AntiChlamydia Vaccine From Bench to Bedside Ashlesh Murthy

Anti-Chlamydia Vaccine: From Bench to Bedside Ashlesh Murthy, M. B. B. S. , Ph. D Research Assistant Professor University of Texas at San Antonio 1

2

Chlamydia Obligate intracellular pathogen C. trachomatis, C. pneumoniae, C. psittaci, C. pecorum, C. muridarum C. trachomatis multiple serovars: A-K, L 1 -3 3
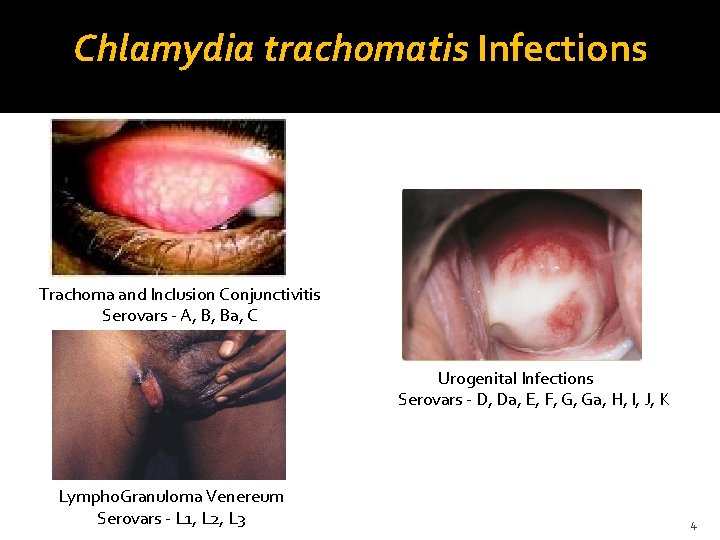
Chlamydia trachomatis Infections Trachoma and Inclusion Conjunctivitis Serovars - A, B, Ba, C Urogenital Infections Serovars - D, Da, E, F, G, Ga, H, I, J, K Lympho. Granuloma Venereum Serovars - L 1, L 2, L 3 4
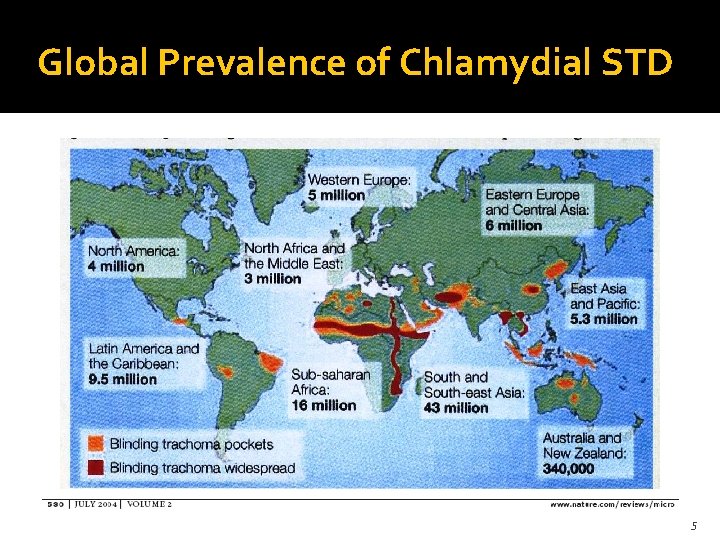
Global Prevalence of Chlamydial STD 5
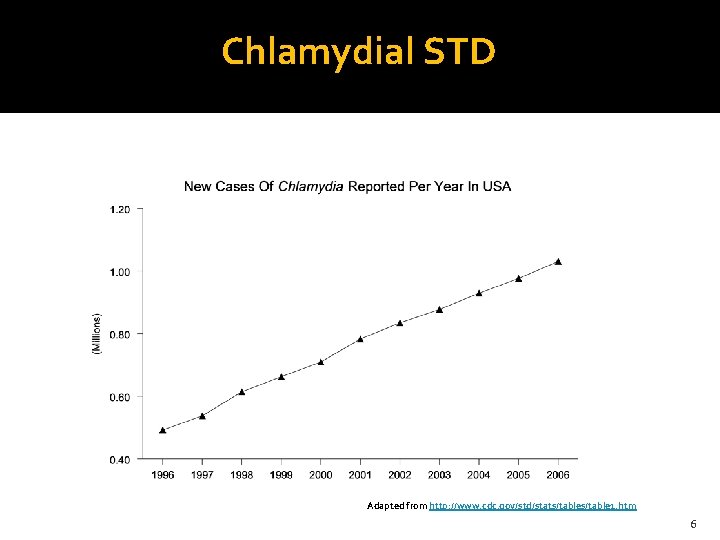
Chlamydial STD Adapted from http: //www. cdc. gov/std/stats/table 1. htm 6
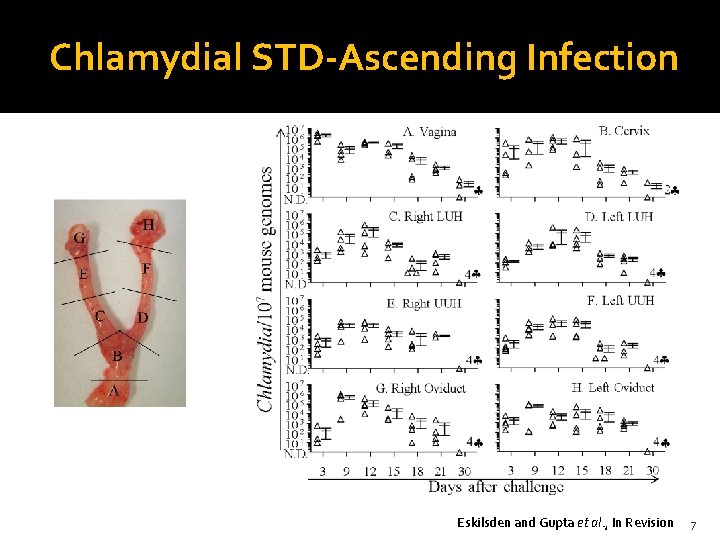
Chlamydial STD-Ascending Infection Eskilsden and Gupta et al. , In Revision 7
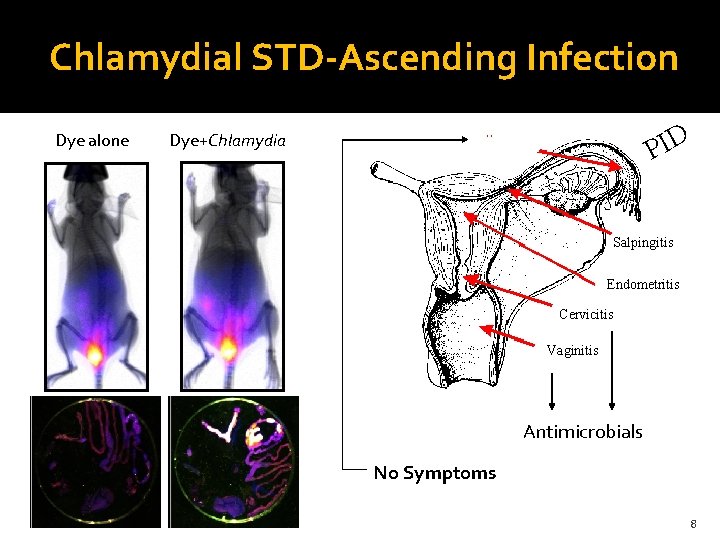
Chlamydial STD-Ascending Infection Dye alone PID Dye+Chlamydia Salpingitis Endometritis Cervicitis Vaginitis Antimicrobials No Symptoms 8
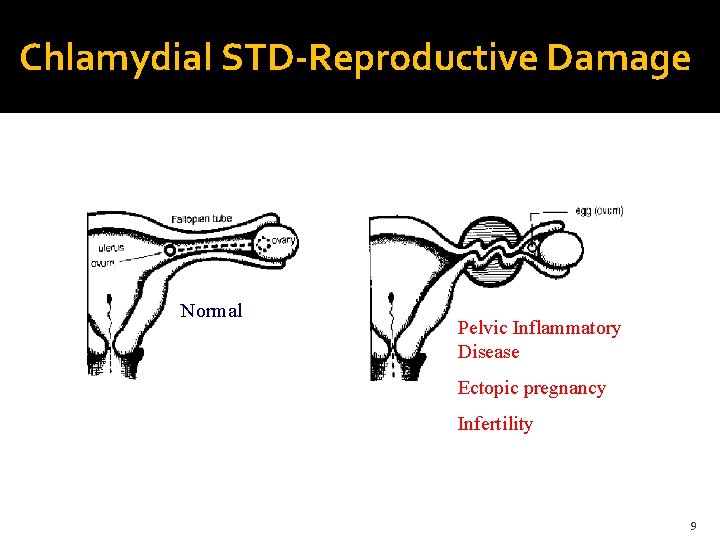
Chlamydial STD-Reproductive Damage Normal Pelvic Inflammatory Disease Ectopic pregnancy Infertility 9

Chlamydial STD- Statistics 90 million cases 50 -70% of patients initially asymptomatic: not treated 40% of untreated females – pelvic inflammatory disease 20% of PID cases- infertility Adapted from www. cdc. gov- 2006 STD statistics 10

Anti-Chlamydial Vaccine 11
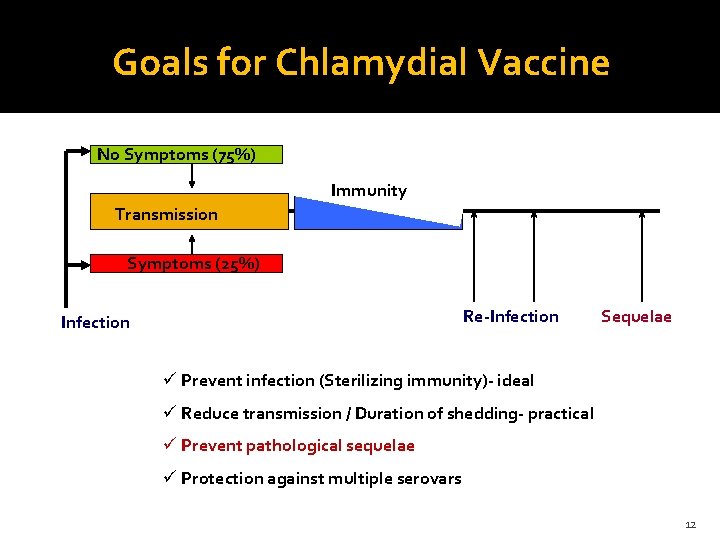
Goals for Chlamydial Vaccine No Symptoms (75%) Immunity Transmission Symptoms (25%) Re-Infection Sequelae ü Prevent infection (Sterilizing immunity)- ideal ü Reduce transmission / Duration of shedding- practical ü Prevent pathological sequelae ü Protection against multiple serovars 12

Chlamydial Vaccine Trials of 1960 s � Formalin-killed whole chlamydial organisms � Reduction in incidence for ~ 1 year � Comparable/exaggerated ocular pathology in vaccinees � Focus on sub-unit vaccines 13
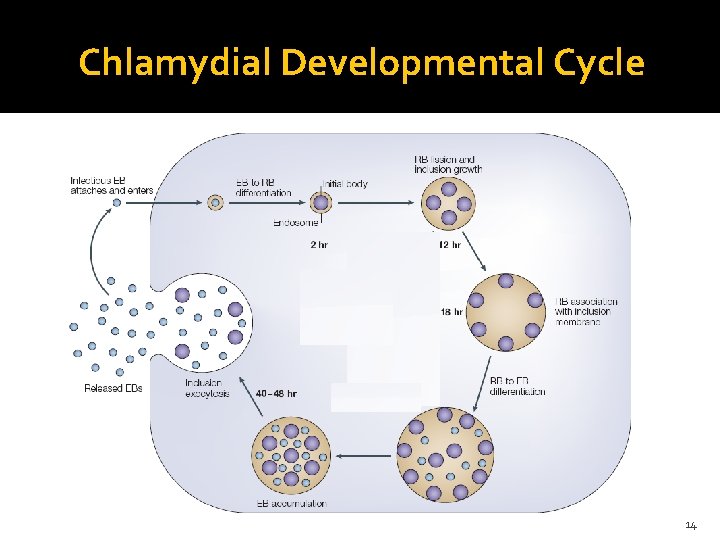
Chlamydial Developmental Cycle 14

Vaccines Against Chlamydial STD Major outer membrane protein (MOMP) ü ü ü Chlamydia-purified MOMP Recombinant MOMP DNA MOMP synthetic peptides MOMP plus outer membrane protein 2 (omp 2) Chlamydia-purified MOMP refolded to native configuration Cons: MOMP - serovar-specific Currently no licensed vaccine against C. trachomatis Need for identification of new vaccine candidates 15
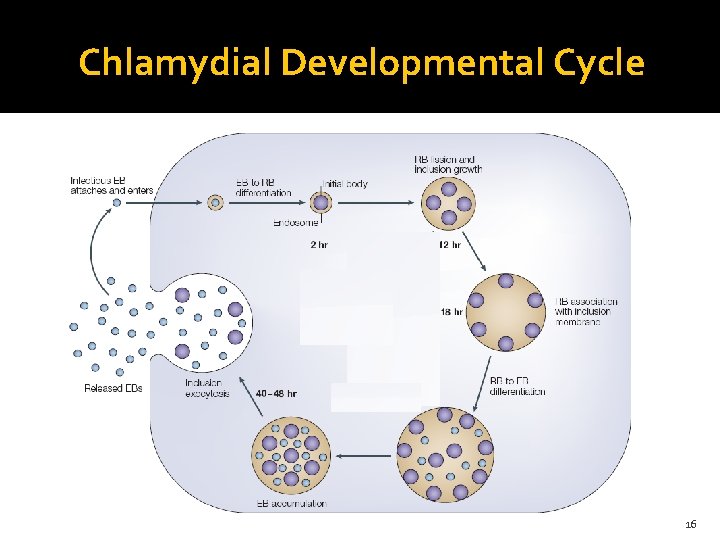
Chlamydial Developmental Cycle 16
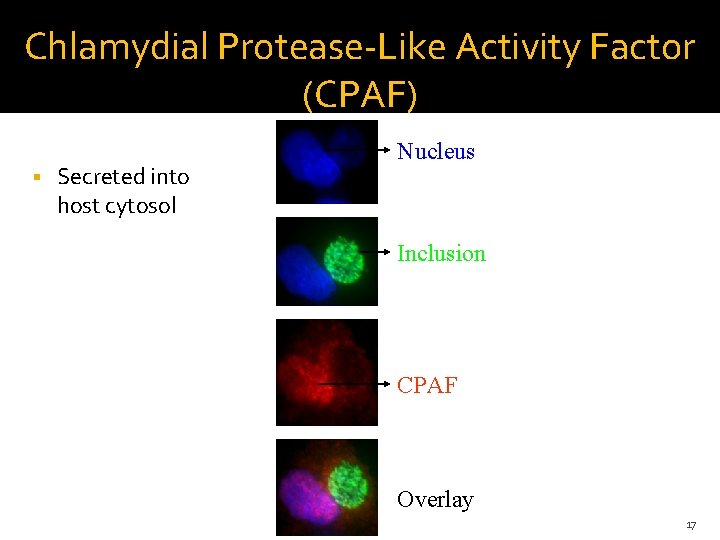
Chlamydial Protease-Like Activity Factor (CPAF) Secreted into host cytosol Nucleus Inclusion CPAF Overlay 17
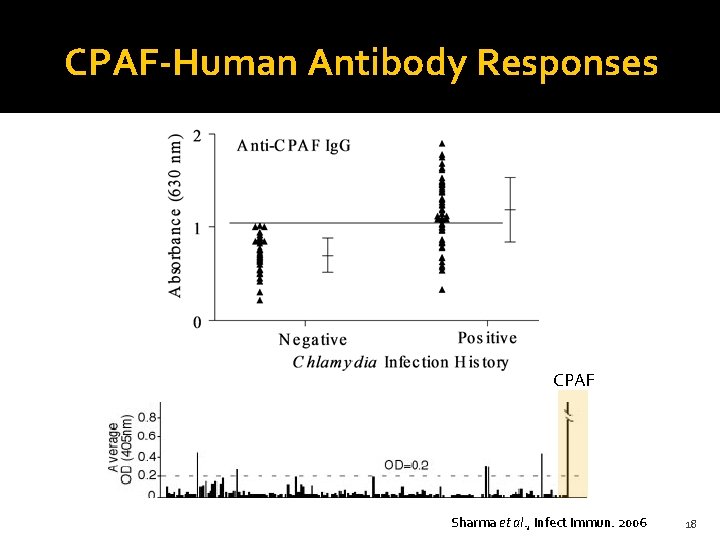
CPAF-Human Antibody Responses CPAF Sharma et al. , Infect Immun. 2006 18
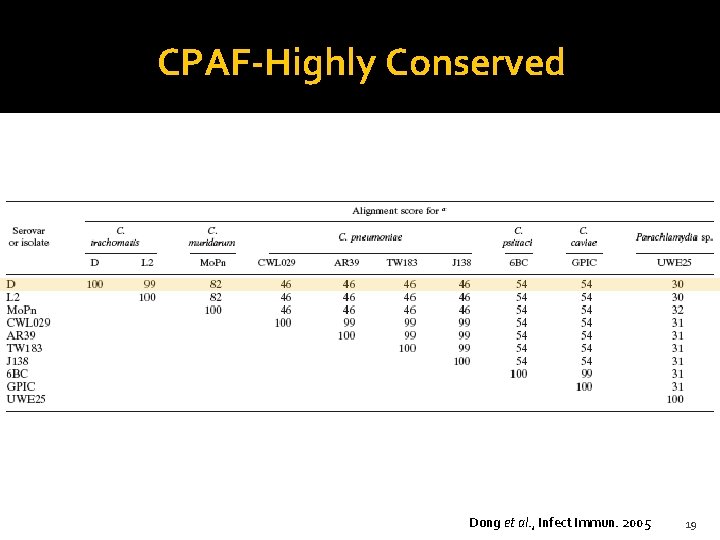
CPAF-Highly Conserved Dong et al. , Infect Immun. 2005 19

CPAF- A Potential Vaccine Candidate 20
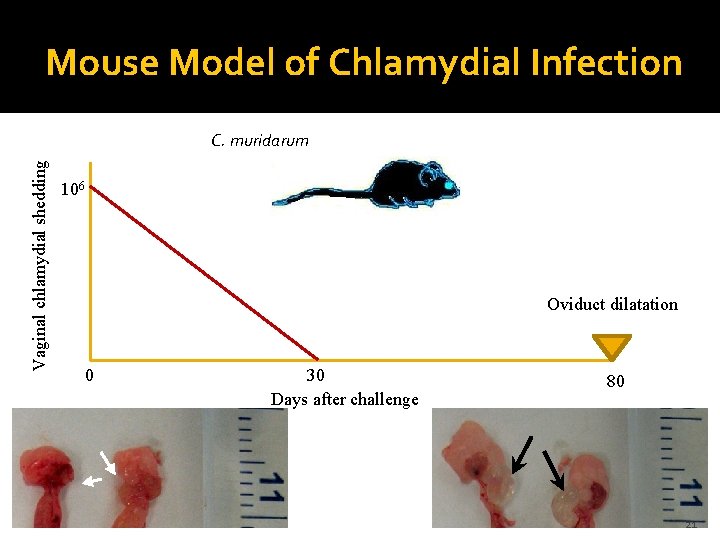
Mouse Model of Chlamydial Infection Vaginal chlamydial shedding C. muridarum 106 Oviduct dilatation 0 30 Days after challenge 80 21
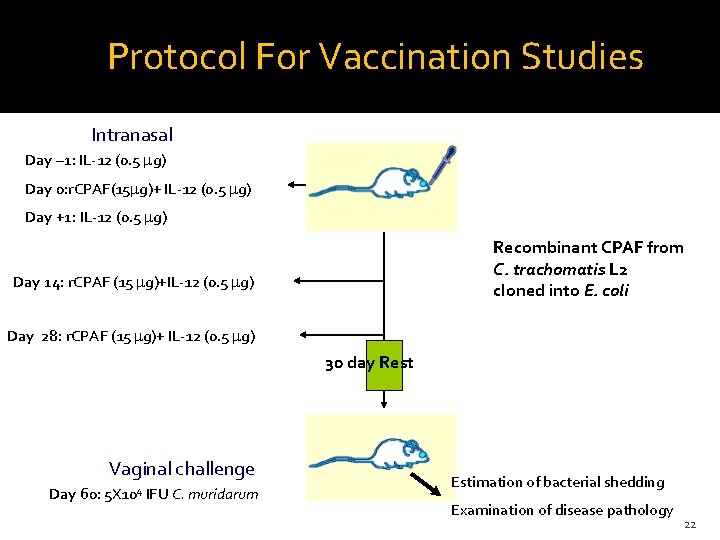
Protocol For Vaccination Studies Intranasal Day – 1: IL-12 (0. 5 mg) Day 0: r. CPAF(15 mg)+ IL-12 (0. 5 mg) Day +1: IL-12 (0. 5 mg) Recombinant CPAF from C. trachomatis L 2 cloned into E. coli Day 14: r. CPAF (15 mg)+IL-12 (0. 5 mg) Day 28: r. CPAF (15 mg)+ IL-12 (0. 5 mg) 30 day Rest Vaginal challenge Day 60: 5 X 104 IFU C. muridarum Estimation of bacterial shedding Examination of disease pathology 22
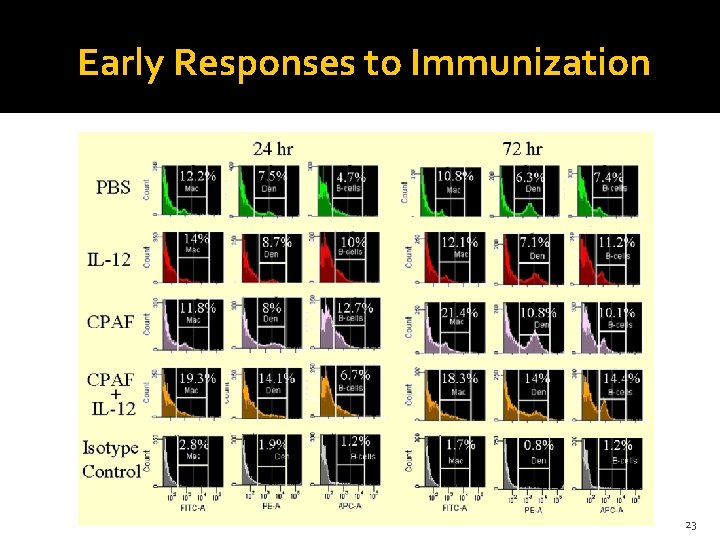
Early Responses to Immunization 23
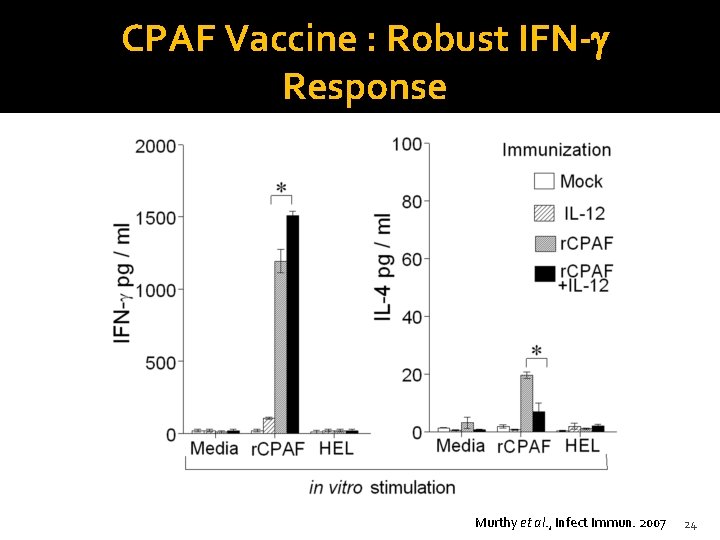
CPAF Vaccine : Robust IFN-g Response Murthy et al. , Infect Immun. 2007 24
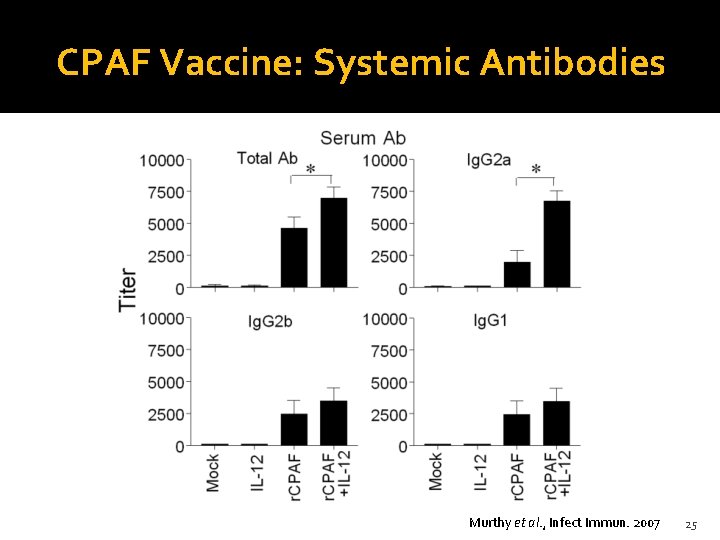
CPAF Vaccine: Systemic Antibodies Murthy et al. , Infect Immun. 2007 25
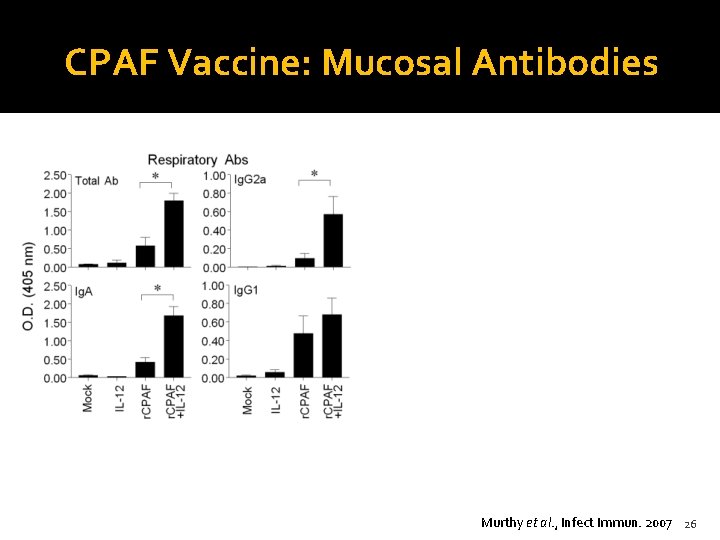
CPAF Vaccine: Mucosal Antibodies Murthy et al. , Infect Immun. 2007 26
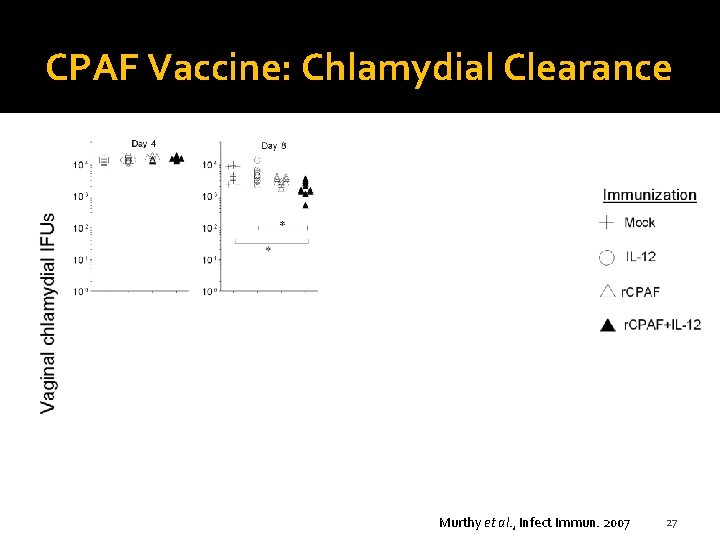
CPAF Vaccine: Chlamydial Clearance Murthy et al. , Infect Immun. 2007 27
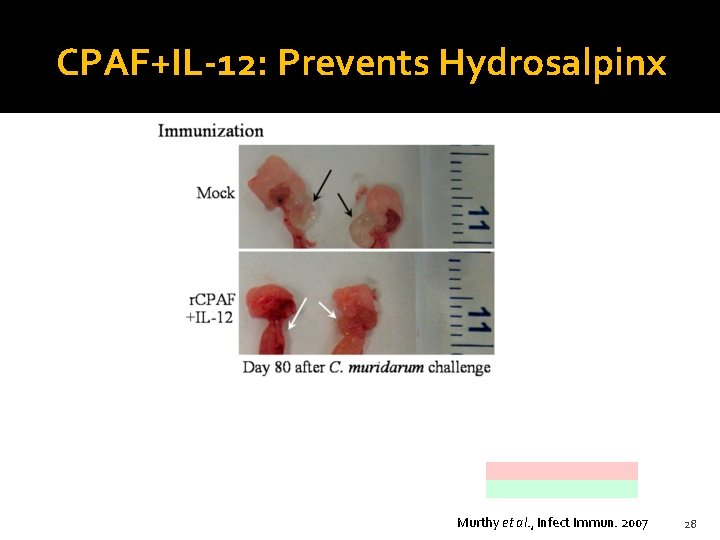
CPAF+IL-12: Prevents Hydrosalpinx Murthy et al. , Infect Immun. 2007 28
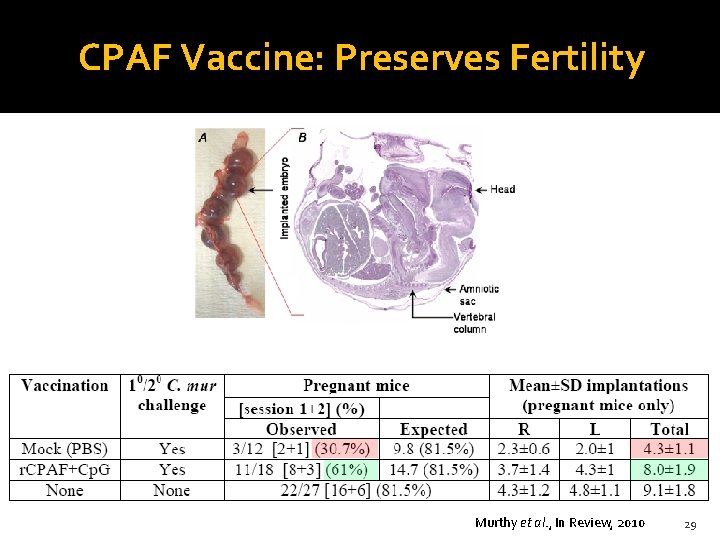
CPAF Vaccine: Preserves Fertility Murthy et al. , In Review, 2010 29

Role of Human HLA-DR Murthy et al. , Infect Immun. 2006 30
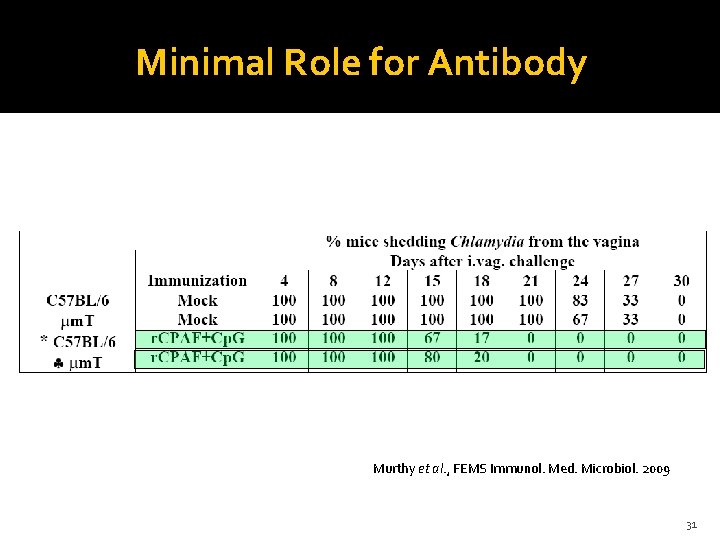
Minimal Role for Antibody Murthy et al. , FEMS Immunol. Med. Microbiol. 2009 31

Role of CD 4+ T-cells CD 4+ T cell adoptive transfer Murphey et al. , Cell. Immunol. 2006 32
Role of IFN-g Murthy et al. , J. Immunol. 2008 33

Summary � CPAF vaccine administered with Th 1 adjuvant: Enhances clearance of infection Protects against severe pathology Induces robust cellular IFN-g response Induces systemic and mucosal antibody Protection is dependent upon CPAF-specific cellular IFN-g responses but not antibody 34

Ongoing Collaborative Effort US Patent Application No. 12/243, 769: Inventors- Arulanandam, Murthy, Zhong 35
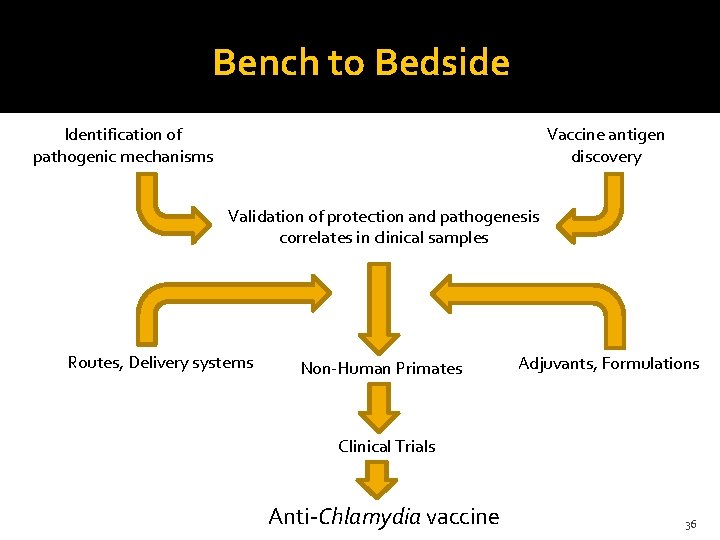
Bench to Bedside Identification of pathogenic mechanisms Vaccine antigen discovery Validation of protection and pathogenesis correlates in clinical samples Routes, Delivery systems Non-Human Primates Adjuvants, Formulations Clinical Trials Anti-Chlamydia vaccine 36

Acknowledgements Bernard Arulanandam, Ph. D. , M. B. A. Weidang Li, M. D. , Ph. D. Bharat Chaganty, MS Sangamithra Kamalakaran, B. Tech Kishan Evani, MS Rishein Gupta, Ph. D. Guangming Zhong, M. D. , Ph. D. , UTHSCSA M. Neal Guentzel, Ph. D. , STCEID, UTSA Aruna Mittal, Ph. D, IOP, India Yu Cong, M. D. Madhulika Jupelli, Ph. D. Cathi Murphey, M. S. 1 R 03 AI 088342 37

38
- Slides: 38