Antibodies Immunoglobulins Chapter 4 Antibody structure and function

Antibodies (Immunoglobulins) Chapter 4
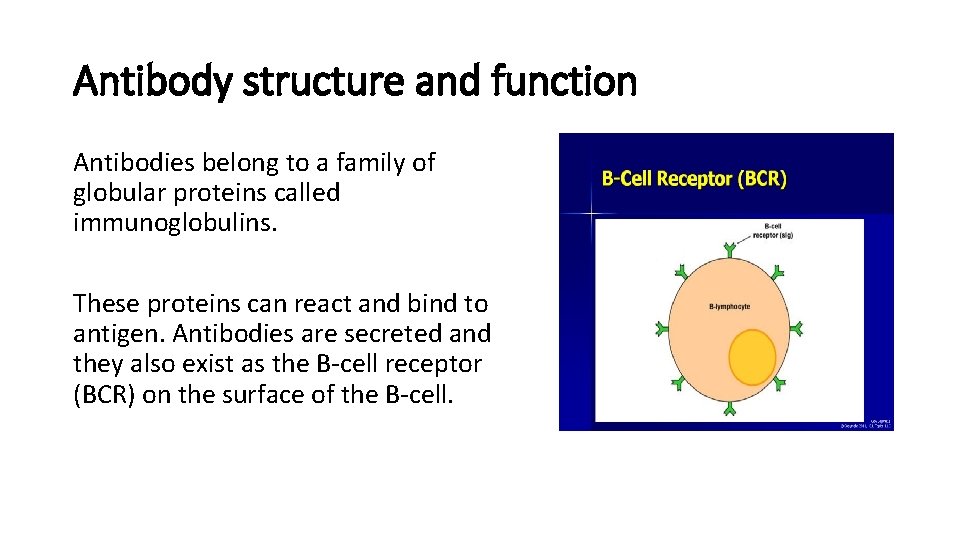
Antibody structure and function Antibodies belong to a family of globular proteins called immunoglobulins. These proteins can react and bind to antigen. Antibodies are secreted and they also exist as the B-cell receptor (BCR) on the surface of the B-cell.
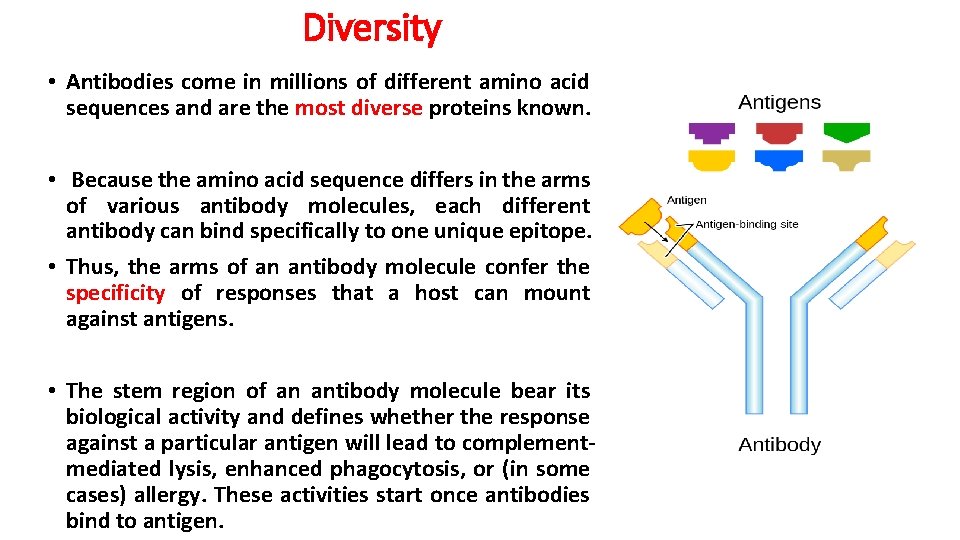
Diversity • Antibodies come in millions of different amino acid sequences and are the most diverse proteins known. • Because the amino acid sequence differs in the arms of various antibody molecules, each different antibody can bind specifically to one unique epitope. • Thus, the arms of an antibody molecule confer the specificity of responses that a host can mount against antigens. • The stem region of an antibody molecule bear its biological activity and defines whether the response against a particular antigen will lead to complementmediated lysis, enhanced phagocytosis, or (in some cases) allergy. These activities start once antibodies bind to antigen.

Biological Activities The ability to trigger protective physiological activities usually after binding to antigen. This would include: -Opsonization, -activation of complement, -allergic responses, -neutralize toxins, -neutralize viruses and other activities
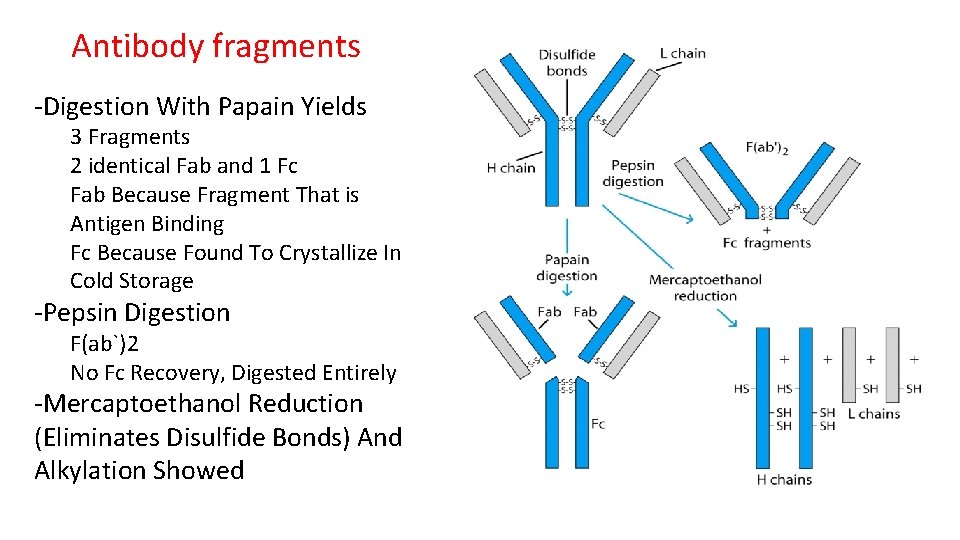
Antibody fragments -Digestion With Papain Yields 3 Fragments 2 identical Fab and 1 Fc Fab Because Fragment That is Antigen Binding Fc Because Found To Crystallize In Cold Storage -Pepsin Digestion F(ab`)2 No Fc Recovery, Digested Entirely -Mercaptoethanol Reduction (Eliminates Disulfide Bonds) And Alkylation Showed
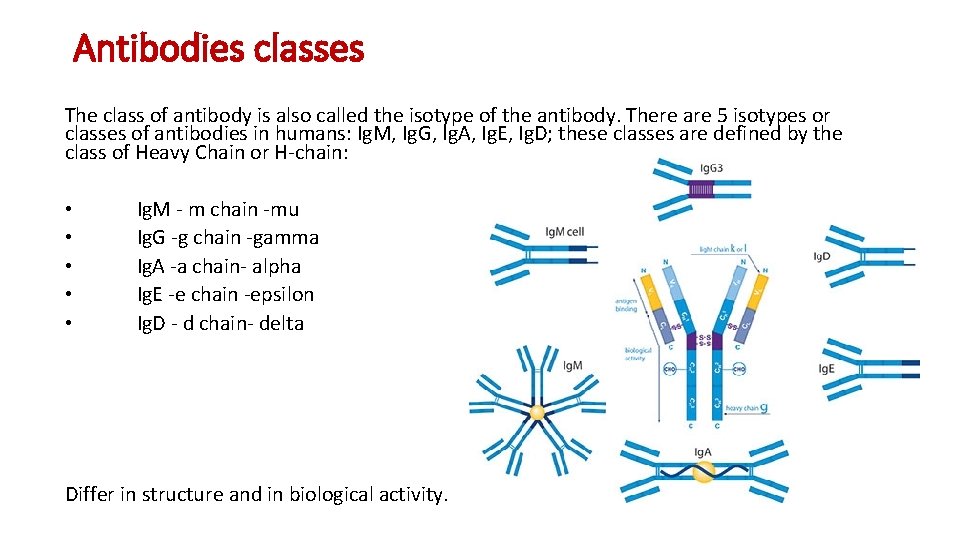
Antibodies classes The class of antibody is also called the isotype of the antibody. There are 5 isotypes or classes of antibodies in humans: Ig. M, Ig. G, Ig. A, Ig. E, Ig. D; these classes are defined by the class of Heavy Chain or H-chain: • • • Ig. M - m chain -mu Ig. G -g chain -gamma Ig. A -a chain- alpha Ig. E -e chain -epsilon Ig. D - d chain- delta Differ in structure and in biological activity.
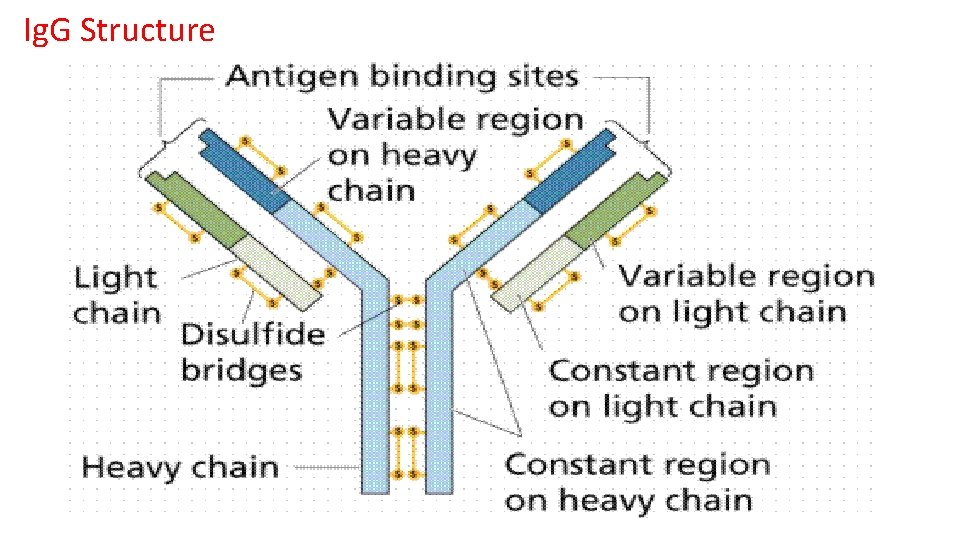
Ig. G Structure

Ig. G Functions § § § Most abundant of all Ig classes Longest half-life of all the Ig classes Agglutinates particulate antigens Precipitates soluble antigens This is the only antibody class that crosses the placenta Opsonizes antigen. Ig. G that is bound to cells or Ig. G that is aggregated in antigen-antibody complexes can activate complement. Ig. G is good at neutralizing toxin Ig. G can interfere with bacterial virulence features such as motility and adherence to tissues Ig. G can block virus ability to adhere to target cells Ig. G is the main antibody found in the secondary immune response

• Ig. G functions continue • Most abundant immunoglobin 80% of serum Ig ~10 mg/m. L • Ig. G 1, 2, 3, 4 (decreasing serum concentration) • Ig. G 1, Ig. G 3 and Ig. G 4 cross placenta • Ig. G 3 Most effective complement activator • Ig. G 1 and Ig. G 3 High affinity for Fc. R on phagocytic cells, good for opsonization
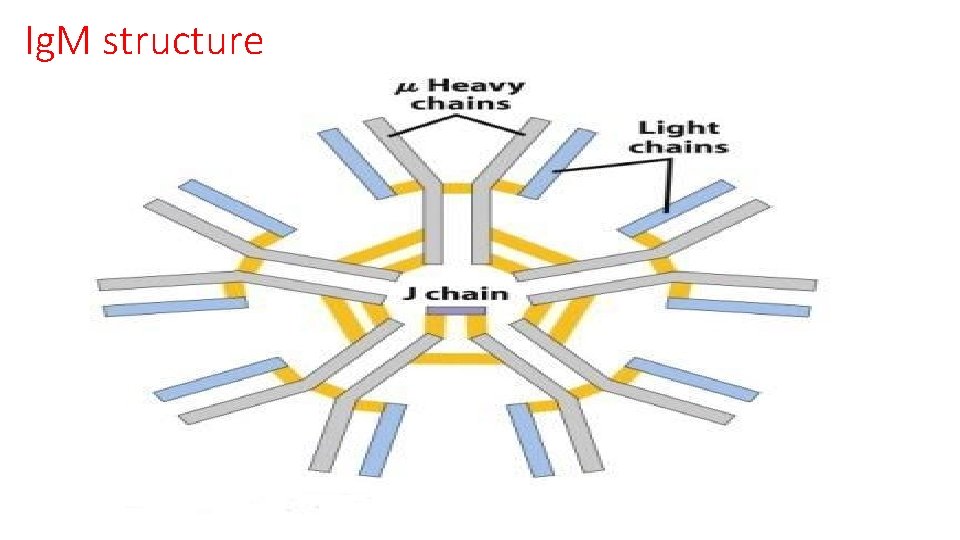
Ig. M structure

Ig. M Function § Ig. M monomer is the B-cell receptor of the mature B-cell § Ig. M is the first antibody made in an immune response, about 7 to 10 days after initial exposure § Ig. M is the first antibody made in life, about 5 months in utero § Ig. M is the most primitive antibody class, found even in primitive vertebrates. § Ig. M is the antibody made to T-independent antigens, polysaccharides § Ig. M is a very good agglutinating antibody § Natural isohemagglutinins are Ig. M class § Ig. M attached to antigen is a very good activator of complement § Ig. M does not cross the placenta • 5 -10% of serum immunoglobulin • 1. 5 mg/m. L

Ig. A Functions • Ig. A is the secretory antibody, found in all secretions including colostrum and milk saliva tears mucus. • Ig. A is particularly important for protection against respiratory and gastrointestinal infectious agents. • Ig. A is important for passively acquired immunity of nursing baby. • Ig. A makes lysozyme work better, especially against gram-negative bacteria • Ig. A neutralizes virus • Ig. A is transported across membranes via the poly-Ig receptor • 10 -15% of serum Ig. G • 5 -15 g of Ig. A released in secretions!!!!

Draw the structure of Ig. A

Ig. E functions • Ig. E is made in response to parasitic worms. When bound to the parasite, it triggers a very strong inflammatory response to the worms. • Ig. E is the antibody that triggers allergy • Ig. E binds to Fc receptors on the surface of mast cells and basophils • Binding causes degranulation (Histamine Release) • Very low serum concentration, 0. 3 g/m. L

Draw the structure of Ig. E?
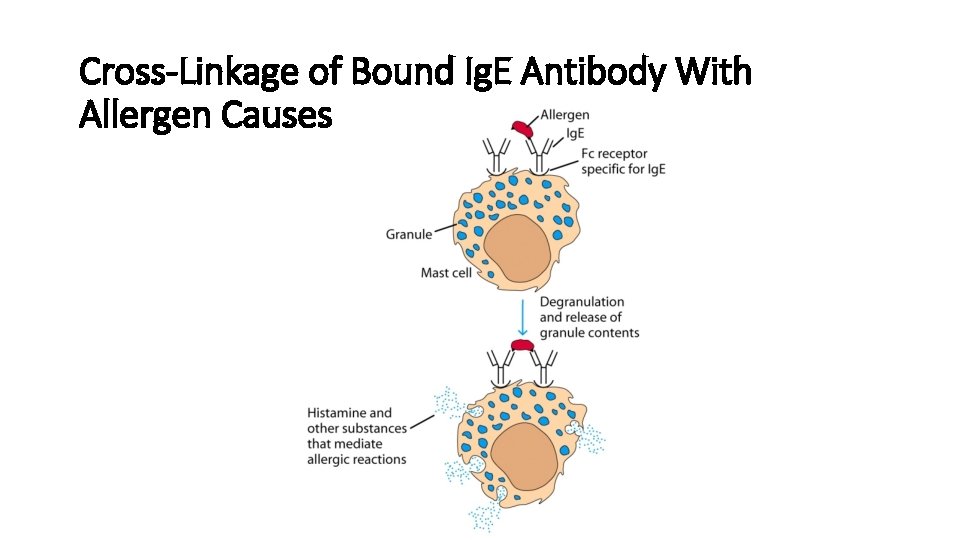
Cross-Linkage of Bound Ig. E Antibody With Allergen Causes

• Ig. D • Expressed on B-cell Surface • Ig. M and Ig. D, Expressed on B-cell Surface • We Do Not Know Any Other Biological Effector Activity • Low serum concentrations, ~30 g/m. L
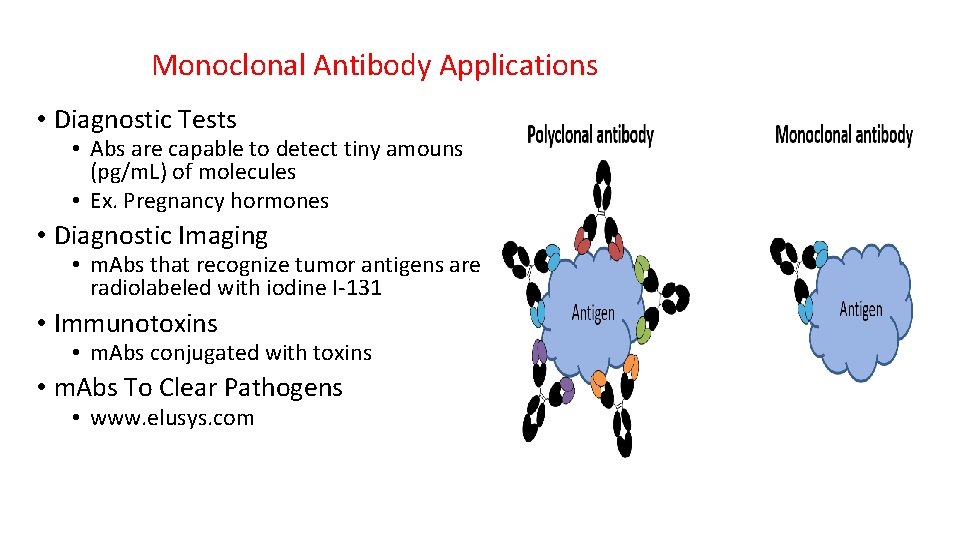
Monoclonal Antibody Applications • Diagnostic Tests • Abs are capable to detect tiny amouns (pg/m. L) of molecules • Ex. Pregnancy hormones • Diagnostic Imaging • m. Abs that recognize tumor antigens are radiolabeled with iodine I-131 • Immunotoxins • m. Abs conjugated with toxins • m. Abs To Clear Pathogens • www. elusys. com
- Slides: 18