ANTIMICROBIAL RESISTANCE How to tackle an issue in

ANTIMICROBIAL RESISTANCE How to tackle an issue in the new legislative for veterinary medicinal products Lucie Pokludová Institute for State Control of Veterinary Biologicals and Medicines Czech Republic EPP Group Public Hearing, Brussells, 22. 4. 2015

Main motivations to adopt new rules for VMPs • Improve availability, in particular for „limited markets“ • Reduce administrative burden for the veterinary pharmaceutical companies • Enhance innovation in veterinary medicine • Strengthen internal market …while not reducing the quality, safety and efficacy standards for veterinary medicinal products
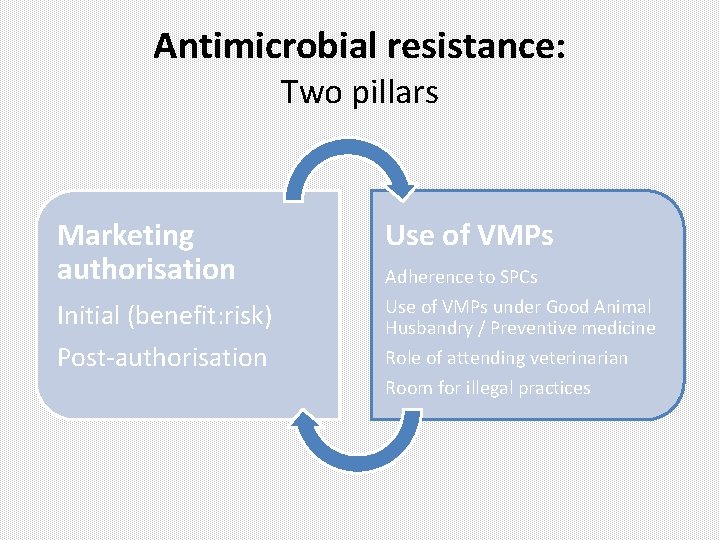
Antimicrobial resistance: Two pillars Marketing authorisation Initial (benefit: risk) Post-authorisation Use of VMPs Adherence to SPCs Use of VMPs under Good Animal Husbandry / Preventive medicine Role of attending veterinarian Room for illegal practices

Example from practice: Dairy cows: ceftiofur (newer, last resort) x amoxicillin (older, first choice) AMR: Prudent use warnings Economy: Withdrawal period Ceftiofur: Milk: zero days MRL : milk: 100 µg/ kg • • • „Ceftiofur VMP“ selects for resistant strains such as bacteria carrying extended spectrum betalactamases (ESBL) => a risk to human health if these strains disseminate to humans. Therefore should be reserved for the treatment of clinical conditions which have responded poorly, or are expected to respond poorly (refers to very acute cases when treatment must be initiated without bacteriological diagnosis), to more narrow spectrum ATMs first line treatment. Official, national and regional antimicrobial policies should be taken into account when the product is used. Increased use, including use of the product deviating from the instructions given in the SPC, may increase the prevalence of resistance to ceftiofur. Whenever possible „ceftiofur VMP“ should only be used based on susceptibility testing. Do not use as prophylaxis in case of retained placenta. „Ceftiofur VMP“ is intended for treatment of individual animals. Do not use for disease prevention or as a part of herd health programmes. Treatment of groups of animals should be strictly limited to ongoing disease outbreaks according to the approved conditions of use. Amoxicillin: ATM to be used based on susceptibility testing or knowledge of epidemiological situation in the herd. Amoxicillin: Milk: max 9 days (differs across VMPs ) MRL : milk: 4 µg/ kg

Antimicrobial resistance: Responsibilities & Stakeholders Pharma industry Regulators / Policy makers MSs / EU Farmers Veterinarians

Antimicrobial resistance: Risk assessment : Risk management RA Availability of data for scientific evaluation – Evidence based Initial MA Post-authorisation Continuous benefit : risk for VMPs Risk (evidence) based Precautionary principle – to be used with caution „One size does NOT fit all“ in case of AMR MA / Use / Supply chain / Records / Inspections RM

How we are going to use the new Regulation in practice in terms of AMR ? • Definition of antimicrobial resistance in the COM draft – based on wild type resistance pattern – ECOFF values to be used as benchmark for regulatory actions ? What do we want to achieve by the new Regulation ? Do we want to use ECOFFs for resistance assessment ? Do we want to use the same criteria for VMPs evaluation in case of animal health, public health and the environment ? What impact will be on innovations / new products development • COM draft does not indicate the goals to be achieved / brings uncertainty … We need to define clearly how the benefit : risk will be assessed for antimicrobial VMPs

AMR in the new regulation on VMPs Marketing authorisation Benefits Risks Neutral Clear requirement: Benefit: risk balance definition: Data to support MA for antimicrobials prescription Benefits: no longer „therapeutic antimicrobials: only medicines benefit“ Consolidates requirements Antimicrobials presented for use as performance enhancer in order to promote the growth of treated animals or to increase yields from treated animals cannot be granted a MA BR not consistently used throughout the Regulation for outcome of assessment MRP/DCP – no dossier (data) available to CMSs for assessment Instructions to be included in the product literature (SPC, PI): Consolidates requirements Ground for refusal MA: Where antimicrobial is reserved for treatment of certain infections in humans; GMP for active substances no Classification criteria / list of longer required Reduced requirements for QP in substances to be defined by the Commission manufacturers GMP requirement for VMPs not Impact difficult to evaluate (avaialability, innovation. . ) explicitly stated

AMR in the new regulation on VMPs Post authorisation / Continuous benefit: risk Benefits Risks Extended data protection period for new antimicrobial VMPs: Will delay cheaper generics and thus reduce exposure of animals to the new compounds Variations to the MA: Major reductions in quality variations foreseen – impact on S / E Legal base for data collection system on sales and use Union interests referral: to include free movement of products BUT: The regulation must be clear what data shall be collected and for such data will be used Establishment of such system – very costly Financial/administrative burden for governments and private sector Rules adopted by EP / Council SPC harmonisation: „group“ approach, administrative not evidence based, direct risks Pharmacovigilance: Requirement significantly reduced, requirement for PSUR (period benefit : risk reevaluation by MAH) has been waived -signal detection Neutral Possibility to require postauthorisation studies in order to ensure that the benefit-risk balance remains positive with a view to the possible development of antimicrobial resistance. In line with the options provided under current legislation, but mentioned specifically for antimicrobials

AMR in the new regulation on VMPs Use of VMPs Benefits Retail of antimicrobials by person qualified to prescribe: Only animals under their care in the amount needed for treatment Risks Wholesale distribution – no definition - risk for illegal activities Internet sales Includes prescription only medicines Neutral

What is missing in the new legislation Marketing authorisation Clear reference to detailed benefit: risk methodology for assessment of VMPs (incl. Data to be submitted for fixed combinations, metaphylactic indications). Post-marketing Clear responsibility for the MAHs to ensure positive benefit: risk balance on a continuous basis – risk based PSURs and proactive pharmacovigilance by MAHs Use of VMPs Prophylactic use of VMPs shall be prohibited Rules for metaphylactic use should be defined Risk based Target pathogens resistance monitoring programme: Allowing to asses trends in AMR Public data Co-financed by EU (as the SPC shall include a clear statement zoonoses resistance monitoring of the benefit-risk that formed the programme) basis of the marketing Shall also include establishment of authorization standardised laboratory methods / interpretation criteria Attending veterinarian shall be defined Detailed knowledge about the animals, clinical diagnosis Reference products for generics applications: reference products with a clearly positive benefit: risk balance should only be used as reference products Role of veterinarians in decision making on use of antimicrobials to be defined Good husbandry practice, including regular visits by the vets Clinical trial parameters, in dossiers, that indicate performance enhancement or growth promotion should not be part of the benefitrisk balance of a veterinary antimicrobial marketing authorization. Obligation to follow instructions in the SPC MSs to draft detailed guidelines on use of antimicrobial VMPs reflecting local conditions

Main concerns: • The new Regulation on VMPs as proposed by COM would lead to deterioration of Quality/Safety/Efficacy standards for VMPs with a direct negative impact on AMR. • Main gaps identified as for: – – Variations SPC harmonisation Reduced pharmacovigilance MRP/DCP • Post-marketing – in consequence to MA : NOT in line with „One Health“ Concept • Proper balance is missing in terms of internal market and animal/public health/environmental protection

QSE of VMPs lifecycle Innovation of VMPs One Health / Responsible use of VMPs / Preventive veterinary medicine / Good animal husbandry practices Restrictions

Thank you for your attention ! Questions/ comments are welcomed ! pokludova@uskvbl. cz
- Slides: 14