Antimicrobial Pharmacotherapy in Children Paul C Walker Pharm

Antimicrobial Pharmacotherapy in Children Paul C. Walker, Pharm. D. Manager, Clinical Pharmacy Services Detroit Medical Center and Clinical Assistant Professor College of Pharmacy and School of Nursing University of Michigan

Classifying Antimicrobial Agents Inhibition of cell wall synthesis n Altering cell membrane permeability n Reversibly inhibiting protein synthesis n Irreversibly disrupting protein synthesis n Disruption of nucleic acid metabolism n Blocking essential metabolic events n
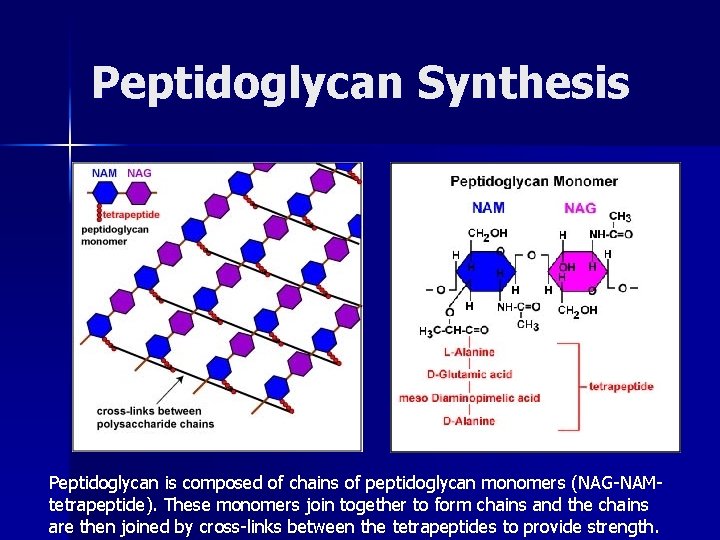
Peptidoglycan Synthesis Peptidoglycan is composed of chains of peptidoglycan monomers (NAG-NAMtetrapeptide). These monomers join together to form chains and the chains are then joined by cross-links between the tetrapeptides to provide strength.

Peptidoglycan Synthesis • • New peptidoglycan synthesis occurs at the cell division plane by way of a collection of cell division machinery known as the divisome. Bacterial enzymes called autolysins, located in the divisome, break both the glycosidic bonds at the point of growth along the existing peptidoglycan, as well as the peptide cross-bridges that link the rows of sugars together. Transglycosidase enzymes then insert and link new peptidoglycan monomers into the breaks in the peptidoglycan. Finally, transpeptidase enzymes reform the peptide cross-links between the rows and layers of peptidoglycan to make the wall strong
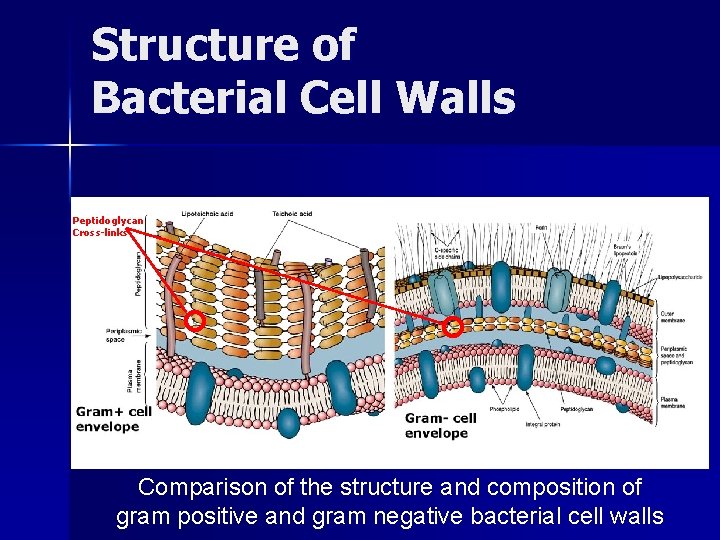
Structure of Bacterial Cell Walls Peptidoglycan Cross-links Comparison of the structure and composition of gram positive and gram negative bacterial cell walls
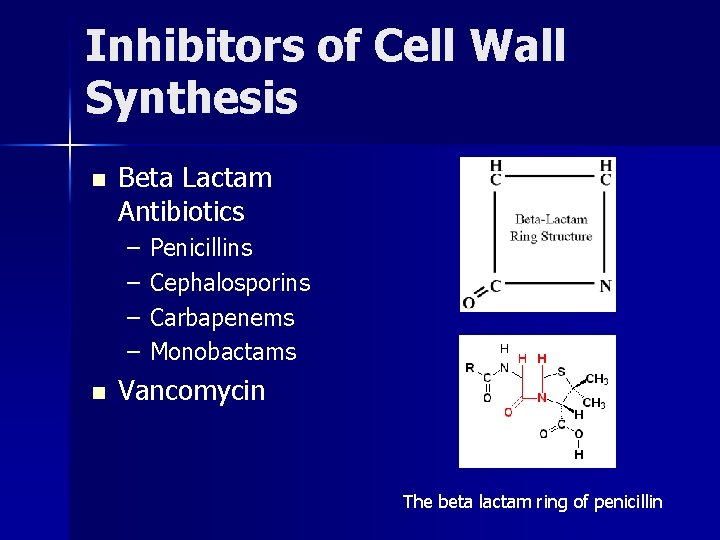
Inhibitors of Cell Wall Synthesis n Beta Lactam Antibiotics – – n Penicillins Cephalosporins Carbapenems Monobactams Vancomycin The beta lactam ring of penicillin
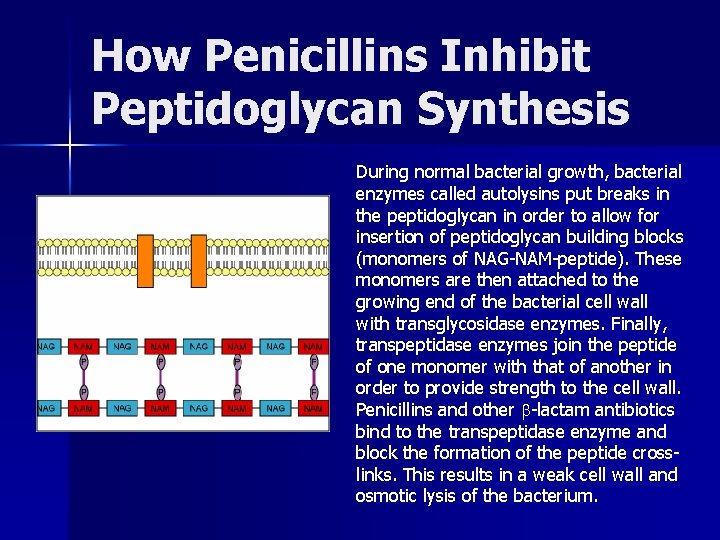
How Penicillins Inhibit Peptidoglycan Synthesis During normal bacterial growth, bacterial enzymes called autolysins put breaks in the peptidoglycan in order to allow for insertion of peptidoglycan building blocks (monomers of NAG-NAM-peptide). These monomers are then attached to the growing end of the bacterial cell wall with transglycosidase enzymes. Finally, transpeptidase enzymes join the peptide of one monomer with that of another in order to provide strength to the cell wall. Penicillins and other -lactam antibiotics bind to the transpeptidase enzyme and block the formation of the peptide crosslinks. This results in a weak cell wall and osmotic lysis of the bacterium.

Beta Lactam Antibiotics: The Penicillins n Natural Penicillins n – Penicillin G – Penicillin V n – – – Aminopenicillins – Ampicillin – Amoxicillin n Carboxypenicillins – Ticarcillin – Carbenicillin Penicillinase-Resistant Penicillins n Cloxacillin Dicloxacillin Methicillin Nafcillin Oxacillin Ureidopenicillins – Mezlocillin – Piperacillin

Beta Lactam Antibiotics: The Cephalosporins n First Generation – – – Cephalothin Cefazolin Cephalexin Cephapirin Cefadroxil Cephradine n Second Generation – – – – Cefaclor Cefoxitin Cefuroxime Cefotetan Cefpoxodime Cefprozil Cefonicid Cefmetazole n Third Generation – – – – – Cefotaxime Ceftriaxone Cefoperazone Cefipime* Cefmenoxime Ceftizoxime Ceftazidime Cefdinir Cefixime Ceftibutin * This is classified as a “fourth” generation agent; it has gram negative activity similar to other third generation agents, but better gram positive coverage.

Beta Lactam Antibiotics: The Carbapenems and Monobactams n Carbapenems – Imipenem/Cilastatin – Meropenem – Ertapenem n Monobactams – Aztreonam

Side Effects and Adverse Reactions n Beta lactam Antibiotics – Hepatic dysfunction – Acute interstitial nephritis n azotemia, hematuria, proteinuria, fever, rash, eosinophilia – Neurotoxicity – Transient blood dyscrasias – Allergic or hypersensitivity reactions – Coagulopathy

Vancomycin Indications: serious gram positive infections where -lactams are inappropriate (MRSA, MRSE, allergy, etc. ) n Toxicities and Side Effects n – Nephrotoxicity – Ototoxicity – Red Man Syndrome

Prokaryotes vs. Eukaryotes: Ribosomes
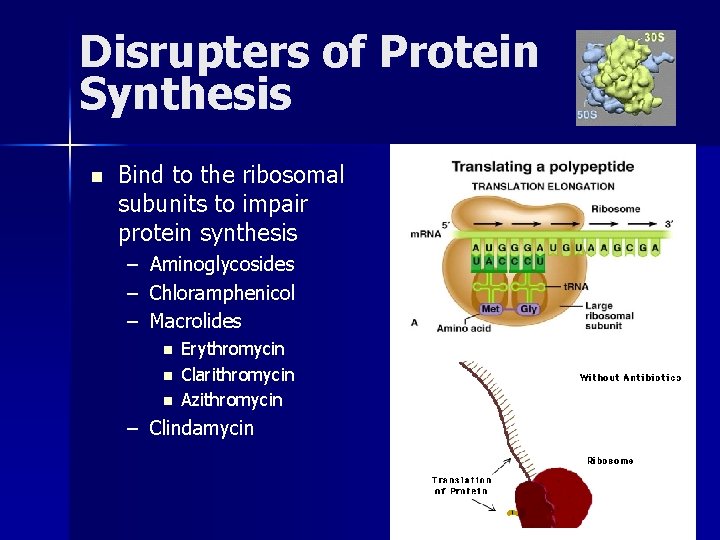
Disrupters of Protein Synthesis n Bind to the ribosomal subunits to impair protein synthesis – – – Aminoglycosides Chloramphenicol Macrolides n n n Erythromycin Clarithromycin Azithromycin – Clindamycin

The Aminoglycosides n n n Kanamycin Gentamicin Tobramycin Amikacin Netilmicin Sisomycin Structure of the antibiotic gentamicin C 1 a bound to its RNA target. Aminoglycoside antibiotics cause misreading of the genetic code. Blocks initiation of protein synthesis Blocks translation to cause premature termination Causes incorporation of incorrect amino acid Aminoglycosides bind to the 30 s subunit to impair protein synthesis.

Agents that Bind to the 50 S Ribosome n Chloramphenicol – spectrum of activity n S. pneumonia n H. influenza n Neisseria spp. n Salmonella n Bordetella n Enterobacteriaceae n some anaerobes

Agents that Bind to the 50 S Ribosome n Macrolides – Erythromycin n S. pneumonia, S. pyogenes, Legionella, Chlamydia trachomatis, M. catarrhalis, H. influenza, Mycoplasma pneumonia – Clarithromycin n MAC – Azithromycin n MAC n Clindamycin – aerobic gram-positive bacteria – anaerobes, especially B. fragilis – used in combination with aminoglycosides to treat intra-abdominal and gynecologic infections

Side Effects and Adverse Reactions n Chloramphenicol – Gray syndrome – Dose-dependent bone marrow suppression – Aplastic anemia, pancytopenia n n Macrolides – – GI complaints Rash – – Diarrhea Pseudomembranous colitis Rash, urticaria Hypotension Clindamycin – –

Disrupters of Nucleic Acid Metabolism n n Metronidazole Quinolones: – – – – Ciprofloxacin Levfloxacin Moxifloxacin Norfloxacin Ofloxacin Trovafloxacin Gatifloxacin Grepafloxacin

Disrupters of Nucleic Acid Metabolism n Metronidazole Participates in redox reactions; it is activated by a reduction of the nitro group to an anion radical. In the case of metronidazole, reduced ferredoxin appears to be the primary electron donor responsible for its reduction The anion radical is highly reactive and will form adjuncts with proteins and DNA leading to a loss of function. In particular, the reactions with DNA result in strand breakage and inhibition of replication and will lead to cell death.

Disrupters of Nucleic Acid Metabolism n Quinolones: inhibit DNA -gyrase and topoisomerase II – – – – Ciprofloxacin Levfloxacin Moxifloxacin Norfloxacin Ofloxacin Trovafloxacin Gatifloxacin Grepafloxacin

Side Effects and Adverse Reactions n Metronidazole – dizziness – paresthesias – peripheral neuropathy – disulfiram-like reaction – blood dyscrasias n Quinolones – headache – rash, photosensitivity – GI complaints – arthralgias – confusion – liver dysfunction

Antimetabolites Trimethoprim n Sulfonamides n – Sulfamethoxazole – Sulfisoxazole Inhibition of folate metabolism by sulfonamides and trimethoprim

Side Effects and Adverse Reactions n Sulfonamides – Dizziness, headache – Rash – Blood dyscrasias – Crystalluria – Acute nephropathy – Bilirubin displacement

Proper Antimicrobial Selection: Factors to Consider Identity of infecting organism n Susceptibility of infecting organism n Host Factors n

Major Mechanisms of Antimicrobial Resistance Target site modification (intracellular or extracellular; -lactams, macrolides, quinolones, glycopeptides) Enzymatic degradation (intracellular or extracellular; -lactams, aminoglycosides) Decreased permeability ( -lactams) X Bypass (TMP/SMX) Efflux (macrolides, quinolones)

Enzyme Inactivation of Penicillins 2 1 1 = Site of action of penicillinase 2 = Site of action of amidase A = Thiazolidine ring B = -lactam ring Structure of penicillins and interaction with beta lactamase
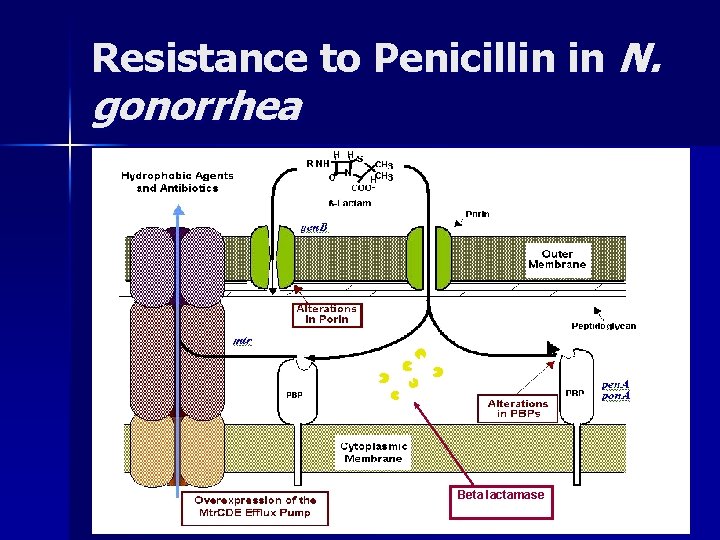
Resistance to Penicillin in N. gonorrhea Beta lactamase

Bacterial Resistance: What Problems are We Seeing? n Gram Negative Organisms – – – H. Influenza M. Catarrhalis Enterobacter Klebsiella Citrobacter Serratia n Gram Positive – Staphylococcus n n S. aureus S. epidermidis – Streptococcus n n S. pneumoniae Vancomycin – Enterococci n n E. faecalis E. faecium – S. aureus

Other Important Factors: MICs and MBCs Fail to Tell the Whole Story n Antimicrobial Pharmacodynamics – attempt to characterize the relationship between ANTIMICROBIAL EXPOSURE (concentration, dose, AUC) and ANTIMICROBIAL EFFECT (eg. , rate, extent, and duration of antimicrobial activity)

Other Important Factors: MICs and MBCs Fail to Tell the Whole Story n Antibiotic Pharmacodynamics – Rate and Extent of Bactericidal Action – Post-antibiotic Effect – Effects of Sub-inhibitory Concentrations – Post-antibiotic Leukocyte Effect – Inoculum Effect

Classification Based on Pharmacodynamic Characteristics – Concentration-Dependent Agents n Bactericidal activity is dependent on concentration above the MIC achieved, increasing with increasing concentration – Time-Dependent Agents n Bactericidal activity is dependent on how long the concentration exceeds the MIC – Bacteriostatic Agents n Abort bacterial growth and allow host defenses to eradicate organisms
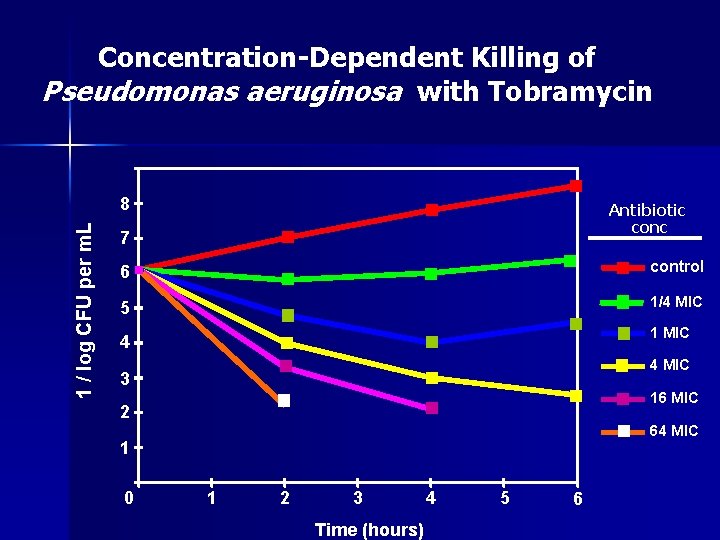
Concentration-Dependent Killing of Pseudomonas aeruginosa with Tobramycin 1 / log CFU per m. L 8 Antibiotic conc 7 6 control 5 1/4 MIC 4 1 MIC 4 MIC 3 16 MIC 2 64 MIC 1 0 1 2 3 Time (hours) 4 5 6
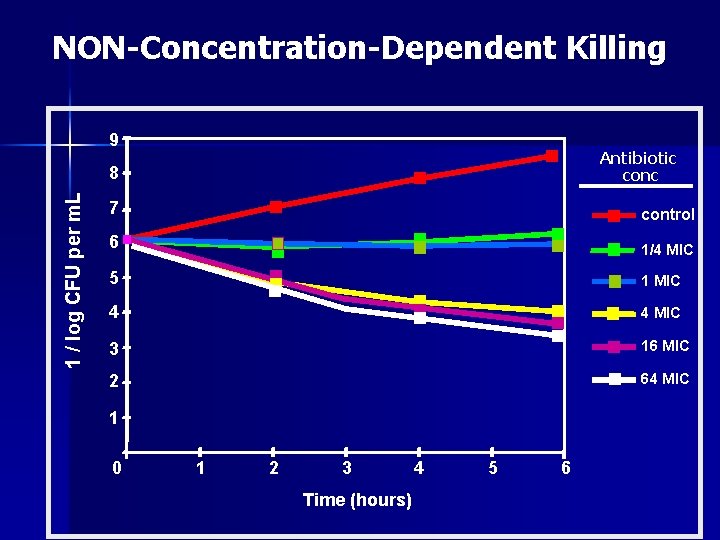
NON-Concentration-Dependent Killing 9 Antibiotic conc 1 / log CFU per m. L 8 7 control 6 1/4 MIC 5 1 MIC 4 4 MIC 3 16 MIC 2 64 MIC 1 0 1 2 3 Time (hours) 4 5 6

Pharmacodynamic Properties by Antibiotic Class CONCENTRATION dependent killing Aminoglycosides Fluoroquinolones Azithromycin? TIME dependent killing β-lactams Glycopeptides Metronidazole Macrolides (except Azithromycin) Chloramphenicol Rifampin Tetracyclines Clindamycin
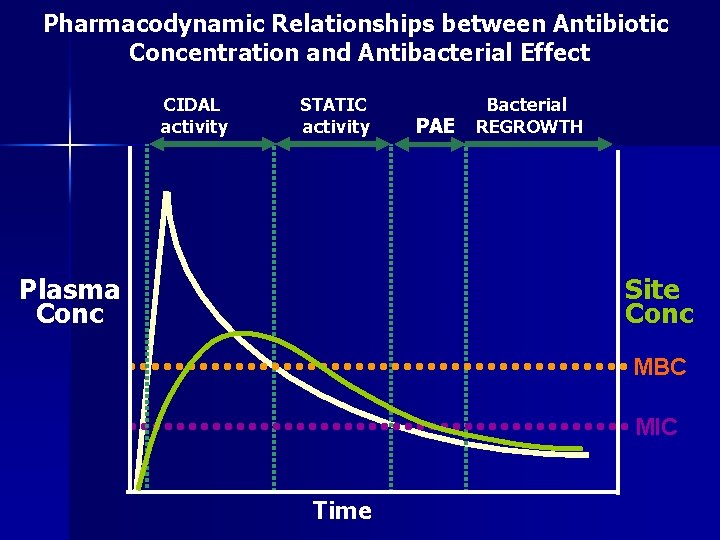
Pharmacodynamic Relationships between Antibiotic Concentration and Antibacterial Effect CIDAL activity STATIC activity Plasma Conc PAE Bacterial REGROWTH Site Conc MBC MIC Time
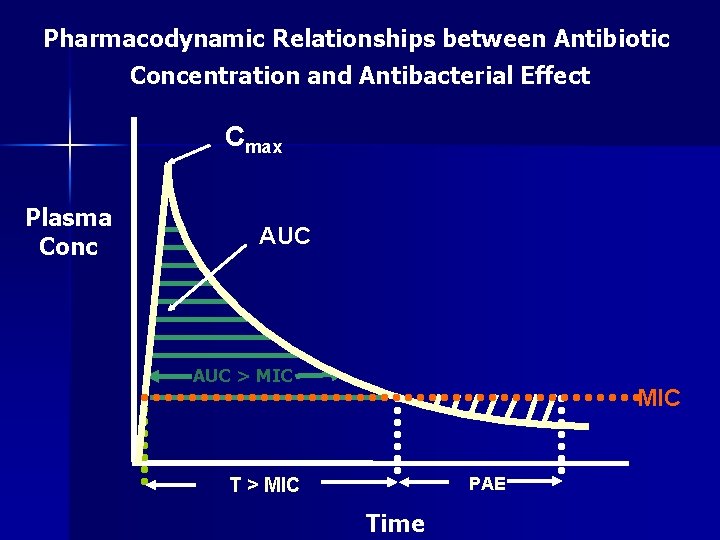
Pharmacodynamic Relationships between Antibiotic Concentration and Antibacterial Effect Cmax Plasma Conc AUC > MIC PAE T > MIC Time
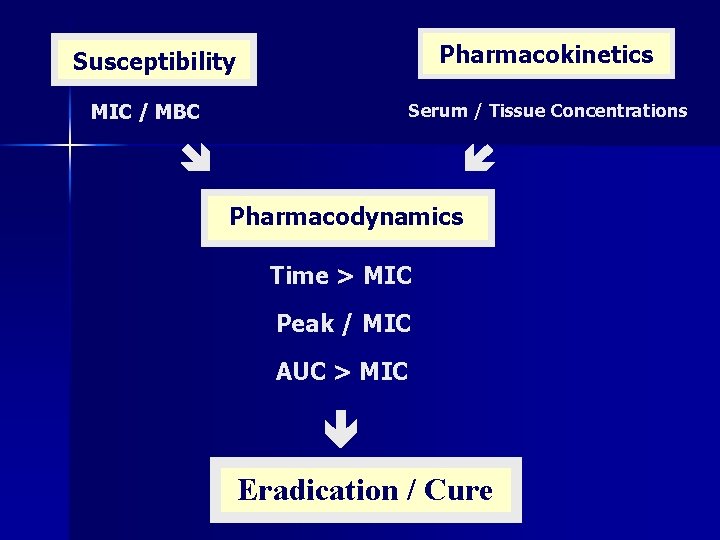
Pharmacokinetics Susceptibility MIC / MBC Serum / Tissue Concentrations Pharmacodynamics Time > MIC Peak / MIC AUC > MIC Eradication / Cure

Antibiotic Pharmacodynamics in Otitis Media: T>MIC Average percentage of time drug concentration exceeds the minimum inhibitory concentration (%T>MIC) for pediatric dosages of oral ß-lactam agents against penicillin-sensitive (black bars) and penicillin-intermediate (hatched bars) Streptococcus pneumoniae. Rodvold. Pharmacoatherapy. 2001; 21(11 s) : 319 s-330 s.
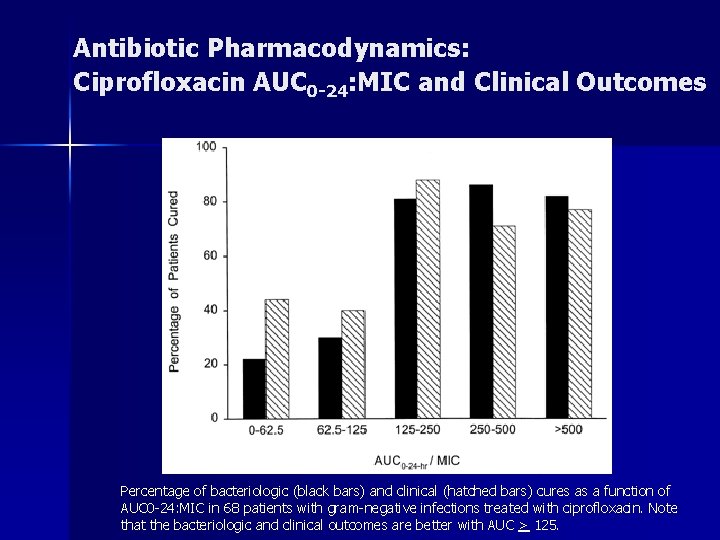
Antibiotic Pharmacodynamics: Ciprofloxacin AUC 0 -24: MIC and Clinical Outcomes Percentage of bacteriologic (black bars) and clinical (hatched bars) cures as a function of AUC 0 -24: MIC in 68 patients with gram-negative infections treated with ciprofloxacin. Note that the bacteriologic and clinical outcomes are better with AUC > 125.
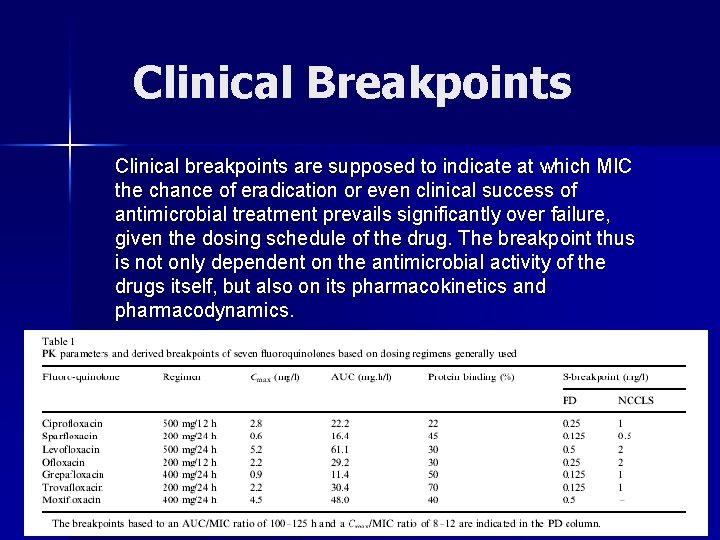
Clinical Breakpoints Clinical breakpoints are supposed to indicate at which MIC the chance of eradication or even clinical success of antimicrobial treatment prevails significantly over failure, given the dosing schedule of the drug. The breakpoint thus is not only dependent on the antimicrobial activity of the drugs itself, but also on its pharmacokinetics and pharmacodynamics.

Postantibiotic effect n The period of time where there is persistent suppression of bacterial growth following exposure to an antimicrobial agent, despite removal of the antimicrobial agent.
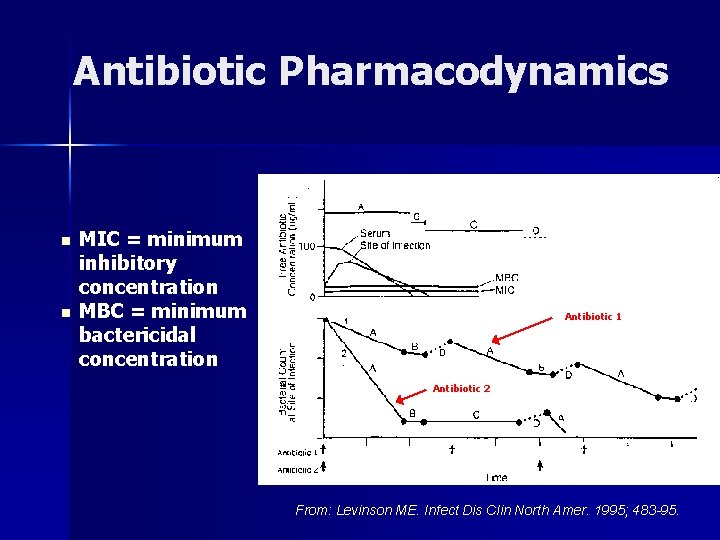
Antibiotic Pharmacodynamics n n MIC = minimum inhibitory concentration MBC = minimum bactericidal concentration Antibiotic 1 Antibiotic 2 From: Levinson ME. Infect Dis Clin North Amer. 1995; 483 -95.

Antibiotic Combinations: Rationale and Indications Additive Effects n Synergistic Effects n Antagonistic Effects n

Antibiotic Synergy and Antagonism

Antibiotic Combinations: Rationale and Indications Prevent emergence of resistance n Polymicrobial infections n Empiric therapy n Reduced drug toxicity n Synergism n

Antibiotic Combinations: Disadvantages of Inappropriate Combination Therapy Antagonism n Increased drug costs n Adverse drug reactions n
- Slides: 47