Anneline Nansen Department of Infectious Disease Immunology Statens

















- Slides: 31

Anneline Nansen Department of Infectious Disease Immunology Statens Serum Institut (SSI) STATENS SERUM INSTITUT DNA Vaccination

A vaccine is a substance that stimulates an immune response that can either prevent an infection or create resistance to an infection STATENS SERUM INSTITUT What is a vaccine?

• Live vaccines • Are able to replicate in the host • Attenuated (weakened) so they do not cause disease • Whole killed vaccines • Subunit vaccines • Part of organism (protein, inactivated toxins) • Genetic Vaccines • Part of genes from organism STATENS SERUM INSTITUT What are the different types of vaccines?

Genetic Vaccines • Injected (Naked) (Intra muscular, i. m. ) • Delivered by Gene gun. Naked DNA Coated on gold particles • Carried by recombinant live vectors: • Vaccinia, adenovirus, or alphaviruses • Intracellular bacteria • Advantages • Easy to produce • Induce cellular (CD 4+T cells and CTL’s) and humoral responses • Disadvantages • Often weak primary responses-need for a boost STATENS SERUM INSTITUT • Introduce DNA or RNA into the host

Genetic Vaccines Live-attenuated or killed Vaccines are not applicable Because: • If there were a manufacturing error and the HIV is not properly killed or attenuated, the poorly-made vaccine could infect people with HIV • Also, because HIV is so highly mutating, there is concern it might be able to mutate out of attenuation and cause disease. • Cancer • A variety of infectious diseases • Tuberculosis • Malaria • HCV STATENS SERUM INSTITUT • HIV

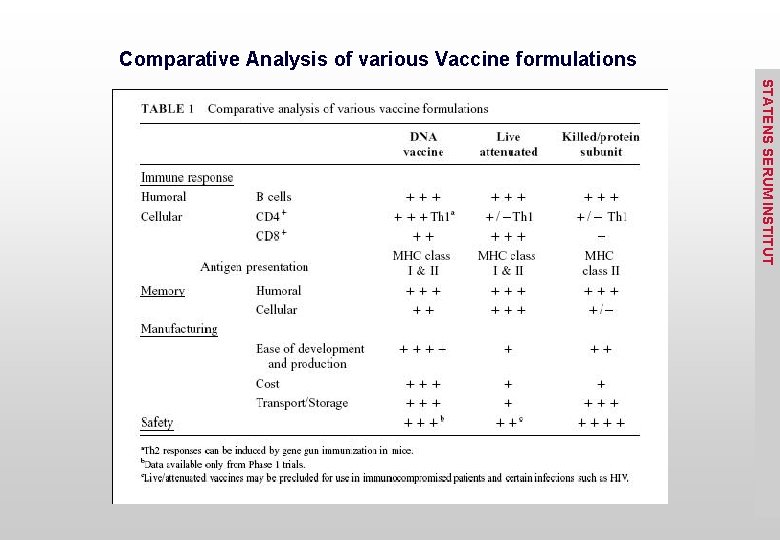
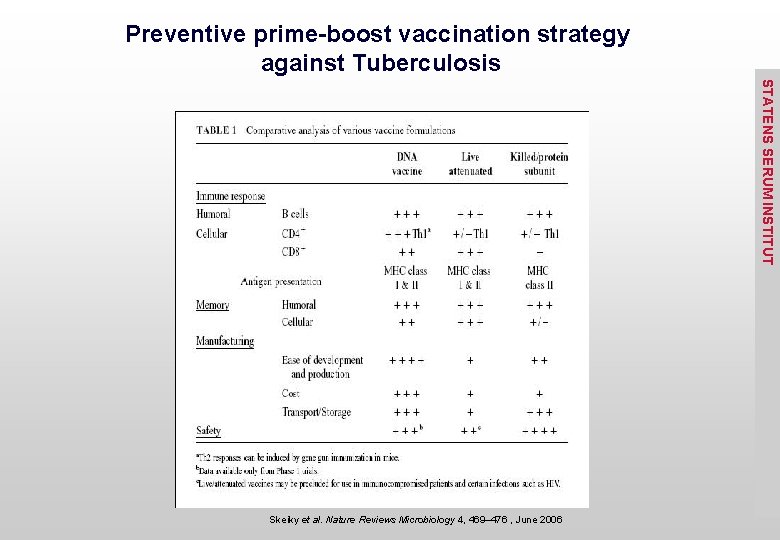
Comparative Analysis of various Vaccine formulations STATENS SERUM INSTITUT

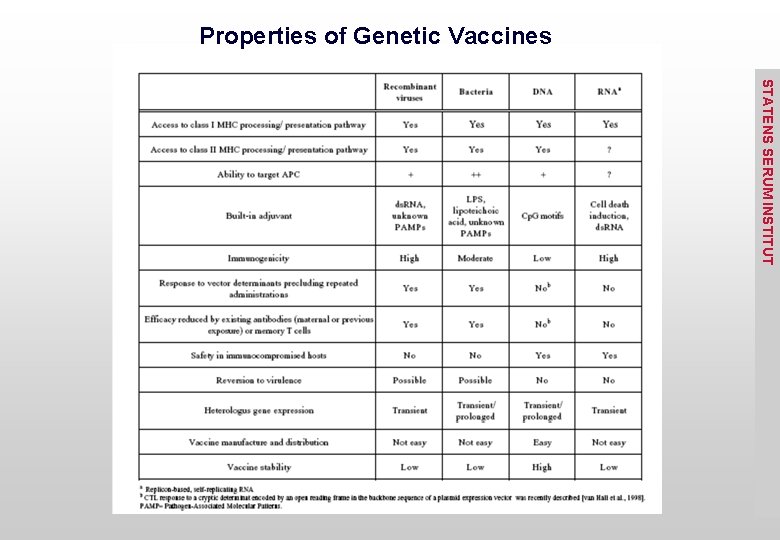
Properties of Genetic Vaccines STATENS SERUM INSTITUT

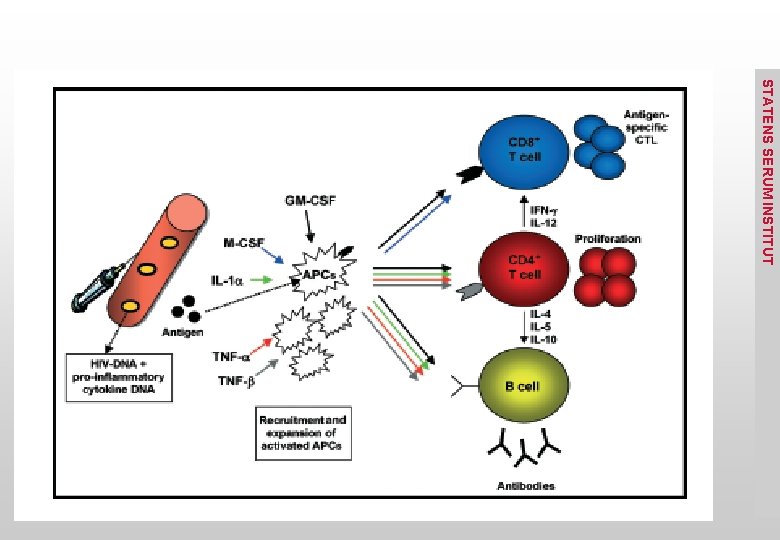
• Pick Genes, epitope(s), of relevance for protection against the disease of interest • Has to be immunogenic in the host • Select a plasmid an expression system • Optimize for expression in eukaryotic cells • Promotor optimization • Synthetic genes with optimized codon usage • Optimize immunogenicity • Insert multiple Cp. G motifs (TLR ligand) • IL-12, IL-15 others… STATENS SERUM INSTITUT DNA Vaccine Design

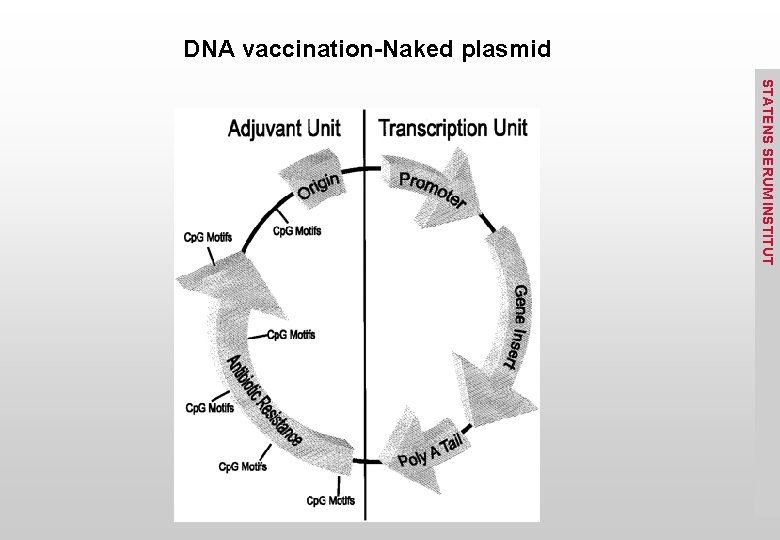
DNA vaccination-Naked plasmid STATENS SERUM INSTITUT

• By Gene Gun • Small amounts of DNA • Th 2 biased immune response • i. m injection • Large amounts of DNA • Th 1 biased immune response STATENS SERUM INSTITUT Delivery of Naked DNA

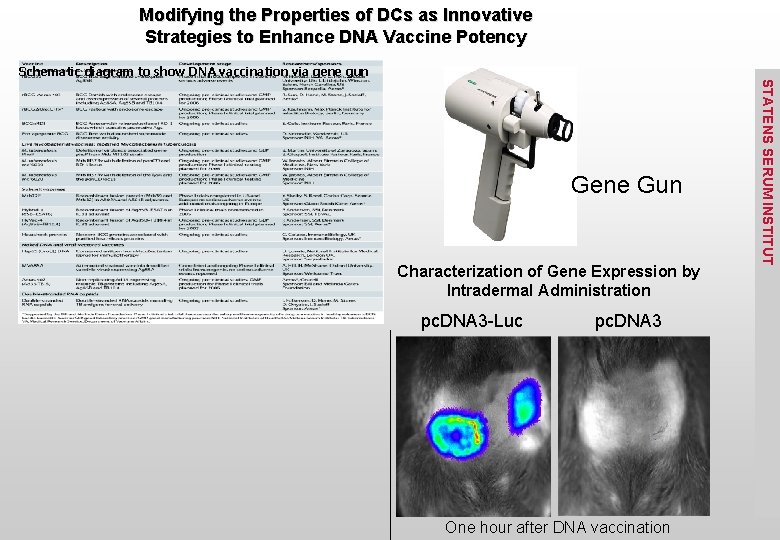
The “gene gun” STATENS SERUM INSTITUT The Helios Gene Gun is a new way for in vivo transformation of cells or organisms (i. e. gene therapy and genetic immunization (DNA vaccination)). This gun uses Biolistic ® particle bombardment where DNA- or RNAcoated gold particles are loaded into the gun and you pull the trigger. A low pressure helium pulse delivers the coated gold particles into virtually any target cell or tissue. The particles carry the DNA so that you do not have to remove cells from tissue in order to transform the cells.

STATENS SERUM INSTITUT

Modifying the Properties of DCs as Innovative Strategies to Enhance DNA Vaccine Potency Schematic diagram to show DNA vaccination via gene gun Characterization of Gene Expression by Intradermal Administration pc. DNA 3 -Luc pc. DNA 3 One hour after DNA vaccination STATENS SERUM INSTITUT Gene Gun

Strategies to Enhance DNA Vaccine Potency Employment of intercellular spreading strategies to increase the number of antigen presenting cells that present antigens encoded by DNA vaccines. Employment of Anti-apoptotic strategies to prolong life span of antigen presenting cells that present antigens encoded by DNA vaccines STATENS SERUM INSTITUT Employment of intracellular sorting signals to improve antigen processing through MHC class I and II pathways.

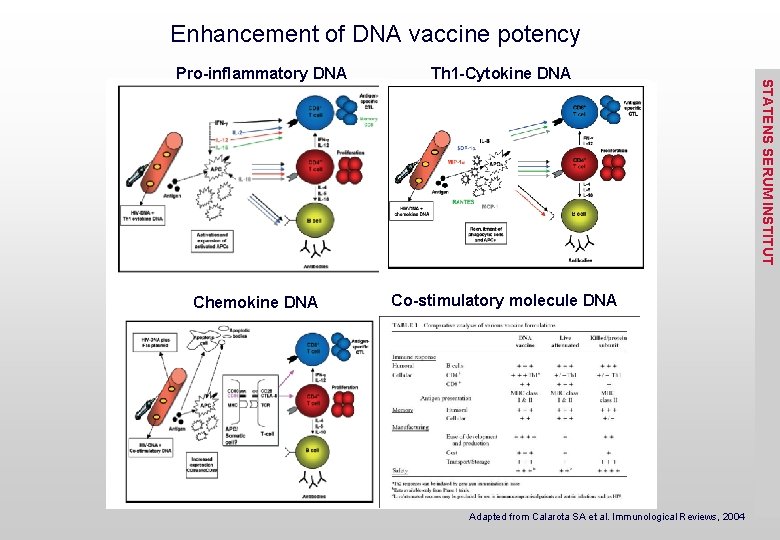
Enhancement of DNA vaccine potency Chemokine DNA Th 1 -Cytokine DNA Co-stimulatory molecule DNA Adapted from Calarota SA et al. Immunological Reviews, 2004 STATENS SERUM INSTITUT Pro-inflammatory DNA

Molecular interactions that contribute to the recruitment, activation, or maturation of DCs in DNA vaccine studies STATENS SERUM INSTITUT Kutzler, M. A. et al. J. Clin. Invest. 2004; 114: 1241 -1244

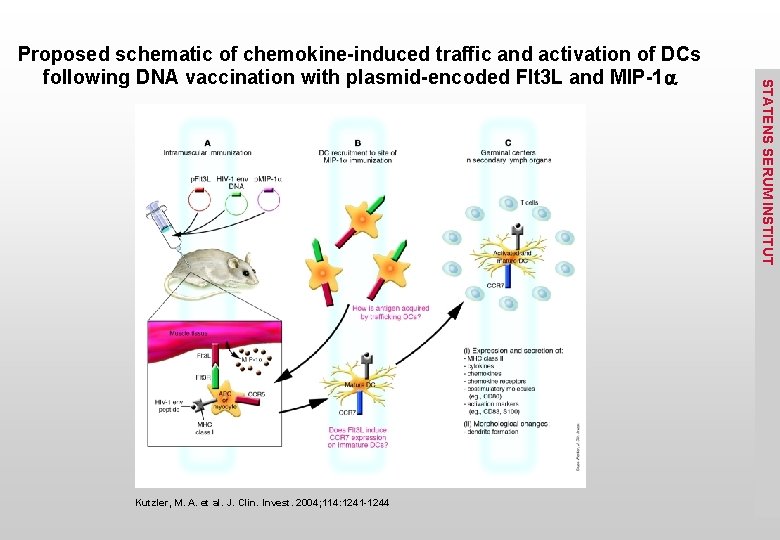
Kutzler, M. A. et al. J. Clin. Invest. 2004; 114: 1241 -1244 STATENS SERUM INSTITUT Proposed schematic of chemokine-induced traffic and activation of DCs following DNA vaccination with plasmid-encoded Flt 3 L and MIP-1 a

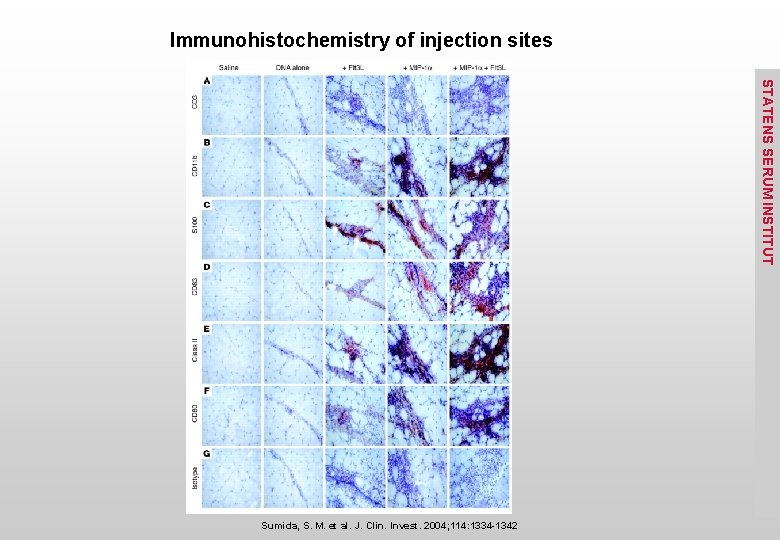
Immunohistochemistry of injection sites STATENS SERUM INSTITUT Sumida, S. M. et al. J. Clin. Invest. 2004; 114: 1334 -1342

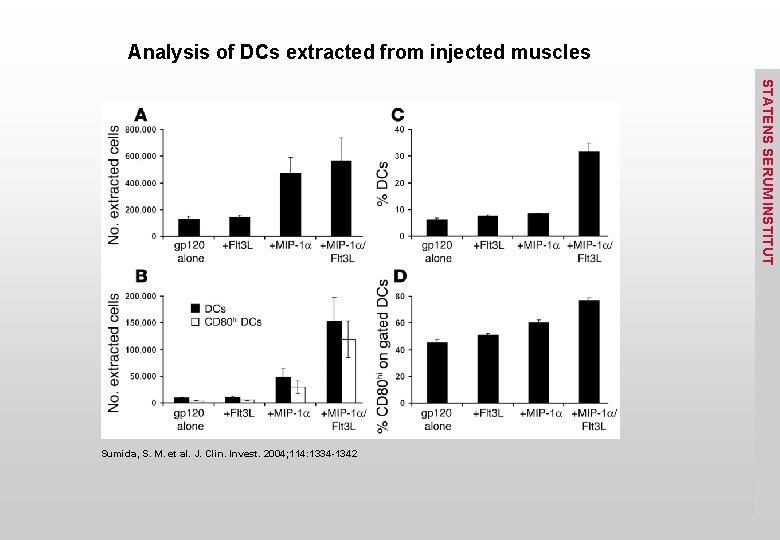
Analysis of DCs extracted from injected muscles STATENS SERUM INSTITUT Sumida, S. M. et al. J. Clin. Invest. 2004; 114: 1334 -1342

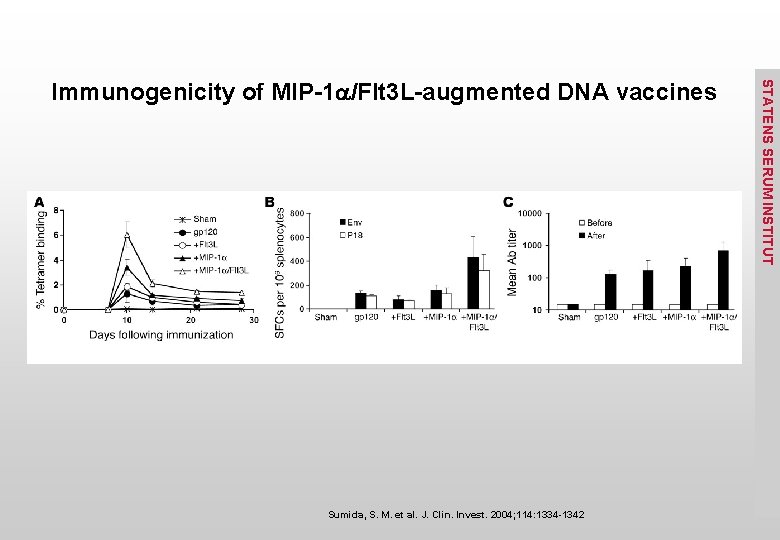
Sumida, S. M. et al. J. Clin. Invest. 2004; 114: 1334 -1342 STATENS SERUM INSTITUT Immunogenicity of MIP-1 a/Flt 3 L-augmented DNA vaccines

STATENS SERUM INSTITUT DNA vaccination by use of live recombinant viruses

Examples of live viral vectors • Adenoviruses • 49 immunologically distinct adenoviral types (serotypes) • Infect many cells types including APC’s • Induce potent CTL responses • Pre-existing immunity against the vector, because of naturally occuring infections • Avipoxviruses • Fowlpox • Not a natural human pathogen- no pre-existing immunity STATENS SERUM INSTITUT • Poxviruses • Vaccinia Virus (VV) • Modified Vaccinia Virus Ankara • MVA replication deficient (very safe, even in immodeficient individuals) • Pre-existing immunity, because VV is used as vaccine against Small Pox

Kinetics of an immune response after a single immunisation with a viral vector or after Prime boost STATENS SERUM INSTITUT Single prime Homologous Prime-Boost Heterologous Prime-Boost Adapted from Rocha CD et al. Int Microbiol, 2004

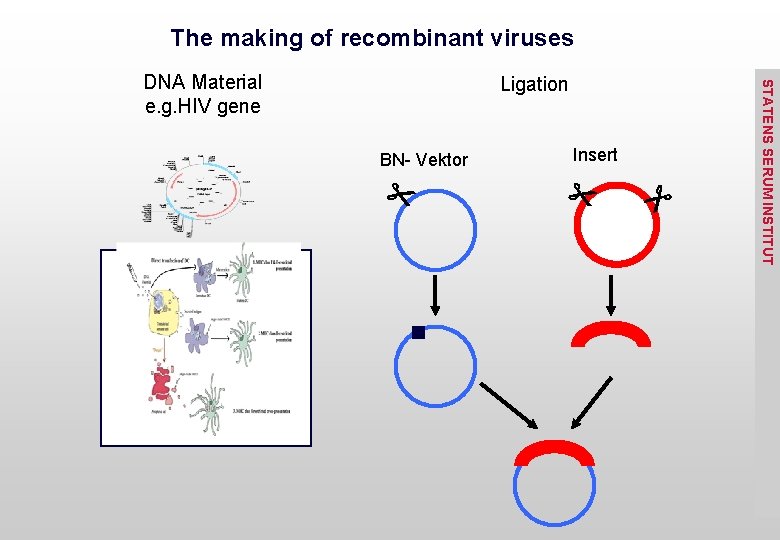
The making of recombinant viruses Ligation Insert BN- Vektor STATENS SERUM INSTITUT DNA Material e. g. HIV gene

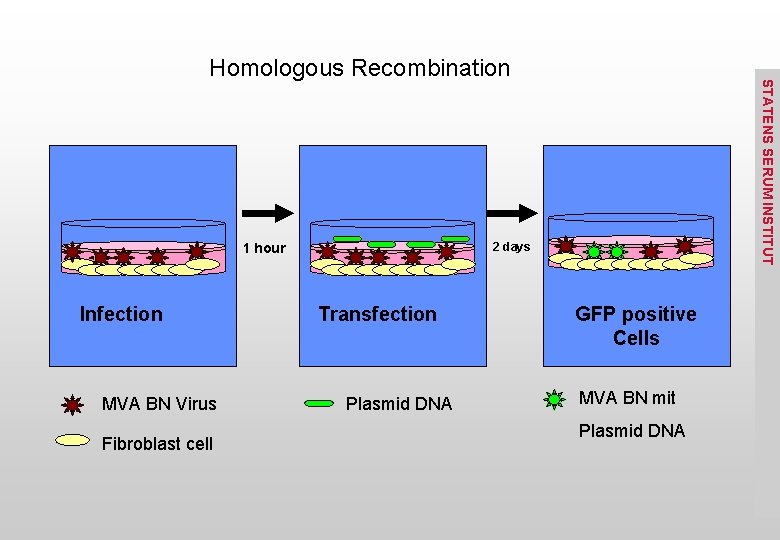
Homologous Recombination STATENS SERUM INSTITUT Adapted from Rocha CD et al. Int Microbiol, 2004

2 days 1 hour Infection MVA BN Virus Fibroblast cell STATENS SERUM INSTITUT Homologous Recombination Transfection Plasmid DNA GFP positive Cells MVA BN mit Plasmid DNA

Naked DNA and Protein • Possible to prime several times, no immunity • Best results if DNA or protein before live viral vector Recombinant Viruses • Only one go-because of immunity against the vector after priming • Often used as a Booster Vaccine • Possible to use different recombinant vectors as prime-boost STATENS SERUM INSTITUT Prime-boost Vaccination strategies

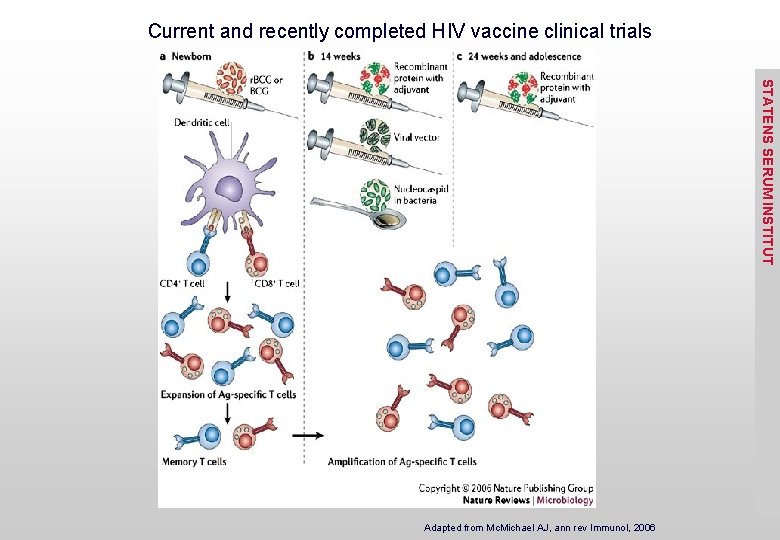
Current and recently completed HIV vaccine clinical trials STATENS SERUM INSTITUT Adapted from Mc. Michael AJ, ann rev Immunol, 2006

On-going Tuberculosis vaccine clinical trials STATENS SERUM INSTITUT Skeiky et al. Nature Reviews Microbiology 4, 469– 476, 2006

Preventive prime-boost vaccination strategy against Tuberculosis STATENS SERUM INSTITUT Skeiky et al. Nature Reviews Microbiology 4, 469– 476 , June 2006

STATENS SERUM INSTITUT THE END