AntiCancer Drugs By Nohad A Atrushi 2932015 INTRODUCTION

(Anti-Cancer Drugs) By Nohad A. Atrushi 29/3/2015

INTRODUCTION • Cancer: group of diseases characterized by uncontrolled growth and spread of abnormal cells that left untreated may lead to death. • Neoplesia: uncontrolled growth of new tissue the product of which is known as tumor & these tumors may be either malignant or benign. • Malignant tumors have the capability of invading surrounding tissues and moving to distant location in the body in process called metastasis that characteristic benign tumors does not posses.

Overview Introduction n Malignant disease accounts for a high proportion of deaths in industrialised countries. n The treatment of anticancer drug is to give palliation, induce remission and, if possible, cure.

Overview Introduction • Cancer occurs after normal cells have been transformed into neoplastic cells through alteration of their genetic material and the abnormal expression of certain genes. • Neoplastic cells usually exhibit chromosomal abnormalities and the loss of their differentiated properties. These changes lead to uncontrolled cell division and many result in the invasion of previously unaffected organs, a process called metastasis.

Advances in Cancer Chemotherapy Treatment options of cancer: • Surgery: • Radiotherapy: • Chemotherapy: kill cells (ABs & anticancer agents ) • Immunotherapy and Gene therapy

• Anticancer = antineoplastic • Chemotherapy = selective cytotoxicity (difficult!!!!!) • Bec. The cell utilized biochemical pathways utilized by normal cells. • Inc. knowledge of intercellular & intracellular communication has let to develop of several new agents. (monoclonal antibodies target overproduction of growth factor receptors & tyrosine kinase(TK) inhibitors)
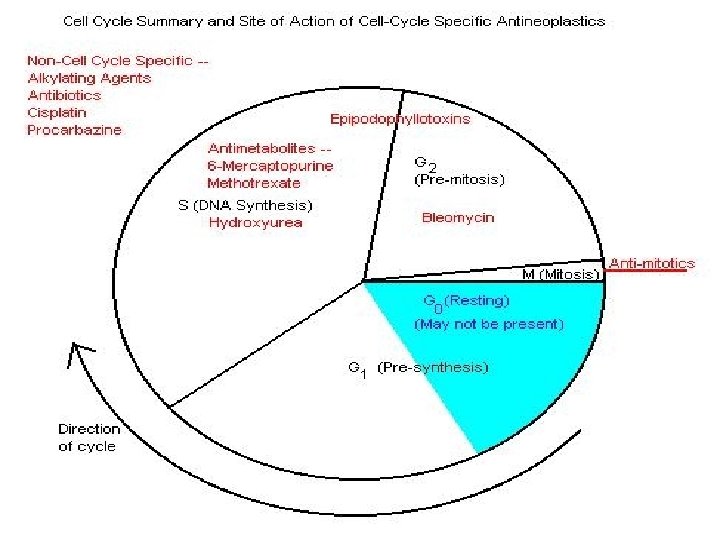

Cell cycle specific agents and Cell cycle Non-specific agents u Cell Cycle Nonspecific Agents (CCNSA) drugs that are active throughout the cell cycle Alkylating Agents p Platinum Compounds p Antibiotics p

Cell cycle specific agents and Cell cycle Non-specific agents u Cell Cycle Specific Agents (CCSA) drugs that act during a specific phase of the cell cycle S Phase Specific Drug: A. Antimetabolites, Topoisomerase Inhabitors M Phase Specific Drug: Vinca Alkaloids, Taxanes G 2 Phase Specific Drug: B. bleomycin

Antineoplastic agents can be divided into main four groups: • • 1. Alkylating agents. 2. Antibiotics. 3. Antimetabolites and natural products, and 4. Tyrosine kinase (TK) inhibitors.

1. Alkylating Agents The alkylating agents are a class of drugs that are capable of forming covalent bonds with important biomolecules. There are several potential nucleophilic sites on DNA, which are susceptible to electrophilic attack by an alkylating agent (N-2, N -3, and N-7 of guanine, N-1, N-3, and N-7 of adenine, 0 -6 of thymine, N-3 of cytosine). The most important of these for many alkylating agents is the N-7 position of guanine whose nucleophilicity may be enhanced by adjacent guanine residues. Alkylation converts the base to an effective leaving group so that attack by water leads to depurination and the loss of genetic information if the resulting depurination is not repaired by the cell (Scheme 10. 1).
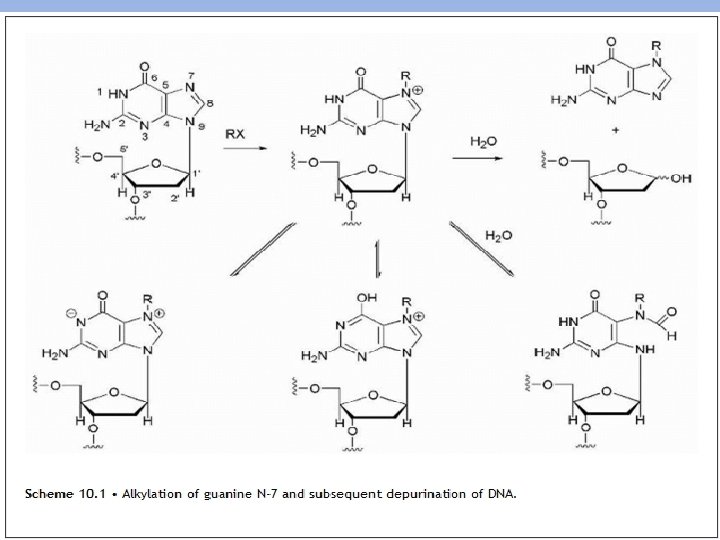

The general mechanism for alkylation involves nucleophilic attack by —N=, — NH 2, —OH, —O—PO 3 H of DNA and RNA, while additional nucleophiles (— SH, COOH, etc. ) present on proteins may also react (Scheme 10. 2). Anion formation increases the reactivity of the nucleophile compared with the un-ionized form (—O- is more nucleophilic than OH). Reaction with water is also possible, because it represents the nucleophile in greatest abundance in the body and this becomes more likely as the electrophile becomes more reactive. Reaction involves displacement of a leaving group on the electrophile by the nucleophile. The reactivity of the electrophile is dependent in part on the ability of the leaving group to stabilize a negative charge.

Alkylating agents • Alkylation is defined as the replacement of H on an atom (usually nucleic acids and proteins) by an alkyl group. Nu-H + alkyl-Y alkyl-Nu + H+ + Y • The reaction rate depends on the nucleophilicity of the atom (S, N, O), of the nucleic acid or protein, which is greatly enhanced if the nucleophile is ionized. • The order of reactivity at physiological p. H: S- > NH 2>PO 4 -3> COO-. • The main group of alkylating agents is nitrogen mustards. • The sulphur mustard (mustard gas) has anti- tumor activity, but it was too nonspecific for clinical use.

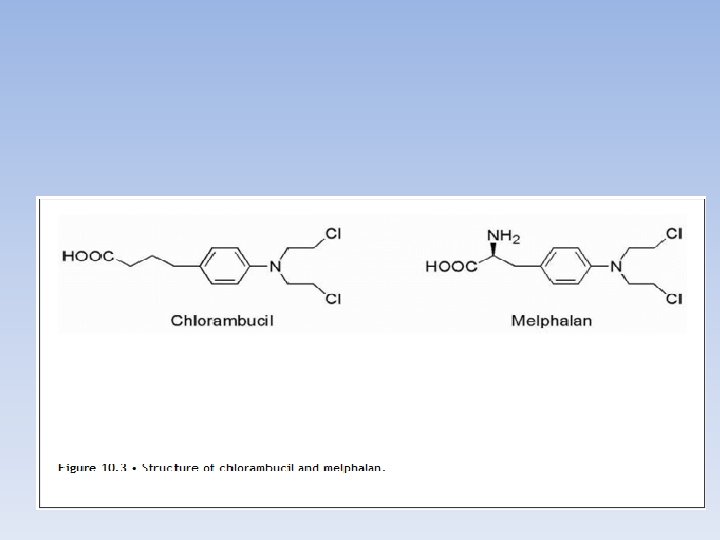
NITROGEN MUSTARDS The nitrogen mustards are compounds that are chemically similar to sulfur mustard or mustard gas developed and used in World War I. Investigation of sulfur mustard revealed that it possessed antineoplastic properties but because the compound existed as a gas at room temperature. Conversion of the sulfide to a tertiary amine allowed for the formation of salts, which exist as solids at room temperature allowing for easier handling and dosing. Chlorambucil, melphalan, cyclophosphamide and ifosfamide. The lack of selectivity of mechlorethamine led to attempts to improve on the agent. One rationale was to reduce the reactivity by reducing the nucleophilicity of nitrogen, thereby slowing aziridinium cation formation. This could be accomplished by replacement of the weakly electron-donating methyl group with groups that were electron withdrawing (-I). This is seen in the case of chlorambucil and melphalan by attachment of nitrogen to a phenyl ring (Fig. 10. 3).
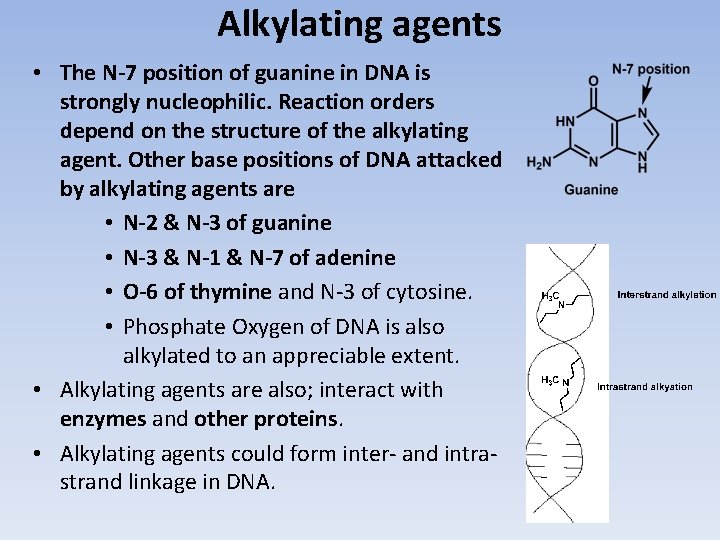
Alkylating agents • The N-7 position of guanine in DNA is strongly nucleophilic. Reaction orders depend on the structure of the alkylating agent. Other base positions of DNA attacked by alkylating agents are • N-2 & N-3 of guanine • N-3 & N-1 & N-7 of adenine • O-6 of thymine and N-3 of cytosine. • Phosphate Oxygen of DNA is also alkylated to an appreciable extent. • Alkylating agents are also; interact with enzymes and other proteins. • Alkylating agents could form inter- and intrastrand linkage in DNA.

Alkylating agents • The cell can repair the damage to their DNA before the next cell division, and the effects of alkylation will not be lethal. • Cells have developed a complex mechanism to discover and accomplish this repair. An endonuclease makes a singlestrand breakage in DNA, i. e. cuts the damaged strand an exonuclease then removes a small segment of DNA containing the damaged bases. • Finally, the DNA is restored to its original structure (backfilled) by replacing the bases and rejoining the strand. Tumor cells with efficient repair mechanism will be relatively resistant to alkylating agents.
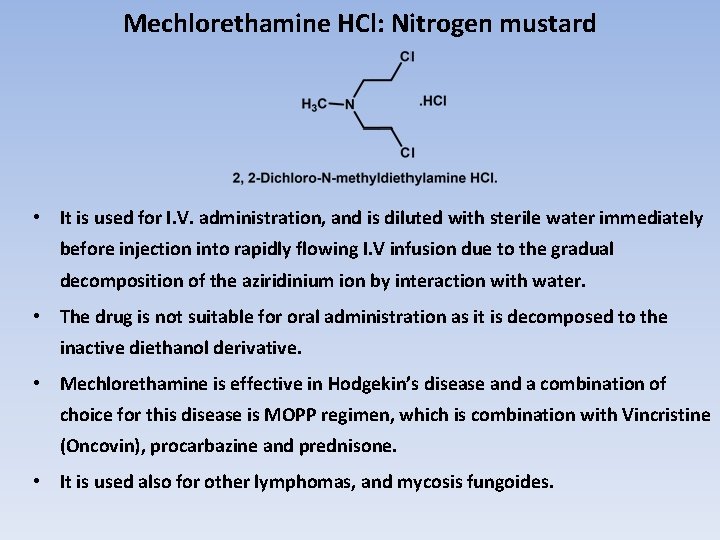
Mechlorethamine HCl: Nitrogen mustard • It is used for I. V. administration, and is diluted with sterile water immediately before injection into rapidly flowing I. V infusion due to the gradual decomposition of the aziridinium ion by interaction with water. • The drug is not suitable for oral administration as it is decomposed to the inactive diethanol derivative. • Mechlorethamine is effective in Hodgekin’s disease and a combination of choice for this disease is MOPP regimen, which is combination with Vincristine (Oncovin), procarbazine and prednisone. • It is used also for other lymphomas, and mycosis fungoides.
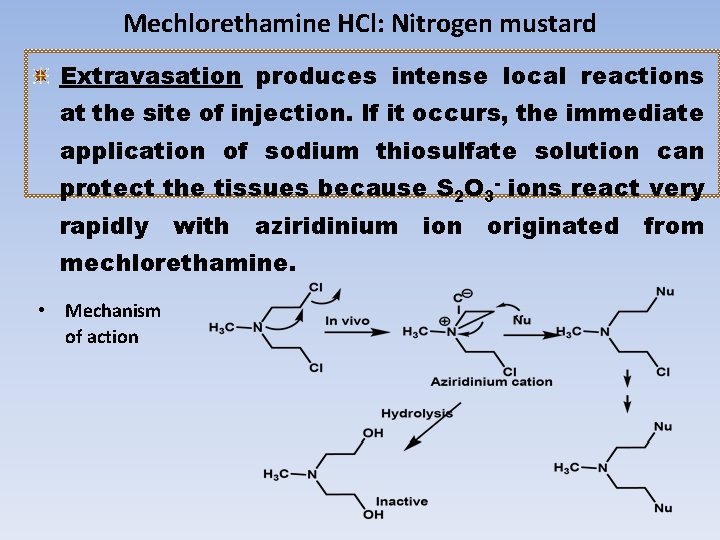
Mechlorethamine HCl: Nitrogen mustard Extravasation produces intense local reactions at the site of injection. If it occurs, the immediate application of sodium thiosulfate solution can protect the tissues because S 2 O 3 - ions react very rapidly with aziridinium mechlorethamine. • Mechanism of action originated from
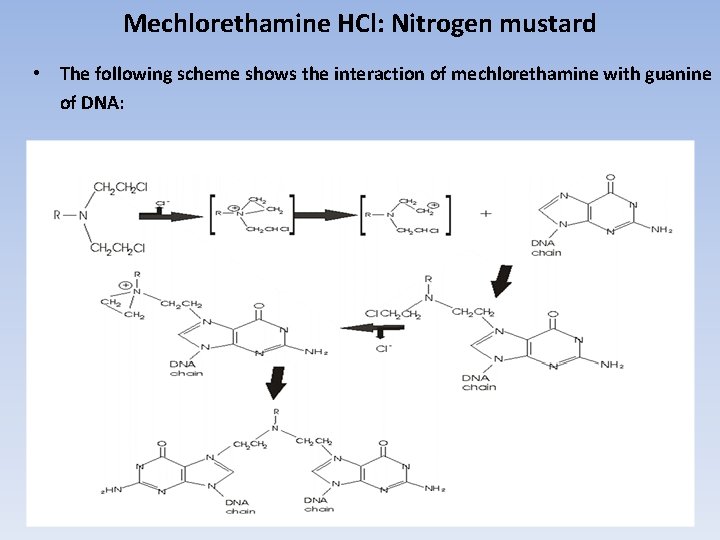
Mechlorethamine HCl: Nitrogen mustard • The following scheme shows the interaction of mechlorethamine with guanine of DNA:
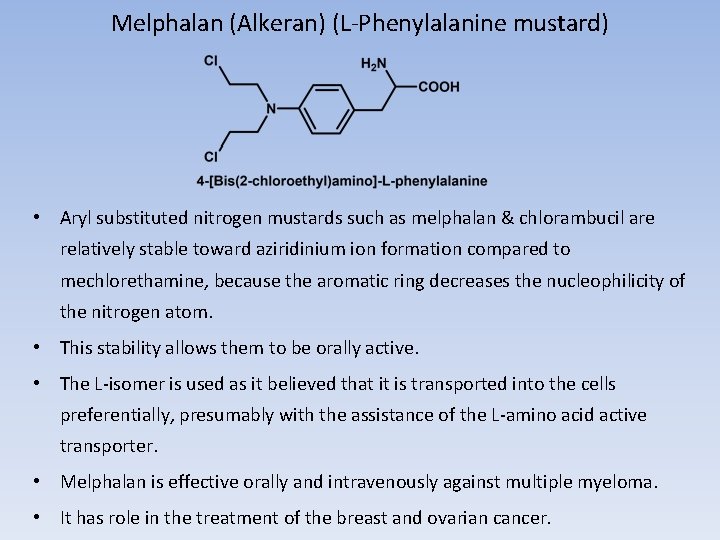
Melphalan (Alkeran) (L-Phenylalanine mustard) • Aryl substituted nitrogen mustards such as melphalan & chlorambucil are relatively stable toward aziridinium ion formation compared to mechlorethamine, because the aromatic ring decreases the nucleophilicity of the nitrogen atom. • This stability allows them to be orally active. • The L-isomer is used as it believed that it is transported into the cells preferentially, presumably with the assistance of the L-amino acid active transporter. • Melphalan is effective orally and intravenously against multiple myeloma. • It has role in the treatment of the breast and ovarian cancer.
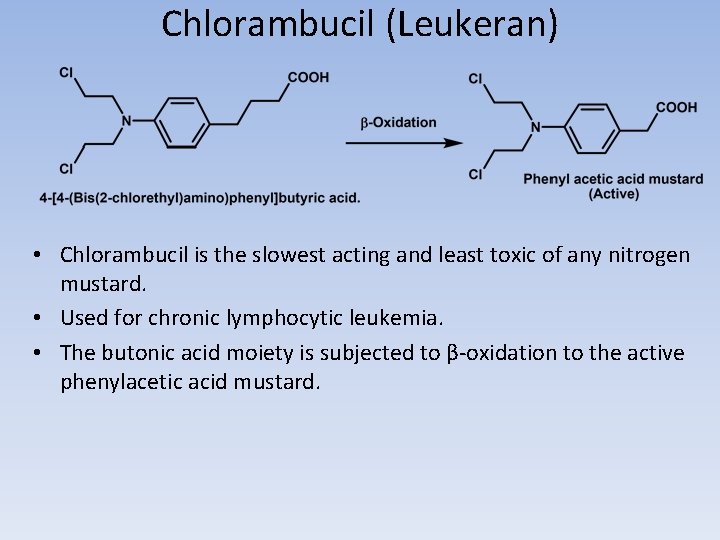
Chlorambucil (Leukeran) • Chlorambucil is the slowest acting and least toxic of any nitrogen mustard. • Used for chronic lymphocytic leukemia. • The butonic acid moiety is subjected to β-oxidation to the active phenylacetic acid mustard.
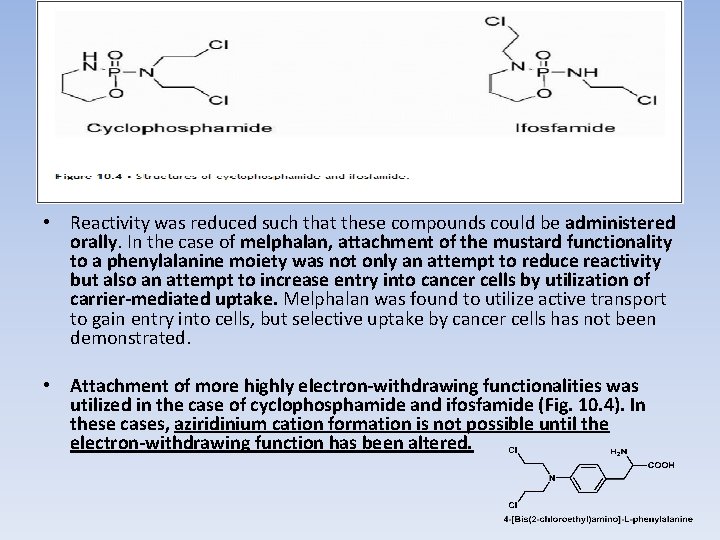
• Reactivity was reduced such that these compounds could be administered orally. In the case of melphalan, attachment of the mustard functionality to a phenylalanine moiety was not only an attempt to reduce reactivity but also an attempt to increase entry into cancer cells by utilization of carrier-mediated uptake. Melphalan was found to utilize active transport to gain entry into cells, but selective uptake by cancer cells has not been demonstrated. • Attachment of more highly electron-withdrawing functionalities was utilized in the case of cyclophosphamide and ifosfamide (Fig. 10. 4). In these cases, aziridinium cation formation is not possible until the electron-withdrawing function has been altered.

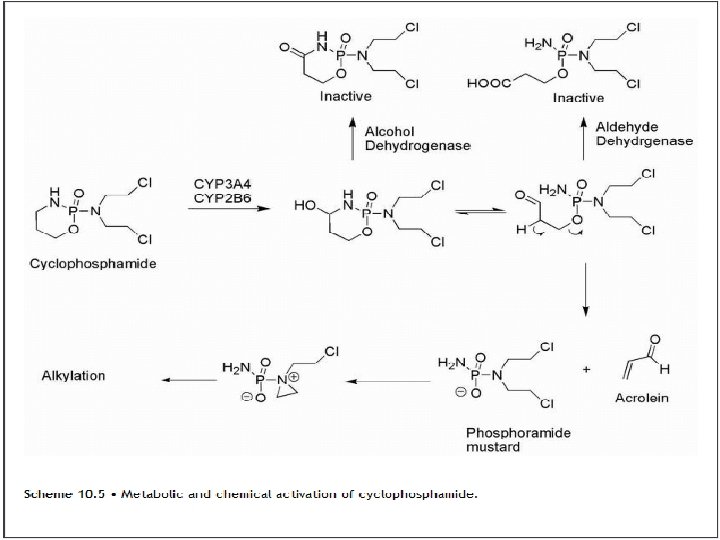
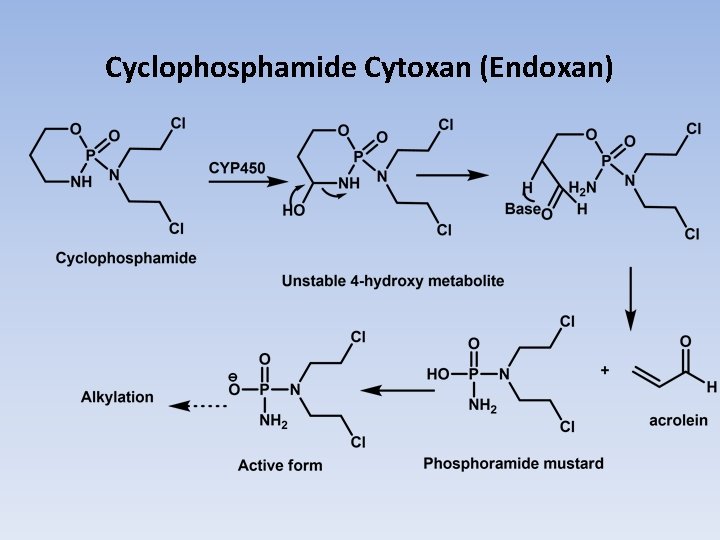
• In the case of cyclophosphamide, it was initially believed that the drug could be selectively activated in cancer cells because they were believed to contain high levels of phosphoramidase enzymes. This would remove the electron-withdrawing phosphoryl function and allow aziridine formation to occur. However, it turned out that the drug was activated by cytochrome P 450 (CYP) isozymes CYP 2 B 6 and CYP 3 A 4/5 to give a carbinolamine that could undergo ring opening to give the aldehyde The increased acidity of the aldehyde α-hydrogen facilitates a retro-Michael decomposition (Scheme 10. 5). • The ionized phosphoramide is now electron-releasing via induction and allows aziridinium cation formation to proceed. Acrolein is also formed as a result of this process, which may itself act as an electrophile that has been associated with bladder toxicity. Alternatively, the agent may be inactivated by alcohol dehydrogenase-mediated oxidation of the carbinolamine to give the amide or • by further oxidation of the aldehyde intermediate to give the acid by aldehyde dehydrogenase.
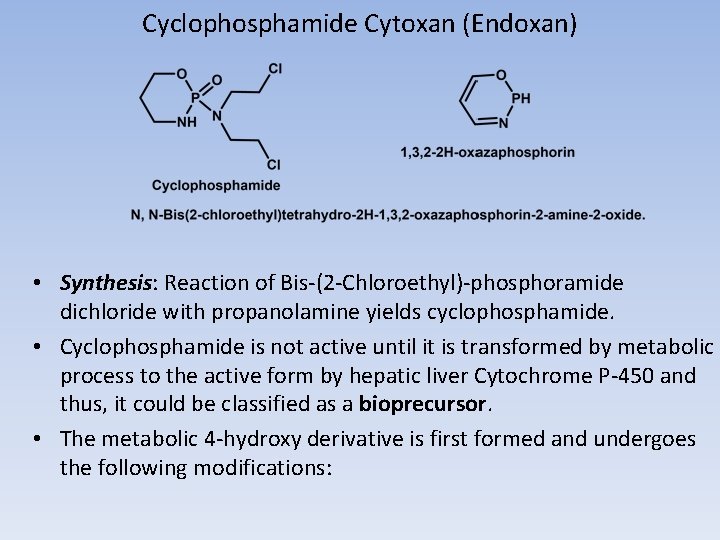
Cyclophosphamide Cytoxan (Endoxan) • Synthesis: Reaction of Bis-(2 -Chloroethyl)-phosphoramide dichloride with propanolamine yields cyclophosphamide. • Cyclophosphamide is not active until it is transformed by metabolic process to the active form by hepatic liver Cytochrome P-450 and thus, it could be classified as a bioprecursor. • The metabolic 4 -hydroxy derivative is first formed and undergoes the following modifications:
Cyclophosphamide Cytoxan (Endoxan)

Cyclophosphamide Cytoxan (Endoxan) • The aziridinium ion is the principle cross linking alkylator formed from cyclophosphamide. • Maximum aziridinium ion formation occurs at p. H 7. 4. It was suggested that selective toxicity toward certain neoplastic cells might be based on their abnormally low p. H. • This would afford lower formation of aziridinium ions and they would persist longer because of decrease in activation by hydroxide ions. • The acrolein formed from the metabolic degradation of cyclophosphamide causes severe hemorrhagic cystitis leading to nephrotoxicity and urotoxicity.
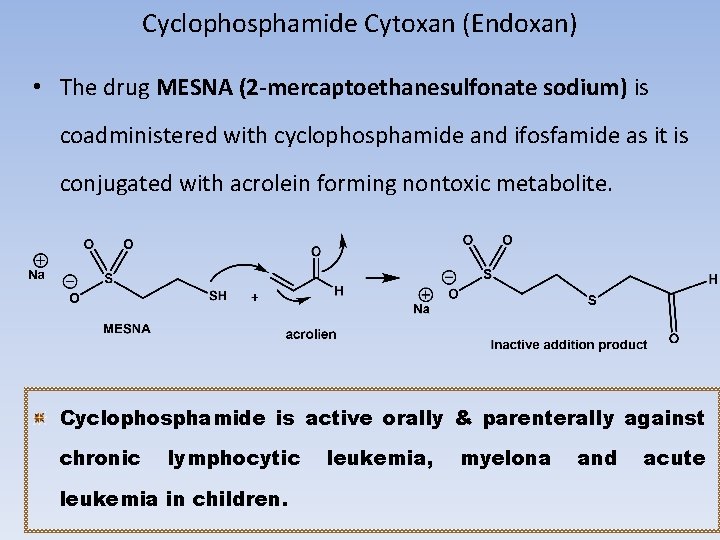
Cyclophosphamide Cytoxan (Endoxan) • The drug MESNA (2 -mercaptoethanesulfonate sodium) is coadministered with cyclophosphamide and ifosfamide as it is conjugated with acrolein forming nontoxic metabolite. Cyclophosphamide is active orally & parenterally against chronic lymphocytic leukemia in children. leukemia, myelona and acute
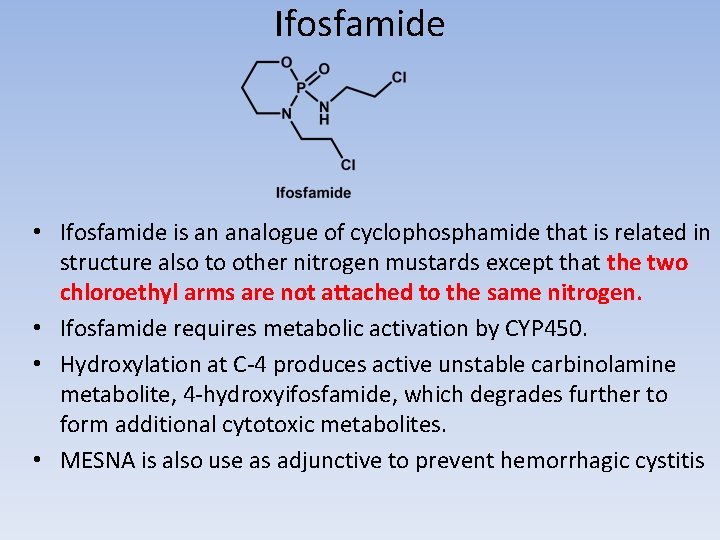
Ifosfamide • Ifosfamide is an analogue of cyclophosphamide that is related in structure also to other nitrogen mustards except that the two chloroethyl arms are not attached to the same nitrogen. • Ifosfamide requires metabolic activation by CYP 450. • Hydroxylation at C-4 produces active unstable carbinolamine metabolite, 4 -hydroxyifosfamide, which degrades further to form additional cytotoxic metabolites. • MESNA is also use as adjunctive to prevent hemorrhagic cystitis
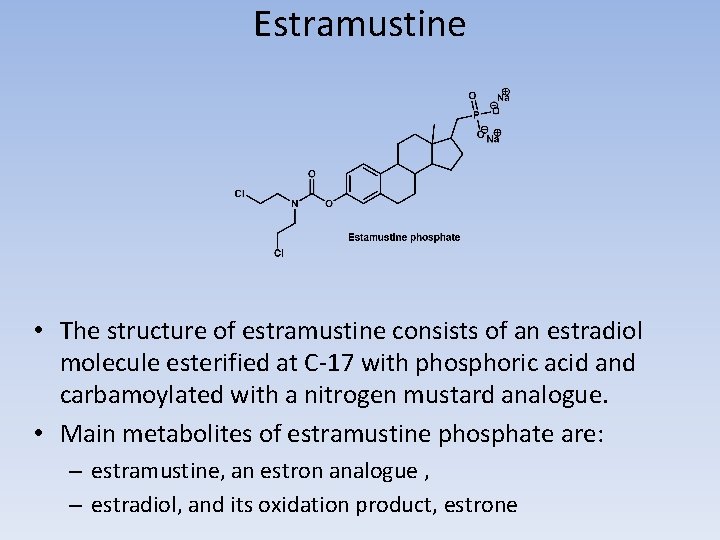
Estramustine • The structure of estramustine consists of an estradiol molecule esterified at C-17 with phosphoric acid and carbamoylated with a nitrogen mustard analogue. • Main metabolites of estramustine phosphate are: – estramustine, an estron analogue , – estradiol, and its oxidation product, estrone

Uracil mustard (Uramustine) • Synthesis: It is prepared by treating 5 -Aminouracil with ethylene oxide followed by thionyl chloride. • Uracil moiety serves as a carrier but little selectivity was shown in this respect. • It is used in Hodgkin’s disease, chronic lymphocytic leukemia, and lymphocarcinoma.

Other alkylating agents Busulfan (Myleran)
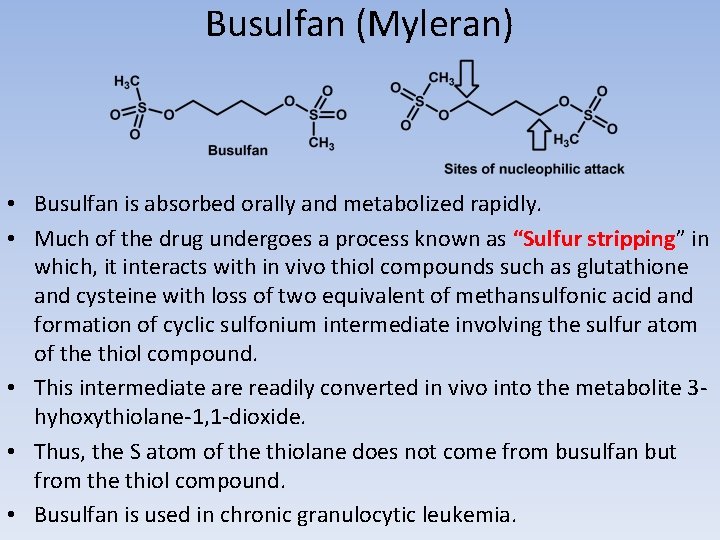
Busulfan (Myleran) • Busulfan is absorbed orally and metabolized rapidly. • Much of the drug undergoes a process known as “Sulfur stripping” in which, it interacts with in vivo thiol compounds such as glutathione and cysteine with loss of two equivalent of methansulfonic acid and formation of cyclic sulfonium intermediate involving the sulfur atom of the thiol compound. • This intermediate are readily converted in vivo into the metabolite 3 hyhoxythiolane-1, 1 -dioxide. • Thus, the S atom of the thiolane does not come from busulfan but from the thiol compound. • Busulfan is used in chronic granulocytic leukemia.
Busulfan (Myleran)

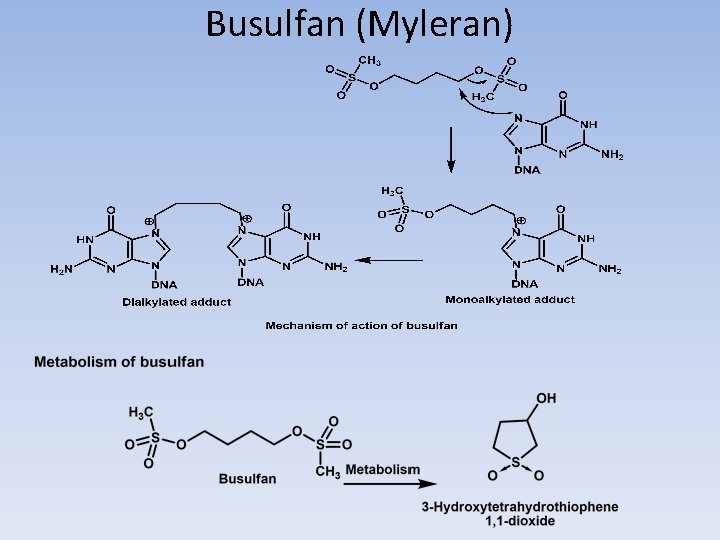
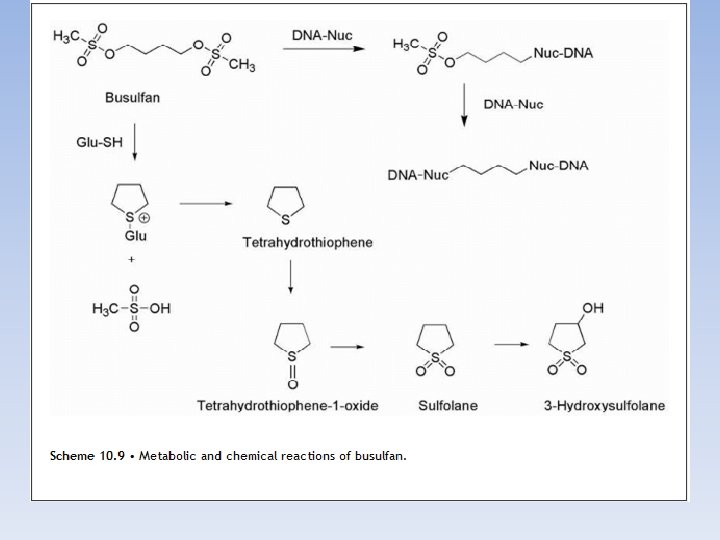
BUSULFAN • As an alternative to utilizing aziridines as electrophilic species. Busulfan utilizes two sulfonate functionalities as leaving groups separated by a four-carbon chain that reacts with DNA to primarily form intrastrand cross-link at 5′-GA-3′ sequences. The sulfonates are also subject to displacement by the sulfhydryl functions found in cysteine and glutathione, and metabolic products are formed as a result of nucleophilic attack by these groups to generate sulfonium species along with methane sulfonic acid. This is followed by conversion to tetrahydrothiophene, and further oxidation products are subsequently produced to give the sulfoxide and sulfone. The cyclic sulfone known as sulfolane may be further oxidized to give 3 -hydroxysulfolane.

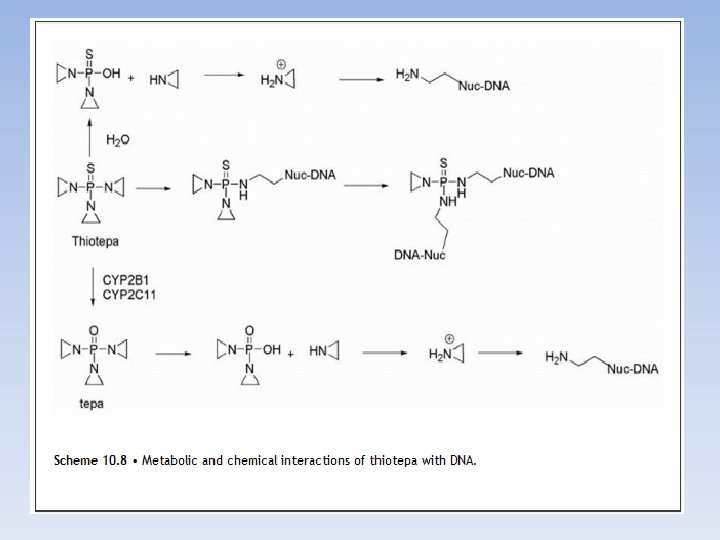
THIOTEPA Thiotepa containing the thiophosphoramide functionality was found to be more stable than the oxa-analog (TEPA) but is metabolically converted to TEPA by desulfuration in vivo. Thiotepa incorporates a less reactive aziridine ring compared with that formed in mechlorethamine. The adjacent thiophosphoryl is electron withdrawing and, therefore, reduces the reactivity of the aziridine ring system. The conclusion that aziridine is the active alkylating agent once thiotepa has been converted to TEPA is based on the fact that when TEPA is incubated with DNA, no cross links are formed and only mono adducts are generated. The reactivity of aziridine generated by either route may be somewhat enhanced within cancer cells, where the p. H is normally reduced 0. 2 to 0. 4 p. H units resulting in an increase in reactivity toward nucleophilic attack.

Other alkylating agents Platinum (IV) Compounds

ORGANO PLATINUM COMPOUNDS There are several organometallic compounds based on platinum that play a central role in many cancer treatment protocols. Movement into the tumor cells is accomplished by passive diffusion or carrier-mediated transport. Once inside the tumor cell, the drug encounters a lower chloride concentration and one chloro group is substituted by a water molecule in a process known as aquation. This serves to “trap” the molecule in the cell as a result of ionization. Reaction with DNA occurs preferentially at the N-7 of guanine of two adjacent guanine residues resulting in primarily intrastrand cross-links.
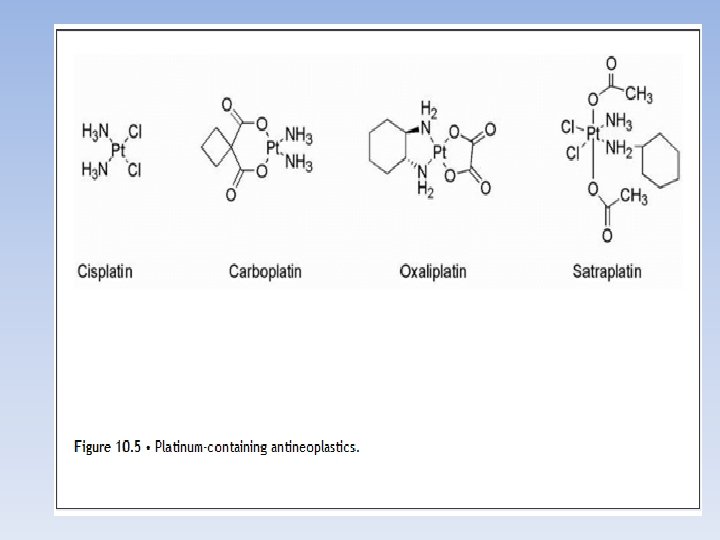
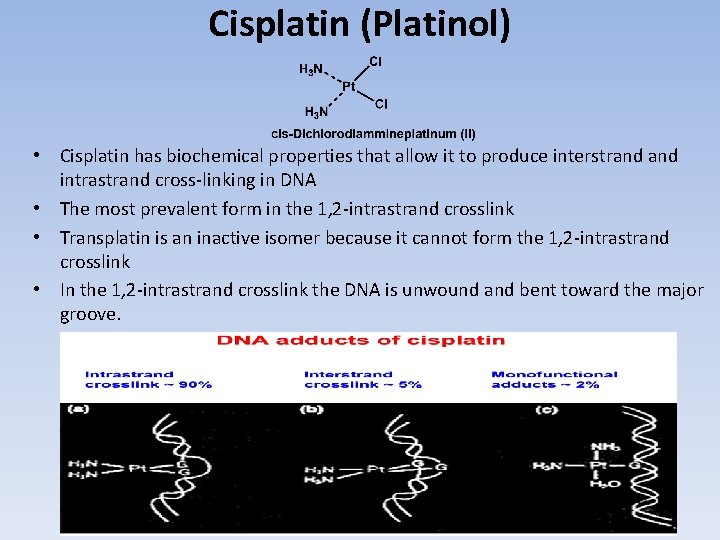
Cisplatin (Platinol) • Cisplatin has biochemical properties that allow it to produce interstrand intrastrand cross-linking in DNA • The most prevalent form in the 1, 2 -intrastrand crosslink • Transplatin is an inactive isomer because it cannot form the 1, 2 -intrastrand crosslink • In the 1, 2 -intrastrand crosslink the DNA is unwound and bent toward the major groove.

Cisplatin (Platinol)
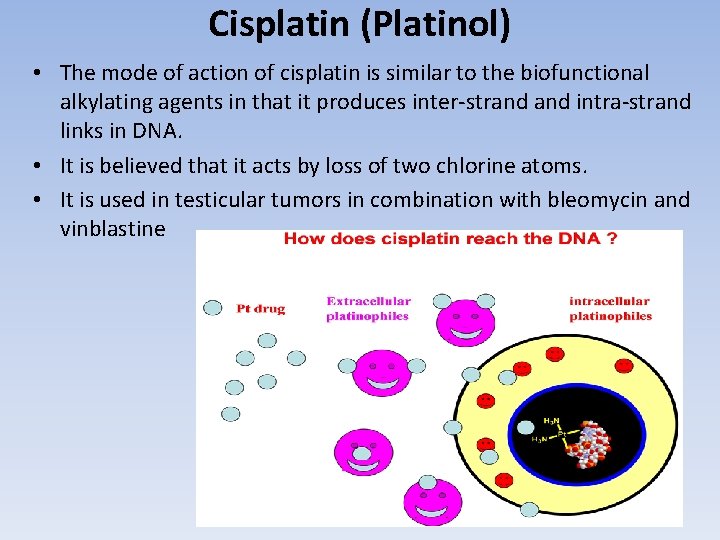
Cisplatin (Platinol) • The mode of action of cisplatin is similar to the biofunctional alkylating agents in that it produces inter-strand intra-strand links in DNA. • It is believed that it acts by loss of two chlorine atoms. • It is used in testicular tumors in combination with bleomycin and vinblastine
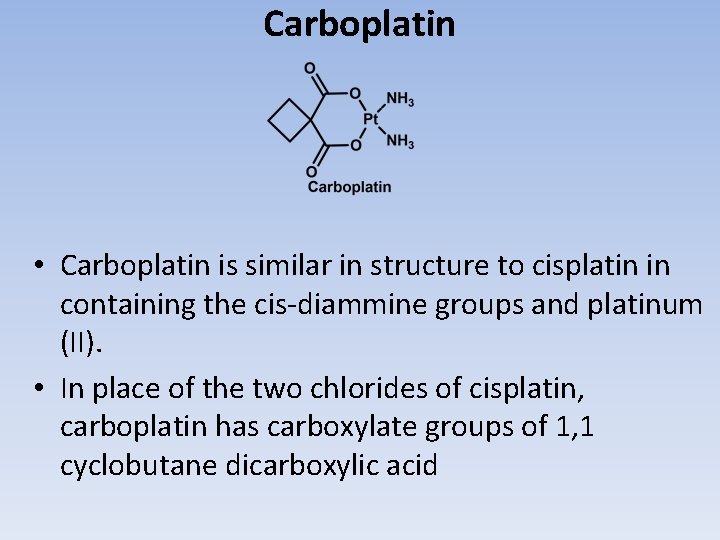
Carboplatin • Carboplatin is similar in structure to cisplatin in containing the cis-diammine groups and platinum (II). • In place of the two chlorides of cisplatin, carboplatin has carboxylate groups of 1, 1 cyclobutane dicarboxylic acid

Other alkylating agents N-alkyl-N-nitrosourea derivatives (Nitrosoureas)

NITROSOUREAS • The nitrosoureas were discovered as a result of drug screening by the Cancer Chemotherapy National Service Center, which identified N-methyl. N'-nitroguanidine as having activity against L 1210 leukemia. Further development of this lead compound was based on the idea that its chemical decomposition was leading to the formation of diazomethane (CH 2 N 2) and subsequent alkylation of DNA. This led to the nitrosoureas, where it was found that activity could be enhanced by attachment of a 2 haloethyl substituent to both nitrogens (Fig. 10. 6). •

These compounds are reasonably stable at p. H = 4. 5 but undergo both acid and base catalyzed decomposition at lower and higher p. H, respectively. There are several pathways of decomposition that are possible for these compounds, but the one that appears to be most important for alkylation of DNA involves abstraction of the NH proton, which is relatively acidic (p. Ka = 8 -9), followed by rearrangement to give an isocyanate and a diazohydroxide. The diazohydroxide, upon protonation followed by loss of water, yields a diazo species that decomposes to a reactive carbocation (Scheme 10. 11).
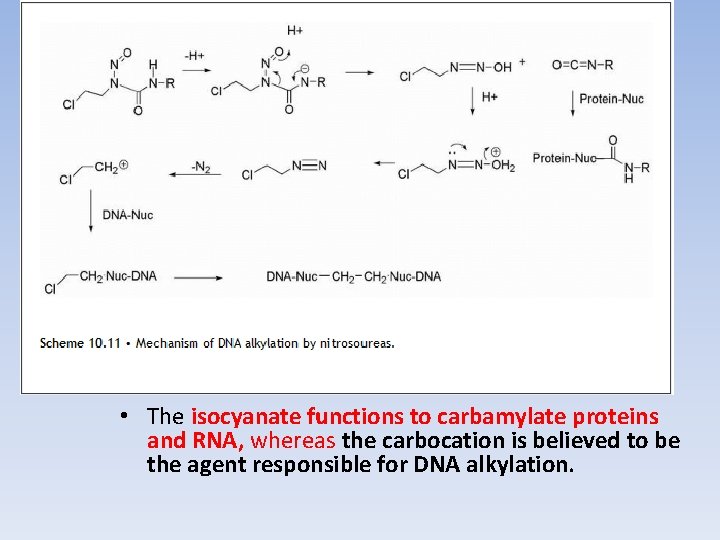
• The isocyanate functions to carbamylate proteins and RNA, whereas the carbocation is believed to be the agent responsible for DNA alkylation.
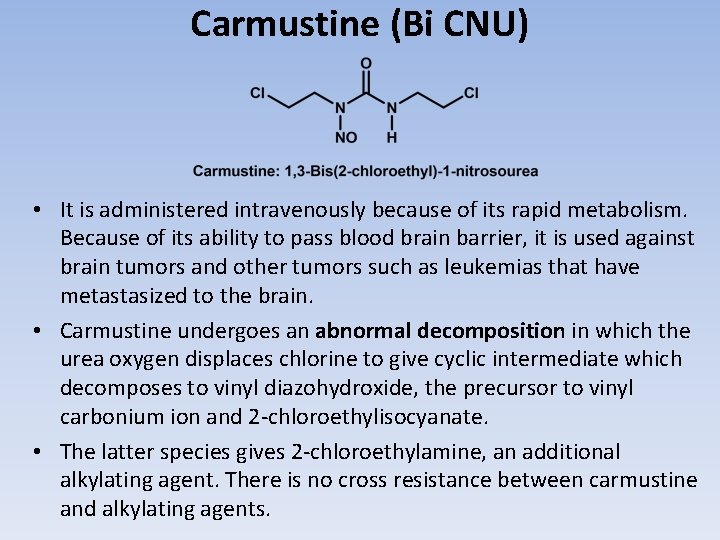
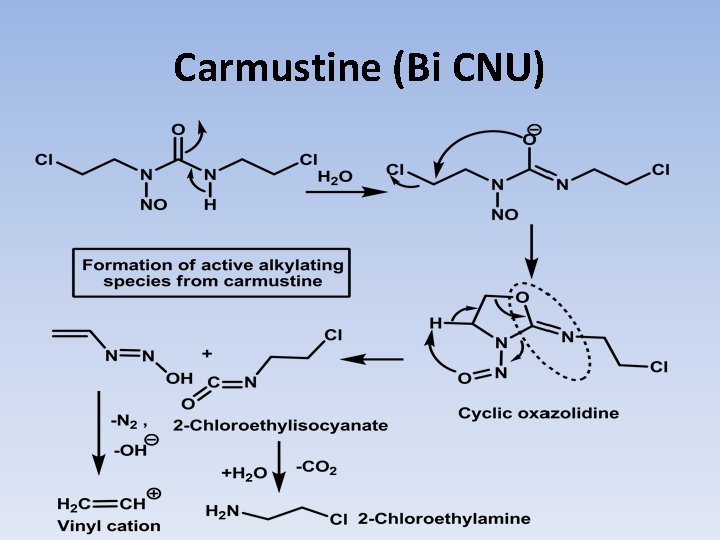
Carmustine (Bi CNU) • It is administered intravenously because of its rapid metabolism. Because of its ability to pass blood brain barrier, it is used against brain tumors and other tumors such as leukemias that have metastasized to the brain. • Carmustine undergoes an abnormal decomposition in which the urea oxygen displaces chlorine to give cyclic intermediate which decomposes to vinyl diazohydroxide, the precursor to vinyl carbonium ion and 2 -chloroethylisocyanate. • The latter species gives 2 -chloroethylamine, an additional alkylating agent. There is no cross resistance between carmustine and alkylating agents.
Carmustine (Bi CNU)

Lomustine (Cee. NU) • The lipid solubility of lomustine allows it to cross the B. B. B. rapidly and used for primary and metasatic brain tumors. • It can be given orally as capsules.
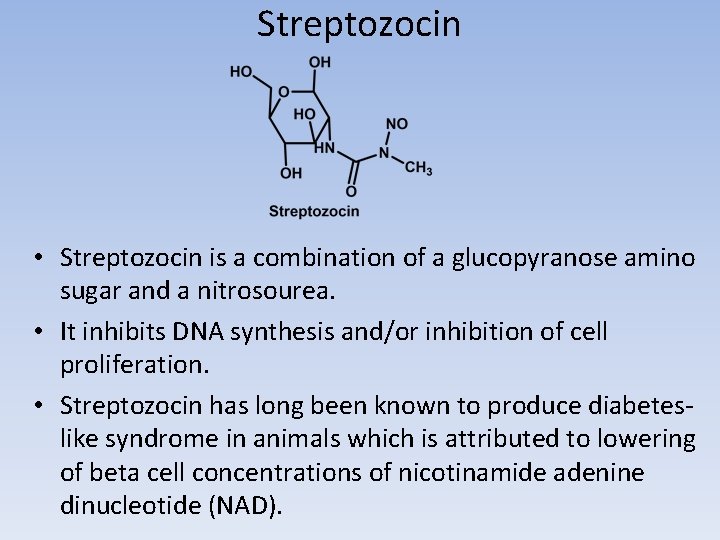
Streptozocin • Streptozocin is a combination of a glucopyranose amino sugar and a nitrosourea. • It inhibits DNA synthesis and/or inhibition of cell proliferation. • Streptozocin has long been known to produce diabeteslike syndrome in animals which is attributed to lowering of beta cell concentrations of nicotinamide adenine dinucleotide (NAD).

Miscellaneous alkylating agents

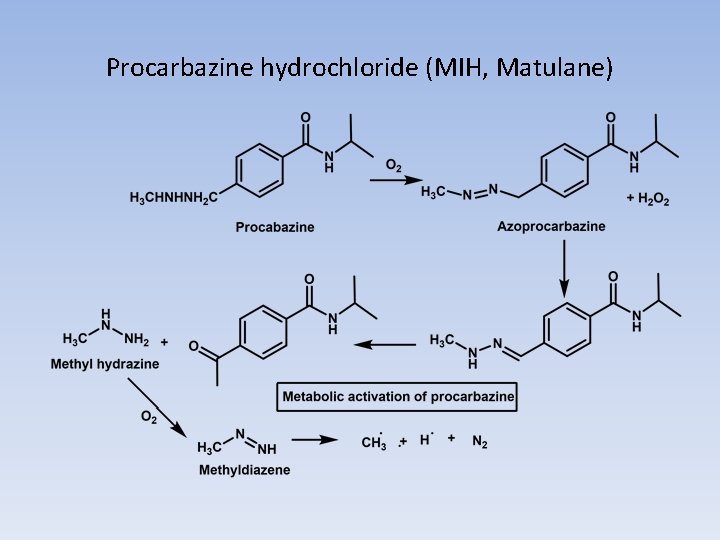
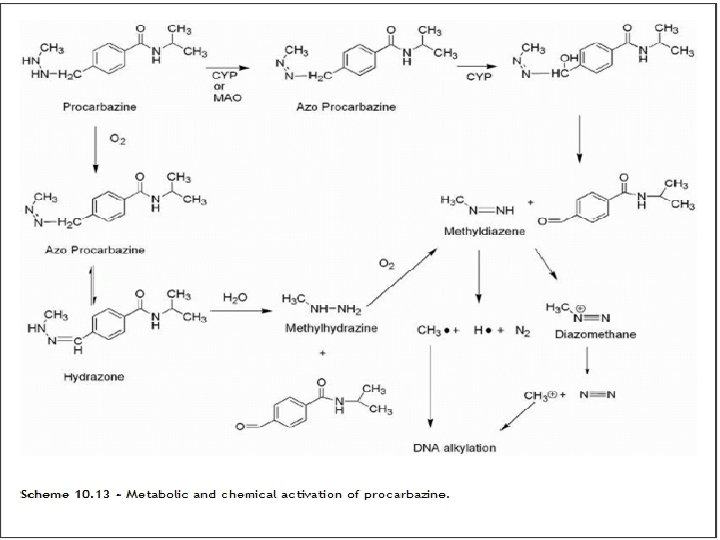
PROCARBAZINE, DACARBAZINE, AND TEMOZOLOMIDE • PROCARBAZINE • The oxidation of procarbazine occur in the liver and is mediated by CYP and monoamine oxidase to give azoprocarbazine. • This compound may also be generated nonenzymatically in an aerobic environment (Scheme 10. 13). • This involves CYP-mediated oxidation of the benzylic methylene carbon with subsequent decomposition to give methyldiazine and the aldehyde. The methyldiazine may then decompose by homolytic bond cleavage to give methyl and hydrogen radicals along with nitrogen gas or be further oxidized to give the diazo compound, which can decompose to give the methyl carbocation.
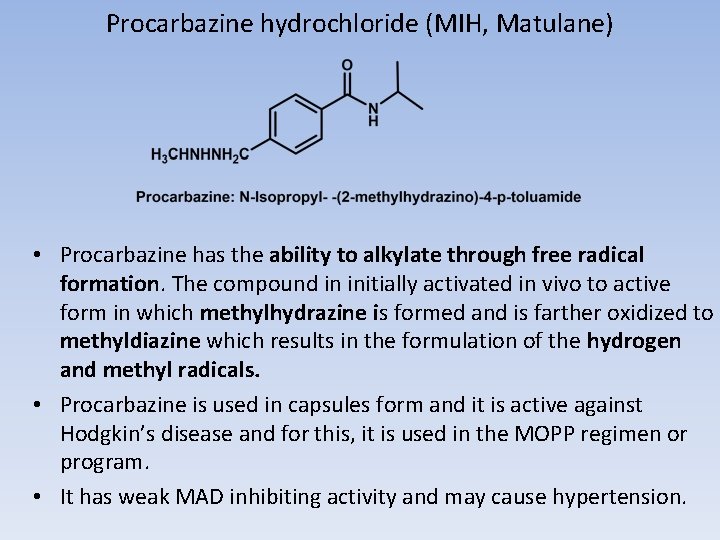
Procarbazine hydrochloride (MIH, Matulane) • Procarbazine has the ability to alkylate through free radical formation. The compound in initially activated in vivo to active form in which methylhydrazine is formed and is farther oxidized to methyldiazine which results in the formulation of the hydrogen and methyl radicals. • Procarbazine is used in capsules form and it is active against Hodgkin’s disease and for this, it is used in the MOPP regimen or program. • It has weak MAD inhibiting activity and may cause hypertension.
Procarbazine hydrochloride (MIH, Matulane)
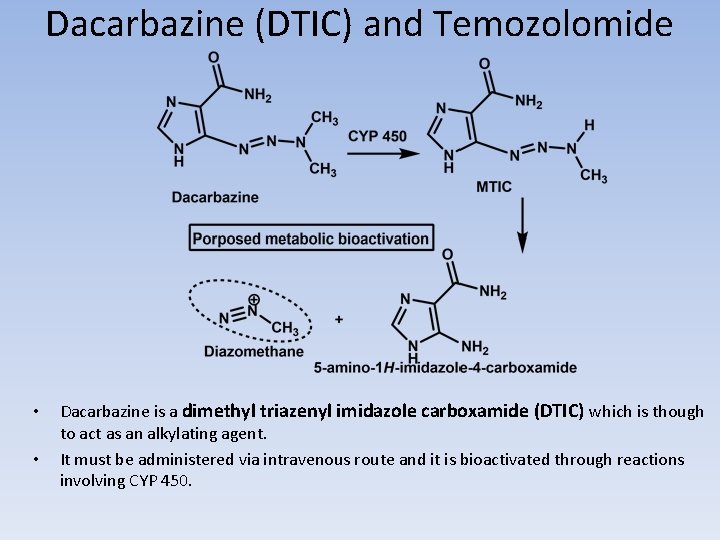
Dacarbazine (DTIC) and Temozolomide • • Dacarbazine is a dimethyl triazenyl imidazole carboxamide (DTIC) which is though to act as an alkylating agent. It must be administered via intravenous route and it is bioactivated through reactions involving CYP 450.

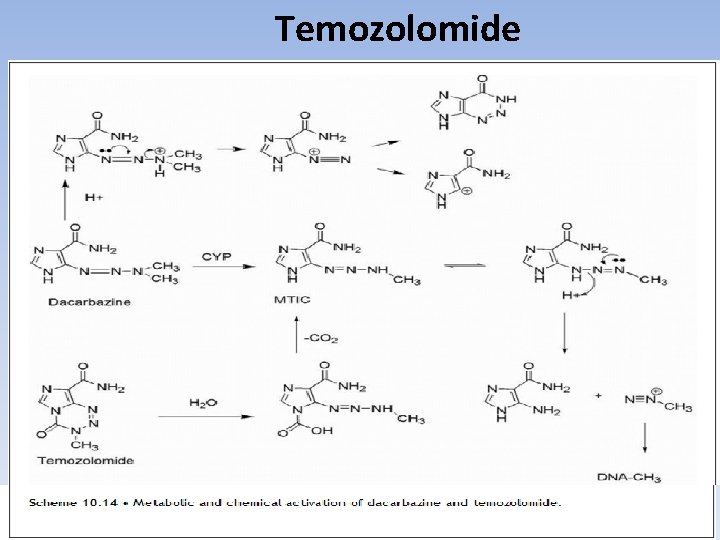
DACARBAZINE • Activation of the agent occurs through the action of CYP (isozymes 1 A 1, 1 A 2, and 2 E 1) to give the demethylated product monomethyl triazeno imidazole carboxamide (MTIC) (Scheme 10. 14). • Tautomerization allows for decomposition to give the aminocarboxamido-imidazole and diazomethane, which is capable of alkylating DNA. An alternative pathway involves acid catalyzed or photoinduced loss of dimethylamine to give an alternative diazo compound (diazo-IC), which may not only generate a carbocation but also undergoes internal cyclization to give 2 -azo-hypoxanthine. • • Formation of diazo-IC has been associated with pain at the injection site, which is often seen during dacarbazine administration. Methylation of DNA occurs at N-7, N-3 and O-6 of guanine among other sites. Dacarbazine proved to be more active against murine tumors than against human tumors. This was attributed to the enhanced ability of mice to metabolize the agent to MTIC and the subsequent conversion to a methylating species.
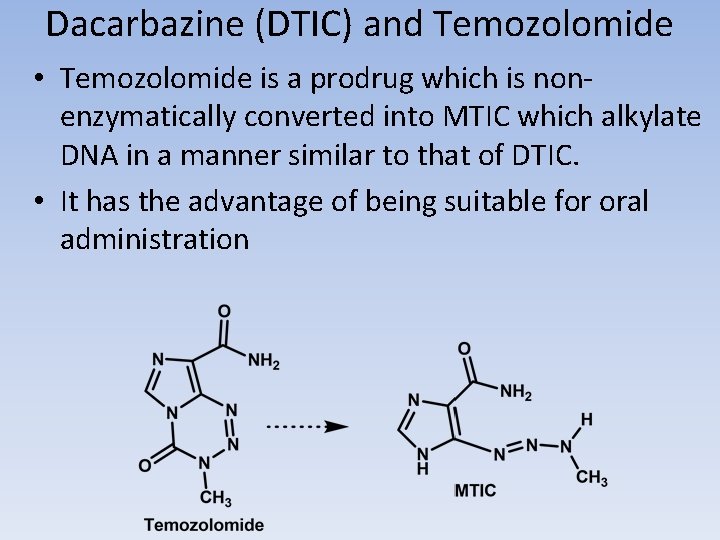
Dacarbazine (DTIC) and Temozolomide • Temozolomide is a prodrug which is nonenzymatically converted into MTIC which alkylate DNA in a manner similar to that of DTIC. • It has the advantage of being suitable for oral administration

Temozolomide • undergoes conversion to the same intermediate, MTIC, as dacarbazine, but it does not require metabolic activation to do so. • Hydrolysis of temozolomide gives the carboxytriazene, which spontaneously loses CO 2 to give MTIC. • Dacarbazine must be administered intravenously; however, the related temozolomide may be administered orally(HW? ? ? )
Temozolomide

Thanks!
- Slides: 66