Anita Olczak Clostridium difficile infection Katedra i Klinika



























- Slides: 37

Anita Olczak Clostridium difficile infection Katedra i Klinika Chorób Zakaźnych i Hepatologii CM UMK Wwykład sponsorowany przez BMS

C. difficile: microbiology Gram positive spore forming bacillus (rods) Obligate anaerobe Part of the GI Flora in ◦ 1 -3% of healthy adult ◦ 70% of children < 12 months Some strains produce toxins A & B Toxins-producing strains cause C. diff Infection (CDI) CDI ranges from mild, moderate, to severe and even fatal illness

C. difficile: microbiology A common cause of nosocomial antibiotic-associated diarrhea (AAD) Most common infectious cause of acute diarrheal illness in LTCFs The only nosocomial organism that is anaerobic and forms spores (survive > 5 months and hard to destroy) Pathogenesis is mainly due to toxins production Infective dose is < 10 spores

Changing Epidemiology. Current strange of C. difficile BI/NAP 1/027, toxinotype III Historically uncommon – epidemic since 2000 More resistant to fluoroquinolones Produces extra toxin called binary toxin More virulent Increased toxin A and B production Change in binding domain of toxin B increase adherence to the gut wall Increased sporulation increase survival • Causes more cases and more severe disease even at low risk populations

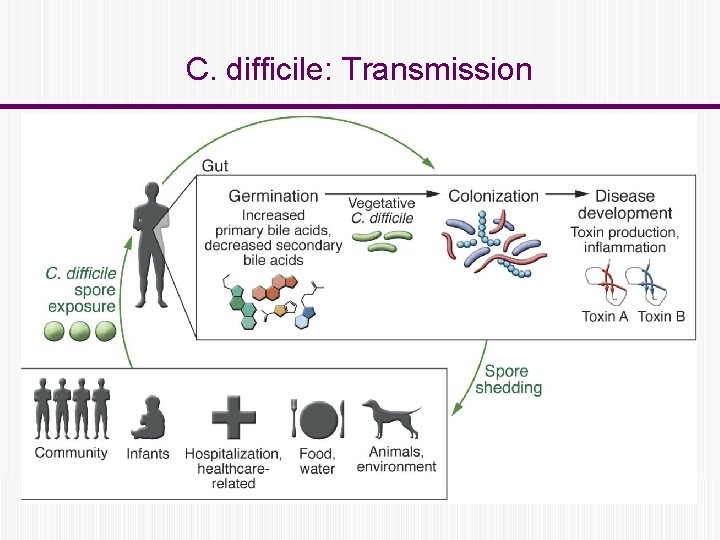
C. difficile: Transmission

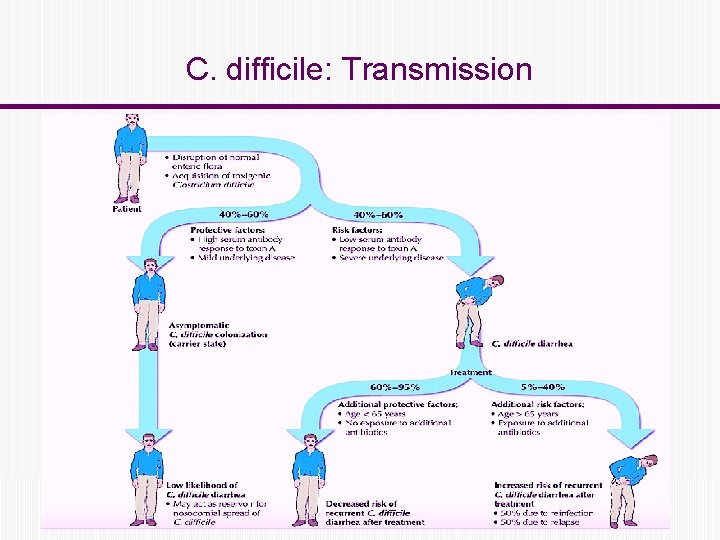
C. difficile: Transmission

C. difficile: Transmission Fecal – oral route ◦ Contaminated hands of healthcare workers ◦ Contaminated environmental surfaces. Person to person in hospitals and LTCFs Reservoir: ◦ Human: colonized or infected persons ◦ Contaminated environment C. diff spores can survive for up 5 months on environmental surfaces.

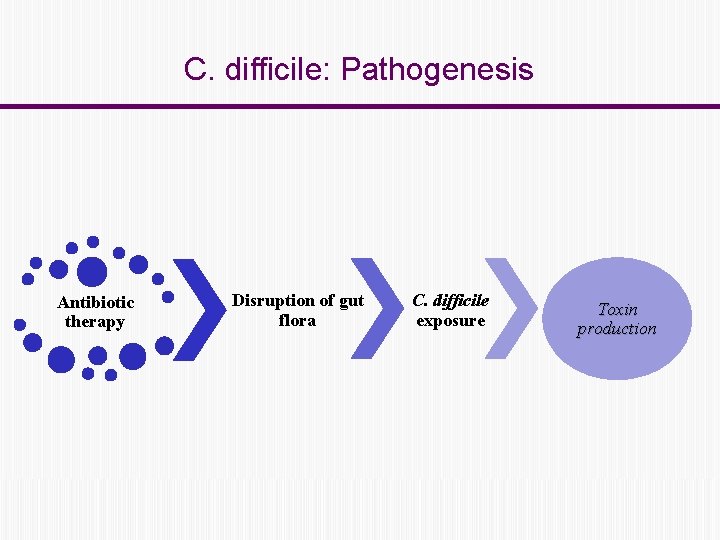
C. difficile: Pathogenesis Antibiotic therapy Disruption of gut flora C. difficile exposure Toxin production

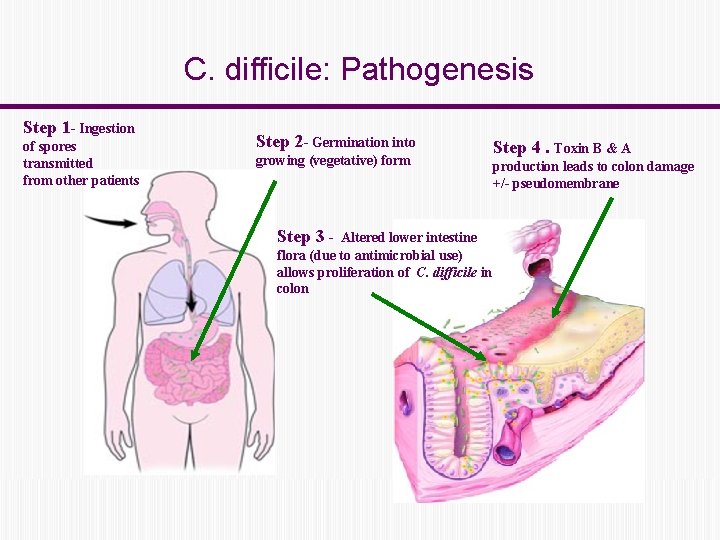
C. difficile: Pathogenesis Step 1 - Ingestion of spores transmitted from other patients Step 2 - Germination into growing (vegetative) form Step 3 - Altered lower intestine flora (due to antimicrobial use) allows proliferation of C. difficile in colon Step 4. Toxin B & A production leads to colon damage +/- pseudomembrane

C. Difficile infection: Risk factors • Antimicrobial exposure • Any and all antibiotics – including surgical prophylaxis • Clindamycin – highest risk for original strain • Fluoroquinolones – highest risk for NAP 1 strain • Sulfamethoxazole-trimethoprim prophylaxis has not been associated with CDAD • Reduced humoral response • Acid suppressant agents

C. Difficile infection: Risk factors Exposure to antimicrobials (prior 2 -3 months) Exposure to healthcare (prior 2 -3 months) Prolonged hospitalization Infection with toxogenic strains of C. difficile Old age > 65 years Severe underlying illness Immunosuppression & HIV Chemotherapy (immunosuppression & antibiotic-like activities) Tube feeds and GI surgery Exposure to gastric acid suppression meds ? ?

Antimicrobials Predisposing to CDI Very commonly related Less commonly related Uncommonly related Clindamycin Ampicillin Amoxicillin Cephalosporins Fluoroquinolons Sulfa Macrolides Carbapenems Other penicillins Aminoglycosides Rifampin Tetracycline Chloramphincol Ø Among symptomatic patients with CDI: • 96% received antimicrobials within the 14 days before onset • 100% received an antimicrobial within the previous 3 months Ø 20% of hospitalized patients are colonized with C. difficile

Acid suppression and CDAD • Clostridium difficile spores are not killed by gastric acid • BUT, vegetative forms which germinates spore form are killed by gastric acid • Clinical trials differ • 2 x higher incidence of CDAD with proton pump inhibitors • Confounded by severity of disease and hospital length of stay Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009, Cunningham R et al. J Hosp Infect 2003, Dubberke ER et al. Clin Infect Dis 200, Loo VG et al. NEJM 2005

Clinical manifestation Illness caused by toxin-producing strains of C. difficile ranges from ◦ Asymptomatic carriers = Colonized ◦ Mild or moderate diarrhea ◦ Pseudo membranous colitis that can be fatal A median time between exposure to onset of CDI symptoms is of 2– 3 days Risk of developing CDI after exposure ranges between 5 -10 days to 10 weeks Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009, Cunningham R et al. J Hosp Infect 2003, Dubberke ER et al. Clin Infect Dis 200, Loo VG et al. NEJM 2005

Clinical manifestation Typical presentation • Watery diarrhea • • Up to 10 -15 bowel movements per day Fever Abdominal cramping, discomfort Unexplained leukocytosis Atypical presentation • Vitals: fever • Physical exam: abdominal pain/distension • Lab values: leukocytosis >30, 000 cells/mm 3 • >50% transplant patients • CT scan: severe colitis Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009, Cunningham R et al. J Hosp Infect 2003, Dubberke ER et al. Clin Infect Dis 200, Loo VG et al. NEJM 2005

Symptoms of CDI Watery diarrhea > 3 unformed stools in 24 or fewer consecutive hours Loss of appetite Fever Nausea Abdominal pain and cramping

Diagnosis • Up to 50% of hospitalized patients are colonized • Only perform diagnostic tests if symptomatic • CDAD is a clinical diagnosis • Colonoscopy for the presence of pseudomembranes • definitive diagnosis • Test for C. difficile toxin in stool • Cytotoxicity cell assay (Gold Standard) • • Expensive, 24 hour turn around time ELISA • • • Inexpensive, rapid turn around time 60 -90% sensitive with a negative predictive value >95% Repeat testing increases risk of false positive Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009

Best strategy for C. difficile testing Testing should be performed only on diarrheal stool Testing asymptomatic patients is not indicated Testing for cure is not recommended Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009

Best strategy for C. difficile testing For clinical use: two-step testing uses initially EIA detection of GDH for screening followed by cytotoxicity assay or toxigenic culture for confirmation Gold standard is stool culture followed by toxigenic culture assay Toxin is very unstable, degrades at room temperature, and undetectable within 2 hours (false negative results) Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009

CDI Surveillance: Case definition Ø Clinical: presence of diarrhea AND Ø Laboratory: A stool test result positive for toxigenic C. diff or its toxins OR colonoscopic / histopathologic findings demonstrating evidence of pseudomembranes Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009

CDI Surveillance: Case definition Surveillance definitions of CDI by time of onset: Ø Healthcare facility (HCF)-onset, HCF-associated CDI Onset > 48 hrs of admission Ø Community-onset, HCF-associated CDI Onset in the community or within 48 hours of admission and within < 4 weeks of the last discharge Ø Community-associated CDI Onset in the community but within more that 12 weeks of last discharge Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009

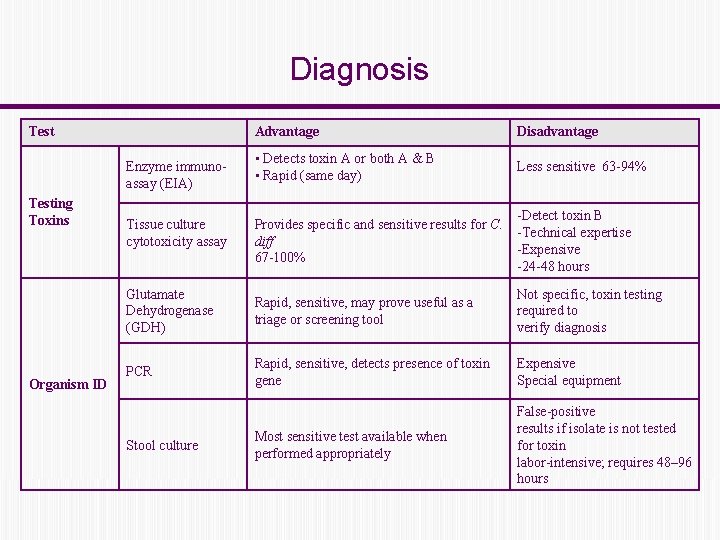
Diagnosis Test Advantage Disadvantage • Detects toxin A or both A & B • Rapid (same day) Less sensitive 63 -94% Provides specific and sensitive results for C. diff 67 -100% -Detect toxin B -Technical expertise -Expensive -24 -48 hours Glutamate Dehydrogenase (GDH) Rapid, sensitive, may prove useful as a triage or screening tool Not specific, toxin testing required to verify diagnosis PCR Rapid, sensitive, detects presence of toxin gene Expensive Special equipment Most sensitive test available when performed appropriately False-positive results if isolate is not tested for toxin labor-intensive; requires 48– 96 hours Enzyme immunoassay (EIA) Testing Toxins Organism ID Tissue culture cytotoxicity assay Stool culture

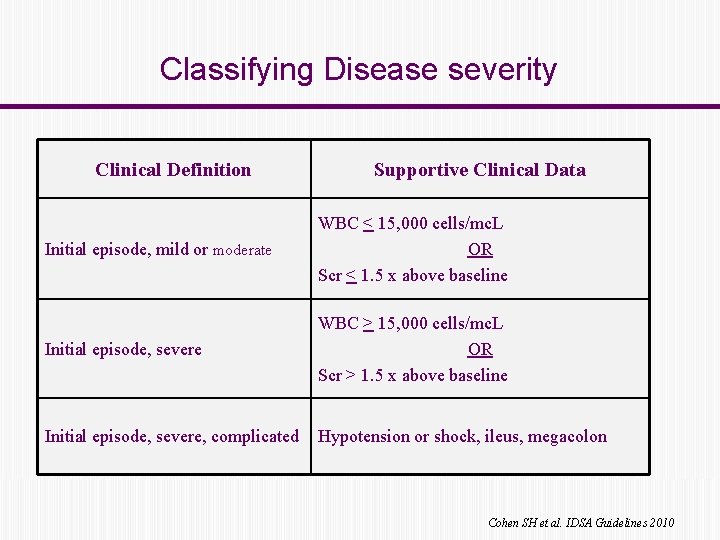
Classifying Disease severity Clinical Definition Supportive Clinical Data Initial episode, mild or moderate WBC < 15, 000 cells/mc. L OR Scr < 1. 5 x above baseline Initial episode, severe WBC > 15, 000 cells/mc. L OR Scr > 1. 5 x above baseline Initial episode, severe, complicated Hypotension or shock, ileus, megacolon Cohen SH et al. IDSA Guidelines 2010

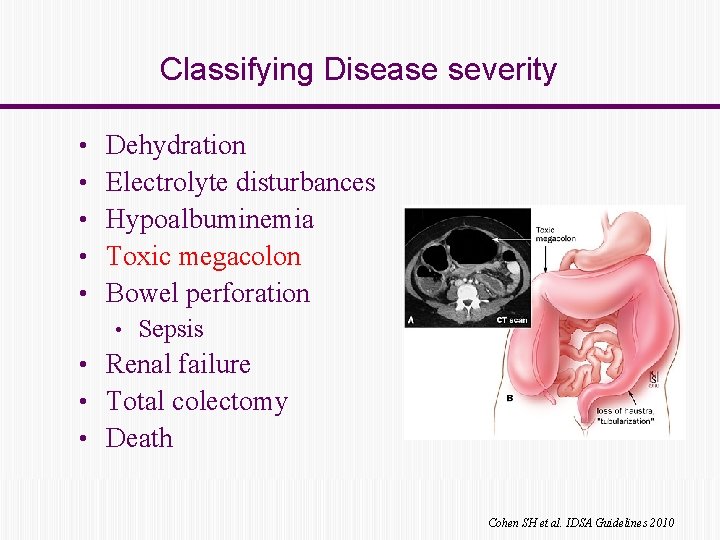
Classifying Disease severity • • • Dehydration Electrolyte disturbances Hypoalbuminemia Toxic megacolon Bowel perforation • Sepsis • Renal failure • Total colectomy • Death Cohen SH et al. IDSA Guidelines 2010

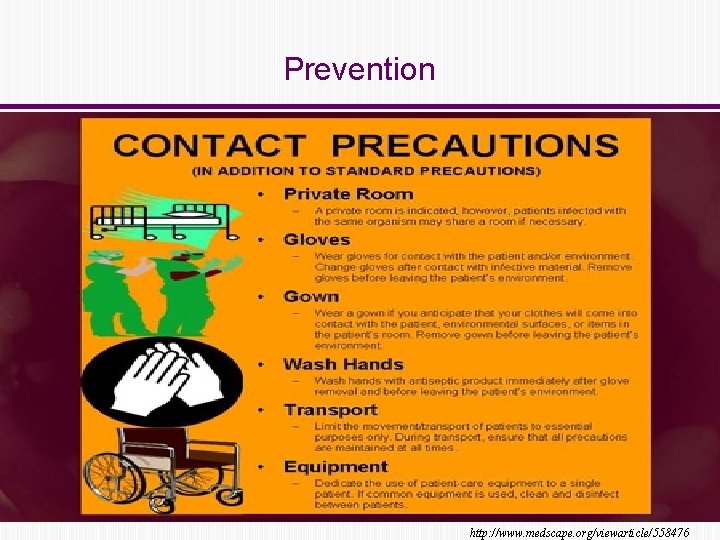
Prevention • Horizontal transmission (IDSA) • • • Hand washing with soap and water Contact precautions: gloves and gown Clean areas with sporicidal agents • Minimize risk factors Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009

Prevention Core Supplemental § Gloves/gowns on room entry § Private room (preferred) or cohort with dedicated commodes § Dedicated equipment § Maintain for duration of diarrhea § Measure compliance § Extend use of contact precautions beyond duration of diarrhea § Presumptive isolation § Universal glove use on units with high CDI rates § Intensify assessment of compliance Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009

Prevention http: //www. medscape. org/viewarticle/558476

Treatment § Stop or narrow antibiotics § Metronidazole (PO, IV) § Vancomycin (PO, PR) § Fidaxomycin (PO) § Alternatives § § § IVIG Rifaximin Nitizoxanide

Metronidazole Not FDA approved for CDAD Mechanism: disrupts protein synthesis resulting in cell death in anaerobic bacteria § Dose: 500 mg PO Q 8 h, 500 mg IV Q 6 -8 h § Pharmacokinetics: rapidly absorbed § Concentration in colon minima § Use: effective for mild to moderate disease and first recurrence § § §Adverse effects §Caution in liver failure §Higher risk for side effects as drug is hepatically metabolized §Neurotoxicity, primarily manifested as paraesthesias §Paraesthesias are more common with prolonged exposure § l

vancomycine n FDA approved for CDAD n Mechanism: inhibits the growth of C. Difficile (bacteroistatic) n Dose: 125 – 500 mg PO Q 6 h; 500 mg PR n n IV administration does not treat CDAD Enema may lead to bacteremia from colonic flora n Administration: n n n Oral capsules (expensive) IV product used orally (in hospital administration) Retention enema

vancomycine n Use: Initial episode of severe or complicated disease and for recurrence n Pharmacokinetics: poorly absorbed in gut n Concentrates in colon n Adverse effects n Compromised gastrointestinal tract may result in systemic absorption

Fidaxomicin • FDA approved for CDAD in May 2011 • Mechanism: macrolide antibiotic • Kills C. difficile (bactericidal) • Postantibiotic effect • Dose: 200 mg PO BID x 10 days • Pharmacokinetics: poor absorption in gut

Fidaxomicin • Use: treatment of C. difficile infections • Non-inferior to oral vancomycin • Lower rate of recurrence of non-NAP 1 strain • Less effect on normal colonic flora • Adverse effects: Nausea, vomiting • Minimal drug interactions • Significant cost: $2800 for 10 day course, poorly covered by insurance providers

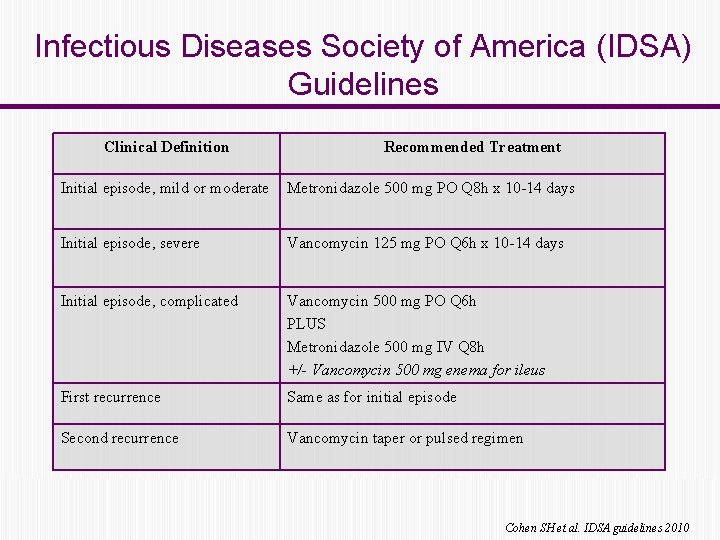
Infectious Diseases Society of America (IDSA) Guidelines Clinical Definition Recommended Treatment Initial episode, mild or moderate Metronidazole 500 mg PO Q 8 h x 10 -14 days Initial episode, severe Vancomycin 125 mg PO Q 6 h x 10 -14 days Initial episode, complicated Vancomycin 500 mg PO Q 6 h PLUS Metronidazole 500 mg IV Q 8 h +/- Vancomycin 500 mg enema for ileus First recurrence Same as for initial episode Second recurrence Vancomycin taper or pulsed regimen Cohen SH et al. IDSA guidelines 2010

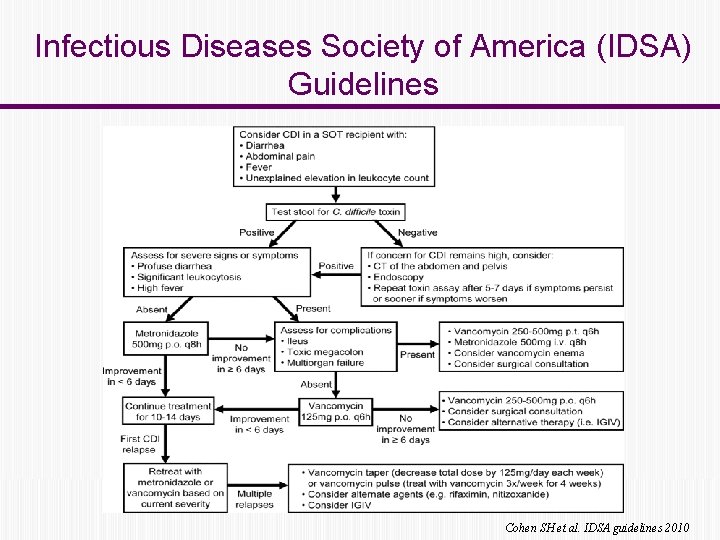
Infectious Diseases Society of America (IDSA) Guidelines Cohen SH et al. IDSA guidelines 2010

Recurrence • 6 – 25% of patients experience 1 episode of recurrence • Definition: relapse of same infection or re-infection from new strain • Risk factors • Age > 65 • Metronidazole (especially in patients > 65) • Use of other antibiotics during or after initial treatment • Impaired immune response to toxin A Cohen SH et al. IDSA guidelines 2010

Treatment of Recurrence • First recurrence: same as initial episode • Second recurrence: vancomycin taper and/or pulse therapy • Taper: slow taper over prolonged period of time • Pulse: high dose given fewer times over a prolonged period of time • Avoid metronidazole for cumulative neurotoxicity • > 2 recurrences: • Alternative therapy Cohen SH et al. IDSA Guidelines 2010, Dubberke ER et al. AJT 2009