ANNOUNCEMENTS Exams will be handed back in lab

ANNOUNCEMENTS Exams will be handed back in lab next week. Quick “open-book” quiz at end of class today.

Lab Information Metal p. H hydroxide solubility titration (on Friday)

An equilibrium will shift in response to change in the system or its environment Le. Chatelier’s Principle If a chemical system at equilibrium experiences a change in concentration, temperature, volume, or partial pressure, then the equilibrium shifts to counteract the imposed change and a new equilibrium is established.
![The effect of concentration changes on equilibria Add product [C] Remove reactant [A] Remove The effect of concentration changes on equilibria Add product [C] Remove reactant [A] Remove](http://slidetodoc.com/presentation_image_h2/e574822b344225ae1b65b94e8f5d8103/image-4.jpg)
The effect of concentration changes on equilibria Add product [C] Remove reactant [A] Remove all of reactant [B] and product [D]
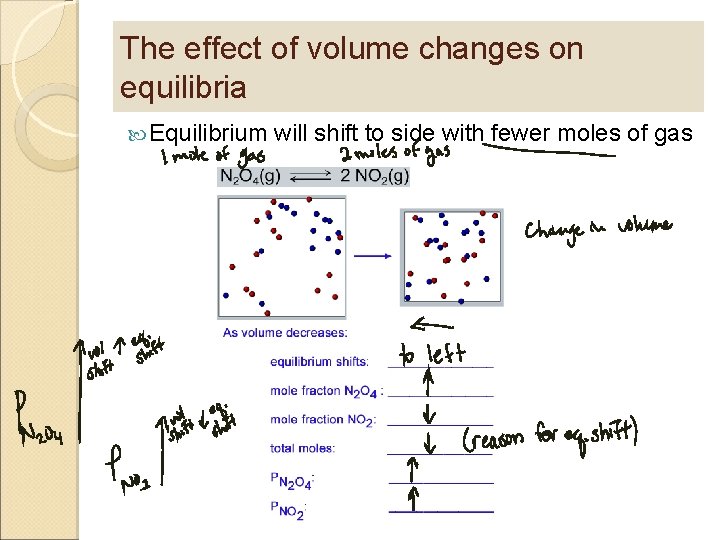
The effect of volume changes on equilibria Equilibrium will shift to side with fewer moles of gas

The effect of temperature changes on equilibria Endothermic Exothermic reactions

Le. Chatelier’s Principle- Quantitative 1 -Liter flask contains 0. 50 mol butane and 1. 25 mol isobutane (at equilibrium). We then add 0. 50 mol and butane. What happens and what are the final concentrations of isobutane and butane?

Chapter 16 ACID-BASE EQUILIBRIA

Acids and Bases- Overview • • • p. H and p. OH Relationship of conjugate pair acid-base strength. When acids or bases control p. H: Ø determine K Ø predict p. H When p. H controls acid/base state: Ø predict acid/base state Ø use acid/base state to determine p. H Buffer action p. H titration curves
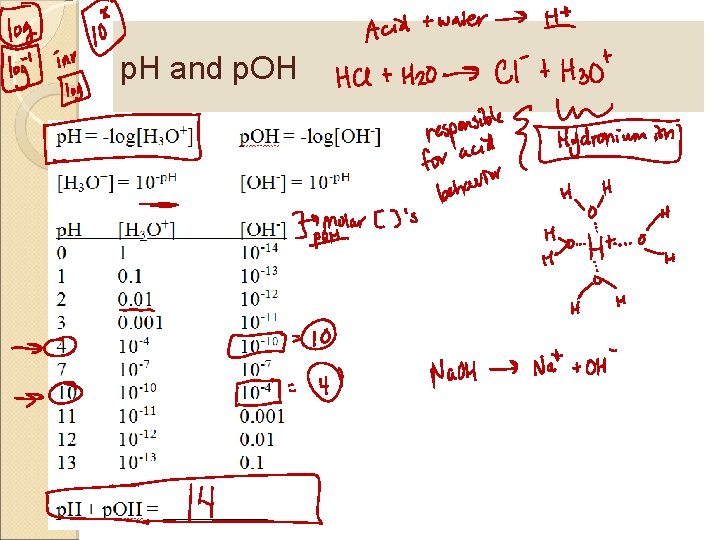
p. H and p. OH

Water autoionization
![p. H and p. OH calculations [H 3 O+][OH-] = 1. 0 x 10 p. H and p. OH calculations [H 3 O+][OH-] = 1. 0 x 10](http://slidetodoc.com/presentation_image_h2/e574822b344225ae1b65b94e8f5d8103/image-12.jpg)
p. H and p. OH calculations [H 3 O+][OH-] = 1. 0 x 10 -14 p. H = -log[H 3 O+] = 10 -p. H + p. OH = 14. 00 p. OH = -log[OH-] = 10 -p. H What is the p. H of [H 3 O+] = 4. 6 x 10 -5? What is the p. OH of [OH-] = 3. 3 x 10 -4? What is the p. OH of [H 3 O+] = 4. 6 x 10 -5? What is the [H 3 O+] when the p. H = 4. 2?
![What is the p. H when [H 3 O+] = 0. 045 M? 0. What is the p. H when [H 3 O+] = 0. 045 M? 0.](http://slidetodoc.com/presentation_image_h2/e574822b344225ae1b65b94e8f5d8103/image-13.jpg)
What is the p. H when [H 3 O+] = 0. 045 M? 0. 045 2. 13. 995 3. -1. 35 4. 1. 35 1.
![What is the p. H when [OH-] = 6. 5 x 10 -4 M? What is the p. H when [OH-] = 6. 5 x 10 -4 M?](http://slidetodoc.com/presentation_image_h2/e574822b344225ae1b65b94e8f5d8103/image-14.jpg)
What is the p. H when [OH-] = 6. 5 x 10 -4 M? 3. 18 2. -3. 18 3. 10. 81 4. 15. 18 1.
![What is the [H 3 O+] when p. H = 3. 66 M? 2. What is the [H 3 O+] when p. H = 3. 66 M? 2.](http://slidetodoc.com/presentation_image_h2/e574822b344225ae1b65b94e8f5d8103/image-15.jpg)
What is the [H 3 O+] when p. H = 3. 66 M? 2. 18 x 10 -4 2. 10. 34 3. 4570 4. 3. 66 1.
![What is the [OH-] when p. H = 5. 84 M? 1. 45 x What is the [OH-] when p. H = 5. 84 M? 1. 45 x](http://slidetodoc.com/presentation_image_h2/e574822b344225ae1b65b94e8f5d8103/image-16.jpg)
What is the [OH-] when p. H = 5. 84 M? 1. 45 x 108 2. 3. 31 x 10 -6 3. 6. 92 x 10 -9 4. 5. 84 1.
![Strong acids and bases ionize completely Strong monoprotic acids [H 3 O+] = [acid] Strong acids and bases ionize completely Strong monoprotic acids [H 3 O+] = [acid]](http://slidetodoc.com/presentation_image_h2/e574822b344225ae1b65b94e8f5d8103/image-17.jpg)
Strong acids and bases ionize completely Strong monoprotic acids [H 3 O+] = [acid] Strong diprotic acid [H 3 O+] = 2[acid] Strong monoprotic base [OH-] = [base] Strong diprotic base [OH-] = 2[base]

QUIZ TIME!
- Slides: 18