Antibacterial agents Traditional targets of antibacterial compounds Nature

Antibacterial agents
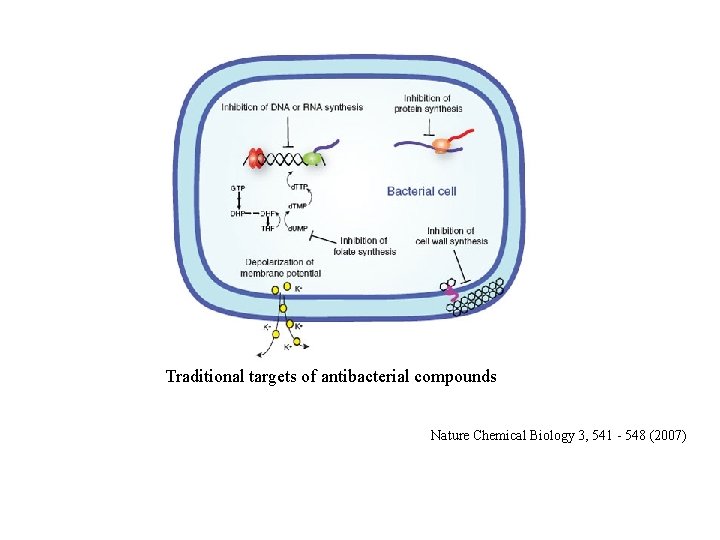
Traditional targets of antibacterial compounds Nature Chemical Biology 3, 541 - 548 (2007)

Cell wall inhibitors oduction • The cell wall of bacteria contains peptidoglycan, a substance that does not occur in eukaryotes • The Gram-positive cell wall is composed of a thick, multilayered peptidoglycan sheath outside of the cytoplasmic membrane • The Gram-negative cell wall is composed of an outer membrane linked by lipoproteins to thin, mainly single-layered peptidoglycan

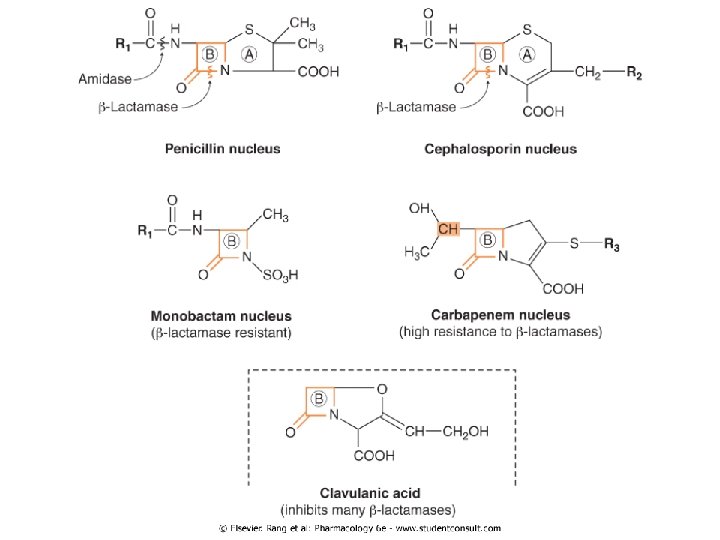
1. Beta lactam antibacterial 1. 2. 3. 4. 5. Penicillins Cephalosporins & Cephamycins Carbapenems: imipenem Monobactams: aztreonam Beta-lactamase inhibitors: clavulanic acid

Mechanism of action • All β-lactam antibiotics interfere with the synthesis of the bacterial cell wall peptidoglycan • β-Lactam antibiotics, structural analogs of the natural DAla-D-Ala substrate, covalently bind to the active site of PBPs • They interfere with the last step of bacterial cell wall synthesis (transpeptidation or cross-linkage) • This results in cell lysis, either through osmotic pressure or through the activation of autolysins • β-Lactam antibiotics are thus bactericidal

Penicillins • The penicillins are a large group of bactericidal compounds • They can be subdivided and classified by their chemical structure and spectrum of activity • The antibacterial spectrum is determined by their ability to cross the bacterial peptidoglycan cell wall to reach the PBPs in the periplasmic space

Penicillins • In general, gram-positive microorganisms, in the absence of resistance, are susceptible to penicillins • Gram-negative microorganisms have an outer lipopolysaccharide membrane (envelope) surrounding the cell wall that presents a barrier to the water-soluble penicillins

Penicillins • The antimicrobial activity of penicillin resides in the β-lactam ring Splitting of the β-lactam ring by either acid hydrolysis or β-lactamases results in the formation of penicilloic acid, a product without antibiotic activity Penicillins may be classified into four groups: 1) Natural penicillins (G and V) 2) Antistaphylococcal penicillins 3) Extended-spectrum penicillins 4) Antipseudomonal penicillins

I. Natural Penicillins • Are obtained from fermentations of the mold Penicillium chrysogenum • The have limited spectrum of antibacterial activity and susceptible to beta-lactmase • Clinical uses include treatment of infection caused by a number of gram positive and gram-negative cocci, gram-positive bacilli, , and spirochetes • Most strains Staphyloccus aureus and significant number of Neiseria gonorrhoea are resistant • Penicillin G is the DOC for syphilis

I. Natural Penicillins • Penicillin G: is an acid-labile compound & is most appropriate for IM or IV therapy • Penicillin G is excreted by the kidneys, with 90% of renal elimination occurring via tubular secretion • Probenecid blocks tubular secretion and has been used to increase the serum concentration and prolong its half-life of penicillin • Depot IM formulations of penicillin G, including procaine penicillin and benzathine penicillin, have decreased solubility, delayed absorption, and a prolonged half-life

I. Natural Penicillins • Penicillin V: • An oral formulation that resists degradation by gastric acid. • Antibacterial spectrum of activity is similar to that of penicillin G • Penicillin V is used to treat streptococcal infections when oral therapy is appropriate and desirable

II. Antisatphylococcal penicillins • Agents: Methicillin, nafcillin (IV), oxacillin, • Are more resistant to bacterial β-lactamases than is penicillin G • Their use is restricted to the treatment of infections known or suspected staphylococcal infections • Methicillin resistant S aureus (MRSA) and S epidermidis (MRSE) are resistant • They are active against staphylococci and streptococci but not against enterococci, anaerobic bacteria, and gram-negative cocci and rods.

III. Extended-spectrum penicillins • Agents: Ampicillin and amoxicillin • Antibacterial spectrum similar to that of penicillin G more effective against gramnegative bacilli, but remains susceptible to penicillinases Ampicillin: a) Treatment of meningitis caused by Listeria monocytogenes. b) Enterococcal endocarditis c) Pneumonia caused by β-lactamase-negative H. influenzae d) Shigellosis

III. Extended-spectrum penicillins 2) Amoxicillin a) Acute nonserious bacterial infections like otitis media and sinusitis. b) Multidrug regimens for the eradication of H. pylori in duodenal and gastric ulcers c) Prophylactically by dentists for patients with abnormal heart valves who are to undergo extensive oral surgery

IV. Antipseudomonal penicillins • Agents: ticarcillin, and piperacillin • They are effective against many gram-negative rods, including Pseudomonas, enterobacter and in SOME CASES klebsiella species • Susceptible to penicillinase • Syngestic effect when used in combination with aminoglycosides

Resistance 1) Elaboration of the enzyme ß-lactamase, which hydrolyzes the ß-lactam ring (loss of bactericidal activity) 2) Alteration of penicillin-binding proteins (PBPs) either by mutation of existing PBP genes or, more importantly, by acquiring new PBP genes (e. g. staphlococcal resistance to methicillin) or by acquiring new "pieces" of PBP genes (e. g. pneumococcal, gonococcal and meningococcal resistance) 3) Resistance seen in gram-negative bacteria, is due to alteration of genes that specify outer membrane proteins (porins) and reduce permeability to penicillins

Adverse Reactions to Penicillins • Hypersensitivity reactions: • Are the most common • All penicillins are cross-sensitizing and crossreacting • Include: Macropapular rash, urticarial rash, fever , bronchospasm , vasculitis , exfoliative dermatitis, stevens-Johnson syndrome, anaphylaxis (very rare-0. 05% of recipients) • GIT disturbances: with oral penicillin caused by direct GIT irritation or overgrowth of gram positive organism or yeast

β-lactamase inhibitors • Agents: Clavulanic acid, sulbactam, & tazobactam • They Contain β-lactam ring but do not have significant antibacterial activity • They bind to & inactivate β-lactamases, thereby protecting the antibiotics that are normally substrates for these enzymes

β-lactamase inhibitors Penicillin plus β-lactamase Inhibitors: 1) 2) 3) 4) Amoxicillin-clavulanic acid Ticarcillin-clavulanic acid Ampicillin-sulbactam Piperacillin-tazobactam

Cephalosporins & Cephamycins • Cephalosporins are similar to penicillins, but more stable to many bacterial β-lactamases and therefore have a broader spectrum of activity • Cephalosporins can be classified into five major groups or generations, depending mainly on the spectrum of antimicrobial activity & resistance to β-lactamases

I. First generation Agents: Cefadroxil, cefazolin, cephalexin, cephalothin, cephapirin, & cephradine Penicillin G substitiutes Have good activity against gram-positive cocci (with the exception of enterococci, methicillin-resistant S. aureus, and S. epidermidis) and relatively modest activity against gram-negative microorganisms Most mouth anaerobes are sensitive, but the B. fragilis group is resistant • Cefazolin penetrates well into most tissues. It is the drug of choice for surgical prophylaxis. Cefazolin DOES NOT penetrate the CENTRAL NERVOUS SYSTEM and cannot be used to treat meningitis

II. Second Generation • Cefaclor, cefamandole, cefonicid, cefuroxime, cefprozil, loracarbef, and ceforanide, cephamycins cefoxitin, cefmetazole, and cefotetan • Have somewhat increased activity against gramnegative microorganisms: – H influenzea – Enterobacter areogenes – Some Niesseria species • As with 1 st generation: none is active against enterococci or P aeuroginosa

Clinical Uses 1. Used to treat otitis, sinusitis and lower RTI (H influenza & B catarrhallis) 2. Cefoxitin, cefotetan or cefmetazole: used to treat mixed anaerobic infections such as peritonitis or diverticulitis 3. Cefuroxime community acquired pneumonia The only to cross the BBB, however less effective 3 rd generation

III. Third Generation • ceftriaxone, cefotaxime, ceftazidime, ceftizoxime, cefoperazone, cefixime, cefpodoxime proxetil, cefdinir, cefditoren pivoxil, ceftibuten, and moxalactam • Expanded gram negative coverage (except cefoperazone) • The ability to cross the BBB • Active against citrobacter, Serratia marscens and providentia (but not the resistant strains) • Active against B-lactamase producing strains of haemophilus and neisseria • Ceftazidime & cefoperazone: the 2 third generation which are active against P aeureginosa

Third Generation-kinetics • They penetrate body fluids and tissues well • Achieve levels in the CSF (With the exception of cefoperazone, cefixime, cefpodoxime proxetil) sufficient to inhibit most pathogens • Excretion of cefoperazone and ceftriaxone is mainly through biliary tract • The rest are excreted by the kidney

Fourth Generation • Cefepime • it may be useful in treatment of enterobacter infections. Otherwise, its clinical role is similar to that of third-generation cephalosporins. • Cefepime has good activity against most penicillin-resistant strains of streptococci • V. Fifth generation • Agents: Ceftaroline fosamil, the prodrug of ceftaroline • Effectively bind to and inhibit penicillin-binding protein 2 a, which mediates methicillin-resistance in staphylococci

Adverse Effects of cephalosporins • Allergy: identical to penicillins- cross allerginicity 5 -10% • Toxicity • Severe pain (IM) • Thrombophelebitis (IV) • Renal toxicity, • Hypoprothrombinemia • Disulfiram like reaction • Superinfection

Monobactams: Aztreonam • Spectrum of activity is limited to aerobic gramnegative rods, including P. aeruginosa • Has no activity against gram-positive bacteria or anaerobes • It is administered either IV or IM and is excreted in the urine • it shows little cross-reactivity with other β-lactam antibiotics • ADRs: phlebitis, skin rash, and abnormal liver function tests

Carbapenems: Imipenem • It is a carbapenem with low susceptibility to B-lactamses • Wide spectrum activity against many gram-negative rods, including P aeruginosa, gram-positive organisms, and anaerobes • It is resistant to most β-lactamases. Administered parenterally • The drug of choice for enterobacter infections • Rapidly metabolised by tubular dehydropeptidase

IMIPENEM-CILASTATIN • CILASTATIN is a peptidase inhibitor that blocks renal degradation of Imipenem • Cilastatin increases the plasma half-life of imipenem and inhibits the formation of potentially nephrotoxic metabolite • ADE: GI distress, skin rash, CNS toxicity (confusion, encephalopathy and seizures) • Partial cross allergenicity with penicillins

Vancomycin • It is active only against gram-positive bacteria, particularly staphylococci • No activity against gram-negative organisms • Vancomycin is poorly absorbed from the intestinal tract and is administered orally only for the treatment of antibioticassociated enterocolitis caused by C difficile

VANCOMYCIN • Mechanism of action: Inhibits bacterial cell wall synthesis by binding firmly to the D-Ala-D-Ala terminus of nascent peptidoglycan pentapeptide • Resistance: modification of the D-Ala-D-Ala binding site of the peptidoglycan in which D-Ala is replaced by D-lactate (enerococci & vancomycin resistant S aureus) • Vancomycin kills staphylococci relatively slowly and only if cells are actively dividing; • The combination of vancomycin and gentamicin is synergistic against Staphylococcus aureus and enterococci

Clinical uses of vancomycin 1. 2. 3. 4. 5. Main indication is: sepsis or endocarditis caused by MRSA Methicillin-susceptible staphylococci in patients who are allergic to penicillins or cephalosporins Vancomycin in combination with gentamicin is an alternative regimen for treatment of enterococcal endocarditis in a patient with serious penicillin allergy Vancomycin (in combination with cefotaxime, ceftriaxone, or rifampin) is also recommended for treatment of meningitis suspected or known to be caused by a highly penicillin-resistant strain of pneumococcus Oral vancomycin is used to treat antibiotic-associated enterocolitis caused by Clostridium difficile
Adverse reactions 1) Phlebitis at site of injection 2) Ototoxicity and Nephrotoxicity are rare with current preparations. 3) AVOID coadministration with aminoglycosides 4) Red man or red neck syndrome: infusion related flushing due to histamine release (slow infusion over 2 hrs, increase dilution volume, or pretreat with antihistamine)

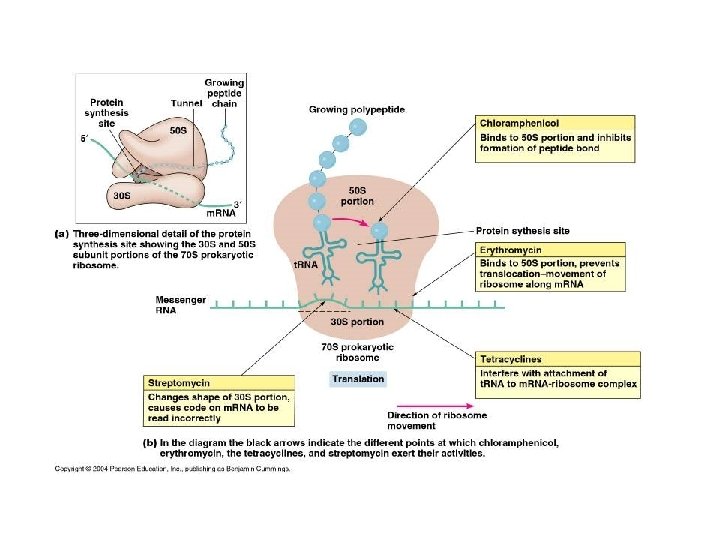
Protein Synthesis Inhibitors • Target the bacterial ribosome, which has components that differ structurally from those of mammalian cytoplasmic ribosomes • The bacterial ribosome consists of a 50 S subunit and a 30 S subunit, whereas in the mammalian ribosome the subunits are 60 S and 40 S 1. Chloramphenicol 2. Tetracyclines 3. Macrolides 4. Clindamycin 5. Aminoglycosides 6. Linezolide 7. Streptogramines

Aminoglycosides • Agents: streptomycin, neomycin, kanamycin, amikacin, gentamicin, tobramycin, sisomicin, netilmicin • Aminoglycosides have been used in paediatric patients for more than 60 years • They have concentration-dependent killing & a significant postantibiotic effect • They are not available for oral administration, so they are mainly used in parenteral form • Predominantly used to treat gram-negative treat bacteria infections including Pseudomonas spp, • They are useful in the treatment of UTIs, intra-abdominal infections, neonatal sepsis, complicated infections in cystic fibrosis, and as empiric therapy for febrile neutropenic patients

Aminoglycosides • Aminoglycosides have concentration-dependent killing & a significant postantibiotic effect • Optimal dosing strategies of aminoglycosides include high doses administered once daily (extended-interval dosing) • The three most commonly used agents are: gentamicin, tobramycin, and amikacin • Aminoglycosides frequently exhibit synergism with beta lactams

Adverse effects 1. 2. Ototoxicity Nephrotoxicity 3. Neuromuscular paralysis: • Most often occurs after direct intraperitoneal or intrapleural application of large doses of aminoglycosides • Caused by a decrease in both the release of acetylcholine from prejunctional nerve endings and the sensitivity of the postsynaptic site • Reversible by calcium gluconate (promptly) or neostigmine

Tetracylcins • Agents: Tetracyclin, minocycline, doxycycline, demeclocycline, & oxytetracycline • Bacteriostatic broad spectrum antibiotics: active against a wide range of aerobic and anaerobic gram-positive and gram-negative bacteria, as well as against some protozoa, eg, amebas • Bind reversibly to 30 S of bacterial ribosome, blocking the binding of aminoacyl-t. RNA to the acceptor site on the m. RNA-ribosome complex

Tetracylcins • The antibacterial activities of most tetracyclines are similar except that tetracycline-resistant strains may be susceptible to doxycycline, minocycline, and tigecycline • Absorption is impaired by divalent cations (Ca 2+, Mg 2+, Fe 2+) or Al 3+; by dairy products and antacids, and by alkaline p. H • Tetracyclines cross the placenta to reach the fetus and are also excreted in milk
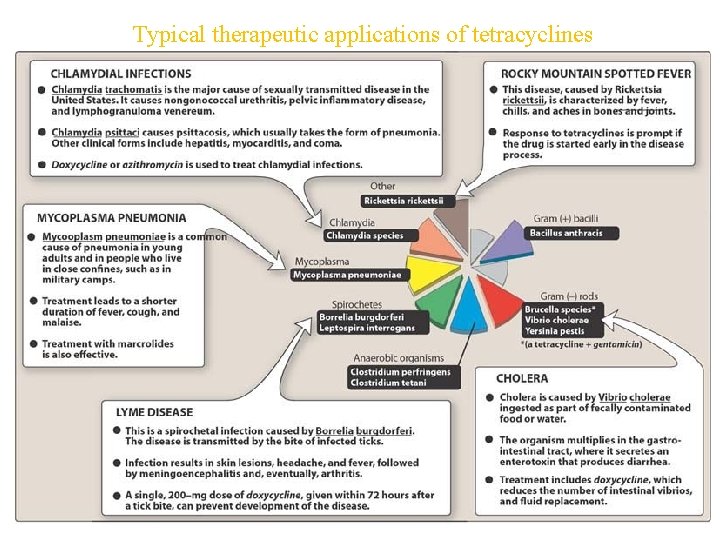
Typical therapeutic applications of tetracyclines
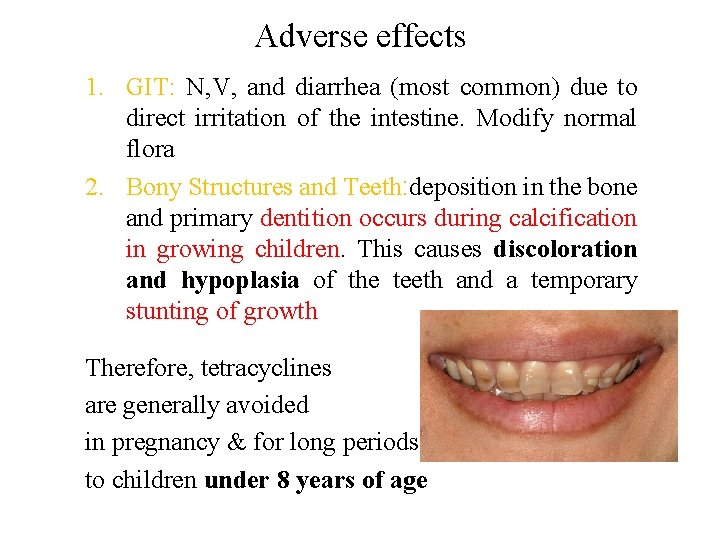
Adverse effects 1. GIT: N, V, and diarrhea (most common) due to direct irritation of the intestine. Modify normal flora 2. Bony Structures and Teeth: deposition in the bone and primary dentition occurs during calcification in growing children. This causes discoloration and hypoplasia of the teeth and a temporary stunting of growth Therefore, tetracyclines are generally avoided in pregnancy & for long periods to children under 8 years of age

Adverse effects 3. Fatal hepatotoxicity: especially during pregnancy 4. Phototoxicity: such as severe sunburn, occurs when a patient receiving a tetracycline , especially demeclocycline, is exposed to sun or ultraviolet rays 5. Local Tissue Toxicity (IV & IM injections) 6. Vestibular Reactions (dizziness, vertigo, N, & V) particularly with minocycline 7. Superinfections: Overgrowths of Candida (for example, in the vagina) or of resistant staphylococci (in the intestine)

Macrolides • Agents: erythromycin, clarithromycin, azithromycin, telithromycin (Ketolide) • The macrolides, in general, are bacteriostatic • Erythromycin may be bactericidal, particularly at higher concentrations, for susceptible organisms • Inhibition of protein synthesis occurs via binding to the 50 S ribosomal RNA • Erythromycin base is destroyed by gastric acid. Thus, either enteric-coated tablets or esterified forms of the antibiotic are administered
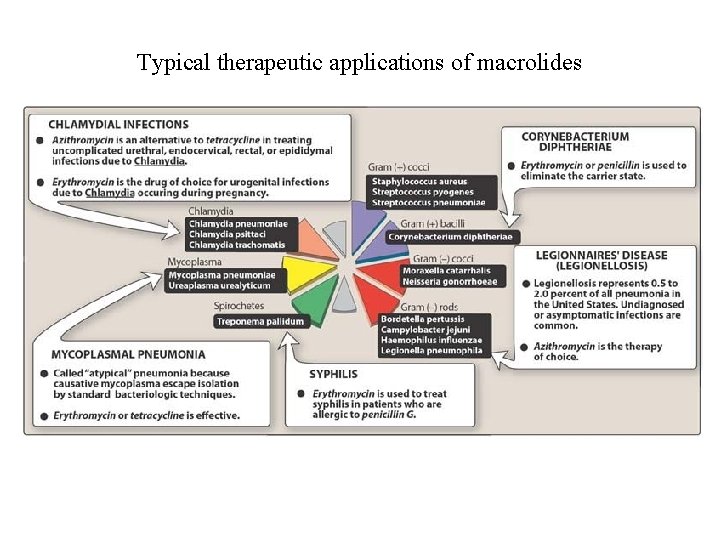
Typical therapeutic applications of macrolides

Macrolides-ADRs • Epigastric distress: Anorexia, nausea, vomiting, and diarrhea. Can lead to poor patient compliance for erythromycin • Cholestatic jaundice: especially with the estolate form of erythromycin as the result of a hypersensitivity reaction • Prolongate the QTc interval in some patients (Telithromycin): avoided in patients with congenital prolongation of the QTc interval and in those patients with proarrhythmic conditions

Lincosamides • Agents: Clindamycin • Similar mechanism of action to macrolides • Clindamycin is employed primarily in the treatment of infections caused by ANAEROBIC bacteria, such as Bacteroides fragilis, which often causes abdominal infections associated with trauma • It is also significantly active nonenterococcal, gram-positive cocci against

Clindamycin • ADRs: 1) Skin rash 2) Pseudomembranous colitis caused by overgrowth of C. Difficile characterized as potentially fatal diarrhea coupled with abdominal cramping and excretion of blood or mucus • Oral administration of either vancomycin or metronidazole is usually effective in controlling this serious condition 3) Impaired liver function

Chloramphenicol • Chloramphenicol is a broad-spectrum antibiotic that is active against both aerobic and anaerobic gram-positive and gram-negative organisms • It binds reversibly to the 50 S of bacterial ribosomes • It is active also against rickettsiae • The drug is either bactericidal or (more commonly) bacteriostatic, depending on the organism Clinical Uses • Topically in the treatment of eye infections b/c of its broad spectrum and its penetration of ocular tissues and the aqueous humor

Clinical Uses 1) Alternative to a β-lactam antibiotic for treatment of meningococcal meningitis occurring in patients who have major hypersensitivity reactions to penicillin or bacterial meningitis caused by penicillin-resistant strains of pneumococci 2) Treatment of serious rickettsial infections such as typhus Adverse Effects 1) GIT disturbances: NVD 2) Oral or vaginal candidiasis: overgrowth of Candida albicans

Chloramphenicol Adverse Effects 3. Bone Marrow Disturbances: dose-related reversible suppression of RBCs production & aplastic anemia especially in patients with low levels of glucose 6 phosphate dehydrogenase 4. Gray baby syndrom: vomiting, flaccidity, hypothermia, gray color, shock, and collapse • Neonates have a decreased ability to excrete the drug, which accumulates to levels that interfere with the function of mitochondrial ribosomes

Streptogramines • A combination of 2 antibiotics: quinupristin & dalfopristin in a 3: 7 wt/wt ratio • Each component bind to a separate site on the 50 S bacterial ribsome, forming a stable ternary compound (synergistic effect) • Bactericidal primarily against G+ve cocci including those resistant to other antibiotics (MRSA and VERSA) • Bacteriostatic against Enterococcus faecium • Approved for treatment of infections caused by staphylococci or by vancomycin-resistant strains of E faecium

Streptogramines- ADRs 1) Pain at infusion site 2) Arthralgia & myalgia (high doses) 3) Hyperbilirubinema (25% of patients)

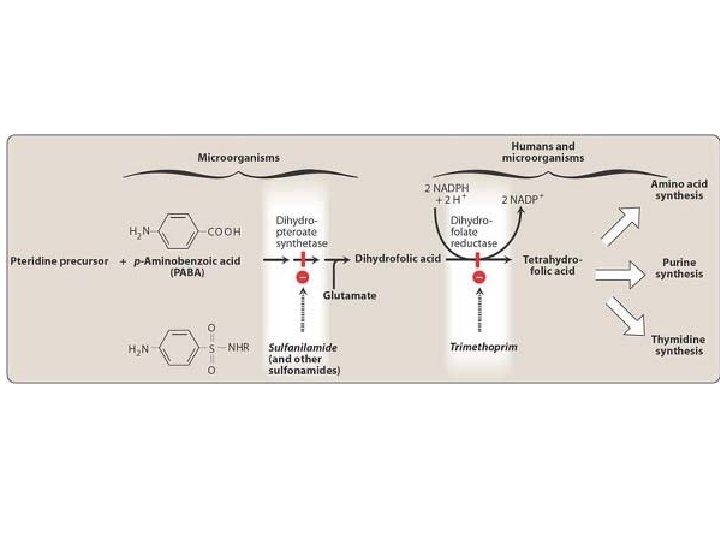
Antimetabolites Sulfonamides and trimethoprim Indirect nucleic acid synthesis inhibitors Sulfonamides: • Bacteriostatic rather than bactericidal • Strctural/ synthetic analogus of p-aminobenzoic acid: they are competitive inhibitors of dihydropteroate synthase • The selective toxicity of sulfonamides results from the inability of mammalian cells to synthesize folic acid; they must use preformed folic acid that is present in the diet
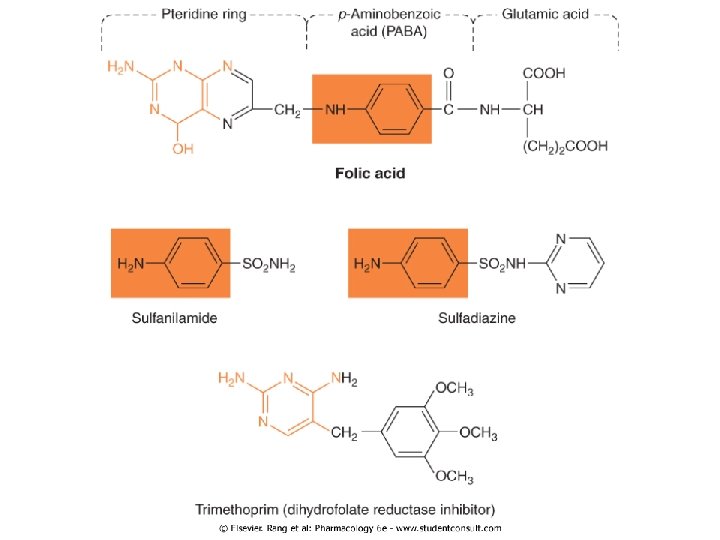

Sulfonamides • The sulfonamides may be classified as: 1. Topical (not commonly b/c of the risk of sensitization) 2. Oral nonabsorbable (Sulfasalazine) 3. Oral absorbable – Short-acting (eg, sulfisoxazole) – Intermediate-acting (eg, sulfamethoxazole) – Long-acting (eg, sulfadoxine)

Sulfonamides – Clinical uses 1) Oral abosrbable: • Toxoplasmosis: Oral sulfadiazine plus pyrimethamine (a DHF reductase inhibitor) plus folinic acid • Simple urinary tract infections: Sulfisoxazole and sulfamethoxazole 2) Topical agents: • • Bacterial conjunctivitis: sodium sulfacetamide ophthalmic solution or ointment Prevention of infection of burn wounds: silver sulfadiazine 3) Oral nonabsorbalbe: ulcerative colitis, rheumatoid arthritis: Oral (sulfasalazine; sulfapyridine and 5 -aminosalicylic acid (5 -ASA))

Sulfonamides – Adverse effects 1) • Hypersensitivity: Allergic reactions, including skin rashes and fever Cross-allergenicity between the individual sulfonamides should be assumed and may also occur with chemically related drugs (eg, oral hypoglycemics, thiazides) 2) GIT: NVD 3) Hematotoxicity: granulocytopenia, aplastic anemia, & thrombocytopenia • Acute hemolysis may occur in persons with glucose-6 -phosphate dehydrogenase deficiency 4. Crystalluria: – Sulfonamides may precipitate in the urine at acidic p. H – Adequate hydration and alkalinization of urine prevent the problem 5. Kernicterus: Sulfonamides can displace bilirubin from plasma proteins & bilirubin is then free to pass into the CNS – Should be avoided in newborns and infants less than 2 months of age as well as in pregnant women at term

Trimethoprim – Mechanism of action • An analog of dihydrofolic acid Trimethoprim is a selective inhibitor of bacterial dihydrofolate reductase that prevents formation of the active tetrahydro- form of folic acid Bacterial dihydrofolate reductase is four to five orders of magnitude more sensitive to inhibition by trimethoprim than the mammalian enzyme Alone can be given in acute urinary tract infections • Adverse effects may cause the predictable adverse effects of an antifolate drug, including 1) Megaloblastic anemia 2) Leukopenia 3) Granulocytopenia • These effects are usually ameliorated by supplementary folinic acid

Trimtethoprim plus sulfamethoxazole (TMPSMZ) • The combination of trimethoprim with sulfamethoxazole is called cotrimoxazole • When the two drugs are used in combination, antimicrobial synergy results from the sequential blockade of folate synthesis • The drug combination is bactericidal against susceptible organisms • Adverse effects • The combination of trimethoprim-sulfamethoxazole may cause any of the adverse effects associated with the sulfonamides • AIDS patients given TMP-SMZ have a high incidence of adverse effects, including fever, rashes, leukopenia, and diarrhea.
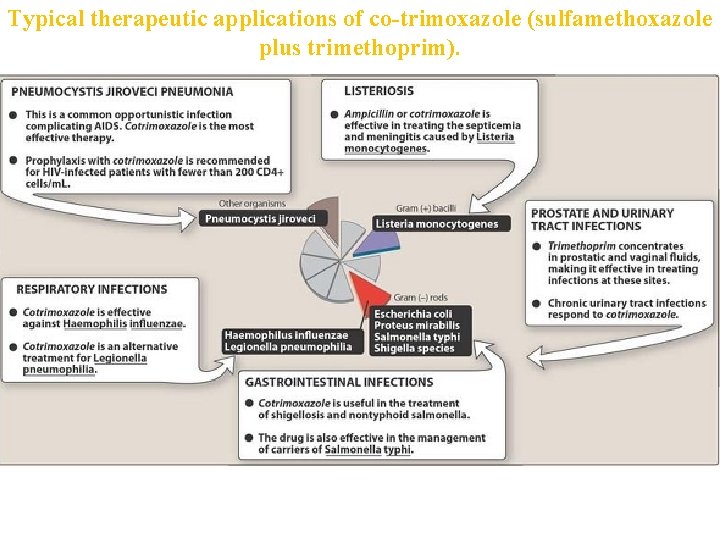
Typical therapeutic applications of co-trimoxazole (sulfamethoxazole plus trimethoprim).

Quinolone Antimetabolite Fluoroquinolones Ciprofloxacin Norflxacin Moxifloxacin Levofloxacin Direct nucleic acid synthesis inhibitors

Fluoroquinolones • Broad-spectrum antibiotic effective against a variety of gram-positive and gram-negative bacteria • They block bacterial DNA synthesis by inhibiting bacterial: 1) Topoisomerase II (DNA gyrase): prevents the relaxation of positively supercoiled DNA that is required for normal transcription and replication 2) Topoisomerase IV: interferes with separation of replicated chromosomal DNA into the respective daughter cells during cell division • Originally developed because of their excellent activity against gram-negative aerobic bacteria; they had limited activity against gram-positive organisms

Fluoroquinolones • Several newer agents have improved activity against gram-positive cocci • The relative activity against gram-negative vs. gram-positive species is useful for classification of these agents. • Clinical uses • UTI even when caused by multidrug-resistant bacteria, eg, pseudomonas • Bacterial diarrhea caused by shigella, salmonella, toxigenic E coli, and campylobacter

Clinical uses • Infections of soft tissues, bones, and joints and in intraabdominal and respiratory tract infections, including those caused by multidrug-resistant organisms such as pseudomonas and enterobacter (except norfloxacin) • Quinolones – Clinical uses • Ciprofloxacin is a DOC for prophylaxis and treatment of anthrax • Ciprofloxacin and levofloxacin are effective in treating chlamydial urethritis or cervicitis • Ciprofloxacin, levofloxacin, or moxifloxacin is occasionally used for treatment of tuberculosis and atypical mycobacterial infections • Respiratory fluoroquinolones (e. g. levofloxacin, gatifloxacin, gemifloxacin, & moxifloxacin): upper and lower RTI

Adverse effects 1) GIT: NVD (most common) 2) CNS: headache and dizziness or light-headedness 3) Phototxicity 4) Connective tissue problems: may damage growing cartilage and cause an arthropathy. Thus, these drugs are not routinely recommended for patients under 18 years of age 5) QT interval prolongation: with gatifloxacin, levofloxacin, gemifloxacin, and moxifloxacin
- Slides: 70