Antiepileptic Medication By Ehsan Mohammad Hosseini Neurosurgery resident


Antiepileptic Medication By: Ehsan Mohammad Hosseini Neurosurgery resident at Isfahan University of Medical Science Advisor: Homayoun Tabesh M. D Assistant Professor of Neurosurgery

Classification of Epileptic Seizures Partial (focal) Seizures Simple Partial Seizures Complex Partial Seizures Generalized Tonic-Clonic Seizures Absence Seizures Tonic Seizures Atonic Seizures Clonic Seizures Myoclonic Seizures Infantile Spasms
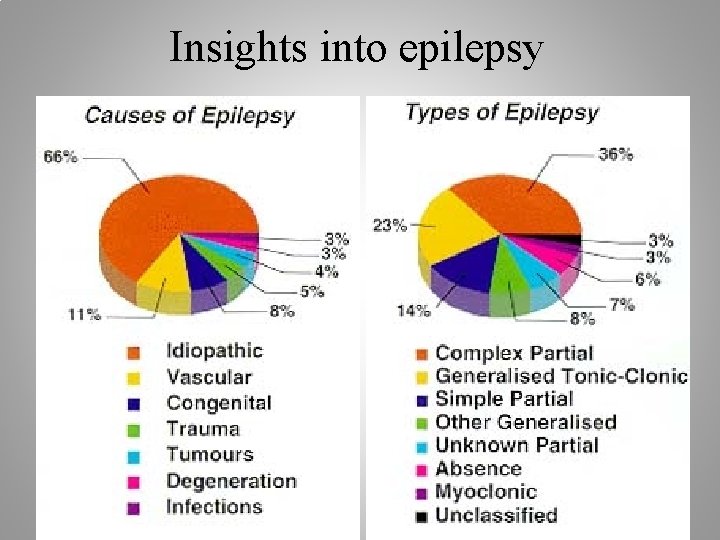
Insights into epilepsy

1857 - Bromides 1912 - Phenobarbital 1937 - Phenytoin 1954 - Primidone 1960 – Ethosuximide 1974 - Carbamazepine 1975 – Clonazepam (benzodiazapine) 1978 - Valproate 1993 - Felbamate, Gabapentin 1995 - Lamotrigine 1997 - Topiramate, Tiagabine 1999 - Levetiracetam 2000 - Oxcarbazepine, Zonisamide Vigabatrin—not approved in US

Classification of Anticonvulsants Classical Phenytoin Phenobarbital Primidone Carbamazepine Ethosuximide Valproic Acid Trimethadione Newer Lamotrigine Felbamate Topiramate Gabapentin Tiagabine Vigabatrin Oxycarbazepine Levetiracetam Fosphenytoin Others
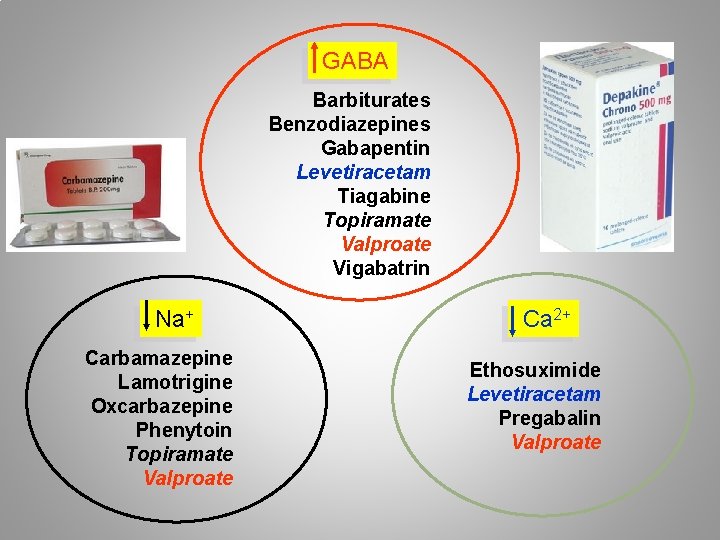
GABA Barbiturates Benzodiazepines Gabapentin Levetiracetam Tiagabine Topiramate Valproate Vigabatrin Na+ Carbamazepine Lamotrigine Oxcarbazepine Phenytoin Topiramate Valproate Ca 2+ Ethosuximide Levetiracetam Pregabalin Valproate

PHENYTOIN therapeutic plasma concentration range is 10– 20 mg/L. Phenytoin is a potent inducer of hepatic metabolizing enzymes affecting itself and other drugs (carbamazepine, warfarin, adrenal and gonadal steroids, thyroxine, tricyclic antidepressant, doxycycline, vitamin D, folate). Drugs that inhibit phenytoin metabolism include: valproic acid, cimetidine, co-trimoxazole, isoniazid, chloramphenicol, some NSAIDs, disulfiram.

Phenytoin is 90% bound to plasma albumin and small changes in binding will result in a higher concentration of free active drug. It is used to prevent all types of partial seizure, generalized seizure, and st. epilepticus. It is not used for absence attacks. ADR: impairment of cognitive function (which has led many physicians to prefer carbamazepine and valproate), sedation, hirsutism, skin rashes, gum hyperplasia (due to the inhibition of collagen metabolism), hyperglycemia, anaemia, osteomalacia

Fosphenytoin A Prodrug. Given i. v. or i. m. and rapidly converted to phenytoin in the body. Avoids local complications associated with phenytoin: vein irritation, tissue damage, pain and burning at site, muscle necrosis with i. m. injection, need for large fluid volumes. Otherwise similar toxicities to phenytoin.
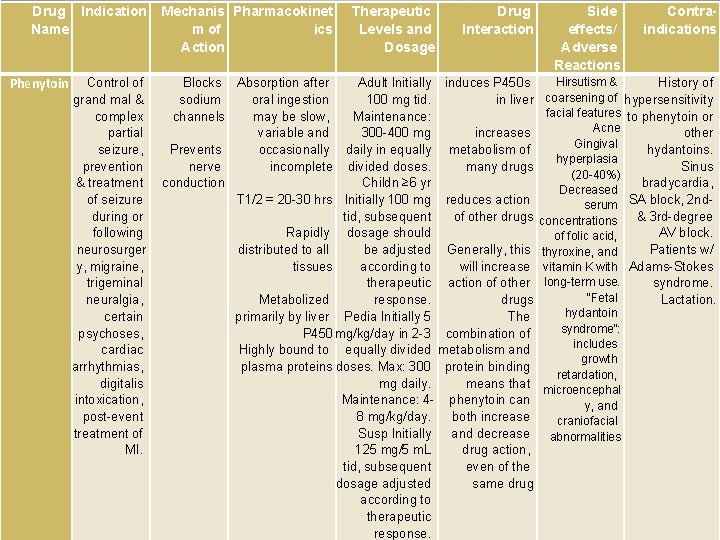
Drug Indication Name Phenytoin Control of grand mal & complex partial seizure, prevention & treatment of seizure during or following neurosurger y, migraine, trigeminal neuralgia, certain psychoses, cardiac arrhythmias, digitalis intoxication, post-event treatment of MI. Mechanis Pharmacokinet m of ics Action Therapeutic Levels and Dosage Drug Interaction Blocks Absorption after Adult Initially induces P 450 s sodium oral ingestion 100 mg tid. in liver channels may be slow, Maintenance: variable and 300 -400 mg increases Prevents occasionally daily in equally metabolism of nerve incomplete divided doses. many drugs conduction Childn ≥ 6 yr T 1/2 = 20 -30 hrs Initially 100 mg reduces action tid, subsequent of other drugs Rapidly dosage should distributed to all be adjusted Generally, this tissues according to will increase therapeutic action of other Metabolized response. drugs primarily by liver Pedia Initially 5 The P 450 mg/kg/day in 2 -3 combination of Highly bound to equally divided metabolism and plasma proteins doses. Max: 300 protein binding mg daily. means that Maintenance: 4 - phenytoin can 8 mg/kg/day. both increase Susp Initially and decrease 125 mg/5 m. L drug action, tid, subsequent even of the dosage adjusted same drug according to therapeutic response. Side effects/ Adverse Reactions Contraindications Hirsutism & History of coarsening of hypersensitivity facial features to phenytoin or Acne other Gingival hydantoins. hyperplasia Sinus (20 -40%) bradycardia, Decreased serum SA block, 2 nd- concentrations & 3 rd-degree AV block. of folic acid, Patients w/ thyroxine, and vitamin K with Adams-Stokes long-term use. syndrome. “Fetal Lactation. hydantoin syndrome”: includes growth retardation, microencephal y, and craniofacial abnormalities

CARBAMAZEPINE Due to outoinduction the t 1/2 of the drug falls from 35 to 20 hr over the first few weeks of therapy, therefor incresae dose from 100 -200 mg/day to 600 -800 mg/day in 3 -4 week. Carbamazepine effective for focal and secondary generalized Epilepsy but aggravates myoclonic and absence seizure. It is useful for the treatment of trigeminal neuralgia, postherpetic pains, etc. Adverse reactions (ARs): reversible blurring of vision, diplopia dizziness, ataxia, depression of AV conduction, skin rashes, liver, and kidney dysfunction.
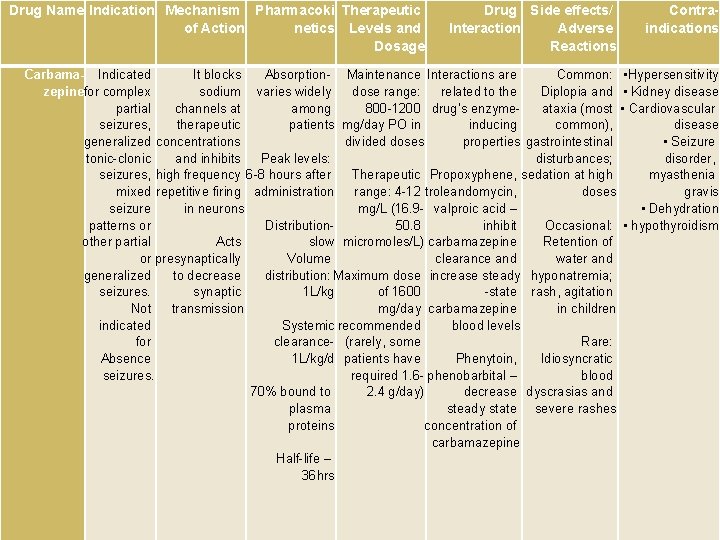
Drug Name Indication Mechanism Pharmacoki Therapeutic of Action netics Levels and Dosage Drug Side effects/ Interaction Adverse Reactions Contraindications Carbama- Indicated It blocks Absorption- Maintenance Interactions are Common: • Hypersensitivity zepinefor complex sodium varies widely dose range: related to the Diplopia and • Kidney disease partial channels at among 800 -1200 drug’s enzymeataxia (most • Cardiovascular seizures, therapeutic patients mg/day PO in inducing common), disease generalized concentrations divided doses properties gastrointestinal • Seizure tonic-clonic and inhibits Peak levels: disturbances; disorder, seizures, high frequency 6 -8 hours after Therapeutic Propoxyphene, sedation at high myasthenia mixed repetitive firing administration range: 4 -12 troleandomycin, doses gravis seizure in neurons mg/L (16. 9 - valproic acid – • Dehydration patterns or Distribution 50. 8 inhibit Occasional: • hypothyroidism other partial Acts slow micromoles/L) carbamazepine Retention of or presynaptically Volume clearance and water and generalized to decrease distribution: Maximum dose increase steady hyponatremia; seizures. synaptic 1 L/kg of 1600 -state rash, agitation Not transmission mg/day carbamazepine in children indicated Systemic recommended blood levels for clearance- (rarely, some Rare: Absence 1 L/kg/d patients have Phenytoin, Idiosyncratic seizures. required 1. 6 - phenobarbital – blood 70% bound to 2. 4 g/day) decrease dyscrasias and plasma steady state severe rashes proteins concentration of carbamazepine Half-life – 36 hrs

VALPROIC ACID (Sodium valproate) Valproic acid has t 1/2 13 h and 90% bound to plasma albumin. It is a nonspecific inhibitor of metabolism, and inhibits own metabolism. Phenobarbital, phenytoin and carbamazepine reduce serum Level of valporate. Valproic acid is effective for treatment of generalized and partial epilepsy, febrile convulsion and post-traumatic epilepsy. ADR can be troublesome: weight gain, teratogenicity, polycystic ovary syndrome, and loss of hair, nausea can be a problem, rarely, liver failure (risk maximal at 2– 12 weeks).
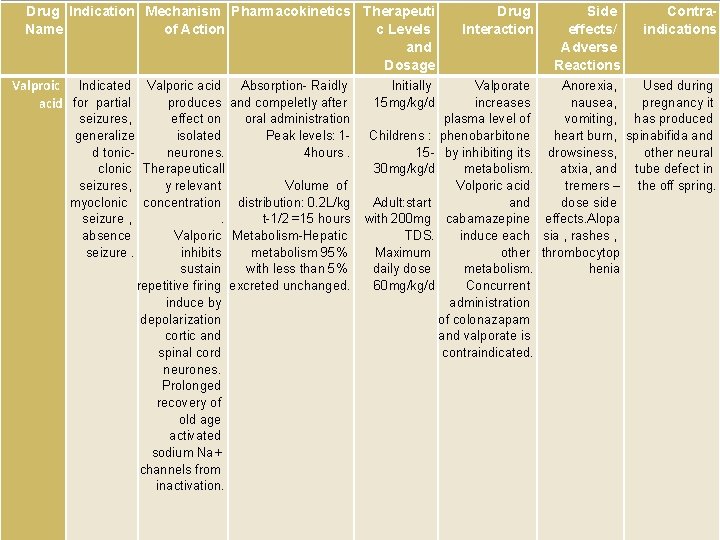
Drug Indication Mechanism Pharmacokinetics Therapeuti Name of Action c Levels and Dosage Initially Valproic Indicated Valporic acid Absorption- Raidly produces and compeletly after 15 mg/kg/d acid for partial seizures, effect on oral administration generalize isolated Peak levels: 1 d tonicneurones. 4 hours. clonic Therapeuticall seizures, y relevant Volume of myoclonic concentration distribution: 0. 2 L/kg seizure , . t-1/2 =15 hours absence Valporic Metabolism-Hepatic seizure. inhibits metabolism 95% sustain with less than 5% repetitive firing excreted unchanged. induce by depolarization cortic and spinal cord neurones. Prolonged recovery of old age activated sodium Na+ channels from inactivation. Drug Interaction Side effects/ Adverse Reactions Contraindications Valporate Anorexia, Used during increases nausea, pregnancy it plasma level of vomiting, has produced Childrens : phenobarbitone heart burn, spinabifida and 15 - by inhibiting its drowsiness, other neural 30 mg/kg/d metabolism. atxia, and tube defect in Volporic acid tremers – the off spring. Adult: start and dose side with 200 mg cabamazepine effects. Alopa TDS. induce each sia , rashes , Maximum other thrombocytop daily dose metabolism. henia 60 mg/kg/d Concurrent administration of colonazapam and valporate is contraindicated.

BARBITURATES Anti epilepsy members include phenobarbital (phenobarbitone( t 1/2 100 h), methyl phenobarbital and Primidone. They are still used for generalized seizures; sedation is usual. Dosage: 2 -5 mg/kg/day Partial and secondary generalized seizure and status epilepticus and prophylaxis of febrile seizure.

Phenobarbital Toxic effects: sedation (early; tolerance develops); nystagmus & ataxia at higher dose; osteomalacia, folate deficiency and vit. K deficiency. In children: paradoxical irritability, hyperactivity and behavioral changes. Deoxybarbiturates: primidone: active but also converted to phenobarbital. Some serious additional ADR’s: leukopenia, SLE-like.

PRIMIDONE (Mysolin) Metabolized to phenobarbital and phenylethylmalonamide (PEMA), both active metabolites. Effective against partial and generalized tonic-clonic seizures. Absorbed completely, low binding to plasma proteins. . Toxicity: . as phenobarbital Same Sedation occurs early. Gastrointestinal complaints.

FELBAMATE Effective against partial seizures but has severe side effects. Because of its severe side effects, it has been relegated to a third-line drug used only for refractory cases and Lennox-Gastaut syn. Dosage 15 mg/kg/day or 1500 mg/day and double and triple in sec and third week. Toxicity: Aplastic anemia Severe hepatitis

Gabapentin Absorption well, excretion unchanged in urine ADR Sedation, dizziness, weight gain INDICATION Simple partial seizure Refractory partial seizures Rolandic Epilepsy Complex partial seizure Dose: 30 mg/kg/day, increase up to 60 -100 mg/kg/day

LAMOTRIGINE (Lamictal) Presently use as add-on therapy with valproic acid (v. a. conc. are be reduced). Almost completely absorbed T 1/2 = 24 hrs Low plasma protein binding Also effective in myoclonic and generalized seizures in childhood and absence attacks and Lennox-Gastaut syn Toxicity: Diplopia Dizziness Headache Nausea Somnolence S. J syn Rash

Phamacokinetics Absorbed well and metabolized in liver Drug Interaction Phenytoin, carbamazepine and phenobarbitorate decrease t½ Valproate increase plasma level ADR Sleepiness, dizziness, ataxia, diplopia and vomiting , Rash – severe reaction
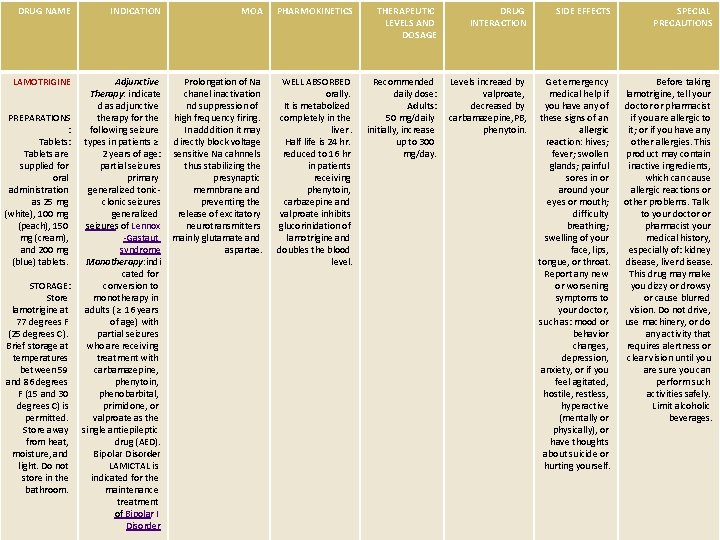
DRUG NAME INDICATION MOA PHARMOKINETICS THERAPEUTIC LEVELS AND DOSAGE DRUG INTERACTION SIDE EFFECTS SPECIAL PRECAUTIONS LAMOTRIGINE Adjunctive Therapy: indicate d as adjunctive therapy for the following seizure types in patients ≥ 2 years of age: partial seizures primary generalized tonicclonic seizures generalized seizures of Lennox -Gastaut syndrome Monotherapy: indi cated for conversion to monotherapy in adults ( ≥ 16 years of age) with partial seizures who are receiving treatment with carbamazepine, phenytoin, phenobarbital, primidone, or valproate as the single antiepileptic drug (AED). Bipolar Disorder LAMICTAL is indicated for the maintenance treatment of Bipolar I Disorder Prolongation of Na chanel inactivation nd suppression of high frequency firing. In adddition it may directly block voltage sensitive Na cahnnels thus stabilizing the presynaptic memnbrane and preventing the release of excitatory neurotransmitters mainly glutamate and aspartae. WELL ABSORBED orally. It is metabolized completely in the liver. Half life is 24 hr. reduced to 16 hr in patients receiving phenytoin, carbazepine and valproate inhibits glucorinidation of lamotrigine and doubles the blood level. Recommended daily dose: Adults: 50 mg/daily initially, increase up to 300 mg/day. Levels increaed by valproate, decreased by carbamazepine, PB, phenytoin. Get emergency medical help if you have any of these signs of an allergic reaction: hives; fever; swollen glands; painful sores in or around your eyes or mouth; difficulty breathing; swelling of your face, lips, tongue, or throat. Report any new or worsening symptoms to your doctor, such as: mood or behavior changes, depression, anxiety, or if you feel agitated, hostile, restless, hyperactive (mentally or physically), or have thoughts about suicide or hurting yourself. Before taking lamotrigine, tell your doctor or pharmacist if you are allergic to it; or if you have any other allergies. This product may contain inactive ingredients, which can cause allergic reactions or other problems. Talk to your doctor or pharmacist your medical history, especially of: kidney disease, liver disease. This drug may make you dizzy or drowsy or cause blurred vision. Do not drive, use machinery, or do any activity that requires alertness or clear vision until you are sure you can perform such activities safely. Limit alcoholic beverages. PREPARATIONS : Tablets are supplied for oral administration as 25 mg (white), 100 mg (peach), 150 mg (cream), and 200 mg (blue) tablets. STORAGE: Store lamotrigine at 77 degrees F (25 degrees C). Brief storage at temperatures between 59 and 86 degrees F (15 and 30 degrees C) is permitted. Store away from heat, moisture, and light. Do not store in the bathroom.

TOPIRAMATE Broad spectrom and rapidly absorbed, bioav. is > 80%, has no active metabolites, excreted in urine. T 1/2 = 20 -30 hrs Toxicity: Fatigue Dizziness Cognitive slowing Paresthesias Nervousness Confusion Met. Acidosis Urolithiasis

Topiramate Dosage: 25 -50 mg/day or 0. 5 -1 mg/kg/day increase weekly to 200 -400 mg/day Or 5 -6 mg/kg/day Indications SPS, CPS , GTCS and Lennox-Gastaut

TIAGABINE MECHANISM OF ACTION inhibit GABA transporter GAT-1 reduces GABA uptake in neurons Pharmacokinetics: Rapid oral absorption Extremely plasma protein bound Liver metabolism by CYP 3 A indication Add on therapy of partial seizures ADR Sedation, abdominal pain

OXCARBAMAZEPINE 10 mono hydroxycarbamazepine. half life 10 -15 hr Advantage –weak enzyme induction Phenytoin, Carbamazepin and phenobarbital Ø 30 -40% reduce level of oxcarbamazepine Dosage: 300 mg BID or 5 -10 mg/kg/day and increase weekly to 1200 -1800 mg or 20 -30 mg/kg/day with range of 10 -35 mg/L USES: In partial seizures and sec generalized ADR same to carbamazepine but hyponatremia is more ferequent

Levetiracetam is a highly effective, broad-spectrum, newer class of AED with a unique mechanism of action. Half life 6 -8 hr It can be used to treat all focal or generalized, idiopathic or symptomatic epileptic syndromes in all age groups.

Indications Treatment of partial seizures with or without secondary generalization in adults, children and infants from 1 month of age with epilepsy. In the treatment of myoclonic seizures in adults and adolescents. In the treatment of primary generalized tonic-clonic seizures in adults and adolescents. Also approved for Rolandic and posthypoxic and postencephalitic myoclonus.

Dosage and titration (Adults) Start treatment with 500 -1000 mg/day (twice-daily dosing), which may be sufficient for seizure control. If needed, Levetiracetam can be titrated in steps of 500 mg/week to a maximum of 3000 mg/day.

Dosage and titration (children) Start with 10 -20 mg/kg/day, which may be sufficient for seizure control. If needed, Levetiracetam can be titrated in steps of 5– 10 mg/kg/week to a usual maintenance dose of 20– 40 mg/kg/day given in two divided doses. Serum level 10 -40 mg/L to max 40 -80 mg/L
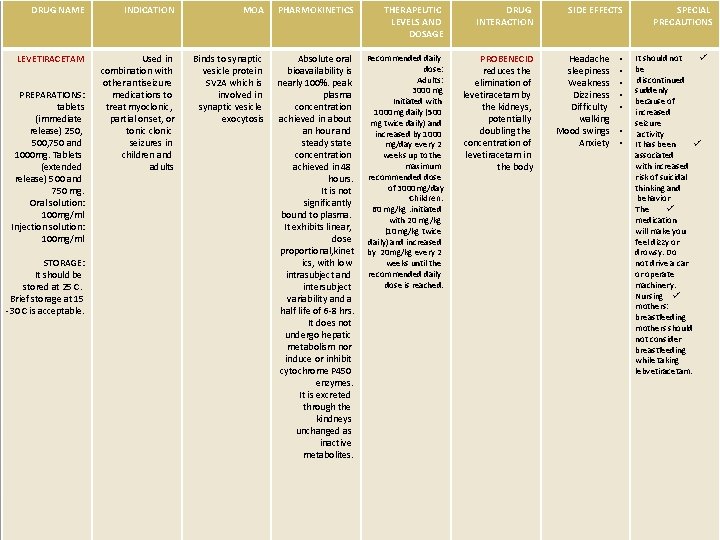
DRUG NAME INDICATION MOA PHARMOKINETICS THERAPEUTIC LEVELS AND DOSAGE DRUG INTERACTION LEVETIRACETAM Used in combination with other antiseizure medications to treat myoclonic, partial onset, or tonic clonic seizures in children and adults Binds to synaptic vesicle protein SV 2 A which is involved in synaptic vesicle exocytosis Absolute oral bioavailability is nearly 100%. peak plasma concentration achieved in about an hour and steady state concentration achieved in 48 hours. It is not significantly bound to plasma. It exhibits linear, dose proportional, kinet ics, with low intrasubject and intersubject variability and a half life of 6 -8 hrs. It does not undergo hepatic metabolism nor induce or inhibit cytochrome P 450 enzymes. It is excreted through the kindneys unchanged as inactive metabolites. Recommended daily dose: Adults: 3000 mg Initiated with 1000 mg daily (500 mg twice daily) and increased by 1000 mg/day every 2 weeks up to the maximum recommended dose of 3000 mg/day Children: 60 mg/kg. initiated with 20 mg/kg (10 mg/kg twice daily) and increased by 20 mg/kg every 2 weeks until the recommended daily dose is reached. PROBENECID reduces the elimination of levetiracetam by the kidneys, potentially doubling the concentration of levetiracetam in the body PREPARATIONS: tablets (immediate release) 250, 500, 750 and 1000 mg. Tablets (extended release) 500 and 750 mg. Oral solution: 100 mg/ml Injection solution: 100 mg/ml STORAGE: It should be stored at 25 C. Brief storage at 15 -30 C is acceptable. SIDE EFFECTS Headache sleepiness Weakness Dizziness Difficulty walking Mood swings Anxiety • • SPECIAL PRECAUTIONS It should not ü be discontinued suddenly because of increased seizure activity It has been ü associated with increased risk of suicidal thinking and behavior The ü medication will make you feel dizzy or drowsy. Do not drive a car or operate machinery. Nursing ü mothers: breastfeeding mothers should not consider breastfeeding while taking lebvetiracetam.

ZONISAMIDE Broad spectrom good bioavailability, low pb. T 1/2 = 1 - 3 days Effective against partial, generalized tonic-clonic seizures, Lennox-Gastaut, Absence, infantile spasm and Myoclonic ataxic epilepsy. Toxicity: Drowsiness Cognitive impairment High incidence of renal stones Hypohydrosis and hyperthermia

zonisamide Mechanism of action Inhibit t type Ca channels Prolong inactivation of Na channel pharmacokinetics Complete oral absorption 40% binding to plasma protein 85% urine excretion

Zonisamide Intraction: Phenytoin, phenobarbitone, carbamazepinedecreases its level Dose 100 mg/day or 1 -2 mg/kg/day to target dose of 100 -600 mg/day or 8 -12 mg/kg/day

Choosing Antiepileptic Drugs Seizure type Epilepsy syndrome Pharmacokinetic profile Interactions/other medical conditions Efficacy Expected adverse effects Cost

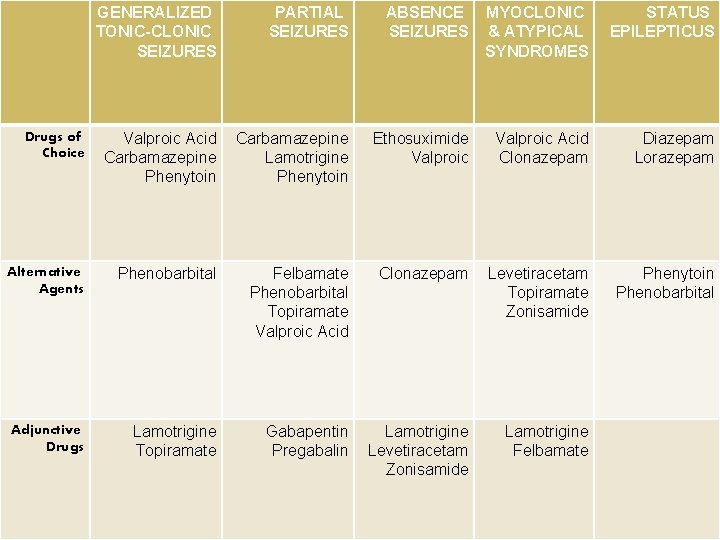
GENERALIZED TONIC-CLONIC SEIZURES PARTIAL SEIZURES ABSENCE SEIZURES MYOCLONIC & ATYPICAL SYNDROMES STATUS EPILEPTICUS Drugs of Choice Valproic Acid Carbamazepine Phenytoin Carbamazepine Lamotrigine Phenytoin Ethosuximide Valproic Acid Clonazepam Diazepam Lorazepam Alternative Agents Phenobarbital Felbamate Phenobarbital Topiramate Valproic Acid Clonazepam Levetiracetam Topiramate Zonisamide Phenytoin Phenobarbital Adjunctive Drugs Lamotrigine Topiramate Gabapentin Pregabalin Lamotrigine Levetiracetam Zonisamide Lamotrigine Felbamate
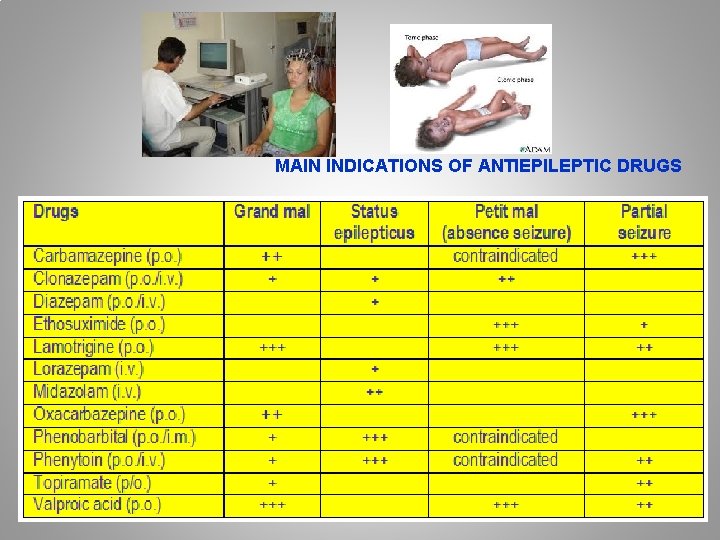
MAIN INDICATIONS OF ANTIEPILEPTIC DRUGS

Side effect issues Sedation - especially with barbiturates Cosmetic - phenytoin Weight gain – valproic acid, gabapentin Weight loss - topiramate Reproductive function – valproic acid Cognitive - topiramate Behavioral – felbamate, leviteracetam Allergic - many

Teratogenicity Valproic acid –neural tube defects Carbamazepine –craniofacial anomalies, spina bifida Phenytoin –fetal hydantoin syndrome

Life-Threatening Toxicity Valproic acid –fatal hepatoxicity Lamotrigine –Stevens-Johnson syndrome Zonisamide –severe skin reactions Felbamate –aplastic anemia, acute hepatic failure

Other Clinical Uses Valproic acid –mania Carbamazepine, Lamotrigine –bipolar disorder Carbamazepine –trigeminal neuralgia Gabapentin –pain of neuropathic origin Topiramate –migraine Pregabalin –neuropathic pain

convulsive drugs of choice rand mal: I choice – valproate or Lamotrigine ative – Carbamazepine, Topiramate or Phenytoin t mal: I choice – Ehosuximide or valproate Alternative – Clonazepam or Lamotrigine Partial seizures: I choice – Carbamazepine or valproate tive – Phenytoin, Lamotrigine, Vigabatrin, Topiramate epilepticus: I choice – Diazepam or Lorazepam (i. v. ) Alternative – Phenobarbital (i. m. /i/v. )

ment of status epilepticus in adults Patient in opisthotonus (grand mal)
Thank you
- Slides: 47