Antibody structure the necessary minimum 1 Antibody structure

Antibody structure: the necessary minimum. 1

Antibody structure • Antibodies occur in the globulin fraction of serum: hence the name immunoglobulins. • All immunoglobulins have the same basic unit structure: 2 identical heavy polypeptide chains and 2 identical light chains i. e. dimer of heavy & light (H 2 L 2). 2
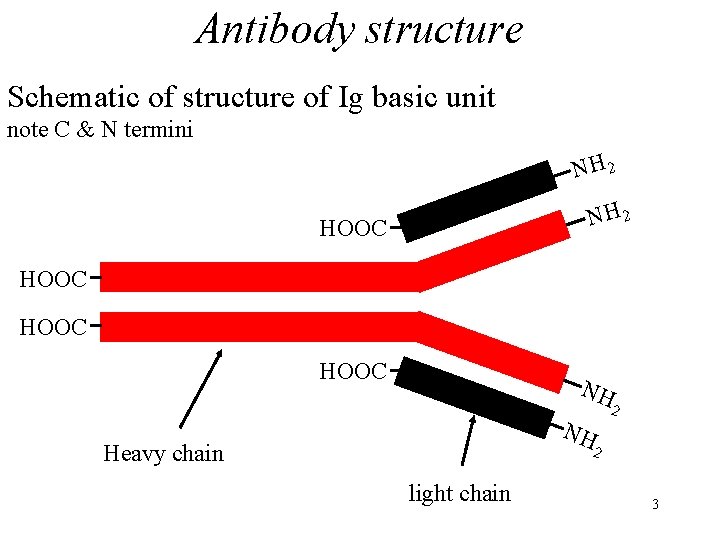
Antibody structure Schematic of structure of Ig basic unit note C & N termini NH 2 HOOC NH NH Heavy chain 2 2 light chain 3

Antibody structure • This dimeric unit has distinct regions with different functions – this was demonstrated by mild proteolytic digestion generating 2 different fragments. 4
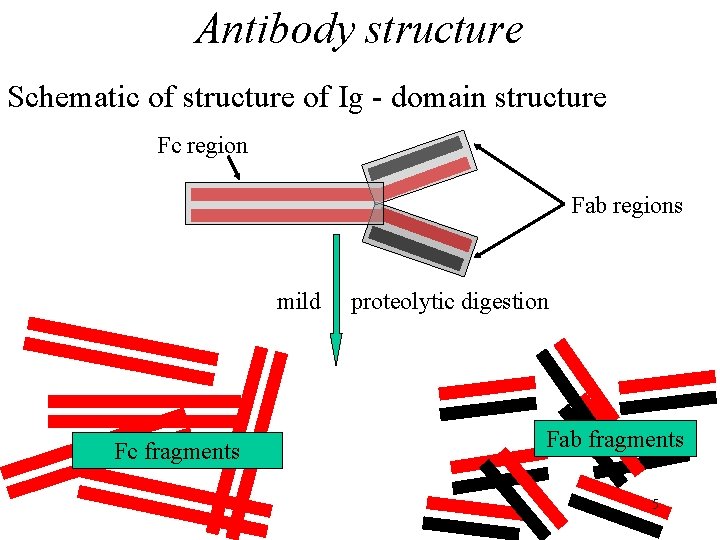
Antibody structure Schematic of structure of Ig - domain structure Fc region Fab regions mild Fc fragments proteolytic digestion Fab fragments 5
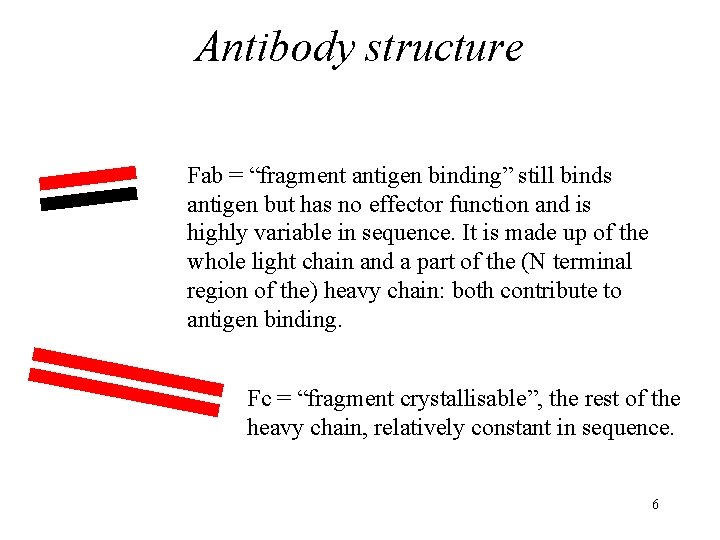
Antibody structure Fab = “fragment antigen binding” still binds antigen but has no effector function and is highly variable in sequence. It is made up of the whole light chain and a part of the (N terminal region of the) heavy chain: both contribute to antigen binding. Fc = “fragment crystallisable”, the rest of the heavy chain, relatively constant in sequence. 6
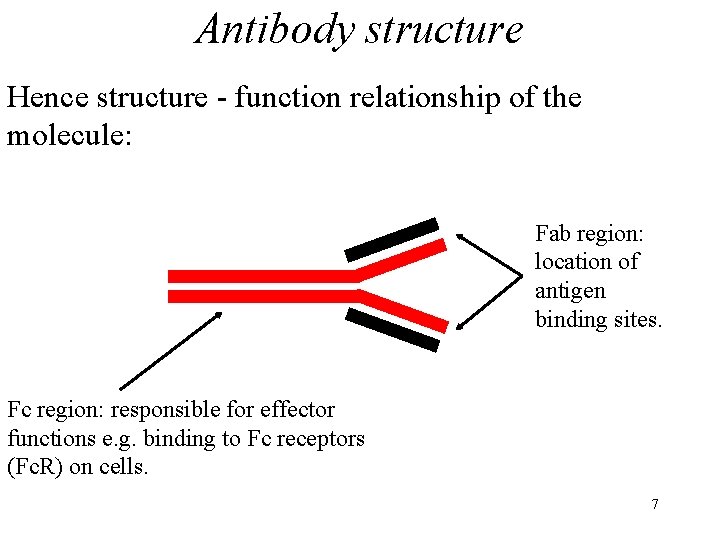
Antibody structure Hence structure - function relationship of the molecule: Fab region: location of antigen binding sites. Fc region: responsible for effector functions e. g. binding to Fc receptors (Fc. R) on cells. 7
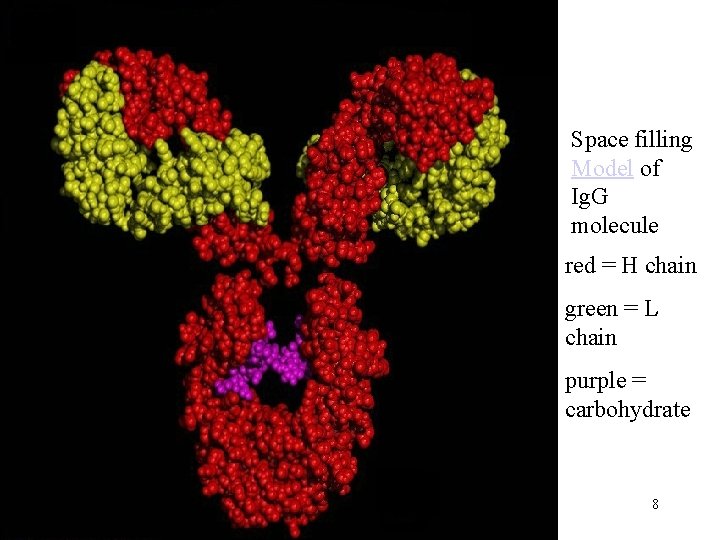
Space filling Model of Ig. G molecule red = H chain green = L chain purple = carbohydrate 8

Antibody structure • Note that the H chain (N terminal region) contributes BOTH to antigen binding AND to effector function • the L chain to antigen binding only. 9
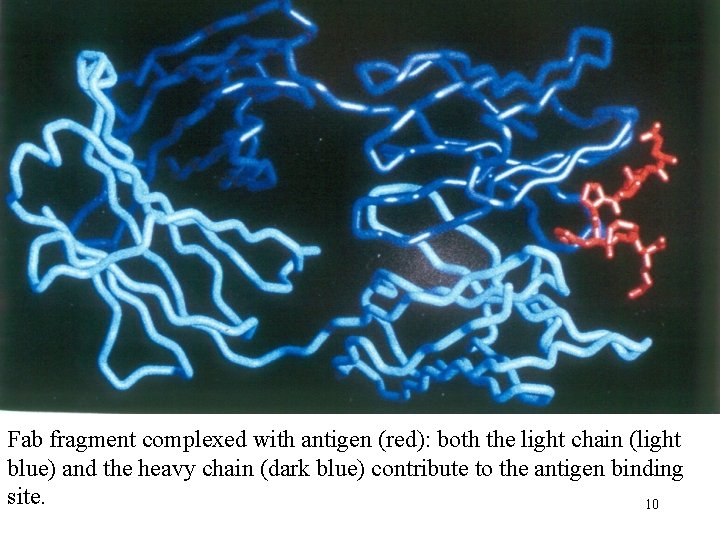
Fab fragment complexed with antigen (red): both the light chain (light blue) and the heavy chain (dark blue) contribute to the antigen binding site. 10
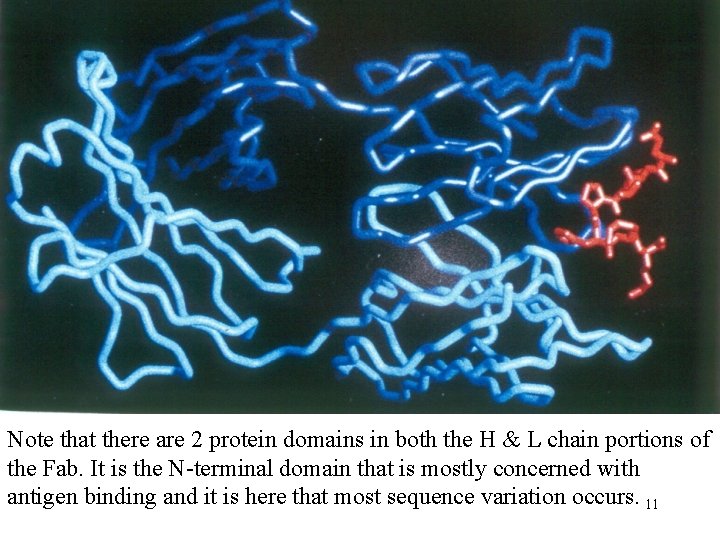
Note that there are 2 protein domains in both the H & L chain portions of the Fab. It is the N-terminal domain that is mostly concerned with antigen binding and it is here that most sequence variation occurs. 11

Antibody structure • In a given B cell (and its descendants - a clone of B cells) only ONE Fab region is expressed. • Both H and L chains in the Fab region are highly variable i. e. the amino acid sequence varies in order to allow binding of a very wide range of antigens – in different B cell clones of course. 12

Antibody structure • During B-lymphopoiesis the mature Ig gene (both H and L) is generated in a random manner so that the final sequence and so specificity of the Ig it encodes can be for any antigen – mechanistic details of how variation is achieved is not necessary. 13

Antibody structure • There are 5 different types of heavy chain – , , ; • these determine the gross molecular structure and effector functions of the different types of antibodies - Ig – M, D, G, E, A; • and 2 light chains which do not have any functional distinctions • . 14

Antibody structure • The Fc region is constant for a given Ig type because it is responsible for fixed effector functions and the gross molecular structure. 15

Antibody structure • Ig. M is a pentamer of the basic unit; • Ig. D, Ig. G and Ig. E are all basic unit structures; • Ig. A is a dimer of the basic unit with an additional polypeptide unit - the secretory component - which facilitates its secretion across an epithelial surface. 16

Antibody structure • In a given B cell the same Fab region of the H chain is successively associated with different Fc regions as the B cell proliferates and differentiates after activation • so an activated proliferating B cell will express Ig. M then G, E or A – again mechanistic details unnecessary. • The same L chain is always expressed. 17

Antibody structure • Go back to main lectures for an account of the immunological functions of immunoglobulins. 18
- Slides: 18