ANNOUNCEMENTS Arnold Orville Beckman April 10 1900 May

ANNOUNCEMENTS Arnold Orville Beckman (April 10, 1900 – May 18, 2004) • Invented the p. H meter in 1934 (because of lemons!) • Founded Silicon Valley (located in Palo Alto so that it was near his friend’s aging mother) • Was a Marine in WWI
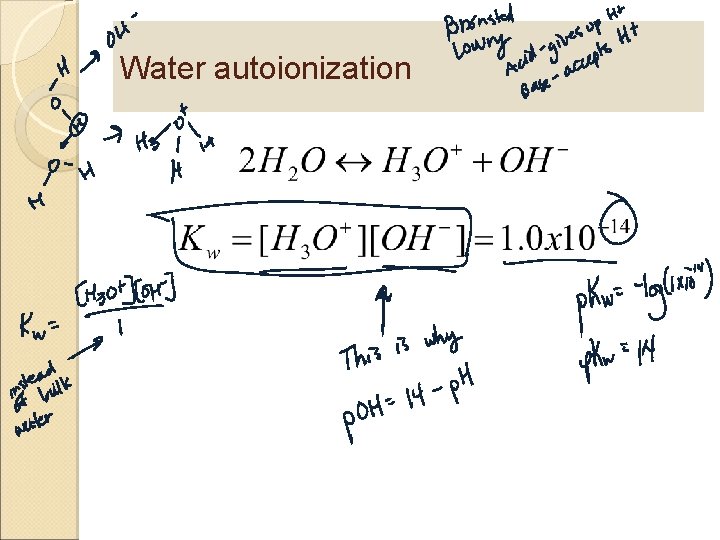
Water autoionization
![p. H and p. OH calculations [H 3 O+][OH-] = 1. 0 x 10 p. H and p. OH calculations [H 3 O+][OH-] = 1. 0 x 10](http://slidetodoc.com/presentation_image_h2/b22f6683179866acaebf1e7fc8e983c3/image-4.jpg)
p. H and p. OH calculations [H 3 O+][OH-] = 1. 0 x 10 -14 p. H = -log[H 3 O+] = 10 -p. H + p. OH = 14. 00 p. OH = -log[OH-] = 10 -p. H What is the p. H of [H 3 O+] = 4. 6 x 10 -5? What is the p. OH of [OH-] = 3. 3 x 10 -4? What is the p. OH of [H 3 O+] = 4. 6 x 10 -5? What is the [H 3 O+] when the p. H = 4. 2?
![The p. H Square p. H [H 3 O+] p. H+p. OH=14 [H 3 The p. H Square p. H [H 3 O+] p. H+p. OH=14 [H 3](http://slidetodoc.com/presentation_image_h2/b22f6683179866acaebf1e7fc8e983c3/image-5.jpg)
The p. H Square p. H [H 3 O+] p. H+p. OH=14 [H 3 O+]][OH-]=10 -14 p. OH [OH-]
![What is the p. H when [H 3 O+] = 0. 045 M? 0. What is the p. H when [H 3 O+] = 0. 045 M? 0.](http://slidetodoc.com/presentation_image_h2/b22f6683179866acaebf1e7fc8e983c3/image-6.jpg)
What is the p. H when [H 3 O+] = 0. 045 M? 0. 045 2. 13. 995 3. -1. 35 4. 1. 35 1.
![What is the p. H when [OH-] = 6. 5 x 10 -4 M? What is the p. H when [OH-] = 6. 5 x 10 -4 M?](http://slidetodoc.com/presentation_image_h2/b22f6683179866acaebf1e7fc8e983c3/image-7.jpg)
What is the p. H when [OH-] = 6. 5 x 10 -4 M? 3. 18 2. -3. 18 3. 10. 81 4. 15. 18 1.
![What is the [OH-] when p. H = 5. 84 M? 1. 45 x What is the [OH-] when p. H = 5. 84 M? 1. 45 x](http://slidetodoc.com/presentation_image_h2/b22f6683179866acaebf1e7fc8e983c3/image-8.jpg)
What is the [OH-] when p. H = 5. 84 M? 1. 45 x 108 2. 3. 31 x 10 -6 3. 6. 92 x 10 -9 4. 5. 84 1.
![Strong acids and bases ionize completely Strong monoprotic acids [H 3 O+] = [acid] Strong acids and bases ionize completely Strong monoprotic acids [H 3 O+] = [acid]](http://slidetodoc.com/presentation_image_h2/b22f6683179866acaebf1e7fc8e983c3/image-9.jpg)
Strong acids and bases ionize completely Strong monoprotic acids [H 3 O+] = [acid] Strong diprotic acid [H 3 O+] = 2[acid] Strong monoprotic base [OH-] = [base] Strong diprotic base [OH-] = 2[base]

Calculating the p. H of a strong acid solution What is the p. H of a 0. 150 M solution of HCl?

Changing the p. H of a solution is harder the farther away you are from neutral p. H HCl molar mass=36. 46 g/mol How much acid does it take to go from p. H=1 to p. H=2? How much acid does it take to go from p. H=5 to p. H=6?

What is the real range of p. H’s that are possible? Can p. H be zero? Can p. H be negative? Can p. H be greater than 14?

Conjugate Acid-Base Pairs Different definitions of acids and bases Brønsted-Lowry ◦ Acid = H+ (proton) donor ◦ Base = H+ (proton) acceptor ◦ Acid-base reaction involves proton transfer ◦ Conjugate acid/base = what is left on the other side after the proton has been transferred Acid Conjugate base HF F- NH 4+ NH 3
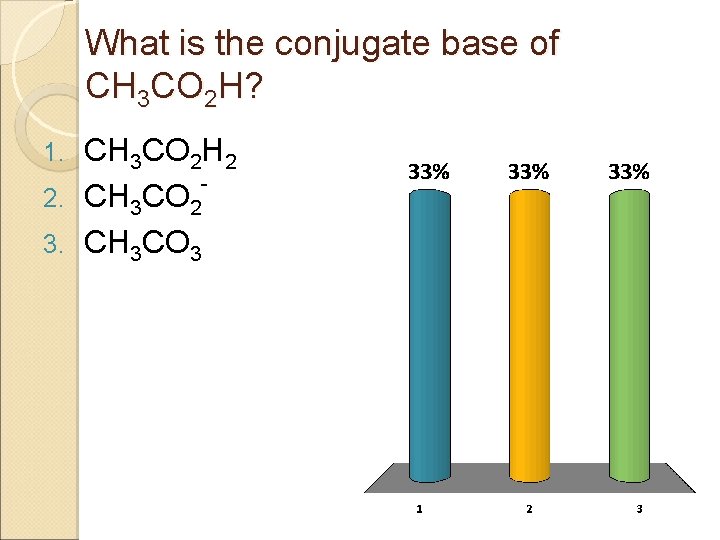
What is the conjugate base of CH 3 CO 2 H? CH 3 CO 2 H 2 2. CH 3 CO 2 3. CH 3 CO 3 1.
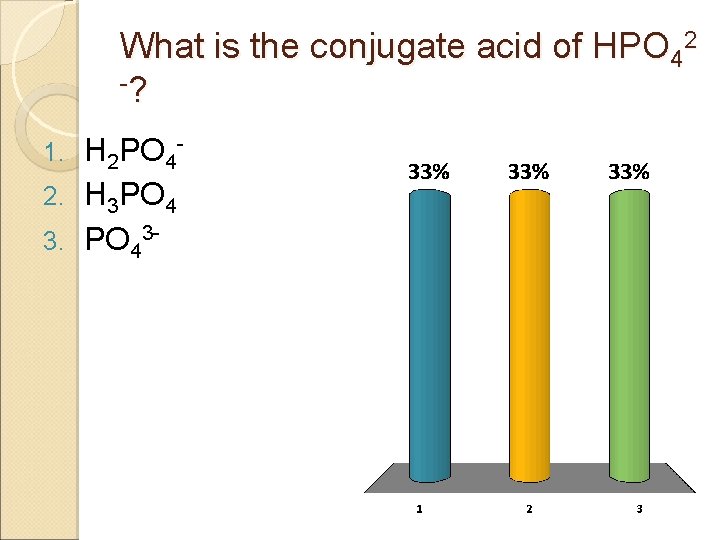
What is the conjugate acid of HPO 42 -? H 2 PO 42. H 3 PO 4 3. PO 431.
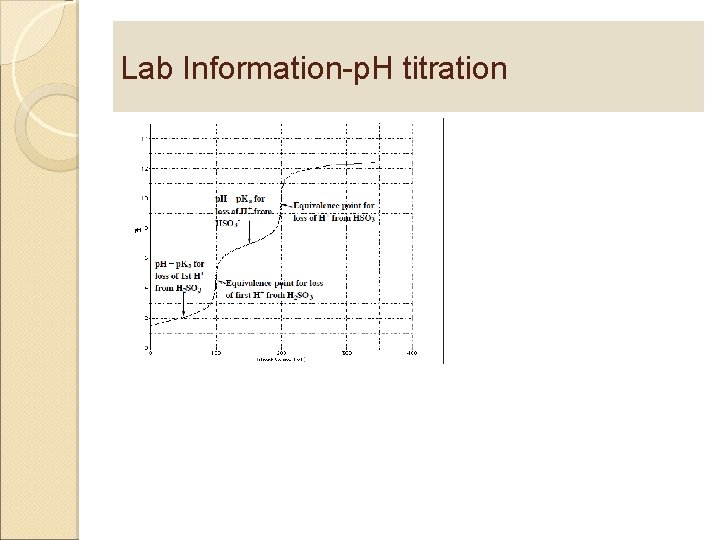
Lab Information-p. H titration

Acid (or base) strength depends on the equilibrium constant
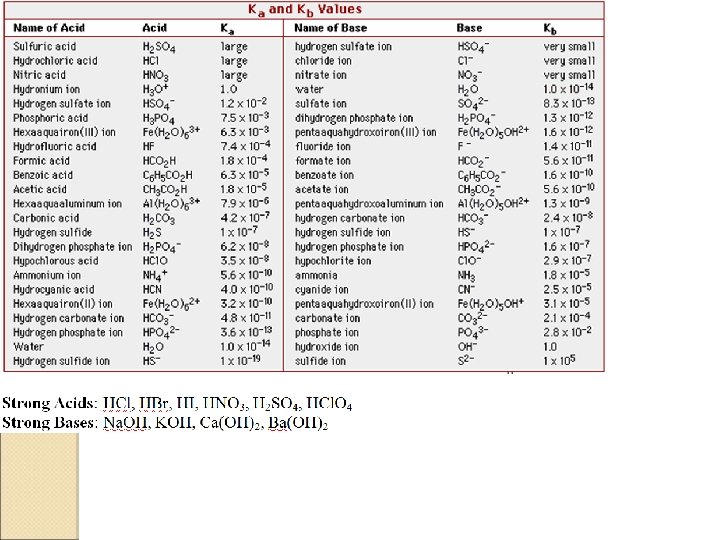

p. H Measurements are used to determine acid strength (Ka) A 0. 10 M solution of oxalic acid has a p. H of 1. 48. What is the Ka value for oxalic acid?

What is the value of Ka for an acid, HA, for which a 0. 240 M solution has a p. H of 3. 28? Is this acid stronger or weaker than oxalic acid? (Ka=0. 016)

Determining Kb from p. H data A 0. 300 M solution of a weak base has a p. H of 9. 20. What is Kb?
Have a great weekend!
- Slides: 22