Announcements 92210 HW solutions 1 8 posted but
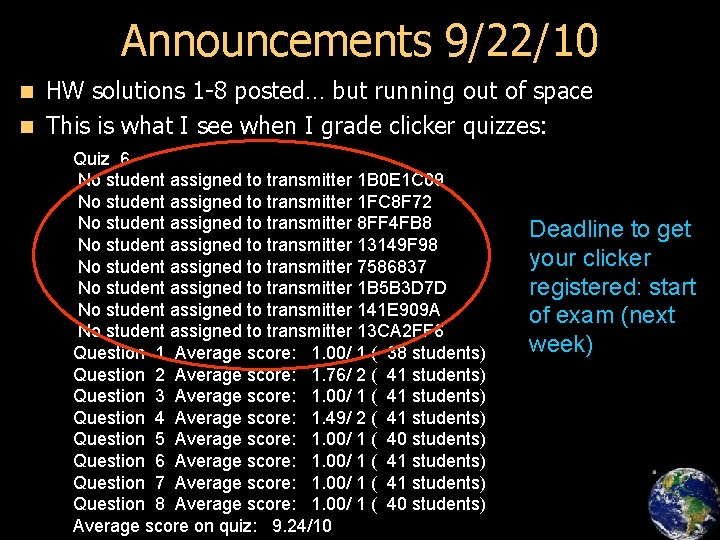
Announcements 9/22/10 HW solutions 1 -8 posted… but running out of space n This is what I see when I grade clicker quizzes: n Quiz 6 No student assigned to transmitter 1 B 0 E 1 C 09 No student assigned to transmitter 1 FC 8 F 72 No student assigned to transmitter 8 FF 4 FB 8 No student assigned to transmitter 13149 F 98 No student assigned to transmitter 7586837 No student assigned to transmitter 1 B 5 B 3 D 7 D No student assigned to transmitter 141 E 909 A No student assigned to transmitter 13 CA 2 FF 6 Question 1 Average score: 1. 00/ 1 ( 38 students) Question 2 Average score: 1. 76/ 2 ( 41 students) Question 3 Average score: 1. 00/ 1 ( 41 students) Question 4 Average score: 1. 49/ 2 ( 41 students) Question 5 Average score: 1. 00/ 1 ( 40 students) Question 6 Average score: 1. 00/ 1 ( 41 students) Question 7 Average score: 1. 00/ 1 ( 41 students) Question 8 Average score: 1. 00/ 1 ( 40 students) Average score on quiz: 9. 24/10 Deadline to get your clicker registered: start of exam (next week)

Quick writing n When I expand a gas from one volume to another, the final pressure will be greater if it is expanded isothermally than if it is expanded adiabatically. Explain why without using equations.

Special Processes: Review n Constant volume n Constant pressure n Constant temperature n No heat added n General process? – – – Eint = ? Won gas = ? Qadded = ?

Last Special Case: Cyclical Processes P State 1 V n Eint =? n net Wby gas or Won gas? |W| = ? n Qadded = ?
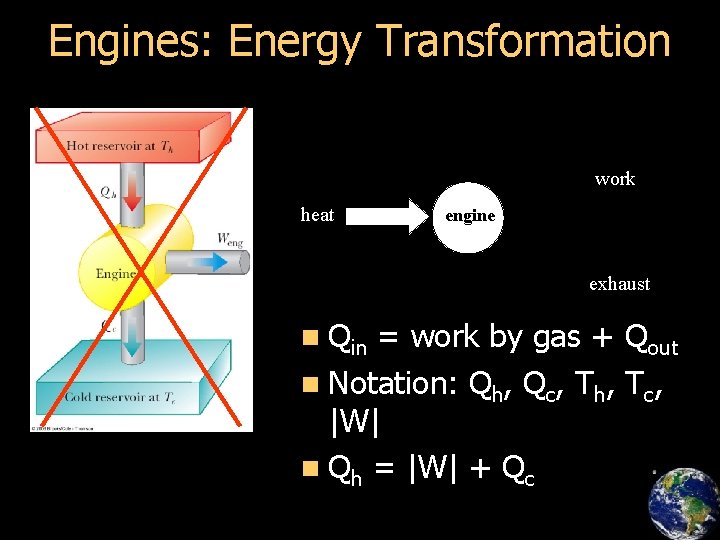
Engines: Energy Transformation work heat engine exhaust n Qin = work by gas + Qout n Notation: Qh, Qc, Th, Tc, |W| n Qh = |W| + Qc
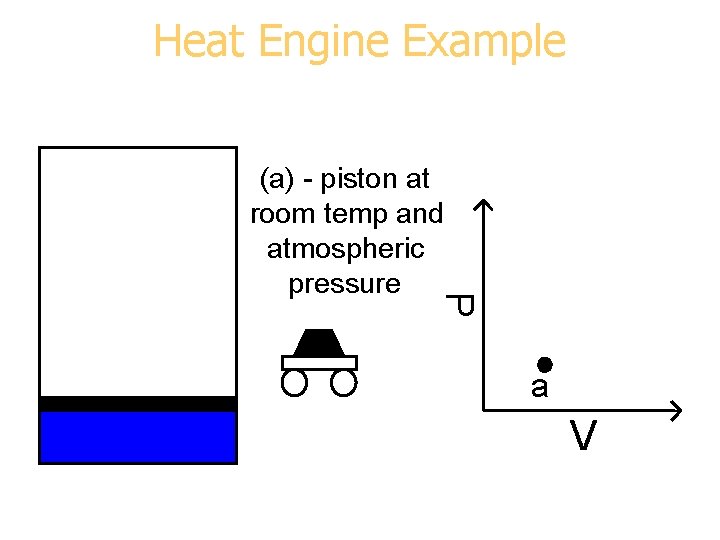
Heat Engine Example P (a) - piston at room temp and atmospheric pressure a V
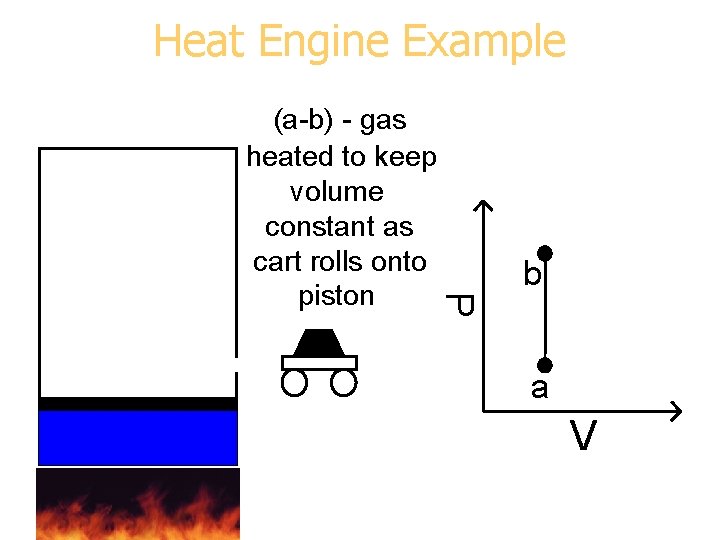
Heat Engine Example P (a-b) - gas heated to keep volume constant as cart rolls onto piston b a V
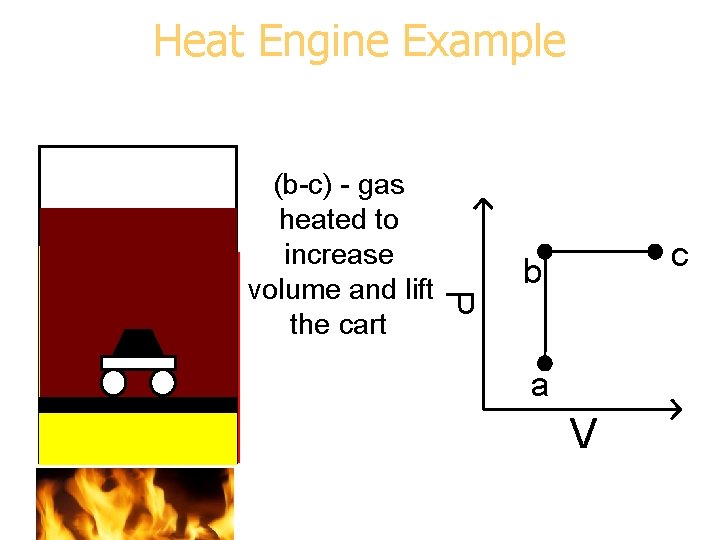
Heat Engine Example P (b-c) - gas heated to increase volume and lift the cart c b a V
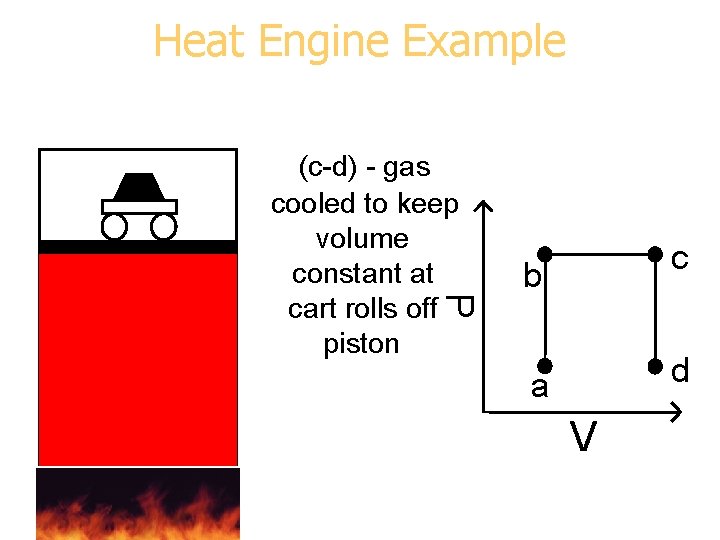
Heat Engine Example P (c-d) - gas cooled to keep volume constant at cart rolls off piston b c a d V
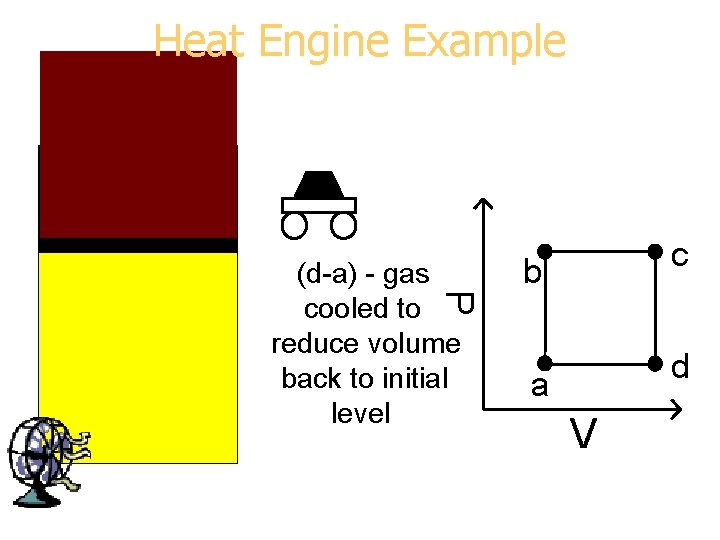
Heat Engine Example P (d-a) - gas cooled to reduce volume back to initial level b c a d V

Efficiency n How good is your engine? n Definition

Worked Problem n. A car engine produces power of 5000 W, at 20 cycles/second. Its efficiency is 20%. What are |W|, Qh, and Qc per cycle? n What do those quantities represent?

Reading Quiz (graded) n What is the name we use for the cycle which models how a gas engine works? – – – Carnot cycle Brayton cycle Diesel cycle Otto cycle Tri cycle
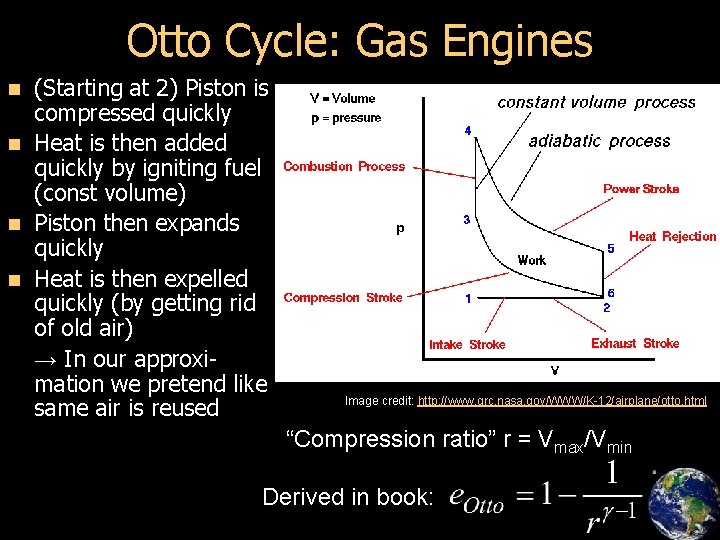
Otto Cycle: Gas Engines (Starting at 2) Piston is compressed quickly n Heat is then added quickly by igniting fuel (const volume) n Piston then expands quickly n Heat is then expelled quickly (by getting rid of old air) → In our approximation we pretend like same air is reused n Image credit: http: //www. grc. nasa. gov/WWW/K-12/airplane/otto. html “Compression ratio” r = Vmax/Vmin Derived in book:

Thought Question n If I replaced all of the nitrogen (N 2) in the air with carbon dioxide (CO 2), what do you think would happen to the efficiency of car engines? – – – They would get more efficient They would get less efficient The efficiency would not change

Diesel Cycle: Diesel Engines n What’s the main difference between gas and diesel engines? n Change to our PV-diagram model n Diesel cycle details… done in HW problem

Worked problem: Class designed n Make up a “three-legged cycle”. n What is the efficiency of this cycle? n Game plan: – – Calculate Q for each leg Calculate Qin, Qout, |W| e = |W|/Qin Test to make sure e < emax
- Slides: 17