Antibacterial therapy By Assis Prof Nader Alaridah Antibacterial

Antibacterial therapy By : Assis. Prof. Nader Alaridah

Antibacterial therapy • 2000 B. C. - "Here, eat this root. " • 1000 B. C. - "That root is heathen, say this prayer. " • 1850 A. D. - "That prayer is superstition, drink this potion. " • 1940 A. D. - "That potion is snake oil, swallow this pill. " • 1985 A. D. - "That pill is ineffective, take this antibiotic. " • 2000 A. D. - "That antibiotic is useless and artificial. Here, eat this root. " ~Author Unknown

Antibacterial therapy Antimicrobial agents are medications used to treat an infection by a microorganism that overcame the body’s defenses. Antibiotics: natural products derived from soil bacteria and fungi Examples: Penicillin from penicillin notatum mold > Semisynthetic agents: Natural compounds that have been chemically modified to increase its activity and improve pharmacokinetics Examples: Amoxycillin, Ampicillin, Cephalosporins and Carbapenems, Rifampicin

Important concepts in antimicrobial therapy • Selective toxicity • Spectrum of activity (Broad vs narrow spectrum) • Static vs cidal (MIC vs MLC) • Synergism: Meaning and example

ANTIMICROBIAL ACTIVITY IN VITRO • Determination of the susceptibility of a bacterial pathogen to antimicrobial drugs can be done by one of two principal methods: dilution or diffusion. • These methods can be used to estimate either the potency of antibiotic in the sample or the susceptibility of the microorganism. • Dilution Method Graded amounts of antimicrobial substances are incorporated into liquid or solid bacteriologic media. • A widely used method in smaller laboratories is the disk diffusion test.
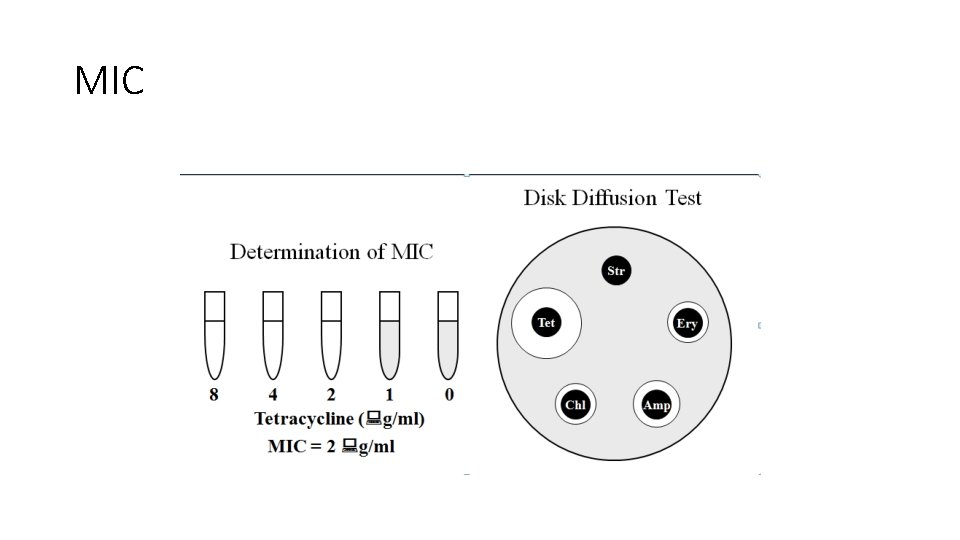
MIC

Mechanism of action v Antimicrobial drugs act in one of several ways: 1. Inhibition of cell wall synthesis (eg : Beta lactam agents and glycopeptides) 2. Inhibition of cell membrane function. (eg: Polymyxin, Colistin) 3. Inhibition of protein synthesis (ie, inhibition of translation transcription of genetic material; ( eg: Aminoglycosides, chloramphenicol, macrolides and tetracyclines ) 4. Inhibition of nucleic acid synthesis (eg: Rifamin , Quinilones)
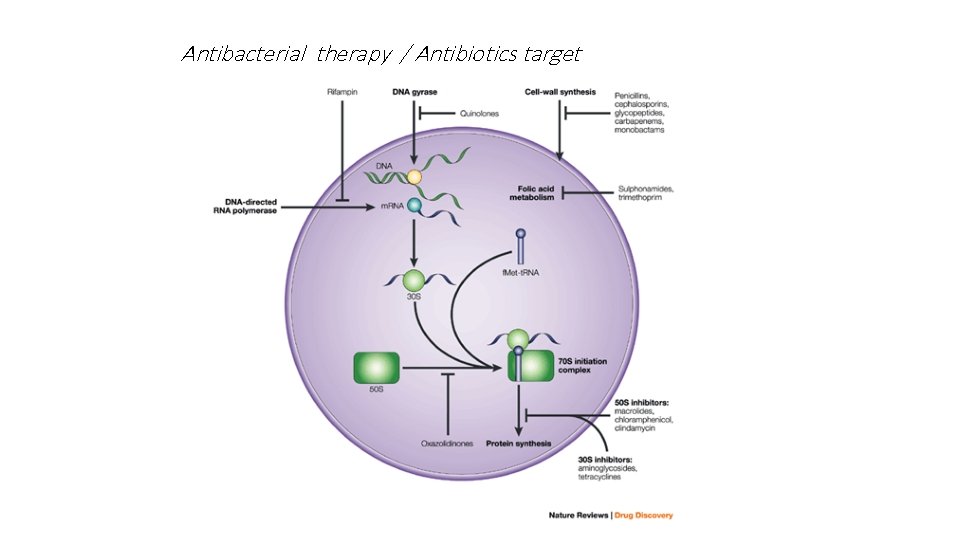
Antibacterial therapy / Antibiotics target

INHIBITION OF CELL WALL SYNTHESIS • Bacteria have a rigid outer layer, the cell wall. The cell wall maintains the shape and size of the microorganism. • The cell wall contains a chemically distinct complex polymer “mucopeptide” (“peptidoglycan”). The polysaccharides regularly contain the amino sugars N- acetylglucosamine and acetylmuramic acid. The latter is found only in bacteria. • The peptidoglycan layer is much thicker in the cell wall of gram-positive than of gramnegative bacteria. • The initial step in drug action consists of binding of the drug to cell receptors (penicillinbinding proteins [PBPs])(eg: Beta lactam agents and glycopeptides )

Antibacterial therapy/Inhibition of cell wall synthesis

Beta lactam agents • Penicillins, cephalosporins, monobactams, carbapenems and beta lactamase inhibitors • Bind penicillin binding proteins preventing cross linking • Beta lactamases are enzymes produced by bacteria that break the antibiotic beta lactam ring > resistance
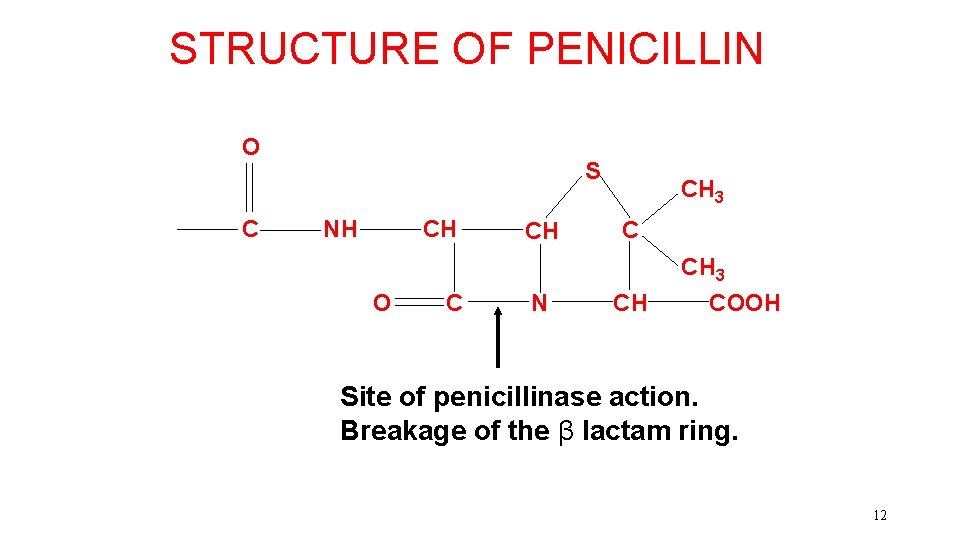
STRUCTURE OF PENICILLIN O C S NH CH O C CH N CH 3 C CH CH 3 COOH Site of penicillinase action. Breakage of the β lactam ring. 12

Beta lactam agents • Penicillins: • • E. g Benzylpenicillin and penicillin G Narrow spectrum Short acting Resistance by beta lactamases • Synthetic penicillins: • Penicillin V • Flucloxacillin and methicillin: Inactivated by S. aureus beta lactamase However, MRSA developed. MRSA? • Ampicillin and amoxicillin: G+ and G • Amoxacillin+Clavulinic Acid (B-lactamase inhibitor) compound)/ Broad Spectrum. . Penicillinase-R

Beta lactam agents • Monobactam/ Aztreonam used mostly against serious aerobic and Facultative G-ve infection. • Carbapenem / imipenem & meropenem. . Broad Spectrum G- G+ aerobic anaerobic. . , Penicillinase-R. • Cephalosporins: • Broader spectrum • Less hypersensitivity reactions • 5 Different generations

Cephalosporins • 1 st (1960) Cephalexin, Cephradine, spectrum G+. . • 2 nd (70 s) Cefoxitin, Cefuroxime, Broad spectrum. . • 3 rd (80 s) Ceftriaxone, Cefotaxime. . mainly G-ve Enteric bacteria. . • 4 th (1990 s) Cefepime. . mainly G-ve, GPC and psudomonas • 5 th generation 2000 s: ceftaroline …MRSA

Glycopeptides • Vancomycin and teicoplanin • Large molecules that are unable to penetrate the outer membrane of Gram-negative bacteria. • The spectrum is consequently restricted to Gram-positive organisms. • Gram-positive cocci with multiple resistance to other drugs such as Enterococci and staphylococci, including MRSA, that exhibit resistance or reduced sensitivity to glycopeptides are being reported more frequently.

Disruption of microbial cell membrane • Essentially, affect cell membrane transportation in and out • Increases permeability of membrane • External influences have greater effect • Microbe death • Examples: Polymyxin, Colistin These agents are more toxic systemically than those agents that inhibit cell wall synthesis.

INHIBITION OF MICROBIAL PROTEIN SYNTHESIS • Act at: • Site of protein synthesis (ribosome) • Whereas bacteria have 70 S ribosomes, mammalian cells have 80 S ribosomes. • Tetracyclines (static), aminoglycosides (cidal), chloramphenicol (static) and macrolides (static), e. g erythromycin, clindamycin (static).

https: //www. google. jo/url? sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0 ah. UKEwi. Tt. KKR 9 f. HWAh. UIWRQKHf. QDBSIQj. Rw IBw&url=http%3 A%2 F%2 Fwww. impactjournals. com%2 Foncotarget%2 Findex. php%3 Fjournal%3 Doncotarget%26 page%3 Darticle%26 op%3 Dvie w%26 path%5 B 0%5 D%3 D 3174%26 path%5 B 1%5 D%3 D 6141&psig=AOv. Vaw 2 m. Qy. NGa. G_bx 7_tip. Md 57 JJ&ust=1508132391528053

Aminoglycosides • Gentamicin, amikacin • G-ve aerobics and synergism with B lactams • Mechanism: ØBind to 30 s subunit and Inhibit formation of ribosomal initiation complex ØMisreading of the m. RNA • Side effects: ototoxicity and nephrotoxicity • Drug level monitoring is required

Chloramphenicol • Broad spectrum • Mechanism: • It acts by blocking the elongation of the peptide chain on the 50 s bacteria subunit • Uses are limited because of the occurrence of a rare but Some side effects; Aplastic anaemia !!!, grey baby syndrome!!!.

Macrolides: ØErythromycin, clarithromycins, azithromycin (good tissue penetration and a long terminal half-life) and telithromycins Øbind to the 50 S subunit of the ribosome, and the binding site is a 23 S r. RNA. • Used mainly as antistaphylococcal and antistreptococcal agents, though some have wider applications (broad spectrum) • Clindamycins: G+ve, pseudomembranous colitis

Tetracycline • Broad spectrum • Inhibit binding of t. RNA to ribosome leading to failure of peptide chain elongation • G-ve, +ve, mycoplasma, rickettsia and malaria • Tetracycline, doxycycline and tigecycline. • Some side effects: • Stain developing teeth, Discolor permanent teeth (yellow-gray -brown), from infancy and childhood to eight years old and adulthood, Affect bone growth of the fetus, so should be avoided during pregnancy

So far the story is: • Tetracycline (30 s) work on almost everything! But lots of resistance tigecycline • Chloramphenicol similar action to it, but has a severe side effect(chlorine) • Aminoglycosides (30 s) work on G-ve and TB but not G+ve or anaerobe • Macrolides help with G+ve and intracellulars but not G-ve (except helicobacter) telithromycin • Clindamycin for anaerobes (doesn’t matter Gram +ve or –ve) • Linezolid is against resistant G+ve cocci • Also streptogramins (the cristins) against resistant G+ve cocci • Fusidic acid and mupirocin are topical agents

INHIBITION OF NUCLEIC ACID SYNTHESIS • Rifampin inhibits bacterial growth by binding strongly to the DNAdependent RNA polymerase of bacteria. Thus, it inhibits bacterial RNA synthesis. Rifampin resistance results from a change in RNA polymerase because of a chromosomal mutation that occurs with high frequency. • All quinolones and fluoroquinolones inhibit microbial DNA synthesis by blocking DNA gyrases, topoisomerase enzymes that play key roles in DNA replication and repair

Inhibition of nucleic acid synthesis and function • A. INHIBITORS OF RNA SYNTHESIS AND FUNCTION • Rifampicin (bactericidal) • a. Mode of action These antimicrobials bind to DNA-dependent RNA polymerase and inhibit initiation of m. RNA synthesis • b. Spectrum of activity They are wide spectrum antibiotics but are used most commonly in the treatment of tuberculosis, leprosy and MRSA c. Combination therapy Since resistance is common, rifampicin is usually used in combination therapy and usually reserved for TB.

Inhibition of nucleic acid synthesis and function • B. INHIBITORS OF DNA SYNTHESIS AND FUNCTION • Quinolones - nalidixic acid (G-ve), ciprofloxacin (plus pseudpmonas and G +ve) and gemifloxacin (and some anaerobes) • Bactericidal • Mode of action These antimicrobials bind to the A subunit of DNA gyrase (topoisomerase) and prevent supercoiling of DNA, thereby inhibiting DNA synthesis. • Absorbed well orally, good body distribution • Children, pregnant and breast feeding!!!

Inhibition of nucleic acid synthesis and function • C. DNA STRAND BREAKAGE • Nitroimidazole e. g metronidazole • Mechanism: At low redoox values they are reduced to a short intermediate form that damages the bacterial DNA So, the expected bacterial activity is against

Inhibition of folate synthesis • Sulphonamides and diaminopyrimidines as Trimethoprim • Trim is less toxic • Inhibition of Precursor Synthesis (Folic acid) • Available as combination, Co-trimoxazole, or separately. • G-ve and +ve aerobes

Anti TB drugs • • • Isoniazid and Ethionamide - chemically related - blocks mycolic acid synthesis Ethambutol blocks arabinogalactan synthesis Rifampicin

Resistance: meaning? Terms: Intrinsic or inherent: no target site or cell wall is impermeable to antibiotics as in gram negative bacteria (vancomycin is too big to cross the cell wall) Acquired resistance: Selection of resistant bacteria by antibiotics Common in areas of heavy antibiotic use e. g hospitals The resistance is initially emerged by genetic process then selected by antibiotics next figure
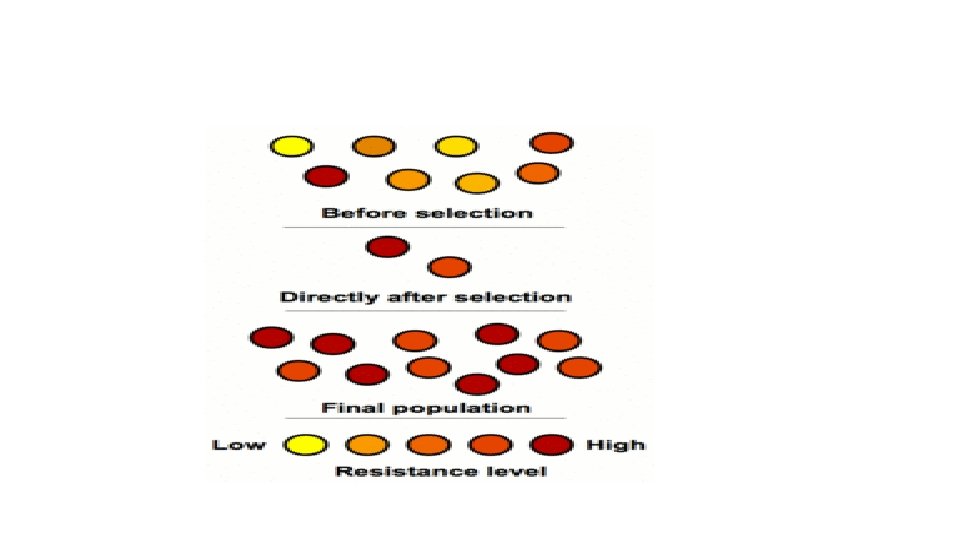

Cross resistance: Resistance to one member of a family will result in resistance or decreased susceptibility to other members within the same family e, . g fluroquinolones, Penicillins and Cephalosporins Multiresistance: Resistance to more than one antibacterial Usually acquired by separate mechanisms

Genetics of resistance: 1. Intrinsic: No target 2. Acquired: A. Chromosomal mutations Single step mutation in the antibiotic target leading to decreased antibiotic efficacy (needs high concentration of antibiotic). Multistep mutations: Leading to complete resistance e. g: penicillin binding protein B. Transferable via Genetic transfer (plasmids )

Mechanism of resistance: 1. Decreased accumulation: Decreased permeability secondary to porins mutations Increased efflux (pumping out the antibacterial using expressed efflux pump)

2. Modification of the target: Sequence mutation leading to target alteration: > Pneumocoocus resistance to penicillins > Quinilones Target bypass: Supplementary enzymes will do the same target function but without binding to the antibacterial agent e. g Meticillin resistant staph aureus MRSA Target hyperproduction: More drug is needed to inactivate the target

3. Inactivation of the antibacterial agent: β lactamase is an enzyme produced by the bacteria This enzyme will destroy the β- lactam ring (this is an essential ring in penicillins and cephalosporins) leading to inactivation of the antibacterial agent > Some types of bacteria produce a β- lactamase with a wide range of activity (ESBLs) Acetylating, adenylating and phosphorylating enzymes: Produced by bacteria (gram negative bacteria) and cause resistance to aminoglycosides and chloramphenicol

Resistance

The End
- Slides: 39