Anticoagulation in Intracerebral Hemorrhage ICH Survivors for Stroke

Anticoagulation in Intracerebral Hemorrhage (ICH) Survivors for Stroke Prevent. Ion and REcovery Webinar May 27, 2020 NINDS U 01 NS 106513 NCT 03907046 27 -May-2020 ASPIRE Webinar

Congratulations to Mayo Clinic Jacksonville for Enrolling Subject #1! Site Principal Investigator: Jason Siegel, MD Sub-Investigator: Ashley Rogers, Chidinma Onweni, Josephine Huang, Lauren Ngtucker, Miichelle Lin, Rocco Cannistraro Study Coordinators: Jaya Vijayan, Meredith Mc. Donald 27 -May-2020 ASPIRE Webinar 1

Outline • • • 27 -May-2020 ASPIRE Site Network COVID-19 Impact Assessment Resumption of Enrollment ASPIRE Remote Procedures for Study Visits Site Progress Discussion ASPIRE Webinar 2

ASPIRE Site Network Status Released to enroll Activation pending Readiness call pending CTA pending c. IRB pending CTA and c. IRB pending Total 27 -May-2020 ASPIRE Webinar No. 52 17 10 5 18 15 117 3

Covid-19 Impact Assessment v 2 Sites Released to Enroll ACTIVE Screening In-person and remote allowed Only remote allowed Neither in-person or remote allowed Enrollment In-person and remote allowed Only in-person allowed Neither in-person or remote allowed Survey not completed 27 -May-2020 ASPIRE Webinar (n=10) SUSPENDED (n=42) 7 3 20 11 4 Submit screen failure forms 7 2 1 10 9 1 15 Change status to active? 7 4

Resumption of Enrollment • Physical safety and overall health of participants and providers/research teams is always the top priority • Continuing to find effective ways to enroll participants is a public health priority • These missions are not mutually exclusive! 27 -May-2020 ASPIRE Webinar 5

Resumption of Enrollment • On May 12 th, c. IRB approved plan to release sites to enroll if they are able to undertake enrollment while minimizing potential risks to study staff and participants and preserving local resources needed for treatment of COVID-19 patients. Before being released to enroll, Site PI must attest that 4 conditions are met: 1. Local institution approves enrollment activities. 2. Local conditions permit enrolling patients safely, in conformity with protocol, and without contributing to shortage of staff or PPE needed for Covid-19 patient care. 3. Study team will conduct study-specific tasks via remote means as much as possible. 4. When in-person contact is needed, study team will follow best practices for infection control in keeping with local rules, including screening patients for COVID-19 symptoms and exposure prior to contact, and using masks, physical barriers, and social distancing to greatest degree possible. 27 -May-2020 ASPIRE Webinar 6

ASPIRE Procedures for Remote Study Visits • Remote study visits – for consent, randomization, and follow-up - are permitted by the ASPIRE protocol. • On 5/11/20, c. IRB reviewed and approved specific procedures to be followed for remote visits in ASPIRE. - ASPIRE Procedures for Remote Study Visits posted in Web. DCU>Toolkit • Remote Informed Consent Implementation Form to be submitted to c. IRB for each site. - Documents if site plans to use remote consent and, if so, procedures to be used. - ASPIRE procedures, or local procedures (compatible with ASPIRE procedures). - c. IRB has regulatory responsibility to ensure adequate IC process. - Sites with c. IRB approval should send completed form to Laura Benken who will handle submission. 27 -May-2020 ASPIRE Webinar 7

Remote Informed Consent Procedures Transmit Consent Materials Method of transmission used will depend on the options approved at the local site and the subject’s circumstances. Consent form (+ stand-alone HIPPA, bill of rights, if applicable) must be sent to subject using a HIPAA and 21 CFR part 11 -compliant method. These include: Method Required by Subject Mail Ability to receive mail Fax Access to fax machine Email* Able to print form to sign or can enter e-signature *Compliant method if subject informed of and agrees to small risk of interception. 27 -May-2020 ASPIRE Webinar 8

E-Consent • E-consent refers to use of a formal, approved electronic application (in-person or remotely) to convey study information and document consent. • Examples of e-Consent - REDCap survey - Other secure software (e. g. , EPIC e. Consent) • Application must be HIPAA and 21 CFR part 11 -compliant to use for ASPIRE. • NCC creating central e-Consent via UCinn’s 21 CFR part 11 -compliant REDCap. - For interested sites, local ICF will be available in REDCap. - NIH Stroke. Net c. IRB e-Consent Webinar on June 1 st 1 -2: 30 pm EDT. • E-consent is not needed to consent patients remotely now! • You can use conventional delivery means, e. g. , mail, fax, and email. 27 -May-2020 ASPIRE Webinar 9

Remote Informed Consent, continued Contact Subject for Consent Discussion Contact subject by telephone or HIPAA-compliant telehealth application and review consent materials as you would in-person. If LAR is providing consent: Confirm LAR resides with patient and will take responsibility for administration of study drug or can identify a person residing with (or responsible for) patient who will take responsibility. - If possible, speak with patient to probe for questions or resistance to participating. If consent obtained, instruct subject how to sign/date and transmit consent back using a locally approved and HIPAA/21 CFR part 11 -compliant method. - Scan or photo of signed printed form may be returned by email (wet-signature consent not required for subject file). - E-signature via Adobe Sign or Docu. Sign may be used if permitted locally. After entire signed and dated consent form received, person obtaining consent should sign and date and transmit copy to subject using approved/compliant method. 27 -May-2020 ASPIRE Webinar 10

Remote Baseline Visit Procedures • Baseline forms may be completed by telephone or HIPAA-compliant telehealth applications if patient can’t return to clinic or be visited. • Modifications are required for the following assessments when completed remotely: - Mo. CA: Follow instructions on form for telephone assessment. - PROMIS Scales: Note telephone administration in Comments section. - Berlin Questionnaire: Record self or surrogate-reported height and weight at baseline contact, if known; otherwise, record latest height in medical record, and latest weight in medical record if obtained within 14 days of baseline contact. Enter sources for height and weight in Comments section. - Vital Signs: Record blood pressure obtained by home measurement device at baseline contact, when available (following guidelines for home BP in ASPIRE Procedures for Remote Visits); otherwise, record latest measures in medical record. Describe source of BP measures, including type of home device if used, in Comments section. 27 -May-2020 ASPIRE Webinar 11

Remote Randomization Procedures If subject able and willing to proceed with participation, • Complete Randomization Form • Obtain Study Drug Kit - Study Drug Kit Assignment (Form 512): Record weight by self or surrogate-report on date of baseline contact, if known; otherwise, use latest recorded weight in medical record. • Ship Study Drug Kit and Participant Materials - Confirm mailing address for shipment to subject. - Mail kit using overnight shipping service following local procedures for shipping drugs; include Participant Information Sheet, Prohibited/Discouraged Medications list, and ASPIRE Alert Card (completed) with kit. - Advise subject (or person responsible if LAR consent) when to expect shipment and instruct them to hold it unopened until contacted. 27 -May-2020 ASPIRE Webinar 12

Starting Study Drug • Contact subject/responsible person when delivery confirmation received (or expected). - Review Participant Information Sheet and Prohibited/Discouraged Meds step-by-step. • Study Drug Dispensing (Form 513): Record date study drug confirmed started. - Must be within 48 hours of randomization. 27 -May-2020 ASPIRE Webinar 13

Remote Follow-Up Visit Procedures • If a subject can’t return to clinic or be visited in-person, complete contact remotely using telephone or HIPAA-compliant telehealth applications and follow procedures for study drug shipping, dispensing, and return below. • Ship Study Drug Kit - Mail kit via overnight shipping service following local procedures for shipping drugs; include Participant Information Sheet & Prohibited/Discouraged Medications list with kit. - Include a return, stamped, self-addressed mailer. - Advise subject/resp person when to expect shipment and to hold it unopened until contacted. • Start New Kit - Contact subject/resp person when delivery confirmation received/expected. - Record date new supply started on Study Drug Dispensing (Form 513). • Study Drug Return - Instruct subject/resp person to return old bottles (with unused tablets) in supplied mailer. 27 -May-2020 ASPIRE Webinar 14

Questions? 27 -May-2020 ASPIRE Webinar 15

Released to Enroll Active 27 -May-2020 Capital Health Regional Med Center Mayo Clinic, Jacksonville Mc. Laren Macomb Med. Star Washington Hospital Center NYP Weill Cornell Med Center Oregon Health & Science U Hospital Peace. Health Sacred Heart Med Ctr - River. Bend St. David's Med Center U of Utah Healthcare WVU Healthcare Ruby Memorial Hospital ASPIRE Webinar 16

Enrollment Suspended 27 -May-2020 Abbott Northwestern Hospital Barnes Jewish Hospital Beth Israel Deaconess Med Ctr Cedars-Sinai Med Ctr Central Du. Page Hospital Chandler Regional Med Ctr Eastern Maine Med Ctr Greenville Hospital System Harborview Med Ctr Hartford Hospital Henry Ford Hospital of U of Pennsylvania Jackson Memorial Hospital Loyola U Med Ctr Maimonides Med Ctr Medical U of South Carolina U Hospital Mercy Health Saint Mary's Metro Health Hospital Montefiore Med Ctr Moses H. Cone Memorial Hospital NY-Pres Brooklyn Methodist Hospital OSF St. Francis Med Ctr OSU Wexner Med Ctr Ochsner Med Ctr - Main Campus ASPIRE Webinar 17

Enrollment Suspended, continued 27 -May-2020 Rhode Island Hospital San Francisco General Hospital St. Mary's Med Ctr Staten Island U Hospital - North Campus Strong Memorial Hospital Tampa General Hospital UAMS Med Ctr UC Irvine Med Ctr UT Southwestern Med Ctr ASPIRE Webinar UVA Med Ctr U of Alabama Hospital U of Cincinnati Med Ctr U of Iowa Hospitals & Clinics U of Michigan U Hospital U of New Mexico Hospital U of Texas HS Ctr San Antonio Wake Forest Baptist Med Ctr Yale New Haven Hospital 18
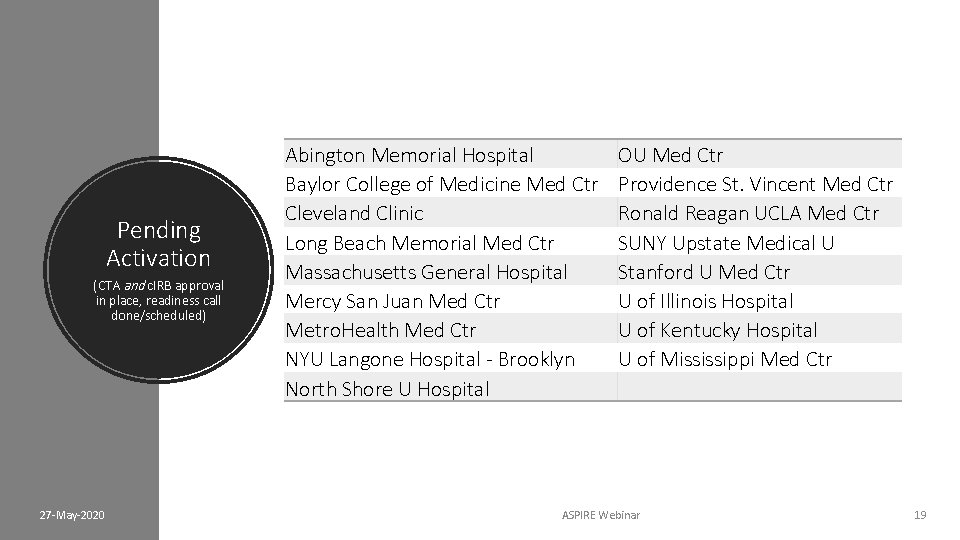
Pending Activation (CTA and c. IRB approval in place, readiness call done/scheduled) 27 -May-2020 Abington Memorial Hospital Baylor College of Medicine Med Ctr Cleveland Clinic Long Beach Memorial Med Ctr Massachusetts General Hospital Mercy San Juan Med Ctr Metro. Health Med Ctr NYU Langone Hospital - Brooklyn North Shore U Hospital OU Med Ctr Providence St. Vincent Med Ctr Ronald Reagan UCLA Med Ctr SUNY Upstate Medical U Stanford U Med Ctr U of Illinois Hospital U of Kentucky Hospital U of Mississippi Med Ctr ASPIRE Webinar 19

Pending Readiness Call (CTA and c. IRB approval in place) 27 -May-2020 Boston Med Ctr Eden Med Ctr Kaiser Permanente Fontana Med Ctr Kaiser Permanente Los Angeles Med Ctr NYP Columbia U Med Ctr Regions Hospital UH Cleveland Med Ctr UPMC Presbyterian Hospital United Hospital U of Minnesota Med Ctr Hospital ASPIRE Webinar 20
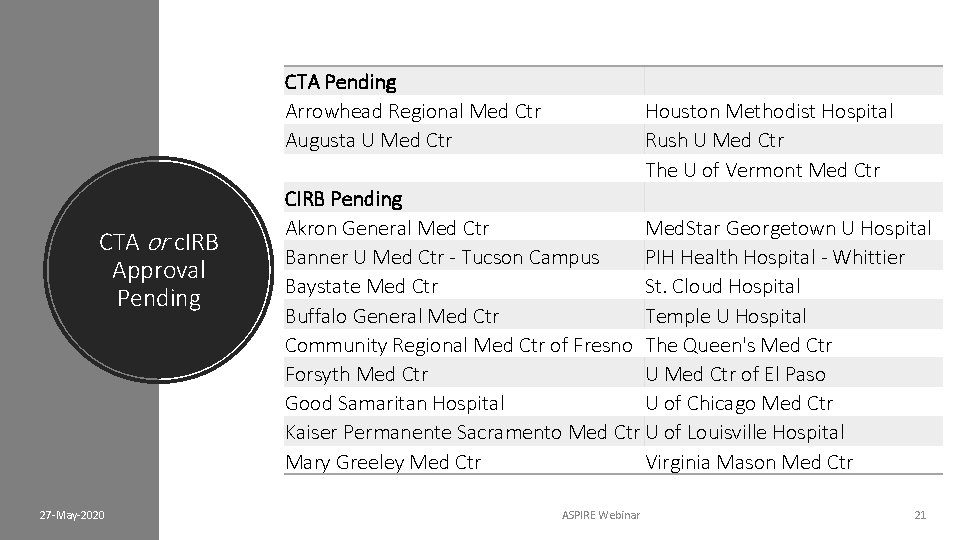
CTA Pending Arrowhead Regional Med Ctr Augusta U Med Ctr CTA or c. IRB Approval Pending 27 -May-2020 Houston Methodist Hospital Rush U Med Ctr The U of Vermont Med Ctr CIRB Pending Akron General Med Ctr Med. Star Georgetown U Hospital Banner U Med Ctr - Tucson Campus PIH Health Hospital - Whittier Baystate Med Ctr St. Cloud Hospital Buffalo General Med Ctr Temple U Hospital Community Regional Med Ctr of Fresno The Queen's Med Ctr Forsyth Med Ctr U Med Ctr of El Paso Good Samaritan Hospital U of Chicago Med Ctr Kaiser Permanente Sacramento Med Ctr U of Louisville Hospital Mary Greeley Med Ctr Virginia Mason Med Ctr ASPIRE Webinar 21

CTA and c. IRB Approval Pending 27 -May-2020 Emory U Hospital Grady Memorial Hospital Hoag Memorial Hospital Presbyterian Javon Bea Hospital - Riverside Kings County Hospital Center Lahey Hospital & Med Ctr Mercy Med Ctr - Des Moines NCH Hospital Park Nicollet Methodist Hospital Robert Wood Johnson U Hospital Sanford Med Ctr Fargo St. Josephs Regional Med Ctr U of Nebraska Med Ctr U of North Carolina Med Ctr U of South Alabama U Hospital ASPIRE Webinar 22

Discussion 27 -May-2020 ASPIRE Webinar 23
- Slides: 24