased in bacterial infections; less so in other types of infections and inflammatory conditions • Procalcitonin decreased antibiotic consumption with no adverse outcomes (11 RCT’s for a variety of conditions including sepsis) • Using a procalcitonin testing algorithm reduces: – Antibiotic treatment duration – Length of ICU stay by up to 2 days Hayashi. Clin Infect Dis, 2011. Hochreiter, et al. Crit Care, 2009.

MALDI-TOF (Matrix-assisted laser desorption/ionization time-offlight ) • MALDI-TOF mass spectrometry: rapidly identifies organisms from clinical specimens • Decreases time to organism ID 1. 2 -1. 5 days compared to traditional culture methods (84. 0 to 55. 9 hours, p < 0. 001) • When MALDI-TOF Is linked with an antimicrobial stewardship program, it can: – Improve time to effective antibiotic (30. 1 to 20. 4 hours, p = 0. 021) – Improve time to streamlined antibiotics (90. 3 to 47. 3 hours, p < 0. 001) – Decrease hospital stay (11. 9 to 9. 3 days, p = 0. 01) – Mean hospital costs ($45, 709 to $26, 162, p = 0. 009) – May also decrease mortality and recurrence of Huang, et al. Clin Infect Dis, 2013. Perez, et al. Arch Pathol Lab Med, 2013.

Objectives • Present the background and rationale for antimicrobial stewardship in healthcare • Demonstrate the value of antimicrobial stewardship programs for improved patient care and controlling costs • Review the role of technology and innovation • Provide theoretical and practical considerations to make the case to hospital leadership for implementing an antimicrobial stewardship program
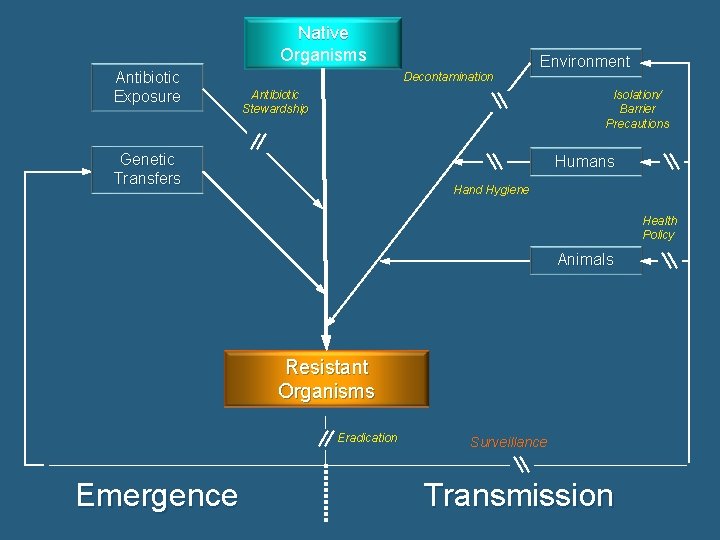
Native Organisms Antibiotic Exposure Decontamination Environment Antibiotic Stewardship Isolation/ Barrier Precautions Genetic Transfers Humans Hand Hygiene Health Policy Animals Resistant Organisms Eradication Emergence Surveillance Transmission

Prevalence of Antimicrobial Stewardship • 48 -60% of US hospitals and long-term care facilities have antimicrobial stewardship programs – ~15. 4% of community hospitals have antimicrobial stewardship or antibiotic subcommittees – Varying degrees of activity: most frequent intervention is automatic stop orders and restricted formulary • Barriers to implementation – Lack of personnel (55%) – Lack of financial resources (36%) – Opposition from prescribers (27%) – Resistance from administration (14%)
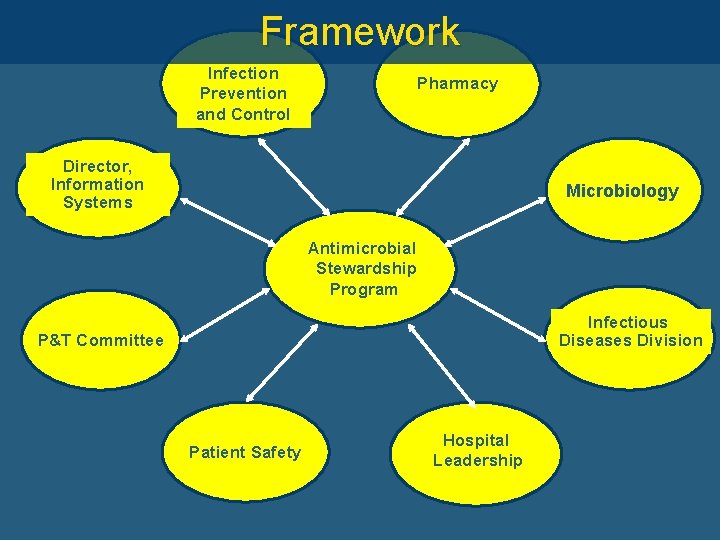
Framework Infection Prevention and Control Pharmacy Director, Information Systems Microbiology Antimicrobial Stewardship Program Infectious Diseases Division P&T Committee Patient Safety Hospital Leadership
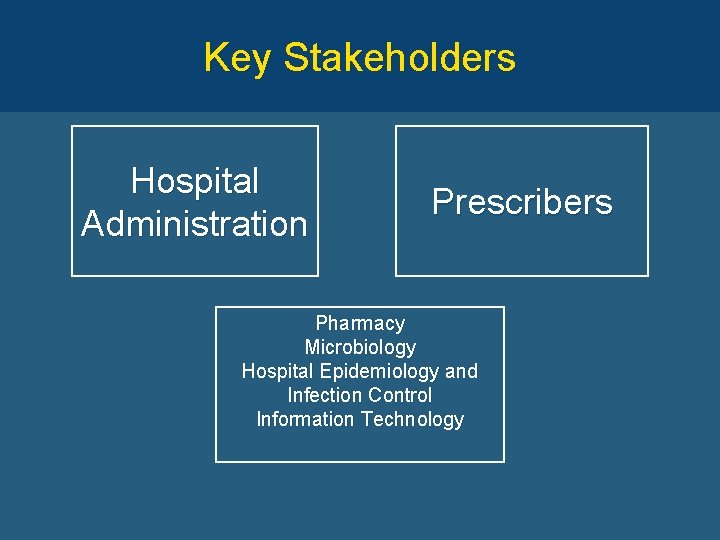
Key Stakeholders Hospital Administration Prescribers Pharmacy Microbiology Hospital Epidemiology and Infection Control Information Technology

The Ideal Stewardship Team “Core members of a multidisciplinary antimicrobial stewardship team include an infectious diseases physician and a clinical pharmacist with infectious diseases training who should be compensated for their time, with the inclusion of a clinical microbiologist, an information system specialist, an infection control professional, and hospital epidemiologist being optimal. ” Guidelines for Developing an Institutional Program to Enhance Antimicrobial Stewardship: CID 2007; 44: 159 -77
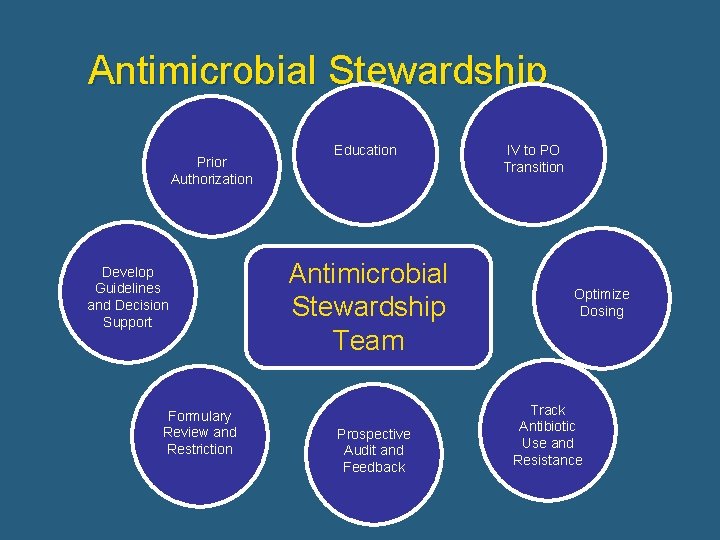
Antimicrobial Stewardship Prior Authorization Develop Guidelines and Decision Support Formulary Review and Restriction Education Antimicrobial Stewardship Team Prospective Audit and Feedback IV to PO Transition Optimize Dosing Track Antibiotic Use and Resistance

Is the Sky Falling?

Is an Institution an Island?
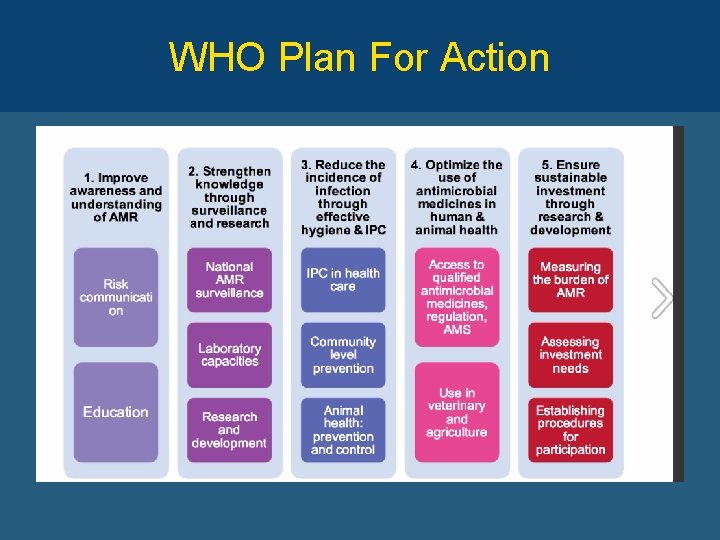
WHO Plan For Action


GCC Plan for Antimicrobial Resistance • Roadmap for region • Measurable elements • Includes – Adherence to infection prevention and control measures – Applying regulations to control the use of antimicrobials in humans and animals – Education – Surveillance Balkhy et al. JIPH 2016; 9: 375 -85

What does this mean for public health and healthcare settings • Enhance laboratory services • Add in performance improvement measures to laboratory processess • Adopt an international system for testing breakpoints • Characterize rates of CRE, MRSA, VRE and C. difficile using GCC standards • Characterize rates for HAIs including CLABSI, CAUTI, VAP • Adopt a unified method to report rates

What does this mean for public health and healthcare settings • Link antimicrobial dispensing to a prescription • Characterize antimicrobial use nationally and at the facility level • Develop robust pediatric and adult vaccination programs • Educate groups about resistance and the appropriate use of antimicrobials

Full Circle? “The time may come when penicillin can be bought by anyone in the shops. Then there is the danger that the ignorant man may easily under dose himself and, by exposing his microbes to non-lethal quantities of the drug, educate them to resist penicillin…. I hope this evil can be averted. ” - Sir Alexander Fleming (1945)

Summary • Resistance is an increasing, emerging and expanding public health problem/crisis. • It is fueled by antimicrobial use in many settings, poor or non existant infection prevention, poor food and water sanitation. • Action is required and the implementation of strong infection prevention and antimicrobial stewardship will be important
- Slides: 72