Animal models for muscular dystrophy Vincenzo Nigro Laboratorio

Animal models for muscular dystrophy Vincenzo Nigro Laboratorio di genetica - Dipartimento di Patologia Generale, Seconda Università degli Studi di Napoli Telethon Institute of Genetics and Medicine, Napoli

muscular dystrophy • MD is a general term that describes a group of inherited and gradually debilitating myogenic disorders • progressive muscle weakness affecting patients since from young age and can lead to early death • pattern of inheritance can be X-linked recessive (DMD/BMD), autosomal dominant (LGMD 1), or autosomal recessive (LGMD 2) • Some underlying genetic defects are well known, others are orphan diseases
dystrophin mutations • DMD Duchenne Muscular Dystrophy - 1/3, 500 boys Onset -- Early childhood - about 2 to 6 years Symptoms -- Generalized weakness and muscle wasting affecting limb and trunk muscles first. Calves often enlarged Progression -- Disease progresses slowly but will affect all voluntary muscles. Survival possible beyond late twenties • BMD Becker Muscular Dystrophy - 1/10, 000 boys Onset -- Adolescence or adulthood Symptoms -- Almost identical to Duchenne but often much less severe. Can be significant heart involvement Progression -- Slower and more variable than Duchenne with survival well into mid to late adulthood

weakness Proximal weakness: the most common site of weakness in a myopathic disorder • Lower extremities – difficulty climbing stairs – arising from a low chair or toilet – getting up from a squatted position • Upper extremities – trouble lifting objects over the head – brushing the hair

fatigue • Much less useful “negative” symptom (non-specific) • Many patients who complain of diffuse global "weakness" or fatigue do not have a disorder of muscle • Abnormal fatigability after exercise: – metabolic and mitochondrial myopathies – define the duration and intensity

disease progression • muscle tissue represent about 40% of the total body mass • respiratory failure can be the cause of premature death as well as heart failure • patients suffer from asymmetries in strength between reciprocal muscles that cause widespread joint and spine deformities requiring timely orthopaedic surgery
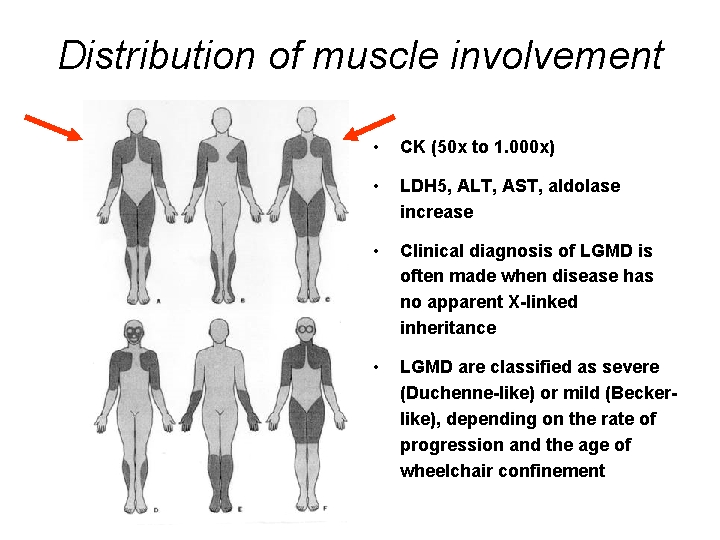
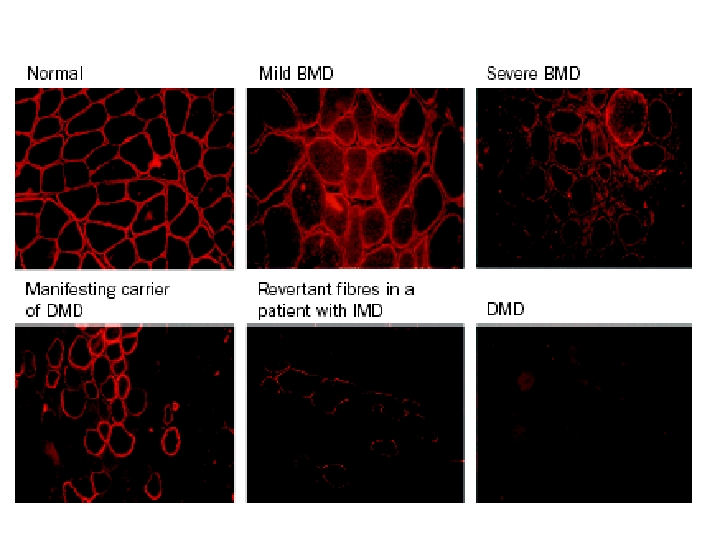
Distribution of muscle involvement • CK (50 x to 1. 000 x) • LDH 5, ALT, AST, aldolase increase • Clinical diagnosis of LGMD is often made when disease has no apparent X-linked inheritance • LGMD are classified as severe (Duchenne-like) or mild (Beckerlike), depending on the rate of progression and the age of wheelchair confinement

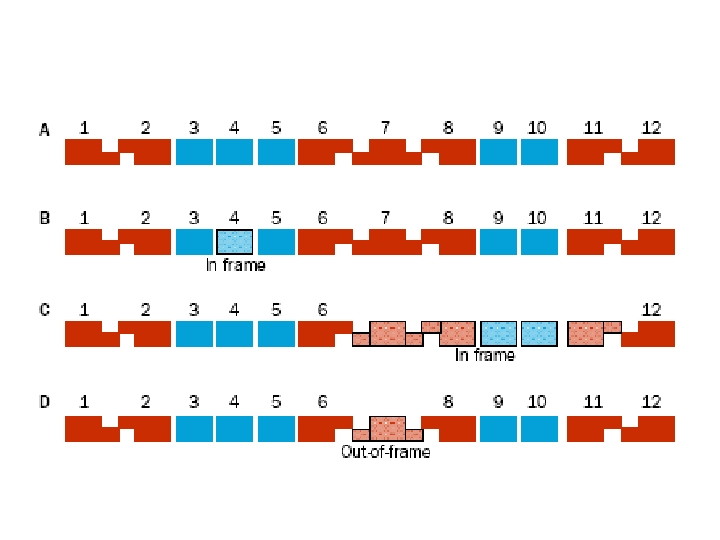
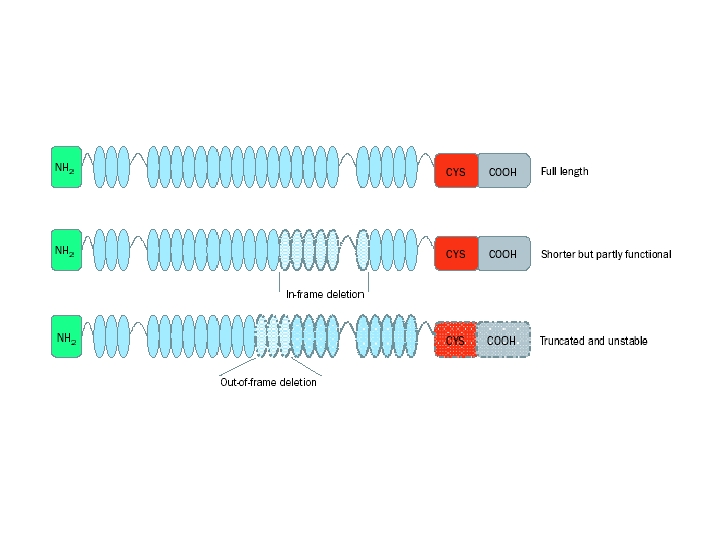
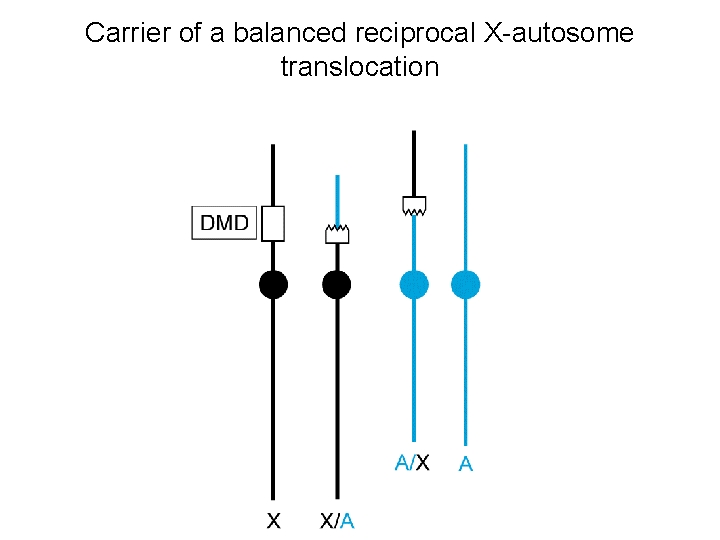
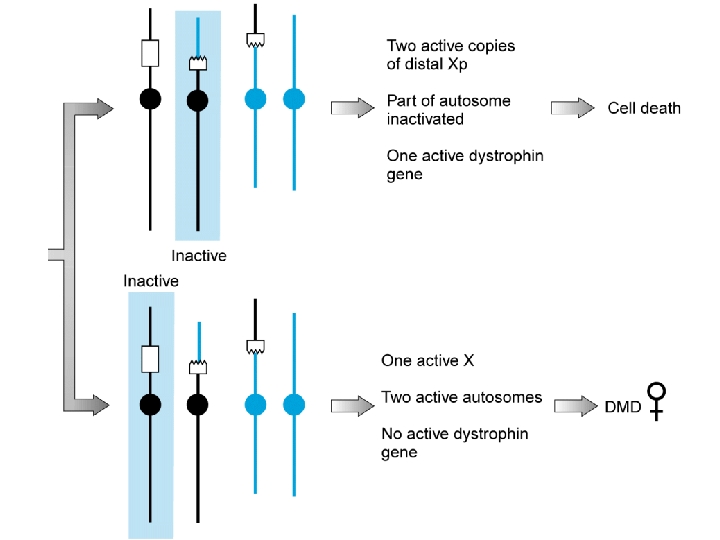
Carrier of a balanced reciprocal X-autosome translocation

management > treatment > therapy > cure Animal models disease should be comparable to human defects Profoundly studied in all pathological characteristics Should allow a reliable prediction of the response

n n n The genetic basis of the disease should be the same as human disease Reiterate key hallmarks of the human disease Animals commercially available, easy to maintain Animal disease well characterized, with abundant literature Robust phenotype that is reproducible over generations

mdx (X-chr MD) mouse C 57 BL/10 Sc. Sn-Dmdmdx • mdx is the best characterized mouse model for muscular dystrophy (>1, 700 papers) since 1984 • mdx has a spontaneous nonsense mutation (stop) in exon 23 of the dystrophin gene and does not produce dystrophin • absence of dystrophin reduces the DGC at the sarcolemma

mdx mouse may be DMD mouse? • mdx shows signs of MD during first 6 weeks of life which results in an increase of the newly differentiated myofibers • it has muscle regeneration with an expansion of the satellite cell population and muscle hypertrophy • Centralized nuclei (50 -60%), heterogeneity in fiber size • Necrosis at early stages, but decreases after 60 days • Plasma creatine kinase is 5. 000 -12. 000 U/L • the most affected muscle (diaphragm) reproduces the degenerative changes of MD
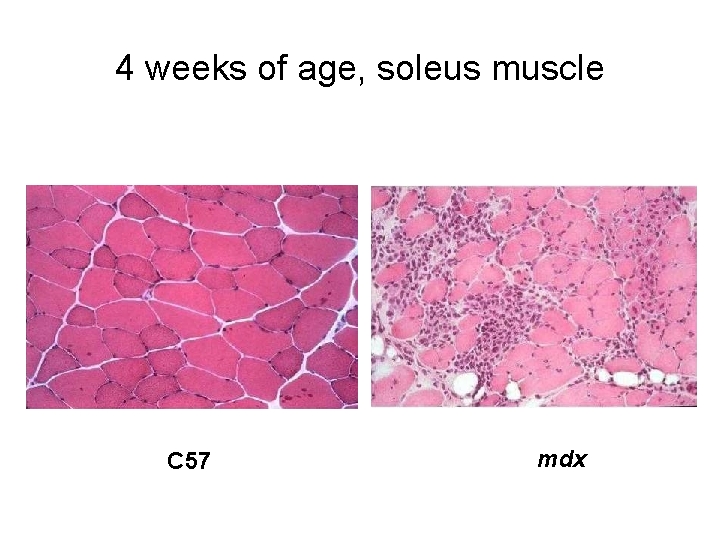
4 weeks of age, soleus muscle C 57 mdx

but

• Fibrosis is only in diaphragm • Absolute muscle force of limb muscles remains similar to unaffected mice • Lifespan is shorter but no so much (-19% in males) • it has muscle regeneration with an expansion of the satellite cell population and muscle hypertrophy • mdx lacking the muscle-specific transcription factor Myo. D or myocyte nuclear factor (expressed in the satellite cells) show more severe MD mdx mouse is a bad DMD mouse model

Double mouse mutants utrophin/dystrophin • Utrophin is a developmentally regulated protein, an autosomal homologue to dystrophin • utrophin is overexpressed when dystrophin is absent • the utrn−/−/mdx mice are severely affected • reduced lifespan • severe muscle weakness with joint contractures, growth retardation, and cardiomyopathy • the phenotype is ameliorated by skeletal-muscle specific expression of utrophin
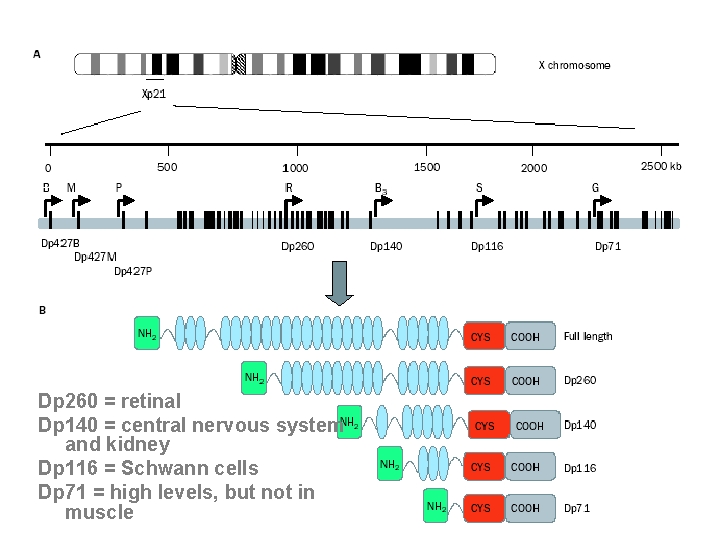
Dp 260 = retinal Dp 140 = central nervous system and kidney Dp 116 = Schwann cells Dp 71 = high levels, but not in muscle

mdx 52 mouse • mdx 52 is dystrophin KO mouse • mdx has a deletion in exon 52 of the dystrophin gene and does not produce dystrophin • in contrast to mdx, this mouse cannot produce also Dp 260(ret) and Dp 140(CNS), maintaining Dp 116 (S) and Dp 71 • it is very similar to mdx mouse with the absence of dystrophin that reduces the DGC at the sarcolemma, but has no cardiomyopathy

mdx 2 cv-5 cv mice • they were generated by chemical mutagenesis using N-ethyl-nitrosurea • mdx 2 cv lacks dys, Dp 260 • mdx 3 cv lacks dys, Dp 260, Dp 140, Dp 116 and Dp 71 • mdx 4 cv lacks dys, Dp 260 and Dp 140 • mdx 5 cv lacks only dystrophin • phenotypes are very similar to mdx mouse and no phenotype worsening

targeted inactivation of Dp 71 only • there is a mouse that cannot produce Dp 71 only • it has normal phenotype
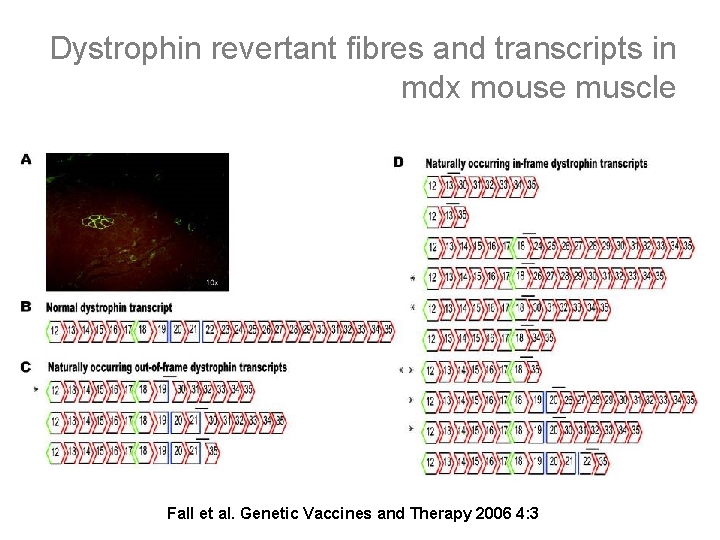
Dystrophin revertant fibres and transcripts in mdx mouse muscle Fall et al. Genetic Vaccines and Therapy 2006 4: 3

the "humanized" h. DMD mouse • “humanised” DMD (h. DMD) mice carry an integrated and functional copy of the full-length human DMD gene • it serves to test the “exon skipping strategy” that is a sequence-specific therapeutic approach

human sequence-specific DMD exon skipping in vivo • the h. DMD mouse model allows the direct testing of human-specific AONs and target sequences in a mouse experimental background • the induction of specific skipping of the h. DMD exons 44, 46, and 49, whilst the endogenous mouse transcripts are not affected [Bremmer-Bout et al. , Mol. Ther. 2004] • this underlines that AONs, based upon specific design, can be highly sequence-specific small molecule drugs.
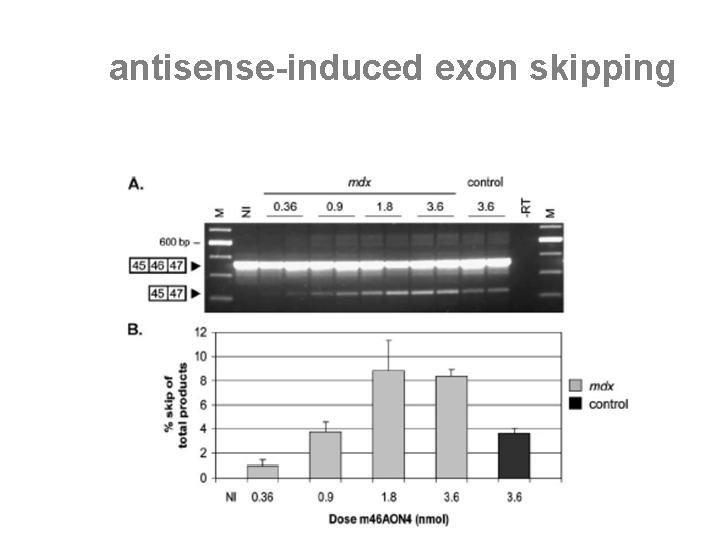
antisense-induced exon skipping
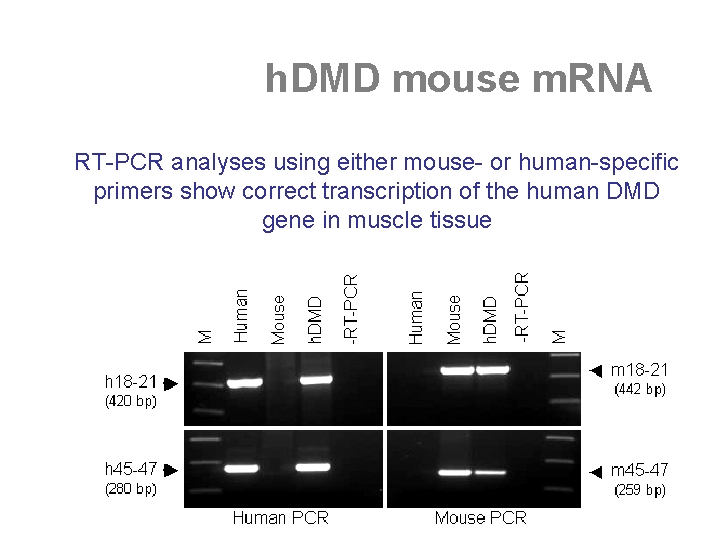
h. DMD mouse m. RNA RT-PCR analyses using either mouse- or human-specific primers show correct transcription of the human DMD gene in muscle tissue
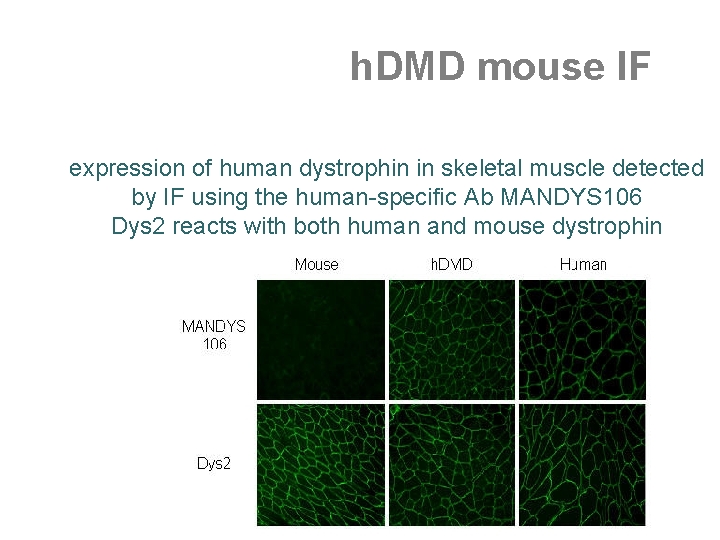
h. DMD mouse IF expression of human dystrophin in skeletal muscle detected by IF using the human-specific Ab MANDYS 106 Dys 2 reacts with both human and mouse dystrophin
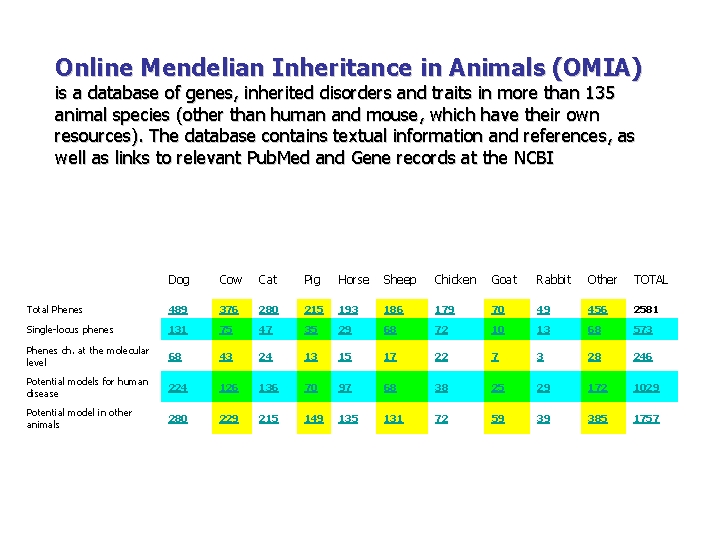
Online Mendelian Inheritance in Animals (OMIA) is a database of genes, inherited disorders and traits in more than 135 animal species (other than human and mouse, which have their own resources). The database contains textual information and references, as well as links to relevant Pub. Med and Gene records at the NCBI Dog Cow Cat Pig Horse Sheep Chicken Goat Rabbit Other TOTAL Total Phenes 489 376 280 215 193 186 179 70 49 456 2581 Single-locus phenes 131 75 47 35 29 68 72 10 13 68 573 Phenes ch. at the molecular level 68 43 24 13 15 17 22 7 3 28 246 Potential models for human disease 224 126 136 70 97 68 38 25 29 172 1029 Potential model in other animals 280 229 215 149 135 131 72 59 39 385 1757

Simple search for: "muscular dystrophy" 10 records found OMIA 000679 Muscular dystrophy in Gallus gallus (chicken) Sub-type: Abnormal muscle; AM Genes: WWP 1 OMIA 000679 Muscular dystrophy in Canis familiaris (dog) OMIA 000679 Muscular dystrophy in Ovis aries (sheep) OMIA 000679 Muscular dystrophy in Meleagris gallopavo (turkey) OMIA 000679 Muscular dystrophy in Mustela lutreola (European mink) OMIA 000679 Muscular dystrophy in Felis catus (cat) OMIA 001081 Muscular dystrophy, Duchenne and Becker types in Felis catus (cat) Genes: DMD OMIA 001081 Muscular dystrophy, Duchenne and Becker types in Canis familiaris (dog) Sub-type: X-linked muscular dystrophy OMIA 000681 Muscular dystrophy, dysphagia-associated in Canis familiaris (dog) OMIA 000828 Progressive muscular dystrophy in Mustela lutreola (European mink)

Golden retriever dog with muscular dystrophy (GRMD) GRMD arises from a mutation in the acceptor splice site of intron 6 of the dystrophin gene Skipping of exon 7 disrupts the m. RNA reading frame and results in premature termination of translation

Golden retriever dog with muscular dystrophy (GRMD) complete absence of the dystrophin, early and severe muscle degeneration with reduction of motility and walking ability Death usually occurs at about 1 year of age as a result of failure of respiratory muscles

dystrophic Golden Retriever dog • gradual weakness and loss of muscle mass • development of contractures, skeletal deformities • significant phenotypic variability among litters

Spitz dogs • Becker-like dystrophy with a truncated form of dystrophin was recently identified in a family of Japanese Spitz dogs

LGMD forms • LGMD have a highly variable onset and progression, but the unifying theme is the proximal muscle involvement • The a. dominant forms (LGMD 1) are generally milder and relatively rare representing less than 10% of all LGMD • The a. recessive forms (LGMD 2) are much more common, having a cumulative prevalence of 1: 14, 0001: 20, 000 with some differences among countries, depending on the carrier distribution and the degree of consanguinity • There are, however, at least 25% of families who can be excluded from any known locus and 40% of typical LGMD cases with no mutation in any known gene

Autosomal dominant LGMD 1 A LGMD 1 B LGMD 1 C LGMD 1 D LGMD 1 E LGMD 1 F LGMD 1 G 5 q 31. 2 1 q 21 3 p 25. 3 6 q 22 7 q 35 7 q 31. 1 4 p 21 myotilin (Hauser, 2000) lamin A/C (Bonne, 1999) caveolin 3 (Minetti, 1997) ? ? Autosomal recessive LGMD 2 A LGMD 2 B LGMD 2 C LGMD 2 D LGMD 2 E LGMD 2 F LGMD 2 G LGMD 2 H LGMD 2 I LGMD 2 J LGMD 2 K LGMD 2 L LGMD 2 M LGMD 2 N LGMD 2 O 15 q 15 2 p 13. 2 13 q 12 17 q 21. 33 4 q 12 5 q 33 17 q 12 9 q 33. 1 19 q 13. 3 2 q 24. 3 9 q 34. 1 9 q 31 1 p 34. 1 14 q 24 11 p 13 -p 12 calpain 3 (Richard, 1995) dysferlin (Bashir, Liu, 1998) g-sarcoglycan (Noguchi, 1995) a-sarcoglycan (Roberds, 1994) b-sarcoglycan (Bonnemann, Lim, 1995) d-sarcoglycan (Nigro, 1996) telethonin (Moreira, 2000) TRIM 32 (Frosk, 2002) FKRP (Brockington, 2001) titin (Udd, 2002) POMT 1 (Balci, 2005) fukutin (Godfrey, 2006) POMGn. T 1 (Clement, 2008) POMT 2 (Biancheri, 2007) ? (Jarry, 2007)



Dy/dy dy 2 J/dy 2 J • Two mouse models for laminin-α 2 deficiency were identified in the Jackson Laboratories (http: //www. jax. org/) • dy/dy (dystrophia-muscularis) mouse • allelic dy 2 J/dy 2 J mouse • Both mice are models for merosin-deficient Congenital MD (CMD 1 A) • Neither of these mouse models exhibits a complete deficiency of laminin α 2 chain

alfa-syntrophin -/- mouse • no defect in muscle • n. NOS and aquaporin-4 are displaced, like in mdx • aquaporin KO are also normal • n. NOS KO are normal • n. NOS(-/-)/mdx are = mdx

alfa-dystrobrevin -/- mouse • maintain the expression of DGC at the sarcolemma • mild muscular dystrophy but not yet in humans • is affected DGC complex signaling? • n. NOS and aquaporin-4 are displaced, like in mdx • aquaporin KO are also normal

SARCOGLICANOPATHY

Mutation in any of the sarcoglycan genes produces a secondary loss of the other components

LGMD 2 C, 2 D, 2 E, 2 F with sarcoglycan gene mutations • Mutation in any of the sarcoglycan genes produces a phenotype very similar to DMD/BMD • Onset between 7 -19 years, variable progression with some patients that never loss deambulation and other that are more severely affected, also with identical mutations • Atrofic muscular dystrophy. Calf hypertrophy. It may involve distal muscles. Little shoulder girdle involvement. Heart is seldom involved. CK is very high

sarcoglycan family members N protein aa MW expression ex. chrom a-sarcoglycan e-sarcoglycan 387 413 50 52 muscle ubiquit. 10 12 17 q 12 7 q 21 C g-sarcoglycan d-sarcoglycan z-sarcoglycan 291 290 299 35 35 36 muscle brain 8 9 9 13 q 12 5 q 33 8 p 22 N b-sarcoglycan 318 43 muscle 6 4 q 12 C

alternative sarcoglycan complexes a b g d e e b g d b z d

K. O. of the e-sarcoglycan gene We deleted exons 6 -9 of e-sarcoglycan encoding Cysrich and transmembrane domains Hind III Eco RV Spe I 1. 3 kb 9. 5 kb b gal Hind III Eco RV Hind III intron 5 e-SG NEO Hind III Eco RV intron 5 e-SG b gal NEO Spe I intron 9 e-SG exx. 7 - 9 ex. 6 vector target locus Spe I intron 9 e-SG K. O.

Mice heterozygous for the deletion in the e-sarcoglycan gene (+/-) are smaller at birth and show tremors in the posterior legs (+/-) (+/+)

cardiomyopathy hamsters (886 items) • The BIO 14. 6 hamster was generated in 1962, when Homburger fixed by repeated inbreeding a spontaneous trait of Syrian hamster characterized by muscular dystrophy and cardiomyopathy • This animal model is one of the most studied models for inherited dilated cardiomyopathy and muscular dystrophy • Its lifespan is shortened to 10 -13 months, because the heart dilation progresses to an ejection fraction below 30 -35% that causes heart failure

Analysis of d-sarcoglycan gene in the cardiomyopathic hamster • strong reduction (>30 times) of d-sarcoglycan m. RNA • identification of the true first exon (24 kb upstream) which is deleted • d-sarcoglycan deletion is identical in all cardiomyopathic hamster strains all over the world

PATHOLOGIC STAGES IN BIO 14. 6 HAMSTERS LIFE Stages of the disease Clinical Effects 20 -30 days Tongue calcifications 30 -40 days Cardiac lesions 40 -50 days Muscular necrotic lesions 100 days Myocardial hypertrophy 150 days Strong myocardial hypertrophy 180 days Fibrosis and calcifications in heart and muscular tissues 7 -12 months Heart failure 14 months Death

wt BIO 14. 6 rescue

- Slides: 56