ANTIBODY IMMUNOGLOBULIN Antibody structure 1 Antibodies belong to

ANTIBODY/ IMMUNOGLOBULIN

Antibody structure 1. Antibodies belong to a class of proteins (glycoprotein) called immunoglobulins 2. Antibody molecules belong to one of five classes i. e. Ig. G, Ig. M, Ig. A, Ig. D & Ig. E 3. Immunoglobulins are “Y” shaped proteins. The “arms” of the “Y” bind antigens. The tail of the “Y” is responsible for biological activity eg. C’ activity or binding to cells 4. Ability of immunoglobulins to bind antigen determined by AA sequence in variable region

Carboxy End N Terminal end Antibody Structure

Antibody Structure • Antibodies Are Made Up Of: – 2 Light Chains (identical/Shorter) ~25 Kda (214 AA) – 2 Heavy Chains (identical) ~50 Kda • Ig Consist of 2 region 1. Constant region 2. Variable region • Each Light Chain Bound To Heavy Chain By Disulfide (H-L) • Based on the Structure of AA sequence Lights chains are Grouped in to 2 Types – • Kappa (K)/ L-type • Lambda (ʎ ) • Approximately 60% Ig are K type • , , 40 % L type
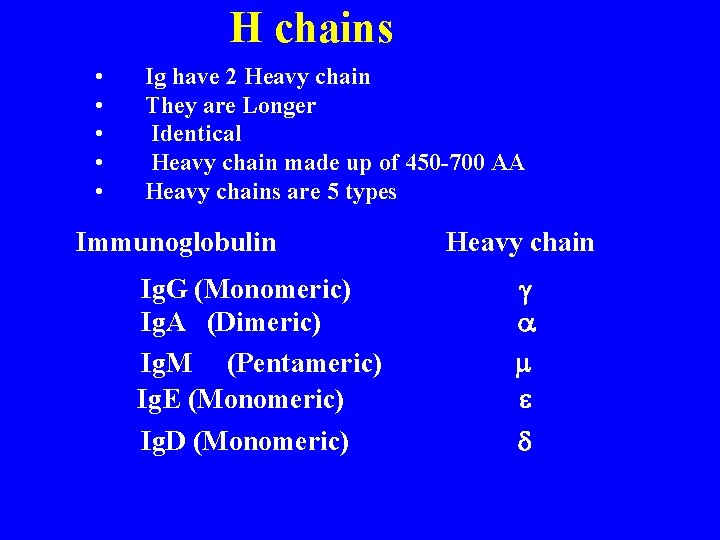
H chains • • • Ig have 2 Heavy chain They are Longer Identical Heavy chain made up of 450 -700 AA Heavy chains are 5 types Immunoglobulin Heavy chain Ig. G (Monomeric) Ig. A (Dimeric) Ig. M (Pentameric) Ig. E (Monomeric) g a m e Ig. D (Monomeric) d
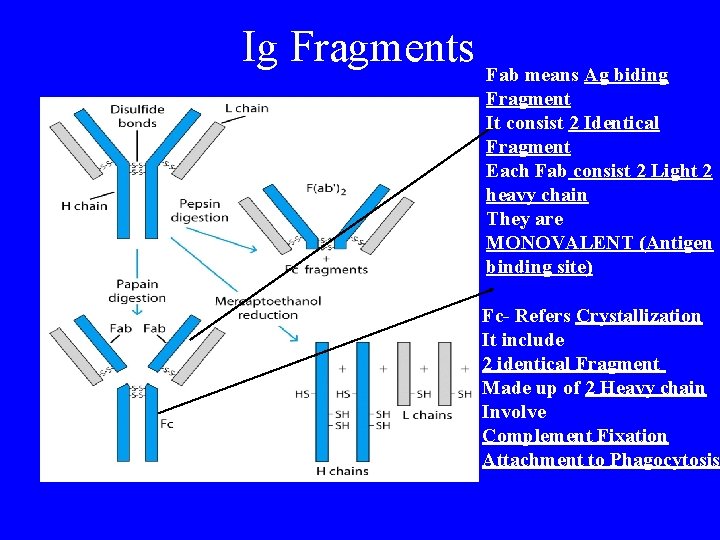
Ig Fragments Fab means Ag biding Fragment It consist 2 Identical Fragment Each Fab consist 2 Light 2 heavy chain They are MONOVALENT (Antigen binding site) Fc- Refers Crystallization It include 2 identical Fragment Made up of 2 Heavy chain Involve Complement Fixation Attachment to Phagocytosis

Functions of Ig • Agglutination of Ag ( Process of clumping of Particulate Ag With AB) • Precipitation of Ag (Ig combines with soluble antigenic agent) • Neutralization of Ag (Ag cover the toxin sites of the antigenic agent and thus NUTRALIZE them ( INACTIVE) • Lysis of Ag (Rupture the cells) • Opsonization ( Process of COATING the BACTERIA with Ab and making SUSPECTIBLE for PHAGOCYTOSIS. • • Activation of Mast cells and basophils Tissue Fixation (Attach to the cells)

Ig G (Monomeric) (g) • Monomer • Made up of 2 light chain 2 heavy chain (g) • Most abundant immunoglobin 80% of serum Ig ~10 mg/m. L • Again classified in to Ig. G 1, 2, 3, 4 • Ig. G 1, Ig. G 3 and Ig. G 4 cross placenta in human • Ig. G 3 Most effective complement activator • Responsible for protection of the newborn during the first mont of life. Because of its relative abundance and excellent specificity toward antigens Properties of Ig. G • Molecular weight: 150, 000 • H-chain type (MW): gamma (53, 000) • Percent of total immunoglobulin: 80% • Half life 25 days • Function: secondary response
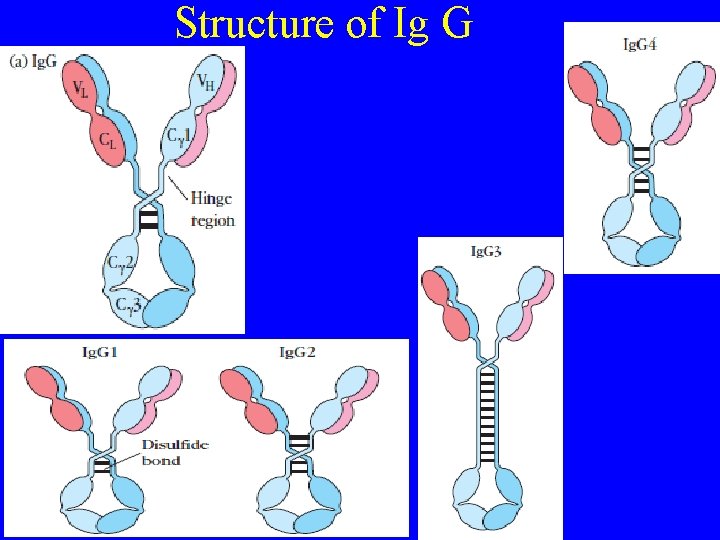
Structure of Ig G

Ig A • Glycoprotein • Dimer • It have 4 Polypeptide chain (2 Light chain (K and lambda) and 2 Heavy chain (Alpha) ) • M. W 160, 000 H-chain type (MW): alpha (55, 000) • Normal serum level 0. 6 -4. 2 mg/ml • Half life 6 -8 days • Predominant Ig in secretions Milk, saliva, tears, mucus • Important role in Mucosal Activity • 3 -5 g of Ig A are secreted in to the intestinal every day • This accumulate 75% of the total immunoglobulin produced in the entire body • Ig A subclass are Ig A 1, Ig A 2

Function • Mucosal paint/ antiseptic paint • Neutralize –local toxin----Promote phagocytosis • Immunity against Tape Worms • Present in the CLOSTRUM protects the baby from intestinal pathogen.
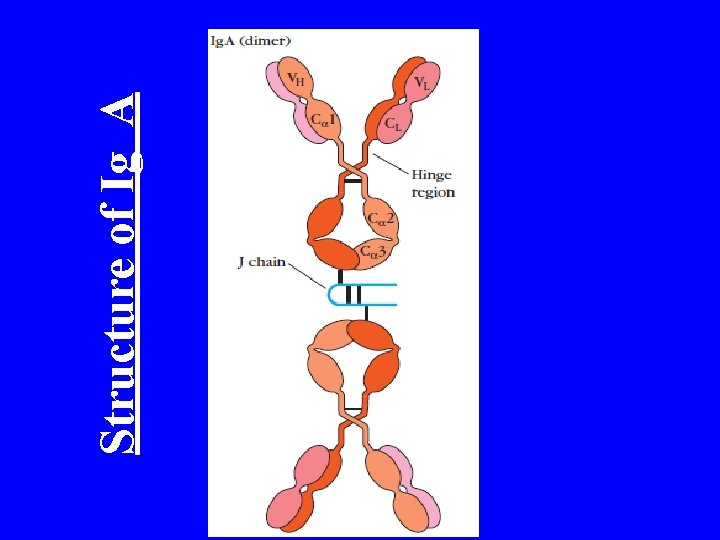
Structure of Ig A

Ig. M • Largest Immunoglobulin • Pentamer Structure ( 5) • Made up of 2 light (Kappa, lambda) and 2 Heavy chain (Mu) • Normally this called Natural Antibody • Multiple antigen binding site • 3 rd highest serum Ig • Produced early in the primary response • It is activate complement pathways • Half life 5 days • Molecular weight: 900, 000 --- mu (65, 000) • H-chain type (MW): mu (65, 000) • Percent of total immunoglobulin: 5 - 10%

Structure of Ig M

Ig E • Monomer • Made up of 2 light (K and Lamda) and 2 heavy chain (Epsilon) • This is the only Ig without hinge Region • M. W 190, 000 epsilon (73, 000) • Heat liable Ig, where as Ig G, Ig. A, Ig. M and Ig. D Heat stable • Its found only trace amount in the serum (0. 2% of total Ig) • This induce Allergic Reaction • Half life 2 -3 days
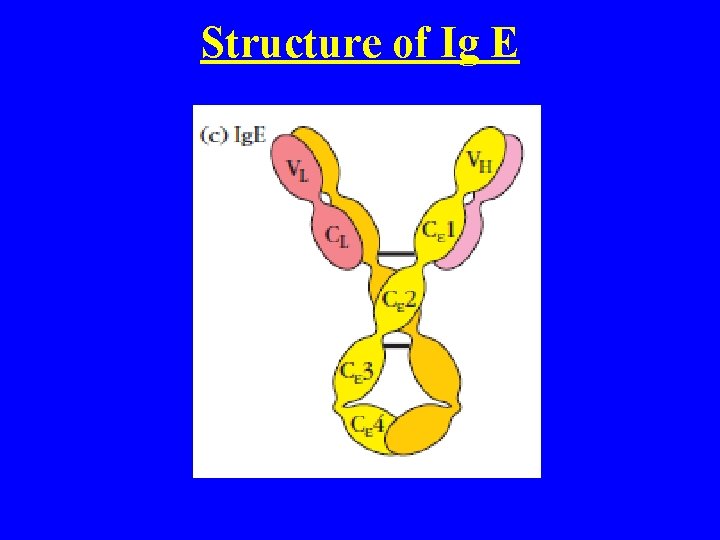
Structure of Ig E

Ig D • It’s a Monomer (Glycoprotein) • Found in Blood serum and Surface of B cell • Made up of 2 Light chain (K and Lamda) and 2 Heavy chain ( Delta) • It s a primitive (ancient) Ab found in fishes to mammals except Birds • Ig D slightly larger than Ig G --- M. W--- 180, 000 (delta (70, 000) • Ig D limited to the Blood serum Average serum level -0. 03 mg/ml • Half life 2 -3 days • Ig D binds to Basophils and mast cells and activates them to secrete ANTIMICEROBIAL Factors
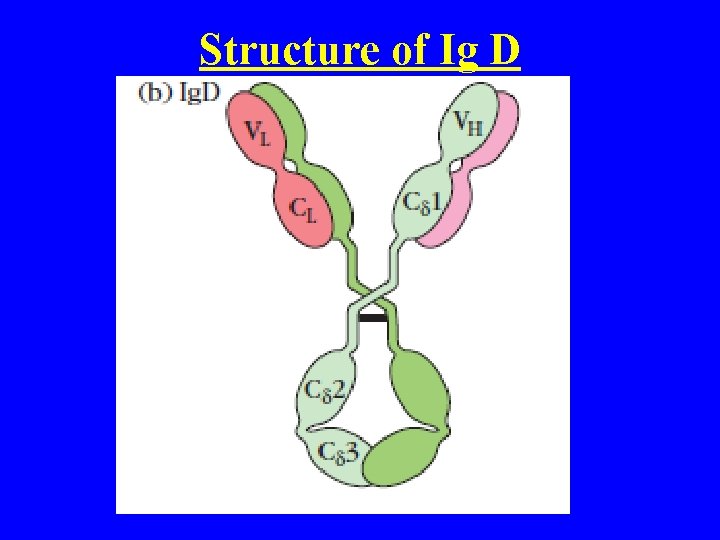
Structure of Ig D

Serum and plasma Serum Blood Plasma Blood + anticoagulant Cells Clot

Isolation & characterization Chromatography Molecular sieving Ion exhange Affinity Salting out/ Dehydration Ammonium sulfate Alcohol precipitation Ultracentrifugation Immunochemical
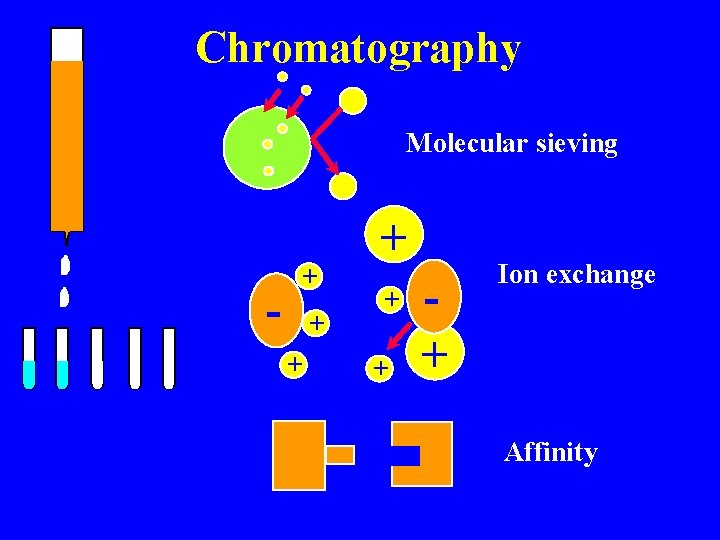
Chromatography Molecular sieving - + + + + Ion exchange Affinity
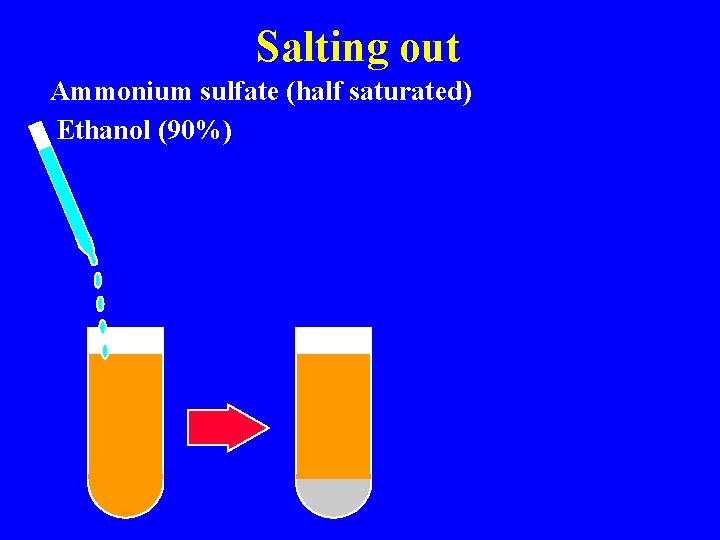
Salting out Ammonium sulfate (half saturated) Ethanol (90%)
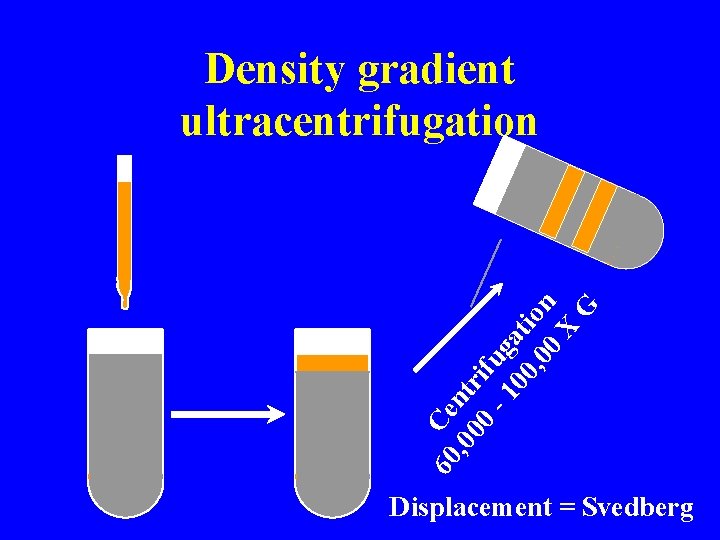
60 Ce , 0 ntr 00 if - 1 ug 00 ati , 0 on 0 X G Density gradient ultracentrifugation Displacement = Svedberg
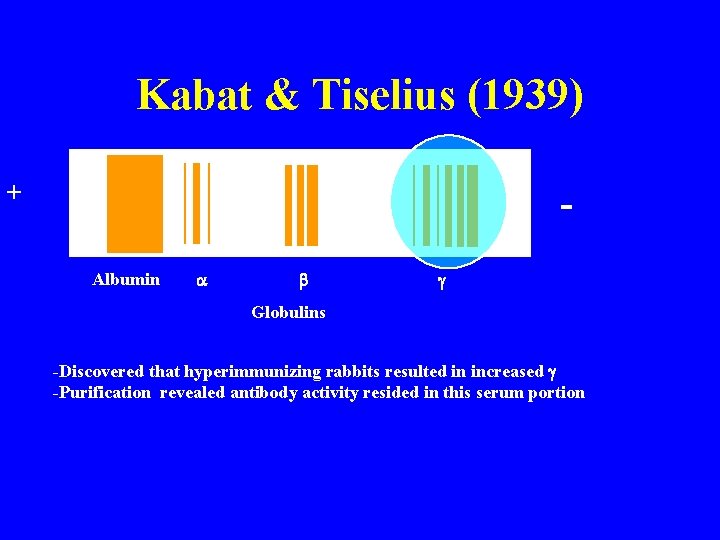
Kabat & Tiselius (1939) + Albumin a b g Globulins -Discovered that hyperimmunizing rabbits resulted in increased g -Purification revealed antibody activity resided in this serum portion
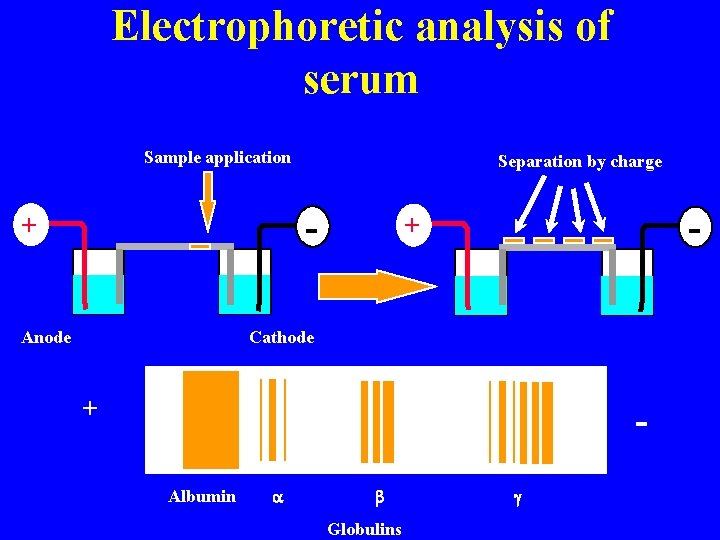
Electrophoretic analysis of serum Sample application Separation by charge - + Anode - + Cathode + Albumin a b Globulins g
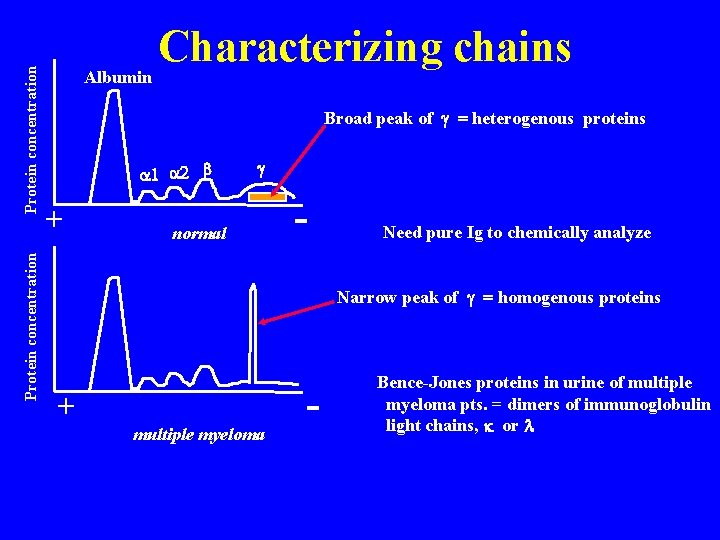
Protein concentration Characterizing chains Albumin Broad peak of g = heterogenous proteins a 1 a 2 b + g normal - Need pure Ig to chemically analyze Narrow peak of g = homogenous proteins + multiple myeloma - Bence-Jones proteins in urine of multiple myeloma pts. = dimers of immunoglobulin light chains, k or l

Porter (England) Treatment with proteolytic enzyme, papain, resulted in three approximately equal sized fragments: 2 capable of ag rx (fragment antigen binding) 1 could be crystallized (fragment- crystallizable) Fab SS Fab specifically bind antigen, univalent (can’t ppt) one binding site each, identical to each other SS SS Fc Fc crystallizable (thus homogenous) can’t bind ag responsible for biological activity of molecule after ag bound to Fab portions

Eddleman (USA) Treatment with mercaptoethanol = 4 chains: (mercaptoethanol breaks S-S bonds) SH HS 2 chains = 53, 000 daltons 2 chains = 22, 000 daltons All immunoglobulins basic unit consisting of 4 polypeptides i. e. 2 H, 2 L
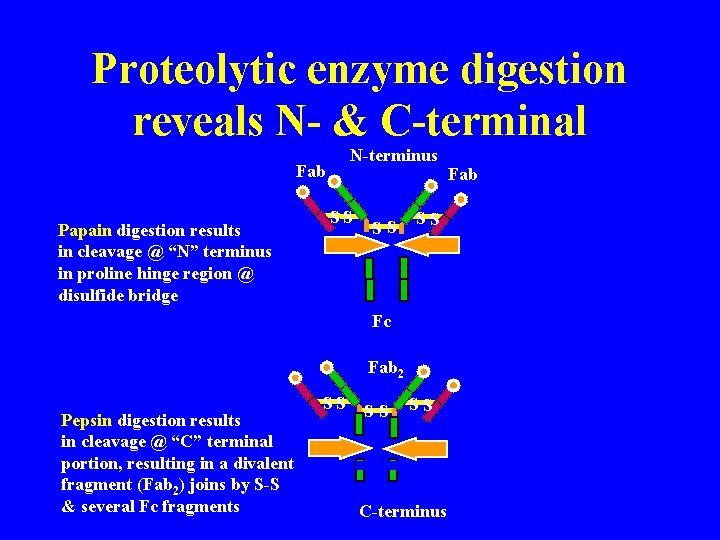
Proteolytic enzyme digestion reveals N- & C-terminal N-terminus Fab Papain digestion results in cleavage @ “N” terminus in proline hinge region @ disulfide bridge SS SS SS Fc Fab 2 Pepsin digestion results in cleavage @ “C” terminal portion, resulting in a divalent fragment (Fab 2) joins by S-S & several Fc fragments SS SS SS C-terminus Fab
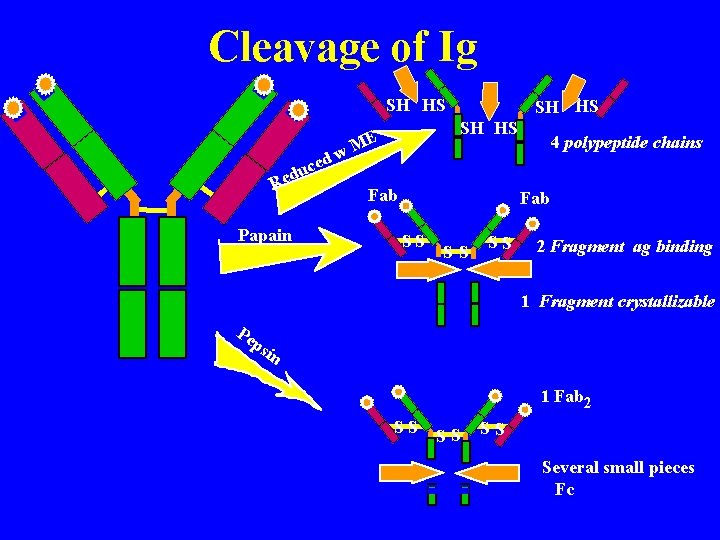
Cleavage of Ig SH HS dw ce u d Re Papain SH HS ME Fab 4 polypeptide chains Fab SS SS SS 2 Fragment ag binding 1 Fragment crystallizable Pe ps in 1 Fab 2 SS SS SS Several small pieces Fc

Myeloma cells Plasma cell becomes tumorous = myeloma produces homogenous (monoclonal) Ig Myeloma Ig = myeloma “proteins” Myeloma proteins may be purified & structurally analyzed AA sequence in L & H chains Normal, non immune Normal, immune Myeloid myeloma
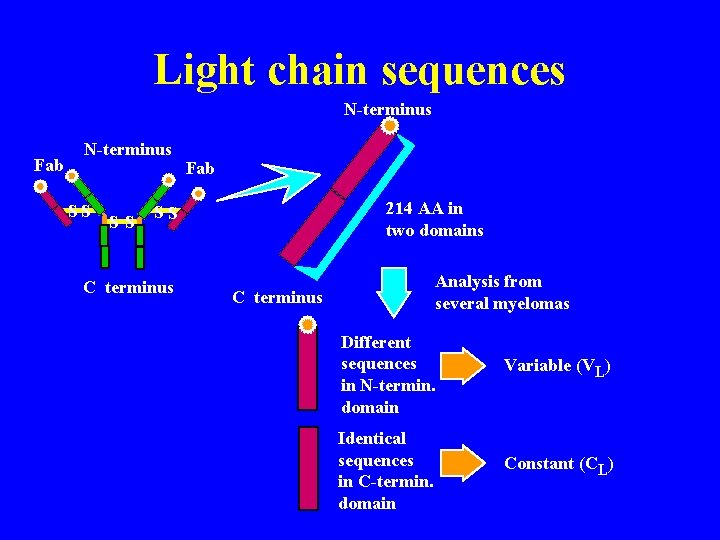
Light chain sequences N-terminus Fab N-terminus SS SS Fab 214 AA in two domains SS C terminus Analysis from several myelomas C terminus Different sequences in N-termin. domain Variable (VL) Identical sequences in C-termin. domain Constant (CL)

Heavy-chain sequence N-terminal domain varies (HV) Other 3 domains constant (HC) Flexible Hinge Region Fab N-terminus SS SS SS C terminus Fab Heavy chain consists of 445 AA in 4 domains
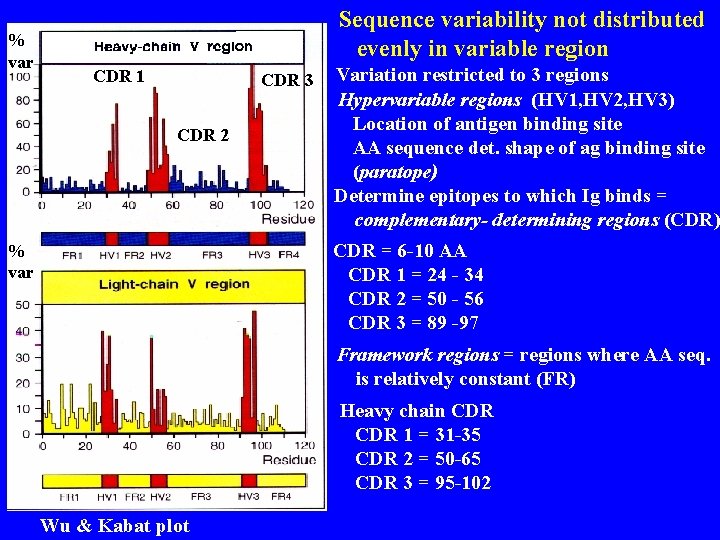
% var Sequence variability not distributed evenly in variable region CDR 1 CDR 3 CDR 2 Variation restricted to 3 regions Hypervariable regions (HV 1, HV 2, HV 3) Location of antigen binding site AA sequence det. shape of ag binding site (paratope) Determine epitopes to which Ig binds = complementary- determining regions (CDR) CDR = 6 -10 AA CDR 1 = 24 - 34 CDR 2 = 50 - 56 CDR 3 = 89 -97 % var Framework regions = regions where AA seq. is relatively constant (FR) Heavy chain CDR 1 = 31 -35 CDR 2 = 50 -65 CDR 3 = 95 -102 Wu & Kabat plot

Structure of variable & constant domains (X-ray crystallography) Light chain C domain Two layers, linked by disulfide bonds Light chain V domain N terminus CDR Disulfide bonds Each layer formed several stretches 3 conformation = b strand Layers = b sheet CDR 1 CDR 2 Order of b strands is characteristic for each sheet 3 D structure = Ig fold C terminus B strands
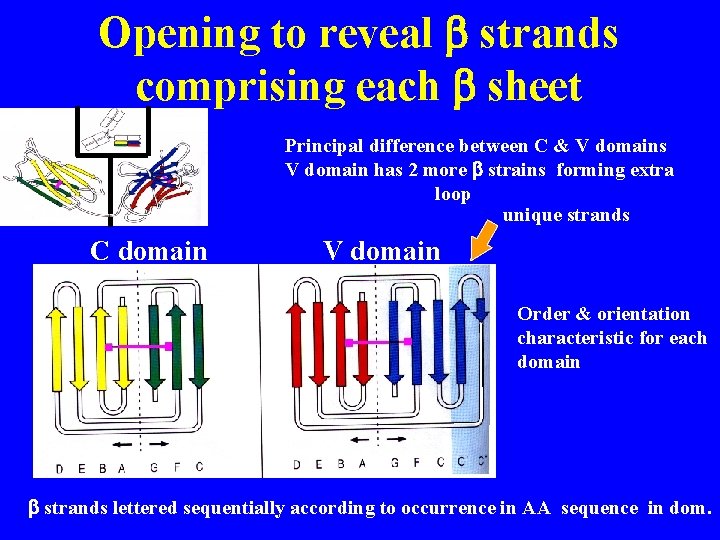
Opening to reveal b strands comprising each b sheet Principal difference between C & V domains V domain has 2 more b strains forming extra loop unique strands C domain V domain Order & orientation characteristic for each domain b strands lettered sequentially according to occurrence in AA sequence in dom.
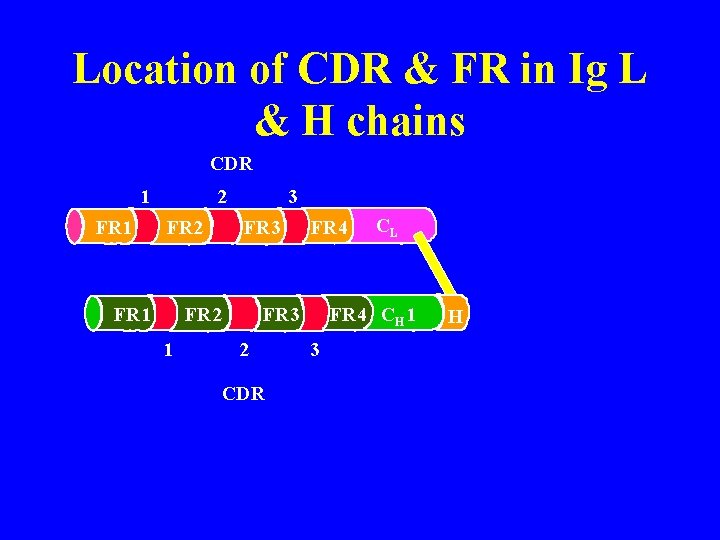
Location of CDR & FR in Ig L & H chains CDR 1 FR 1 2 FR 1 3 FR 2 1 FR 4 CH 1 FR 3 2 CDR CL 3 H
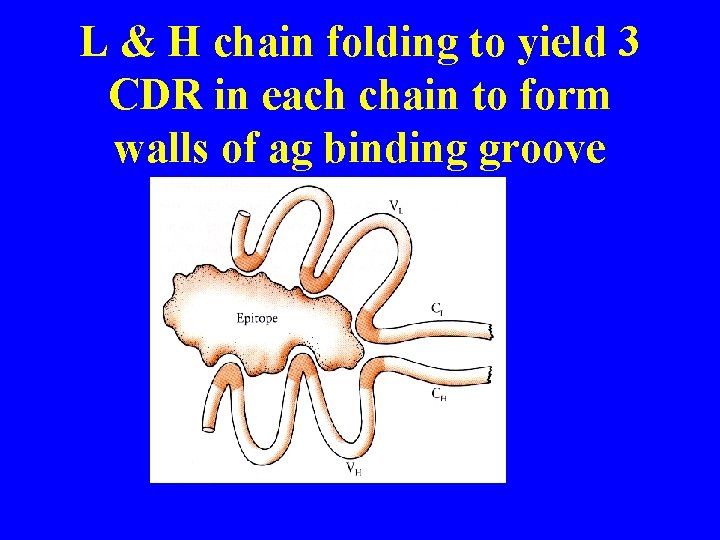
L & H chain folding to yield 3 CDR in each chain to form walls of ag binding groove
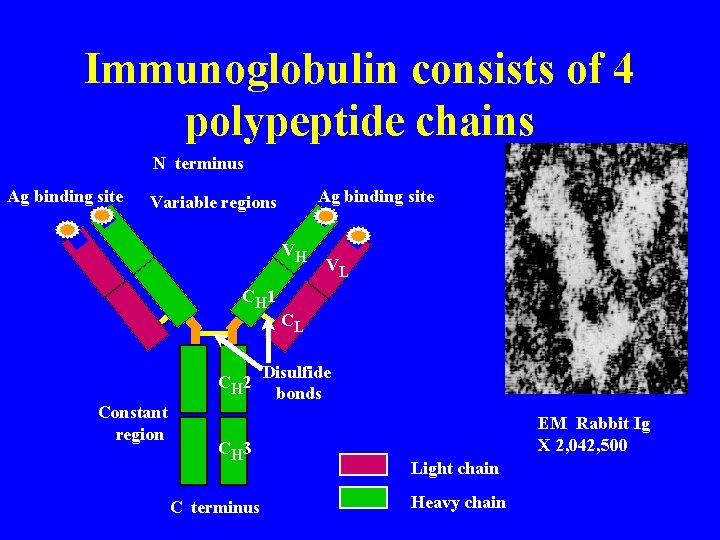
Immunoglobulin consists of 4 polypeptide chains N terminus Ag binding site Variable regions VH CH 1 CH 2 Constant region CH 3 C terminus VL CL Disulfide bonds EM Rabbit Ig X 2, 042, 500 Light chain Heavy chain

Immunologic analysis of immunoglobulins Ig (like most other proteins) stimulate ab in other animal species All species have two major classes of L chains i. e. k, l individual of species produces both types; ratio of k : l varies by species (e. g. mouse 95%k ; human 60% k) in any Ig molecule, both L chains = either k or l, never one of each Ig of all species consist of 5 classes (isotypes) differ in structure of H chains among isotypes differ serologically, CHO content, size & biological function

Isotypic (Class) structure of Ig L H L H Ig. G isotype = k 2 g 2 or l 2 g 2, but not k 1 l 1 g 1 a 1 Ig. E isotype = k 2 e 2 or l 2 e 2 …. etc…. . .

H chains H chain confers unique biologic properties of molecule e. g. 1/2 life, receptor binding , enzyme-, C’ activation with ag Immunoglobulin isotype Ig. M Ig. G Ig. A Ig. D Ig. E Heavy chain m g a d e Individual of species produces all H chains, in proportion characteristic for species, but ab molecule H chains are identical ( i. e. , 2 e, 2 d etc. )

Immunoglobulin consists of 4 polypeptide chains N terminus Ag binding site Variable regions VH CH 1 CH 2 Constant region CH 3 C terminus VL CL Disulfide bonds EM Rabbit Ig X 2, 042, 500 Light chain Heavy chain

Isotypic (Class) structure of Ig L H L H Ig. G isotype = k 2 g 2 or l 2 g 2, but not k 1 l 1 g 1 a 1 Ig. E isotype = k 2 e 2 or l 2 e 2 …. etc…. . .

Hinge region Consists of approx 12 AA between CH 1 & CH 2 No homology between hinge & other Heavy chain domains Angle = 900 0 AA sequence unique for each class Angle = 60 & subclass Angle = 0 o

AA sequence in hinge region Hinge region Light Chain Cys -Arg-Val-Glu-Pro-Lys-Ser-Cys-Asp-Lys-Thr-His-Thr-Cys-Pro-Pro-Cys -Pro-Ala-Pro-Glu. Cys Light Chain Papain Fab region Fc region

Characteristics of hinge region Immunoglobulins (with possible exception of Ig. M & Ig. E) contain hinge between CH 1 & CH 2 No homology between AA sequence of hinge & heavy chains AA sequence differs with different classes Comprised of many cysteine and proline residues Cysteine involved in formation of interchain disulfide bonds Proline prevents folding in a globular structure, allowing flexibility between two Fab arms of the Y-shaped antibody; allows open & close to accommodate binding to two epitopes; because it is open, it can be cleaved by proteases (e. g. papain) to generate the Fab & Fc fragments
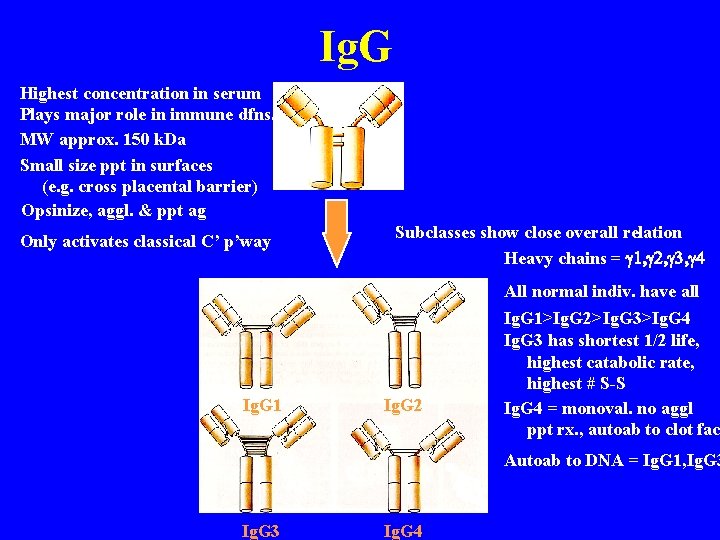
Ig. G Highest concentration in serum Plays major role in immune dfns. MW approx. 150 k. Da Small size ppt in surfaces (e. g. cross placental barrier) Opsinize, aggl. & ppt ag Only activates classical C’ p’way Ig. G 1 Subclasses show close overall relation Heavy chains = g 1, g 2, g 3, g 4 Ig. G 2 All normal indiv. have all Ig. G 1>Ig. G 2>Ig. G 3>Ig. G 4 Ig. G 3 has shortest 1/2 life, highest catabolic rate, highest # S-S Ig. G 4 = monoval. no aggl ppt rx. , autoab to clot fac Autoab to DNA = Ig. G 1, Ig. G 3 Ig. G 4
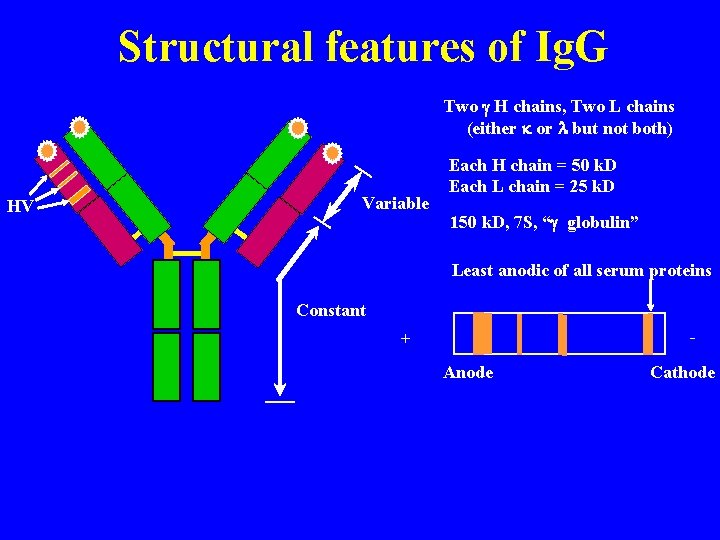
Structural features of Ig. G Two g H chains, Two L chains (either k or l but not both) HV Variable Each H chain = 50 k. D Each L chain = 25 k. D 150 k. D, 7 S, “g globulin” Least anodic of all serum proteins Constant - + Anode Cathode

Structural features of Ig. M SS SS Pentamer (5) SS First Ig produced following immun. Macroglobulin (M) SS J chain S S SS SS SS 900 k. D, 19 S Doesn’t have hinge region has additional H domain Has a J chain (one of 2 Ig isotypes) 15 k. D SS
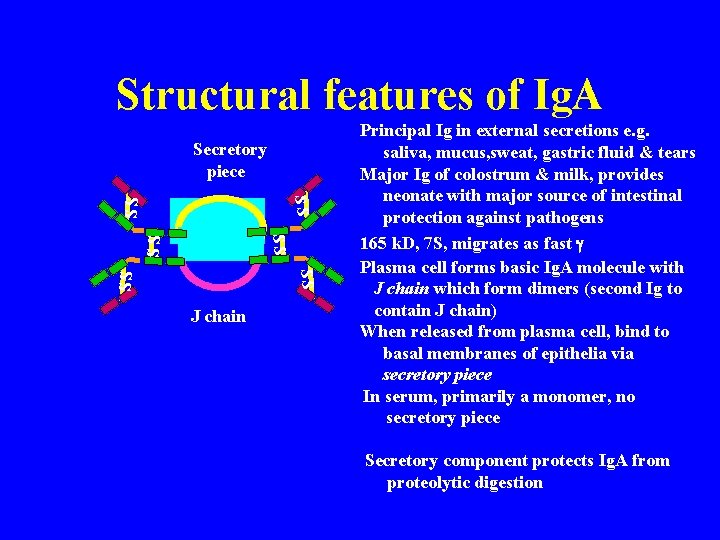
Structural features of Ig. A Secretory piece SS SS SS J chain Principal Ig in external secretions e. g. saliva, mucus, sweat, gastric fluid & tears Major Ig of colostrum & milk, provides neonate with major source of intestinal protection against pathogens 165 k. D, 7 S, migrates as fast g Plasma cell forms basic Ig. A molecule with J chain which form dimers (second Ig to contain J chain) When released from plasma cell, bind to basal membranes of epithelia via secretory piece In serum, primarily a monomer, no secretory piece Secretory component protects Ig. A from proteolytic digestion

Structural features of Ig. D Primarily a B cell antigen receptor Long exposed hinge region 170 k. D, 7 S, migrates as fast g No interchain S-S bridges in H chains Readily denatured

Structural features of Ig. E Sometimes called reaginic ag Mediates allergies (Type I hypersensitivies) 190 k. Da, 8 S, migrates as fast g Contains an extra domain (CH 4) which binds to mast cells & basophils May remain attached for long time when ag reappears, cross links Ig. E on mast cell surface, release mast-cell granules & signs of anaphylaxis
Immunoglobulin variations Isotype = class of Ig, all people have Allotype = genetic difference, depends on existence of allelic forms of the Ig , result of different forms of the same gene, involve changes of 1 -3 AA in constant region; known allotypes Ig. G: Gm (Ig Markers) Km (markers on k chain Idiotype = one of several thousand Ig, each of which directed toward specific epitope
- Slides: 54