ANTIBODY Babitha Elias DEFINITION Antibodies are substances which

ANTIBODY Babitha Elias

DEFINITION Antibodies are substances which are formed in the serum or tissue fluids in response to an antigen. n Antibodies react with antigen specifically and in an observable manner. Immunoglobulin – proteins of animal origin, endowed with known Ab activity & for certain other proteins related to them by chemical structure.

Properties of antibodies n Chemical nature of antibodies is globulin – immunoglobulin n Constitute about 20 – 25% total serum proteins. n Based on sedimentation studies – 7 S (M. W – 1, 50, 000 - 1, 80, 000 KD). Heavier antibodies – 19 S globulins (M. W – 900, 000) n Kabat showed, on electrophoretic mobility, the antibodies belong to gammaglobulins


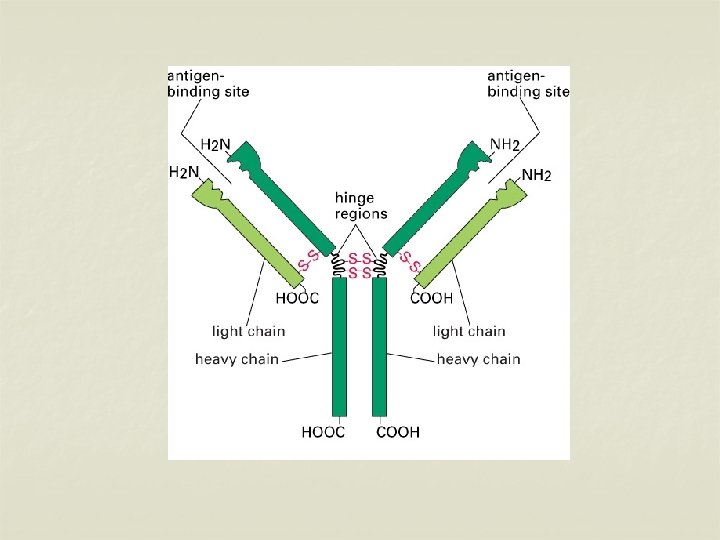
Structure of immunoglobulins n Porter, Edelman and Nisonoff n Antibody molecule consists of two identical heavy chains (H) and two identical light chains (L). n Heavy chains are longer & light chains are shorter. n Both chains are polypeptide in nature. n 2 heavy chains are held together by disulphide bonds. n Each light chain is attached to a heavy chain by disulphide bond.
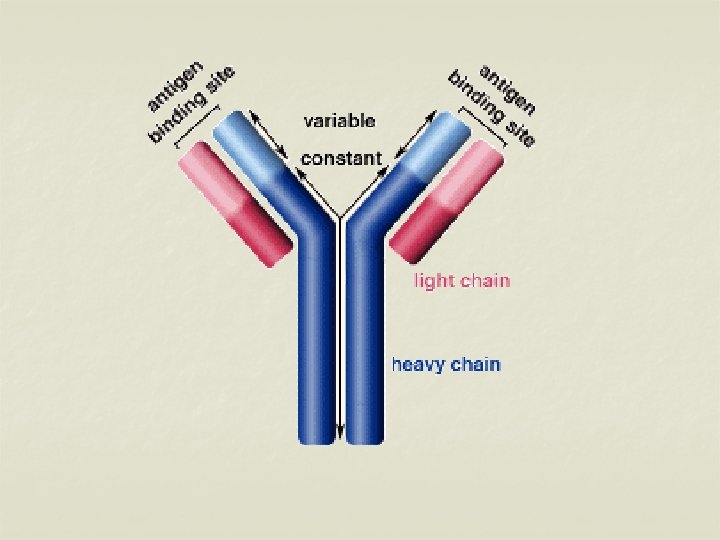
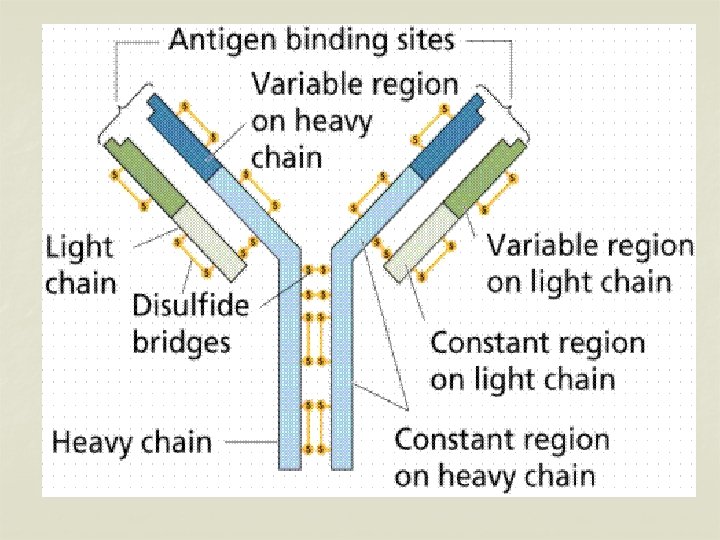

n n The H chains are structurally and antigenically distinct in different classes of immunoglobulins. 5 different classes of immunoglobulins depending on the presence of heavy chain. Ig G – γ n Ig M - μ n Ig A - α n Ig E - ε n Ig D - δ n

n n The L chains are similar in all classes of immunoglobulins. Present in two forms – kappa (K) and lambda (L). Effect of enzymes on Immunoglobulins Papain Digestion n Papain can digest immunoglobulin molecule into 3 fragments – one Fc (Fragment crystallisable) and 2 identical Fab (fragment antigen binding) n The 2 Fab fragments possess the antigen binding sites but the Fc fragment lacks the ability to bind antigen.
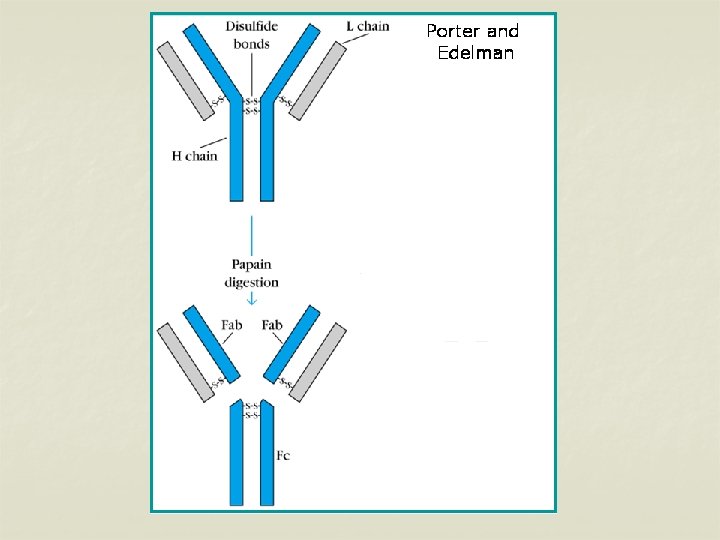

n n Each Fab fragment consists of a light (L) chain & a part of H chain Portion of H chain in Fab fragment – Fd region Fc fragment consists of both the H chains. It determines the biological properties of the Ig – complement fixation, placental transfer etc.

Pepsin digestion n Pepsin cleaves immunoglobulin into 1 Fc portion & 2 Fab fragments held together in position. n The Fab is bivalent & still it can precipitate with antigen – F(ab’)2 n Pepsin degrades the Fc portion into smaller fragments.
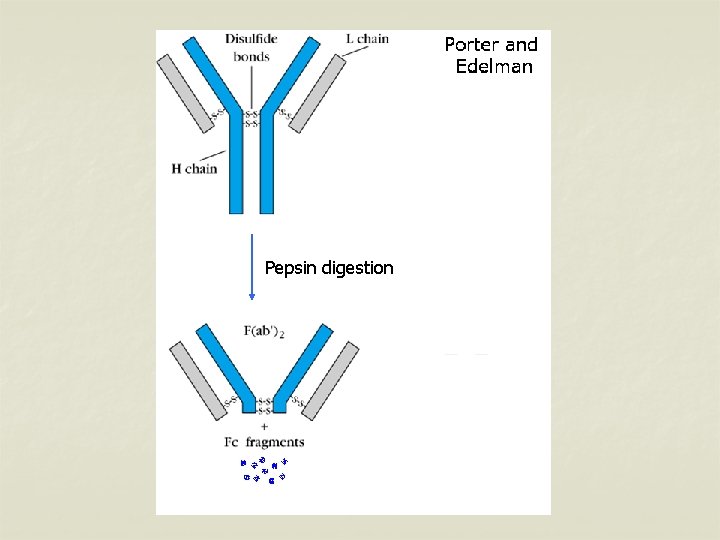
Pepsin digestion

H chains & L chains n Consists of two portions – a variable region (V) & a Constant region (C). n In the L chain, the 2 regions are of equal length, while in the H chain, the V region constitute only a fifth of the chain. n Variable regions are in the amino terminus (NH 2) and Constant region at carboxyterminus (COOH)

IMMUNOGLOBULIN CLASSES IMMUNOGLOBULIN G (Ig G) n n n Major serum immunoglobulin – 80% of total amount. The normal serum concentration – 8 -16 mg/ml. Molecular weight – 150, 000 (7 S) Half life – 23 days It can cross the placenta – Transplacental

n n It is distributed equally between the intravascular & extravascular compartments. Ig G appears late but persists long. It appears after the initial immune response. It participates in precipitation, complement fixation, & neutralization of toxin & viruses. Ig G binds to microorganisms & enhances phagocytosis.

n Catabolism of Ig G – n n n When Ig G level is raised, the synthesis of Ig G against that particular Ag is catabolised rapidly & result in deficiency of particular Ab. Eg: myeloma & kala-azar. In hypogammaglobulinaemia, Ig G antibody given for therapeutic purpose will be catabolised slowly. Passively administered Ig G suppresses the homologous antibody synthesis by feed back mechanism. 4 sub classes of Ig G – Ig G 1, Ig G 2, Ig G 3, Ig G 4 It is protective against microorganisms which are active in the blood and tissues.

IMMUNOGLOBULIN A (Ig A) n n n Second major serum immunoglobulin – 10 - 13%. Normal serum concentration – 0. 6 -4. 2 mg/ml. Half life – 6 – 8 days. Ig A occurs in two forms – Serum Ig A & Secretory Ig A. Serum Ig A is a monomeric 7 S molecule. (MW: 160, 000), While Ig A in the mucosal surfaces & secretions (Secretory Ig A, MW: 400, 000) is a dimer. It is formed by two monomer units joined together by a glycoprotein – J chain
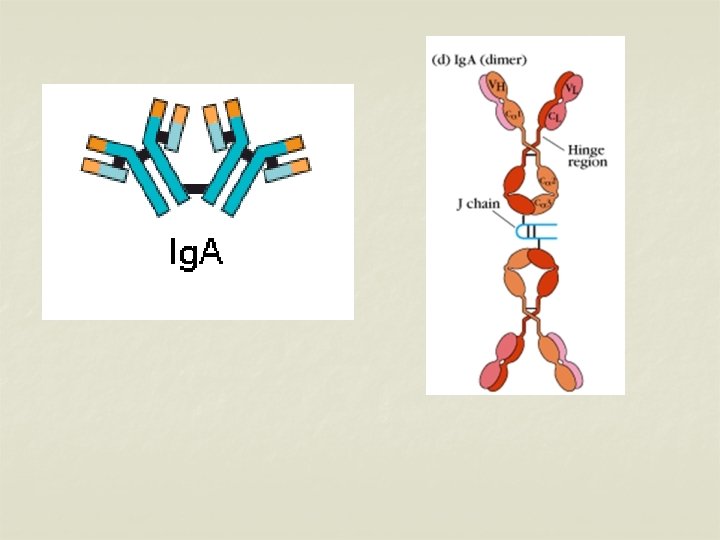

n n n J chain – joins the 2 monomers at their carboxy terminals. Secretory Ig A & J chain are produced by the plasma cells. Secretory Ig A contains a secretory piece, S piece is synthesised by mucosal & glandular epithelial cells. The S piece protects Ig A from denaturation by bacterial proteases in sites such as intestinal mucosa. Ig A is present in secretions such as milk, saliva, tears, sweat, nasal fluids etc. It protects the mucus membranes against microorganisms. .

n n n Ig A cannot fix complement, but can activate alternative complement pathway. Ig A is synthesised locally by plasma cells. 2 subclasses of Ig A – Ig A 1 & Ig A 2

IMMUNOGLOBULIN M (Ig M) n Ig M is a pentamer consisting of 5 monomers joined by a J chain. n It constitutes about 5 -8% of total serum concentration. n Normal level – 0. 5 -2 mg/ml n Half life – 5 days. n Molecular weight – 19 S (900, 000 -1, 000) n ‘Millionaire molecule’ n Distributed intravascularly.
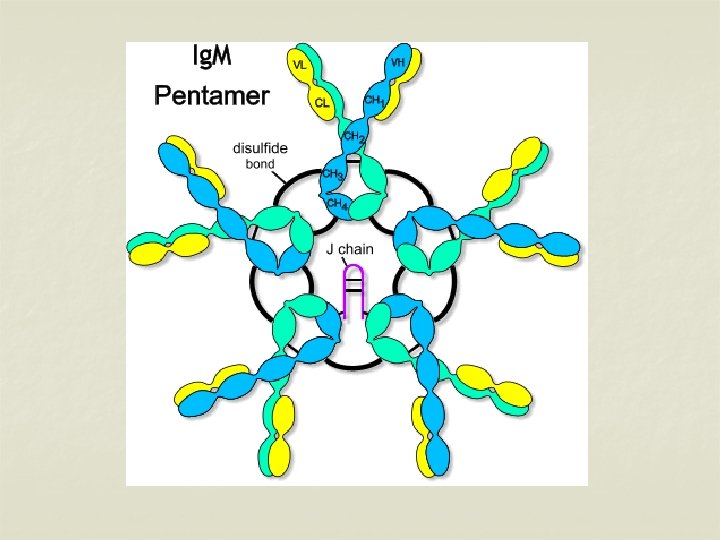

n n n It is the earliest synthesised Ig by foetus in about 20 weeks of age. It appears early in the infection before Ig G. Ig M Abs are short lived – presence indicates recent infection. It cannot cross placenta – presence in new born indicates congenital infection. It participates in agglutination, complement fixation, opsonisation & immune hemolysis Ig M provides protection against blood invasion by microorganisms.

n n Ig M monomers appear on the surface of unstimulated B lymphocytes & act as receptors for antigens. Two subclasses – Ig M 1 & Ig M 2.

IMMUNOGLOBULIN E (Ig. E) n Produced in the lining of respiratory & intestinal tracts. n Also known as “Reagin” n MW- 190, 000 (8 S) n Half life 2 -3 days n Resembles Ig G structurally n Heat labile (Inactivated at 56 o C for 1 hr) n It has affinity for surface of tissue cells – Mast cells. (Homocytotropism)

n n Ig E mediates Type-I hypersensitivity Responsible for asthma, hay fever, eczema, Prausnitz-Kustner (P-K) reaction. Cannot cross the placental barrier. Plays role in defense against parasitic infection.

IMMUNOGLOBULIN D (Ig D) n It resembles Ig G structurally. n Serum concentration – 0. 03 mg/ml n Intravascular in distribution. n MW – 180, 000 (7 S) n Half life – 3 days n It also act as receptors for antigen, like monomeric Ig. M. n 2 subclasses –Ig. D 1, Ig. D 2


Role of different Ig classes Ig. G – Protects the body fluid Ig. A - Protects the body surface Ig. M – Protects the blood stream Ig. E - Mediates reaginic hypersensitivity Ig. D- Recognition molecule on the surface of B lymphocytes

ABNORMAL IMMUNOGLOBULINS n Apart from antibodies, other structurally similar proteins may be found in the serum in the following conditions. Multiple myeloma n Heavy chain disease n Cryoglobulinaemia n

Multiple myeloma n It is a plasma cell dyscrasia in which unchecked proliferation of one clone of plasma cells resulting in the excessive production of particular Ig. n Multiple myeloma involve plasma cells synthesizing any of the 5 classes of Ig. n Multiple myeloma involving Ig. M producing plasma cells – “Waldenstrom’s Macroglobulinaemia”

n n Excessive production of light chains – Bence Jones proteins. It can be identified in urine – proteins get coagulated at 50 o. C but redissolve at 70 o. C Heavy chain disease n Abnormal heavy chains are produced in excess. This is due to lymphoid neoplasia.

Cryoglobulinaemia n It is a condition in which there is formation of precipitate on cooling the serum, which redissolves on warming. n Found in macroglobulinaemia, autoimmune conditions like SLE. n Most cryoglobulins consists of either Ig G or Ig M.

THANK YOU
- Slides: 36