Antigen Antibody Reactions w Antigen combines with its


Antigen – Antibody Reactions w Antigen combines with its specific Antibody in observable manner. w Reaction between Ag & Ab specific. w Ag – Ab reactions in – vitro Serological tests. 2

Uses of Antigen – Antibody Reactions In the Body or in Vivo : w Forms the basis of antibody mediated (humoral) immunity against infectious diseases. w In the Lab. or in Vitro: w For diagnosis of infection. w Helpful in epidemiological studies. May lead to tissue injury w For identification of non in some hypersensitivity infectious agents such reactions & autoimmune as enzymes. diseases. w Detection & quantitation of either Ag or Ab. 3
Nature of Ag/Ab Reactions w Lock & Key concept. w Non covalent bonds. - Hydrogen bonds - Electrostatic bonds - Van der Waal forces - Hydrophobic bonds 4

Stages of Antigen – Antibody Reactions w Ag – Ab Reaction occurs in two stages 1. PRIMARY 2. SECONDARY w Primary Stage: w Initial, rapid, reversible, reaction between Ag & Ab without any visible effect, occurring at low temp. w Binding between Ag & Ab by weaker intermolecular forces. w No covalent bonding 5

Secondary Stage w In most but not all primary stage followed by secondary one. w Leads to demonstrable (visible) effects ------* Precipitation. * Agglutination. * Lysis of cell. * Killing of live antigens. * Neutralisation of toxins. * Complement fixation. * Immobilisation of motile organisms. * Phagocytosis. 6
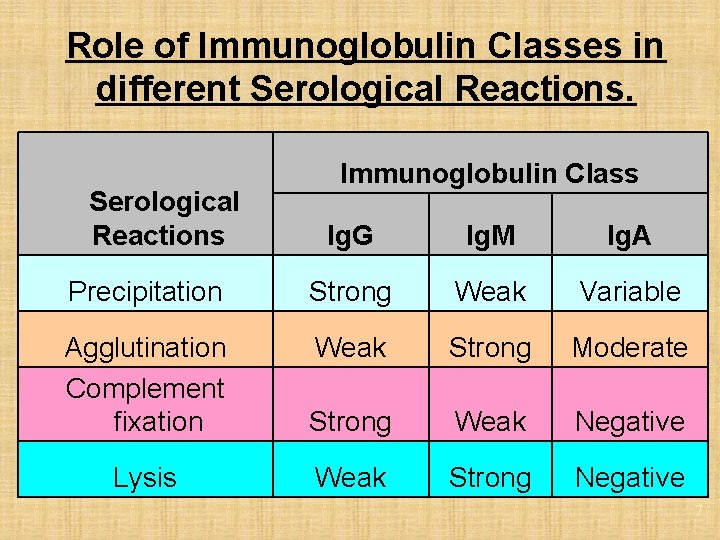
Role of Immunoglobulin Classes in different Serological Reactions Immunoglobulin Class Ig. G Ig. M Ig. A Precipitation Strong Weak Variable Agglutination Complement fixation Weak Strong Moderate Strong Weak Negative Lysis Weak Strong Negative 7

Characteristics of Ag – Ab Reaction w Specific: Ag combines only with its homologous Ab & vice versa. w Specificity not absolute. Cross reactions occur due to antigenic similarity or relatedness. w Entire molecules of Ag & Ab & not fragments react. w No denaturation of Ag or Ab during reaction. w Ag – Ab combination firm but reversible. w Firmness of combination depends on - Affinity & Avidity 8
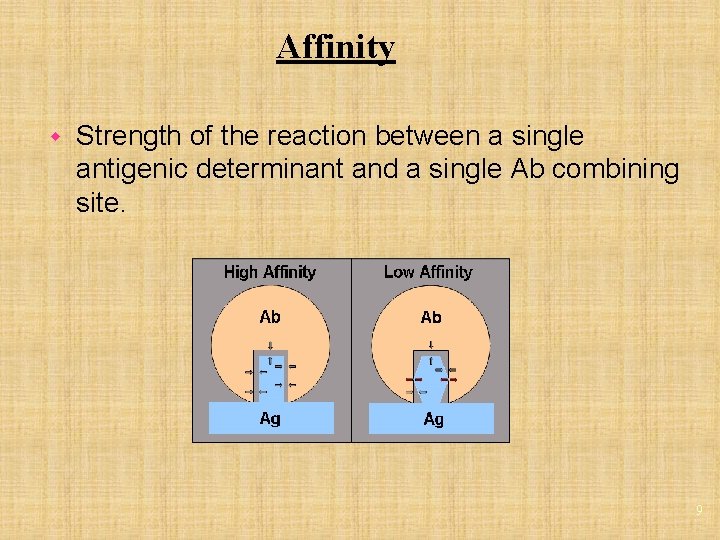
Affinity w Strength of the reaction between a single antigenic determinant and a single Ab combining site. 9

Avidity w The overall strength of binding between an Ag with many determinants and multivalent Abs. 10

Precipitation Tests 11

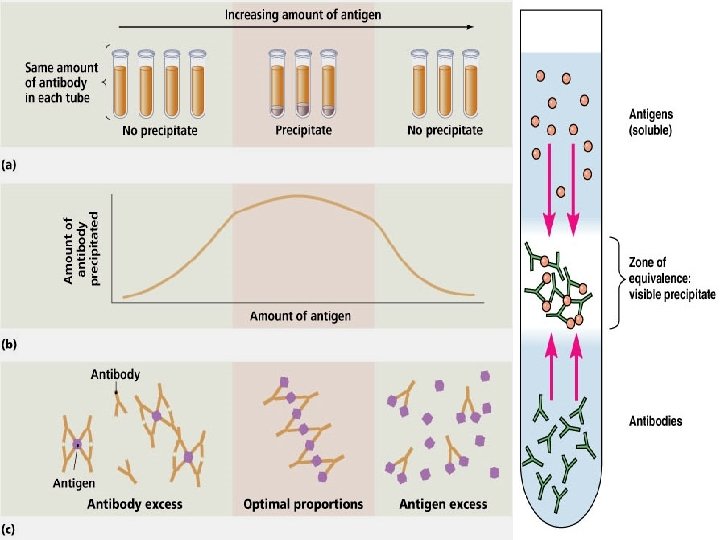
Precipitation Reaction w When a soluble antigen combines with its antibody in presence of electrolytes ( Na. Cl) at suitable temperature & p. H, the Ag – Ab complex forms an insoluble precipitate. w The Ag – Ab complex forms an insoluble precipitate which remains suspended instead of settling down ‘ Flocculation’. 12

Precipitation reactions: w 1. Zone of antibody excess - precipitation is inhibited antibody not bound to antigen can be detected in the supernatant; w 2. Zone equivalence - maximal precipitation in which antibody and antigen form large insoluble complexes and neither antibody nor antigen can be detected in the supernatant; and w 3. Zone of antigen excess - precipitation is inhibited & Ag. not bound to Ab. can be detected in the supernatant.

Applications Qualitative & Quantitative test. w Very sensitive in detecting antigens & as little as 1 g of protein. Less sensitive to detect antibody. 1) Ring test Ascoli’s thermoprecipitin test , Grouping of streptococci by Lancefield technique. w 2) Slide test – VDRL test for syphilis = eg of flocculation test. 3) Tube test – Kahn test eg of tube flocculation test. 4) Quantitative tube flocculation test – used for standardisation of toxins & toxoids. 15

Immunodiffusion ( Precipitation in Gel) w Several advantages in allowing precipitation to occur in gel than in liquid. w Reaction visible as distinct & stable band of precipitation. w Can be stained for preservation. 16
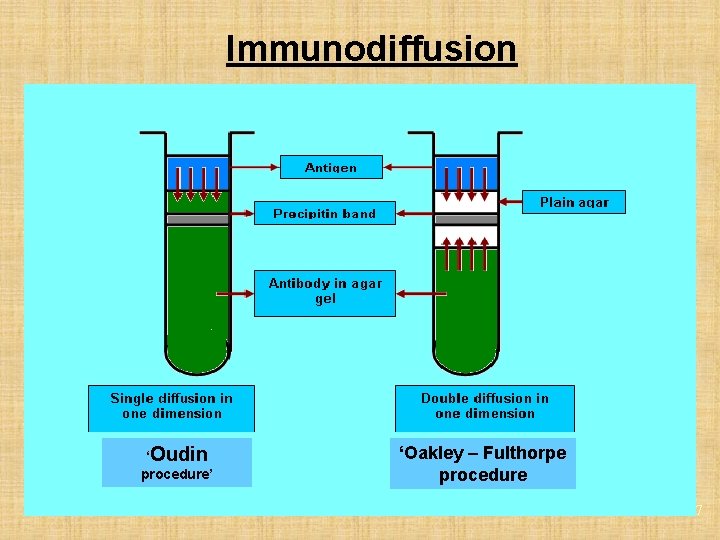
Immunodiffusion ‘Oudin procedure’ ‘Oakley – Fulthorpe procedure 17
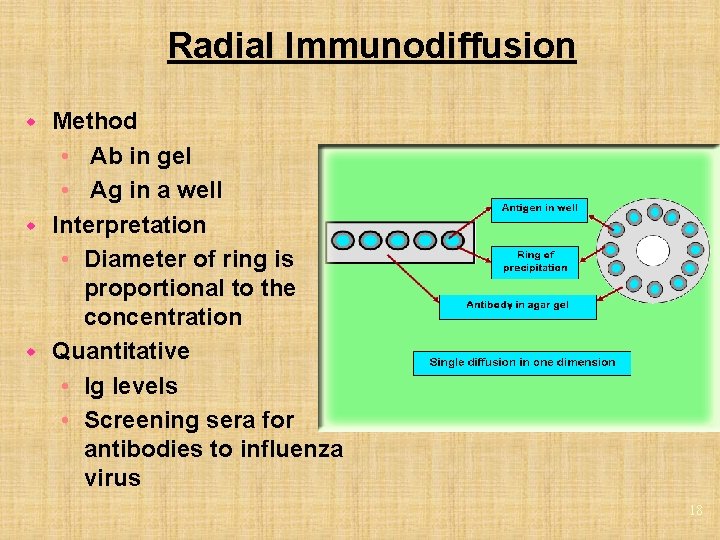
Radial Immunodiffusion Method • Ab in gel • Ag in a well w Interpretation • Diameter of ring is proportional to the concentration w Quantitative • Ig levels • Screening sera for antibodies to influenza virus w 18
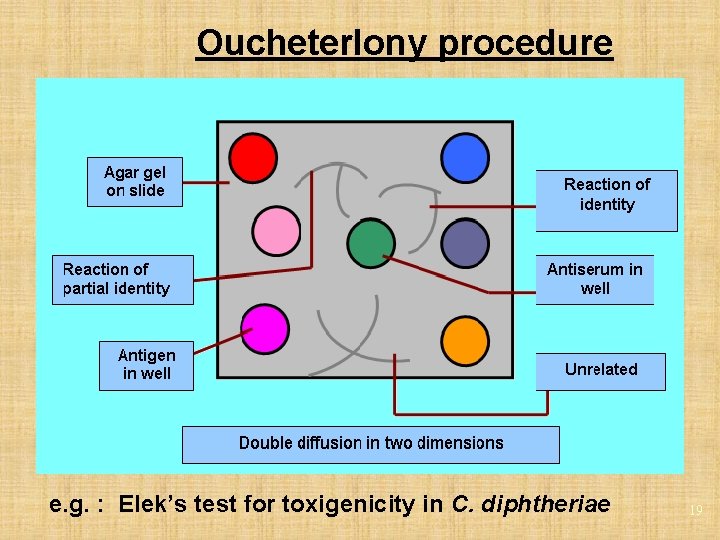
Oucheterlony procedure e. g. : Elek’s test for toxigenicity in C. diphtheriae 19

Immunoelectrophoresis 20

Countercurrent electrophoresis w Method • Ag and Ab migrate toward each other by electrophoresis • Used only when Ag and Ab have opposite charges • Qualitative • For Hepatitis B Ag & Ab , Antigens of Cryptococcus in C. S. F. - + Ag Ab 21
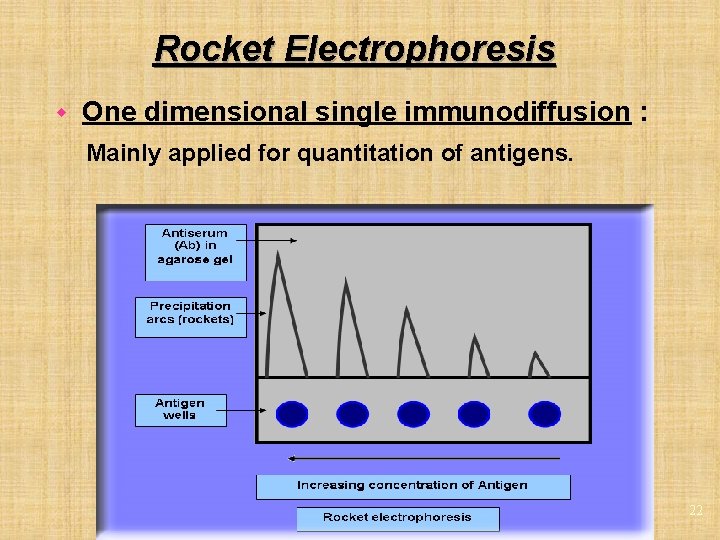
Rocket Electrophoresis w One dimensional single immunodiffusion : Mainly applied for quantitation of antigens. 22

Agglutination Reactions 23

Agglutination Reactions w Particulate antigen combining with its antibody in presence of electrolyte at optimal temp. & p. H, resulting in visible clumping of particles. w Incomplete or monovalent Ab do not cause agglutination. w More sensitive than precipitation for detection of Ab. Better with Ig. M than Ig. G. 24

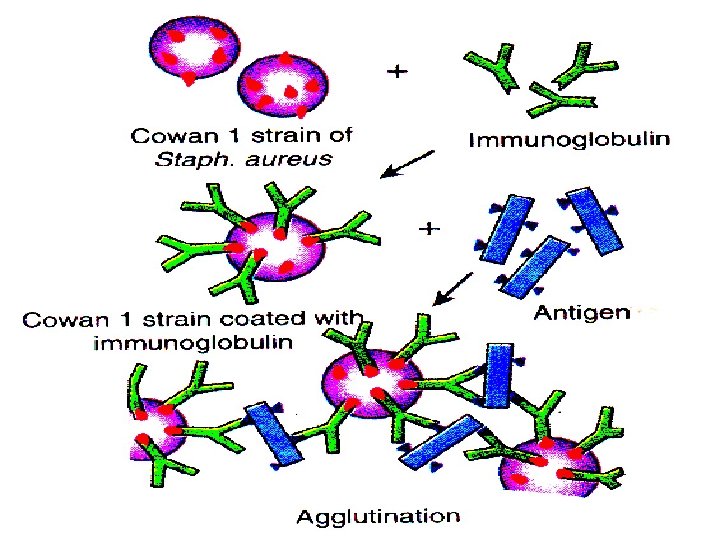
Agglutination Tests w w Agglutination occurs due to the cross-linking of particulate antigens by antibody molecules. Agglutination is the visible clumping of insoluble particles, whereas precipitation involves the aggregation of soluble molecules

TYPES w w w Slide agglutination Tube agglutination The antiglobulin (Coomb’s) test Passive agglutination test Heterophile agglutination test Haemagglutination test 26
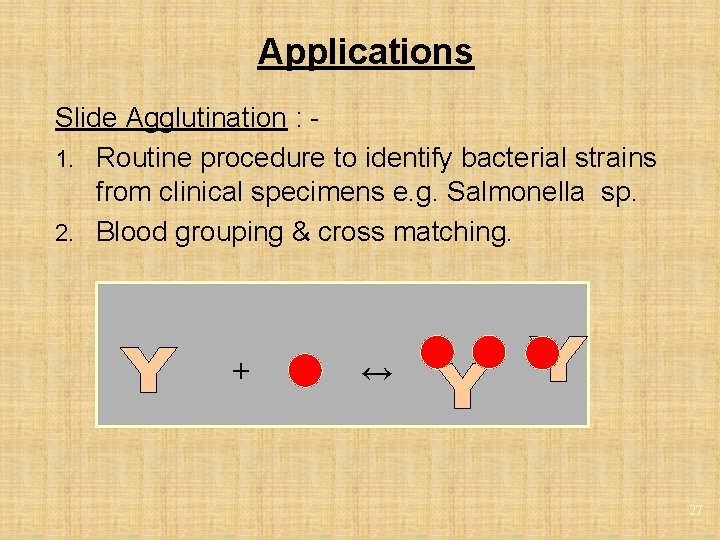
Applications Slide Agglutination : 1. Routine procedure to identify bacterial strains from clinical specimens e. g. Salmonella sp. 2. Blood grouping & cross matching. + ↔ 27

Applications Tube Agglutination test : - standard quantitative method for measurement of antibodies. Fixed volume of particulate Ag+ Equal volume of serial dilutions of antiserum= Agglutination 1. Enteric fever ( Widal test). 2. Typhus fever (Weil Felix test). 3. Infectious mononucleosis ( Paul Bunnel test). 4. Brucellosis. 5. Primary atypical pneumonia. 28

Coombs (Antiglobulin)Tests Incomplete Ab w Direct Coombs Test • Detects antibodies on erythrocytes w + Patient’s RBCs ↔ Coombs Reagent (Antiglobulin) 29
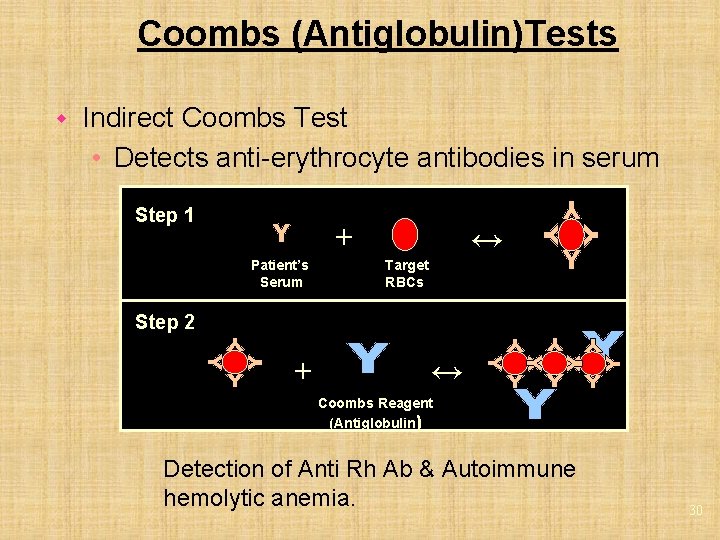
Coombs (Antiglobulin)Tests w Indirect Coombs Test • Detects anti-erythrocyte antibodies in serum Step 1 + Patient’s Serum ↔ Target RBCs Step 2 + ↔ Coombs Reagent (Antiglobulin) Detection of Anti Rh Ab & Autoimmune hemolytic anemia. 30

Passive Agglutination Test w A precipitation reaction can be converted to agglutination test by attaching soluble antigens to the surface of carrier particles, bentonite, latex particles, red blood cells. Latex Agglutination test : w For detection of Hepatitis B Ag. w ASO, w RA factor ( Rose Waller test) 31

Reverse passive Agglutination test w Antibodies are bound to the surface of carrier particles, instead of antigen. Human chorionic Gonadotropin (HCG) w C Reactive Protein (CRP) w 32


Immuno. Fluorescence is the property of absorbing lighter of one particular wavelength (UV) and emitting rays of different wavelength (Visible). w Uses fluorescent dyes as labels • Dyes: Fluorescein, isothiocyanate, lissamine, rhodamine. chemically linked to an antibody • Glows bright green when exposed to fluorescent light w Types: • Direct fluorescent antibody test • Indirect fluorescent antibody tests w
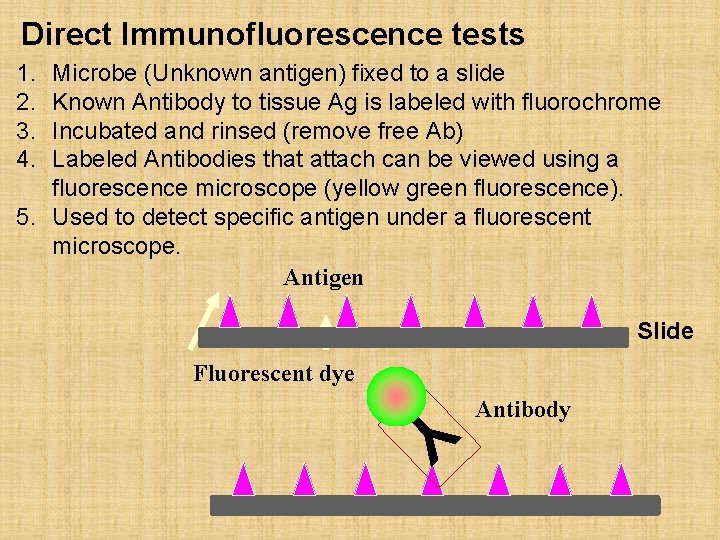
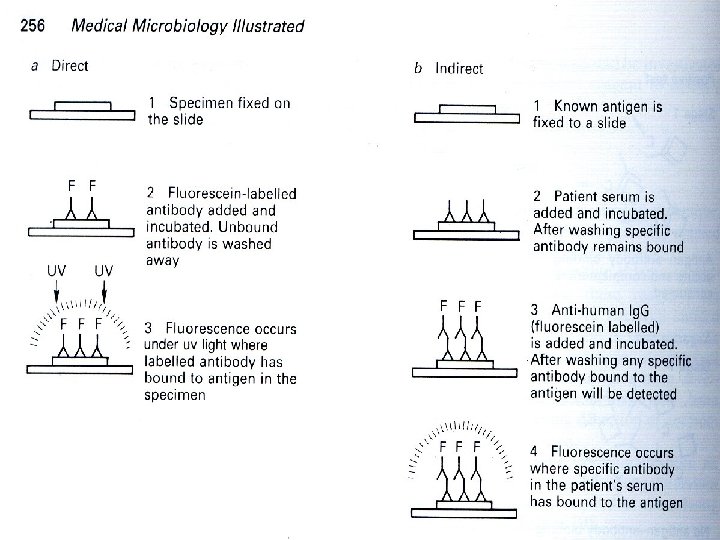
Direct Immunofluorescence tests 1. 2. 3. 4. Microbe (Unknown antigen) fixed to a slide Known Antibody to tissue Ag is labeled with fluorochrome Incubated and rinsed (remove free Ab) Labeled Antibodies that attach can be viewed using a fluorescence microscope (yellow green fluorescence). 5. Used to detect specific antigen under a fluorescent microscope. Antigen Slide Fluorescent dye Antibody Y

Indirect Immunofluorescence test Used to demonstrate the presence of antibody in serum. w Ab to tissue Ag is unlabeled w Fluorochrome-labeled anti-Ig is used to detect binding of the first (serum) Antibody w Unlabeled Ab Fluorochrome Labeled Anti-Ig Ag Tissue Section

Complement mediated serological reactions Comp. fixation test Immune adherence Immobilization test Cytolytic or cytocidal reaction

Complement fixation test Highly sensitive and diagnostically important test for serum antibody. Capable of detecting as little as 0. 04 microgram of Ab and 0. 1 microgram of Antigen. Consists of 2 steps and 5 reagents: Ag, Ab, Complement, Sheep erythrocytes, Amboceptor (Rabbit Ab to sheep RBC)

Principle (Ag+Ab) Complex Complement is activated and there by consumed or utilized. Indicator system (sheep red cells + Anti-sheep red cell Ab) is then added to detect the presence of remaining complement. If is the comp. has been utilized by Ag+Ab reaction No haemolysis (positive) If comp. has not been consumed (sheep RBC+Ab to sheep RBC) +comp. Haemolysis (Negative)

Examples: Wasserman reaction Diagnosis of syphilis. Reagents: i) Ag (cardiolipin from Bovine heart) ii) Serum to be tested for Ab. Decomplemented at 56 o C for 30 mins. iii) Pooled guinea pig serum (source for complement) iv) Haemolytic system (amboceptor)


Complement Fixation • Methodology – Ag mixed with test serum to be assayed for Ab – Standard amount of complement is added – Erythrocytes coated with Abs is added – Amount of erythrocyte lysis is determined No Ag Ag Patient’s serum

Indirect Complement fixation test w Certain avian (duck, turkey) and mammalian (cat, horse) sera don't fix guinea pig complement. w Test set up in duplicate, and after first step std antiserum known to fix complement is added to one set. w If test serum contains Ab the Ag has been used up in the first step and therefore Std Antiserum added later will not be able to fix the complement. w Hemolysis indicates positive test. 45

Other comp. mediated serological reactions T P I (Treponema pllidum Immobilisation test TP (Nichols strain) + serum (Ab) + complement Immobilization of living treponemes seen by dark field microscope. Immune adherence Bacteria (V. cholera + Tr. pallidum) + Sp Ab + Comp. + particulate materials (RBC, platelets etc. ) Bact. are aggregated and adhere to cells Phagocytosis. Cytolytic cytocidal Test: (V. Cholera + Sp Ab + Comp. ) Bacterium is killed.

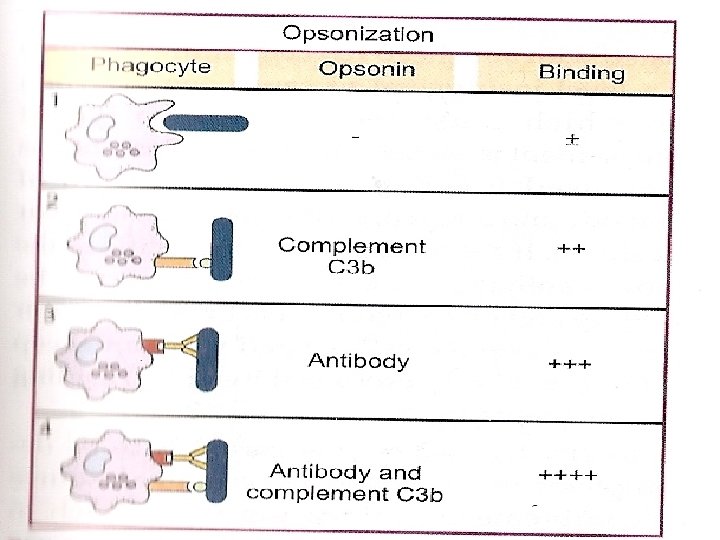
OPSONIZATION Opsonins are the substances (Complement and Ig. M) which coats on the target cells and make them palatable for phagocytosis. The process initiated by opsonin is called Opsonization

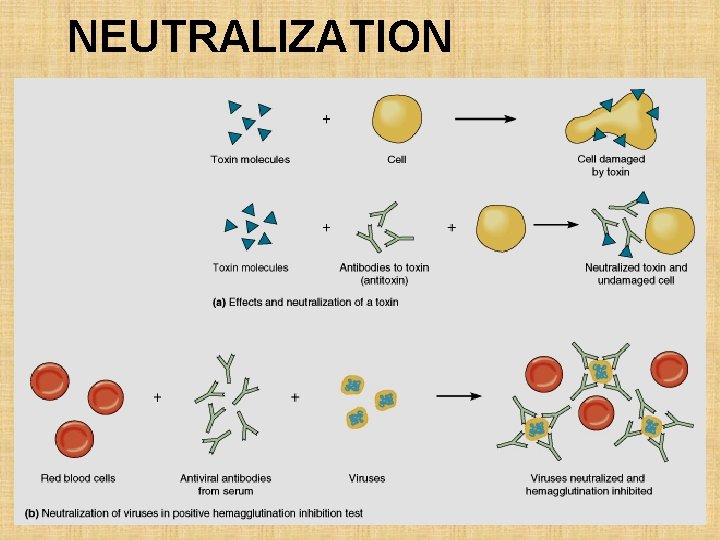
NEUTRALIZATION TEST Virus neutralization: - Neutralization of bacteriophage can be demonstrated in plaque inhibition. - Ab prevents virus adsorption to the receptor. - Viral Heamagglutination inhibition (HI) test HI is used in the diagnosis of influenza, measles, mumps etc. Toxin Neutralization: ATS, ADS in vivo, Schick test, (Invivo), ASO: Invitro
NEUTRALIZATION

Radioimmunoassay (RIA) • Detection of antigen. • Known amounts of radioactively labeled antigen, known antibody and unknown sample mixed together. • If sample has high level of antigen, it will compete with labeled antigen and little radioactive antigen will bind. • If sample has low level of antigen, it will not be able to compete with labeled antigen and much radioactive antigen will bind.
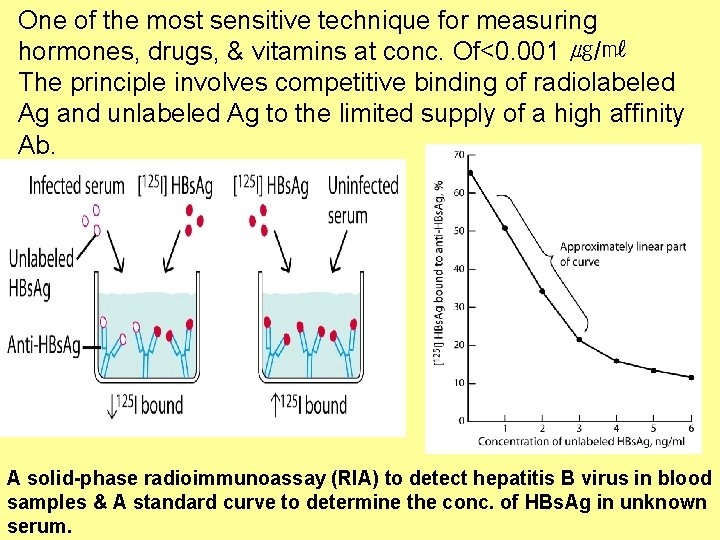
One of the most sensitive technique for measuring hormones, drugs, & vitamins at conc. Of<0. 001 ㎍/㎖ The principle involves competitive binding of radiolabeled Ag and unlabeled Ag to the limited supply of a high affinity Ab. A solid-phase radioimmunoassay (RIA) to detect hepatitis B virus in blood samples & A standard curve to determine the conc. of HBs. Ag in unknown serum.

Assay Procedure • Add known amounts of the test sample + labelled antigen into the microtitre wells • Incubate allow the reaction to reach completion • Decant & wash contents of the well removes all unbound antigens • Radioactivity remaining in the Micro titre wells measured by a Counter [GM counter , Scintillation counter etc] • Intensity of radioactivity is inversely correlated with the conc of antigens in the test sample • Sensitive to very low conc of antigens

Advantages & Disadvantages of RIA • Advantages – Immune reactions are highly specific – Immune reactions are highly sensitive • Disadvantages – Radiation hazards: Uses radiolabeled reagents – Requires specially trained persons – Labs require special license to handle radioactive material – Requires special arrangements for • Requisition, storage of radioactive material • Radioactive waste disposal.

Enzyme-Linked Immunosorbent Assay • Principle of ELISA : • Detection based on enzyme catalysed reaction or Fluorescent probe • ELISA techniques use antibodies linked to an enzyme as horse radish peroxidase or alkaline phosphatase. • Ag-Ab reactions are detected by the enzymesubstrate reaction. • A color change indicates an antigen-antibody reaction has occurred.

Types • The direct ELISAü Sandwich or Capture Assays & ü Competitive ELISA - Used to detect antigens • The indirect ELISA ü Used to detect specific antibodies (i. e. HIV in serum) against antigen bound in a test well.
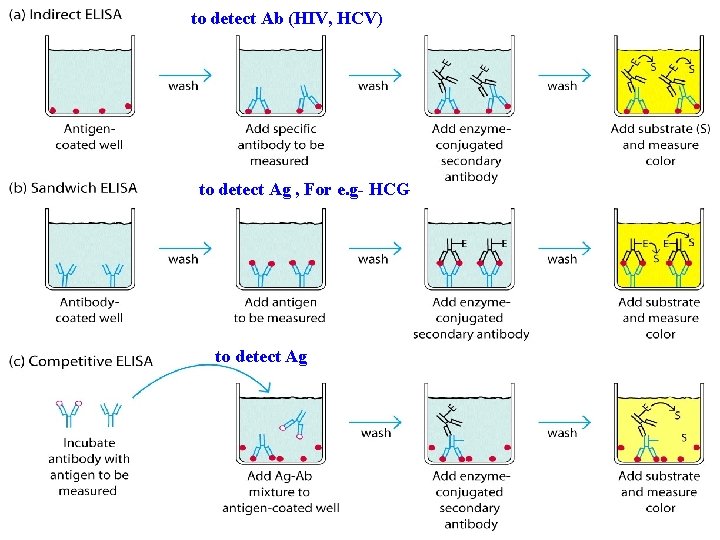
to detect Ab (HIV, HCV) to detect Ag , For e. g- HCG to detect Ag
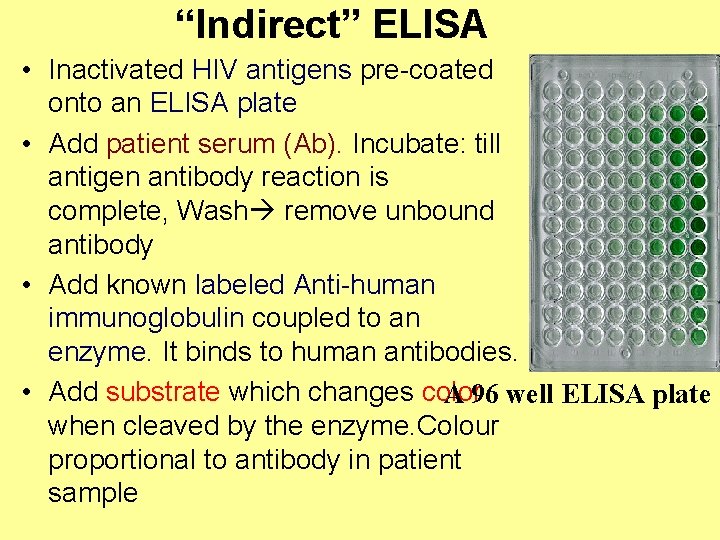
“Indirect” ELISA • Inactivated HIV antigens pre-coated onto an ELISA plate • Add patient serum (Ab). Incubate: till antigen antibody reaction is complete, Wash remove unbound antibody • Add known labeled Anti-human immunoglobulin coupled to an enzyme. It binds to human antibodies. • Add substrate which changes color A 96 well ELISA plate when cleaved by the enzyme. Colour proportional to antibody in patient sample

Noncompetitive ELISA or “Indirect” ELISA Serological Tests

Direct ELISA Sandwich or Capture Assays • Used for antigens with multiple epitopes • Antibody to one epitope fluid, antibody to second epitope fixed. • Enzyme label used to detect reaction Competitive ELISA • Titre wells coated with antibodies • Add known quantities of patient sample containing antigen + antigen labelled with enzyme • Unknown antigen competes with labeled known antigen • Enzyme + Substrate Product measure colour • Colour inversely related to antigen in patient sample
![Applications of Immunoassays [RIA & ELISA] • Analysis of hormones, vitamins, metabolites, diagnostic markers Applications of Immunoassays [RIA & ELISA] • Analysis of hormones, vitamins, metabolites, diagnostic markers](http://slidetodoc.com/presentation_image_h/e93900b6d04a31738606514fae41a031/image-61.jpg)
Applications of Immunoassays [RIA & ELISA] • Analysis of hormones, vitamins, metabolites, diagnostic markers – Eg. ACTH, FSH, T 3, T 4, Glucagon, Insulin, Testosterone, vitamin B 12, prostaglandins, glucocorticoids, • Therapeutic drug monitoring: – Barbiturates, morphine, digoxin, • Diagnostic procedures for detecting infection – HIV, Hepatitis A, Hepatitis B etc. ,

Advantages of ELISA • Sensitive: Nanogram levels or lower • Minimal reagents • Qualitative & Quantitative – Qualitative E. g. HIV testing – Quantitative assays E. g. Theraputic Drug Monitoring • Greater scope : Wells can be coated with Antigens or Antibodies • Suitable for automation high speed • NO radiation hazards

Western Blot • Combines Electrophoresis with ELISA to separate and identify antigens • Confirms positive ELISA results (ELISA may give some false +ve) Steps: • Antigen mixture separated by gel electrophoresis • Blotted onto a filter paper • Serum sample applied to filter • Labels used to interpret results • Used to confirm positive HIV test
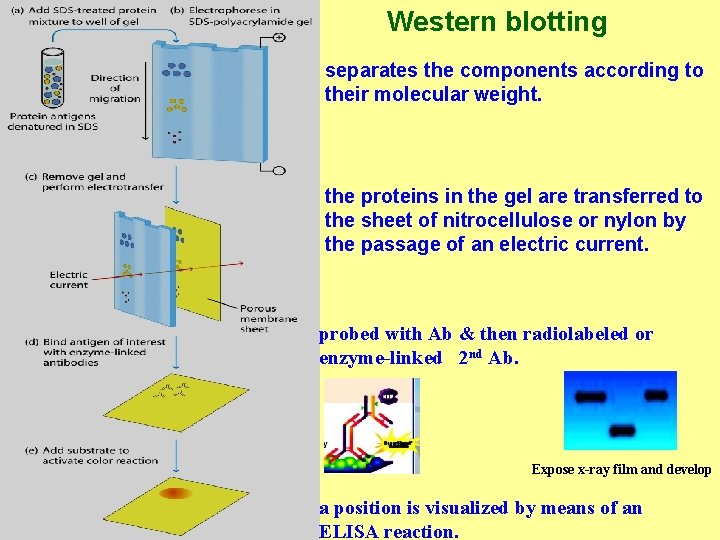
Western blotting separates the components according to their molecular weight. the proteins in the gel are transferred to the sheet of nitrocellulose or nylon by the passage of an electric current. probed with Ab & then radiolabeled or enzyme-linked 2 nd Ab. Expose x-ray film and develop a position is visualized by means of an ELISA reaction.

Western Blotting
- Slides: 65