Animal Cell CB 6 9 Bonds holding atoms
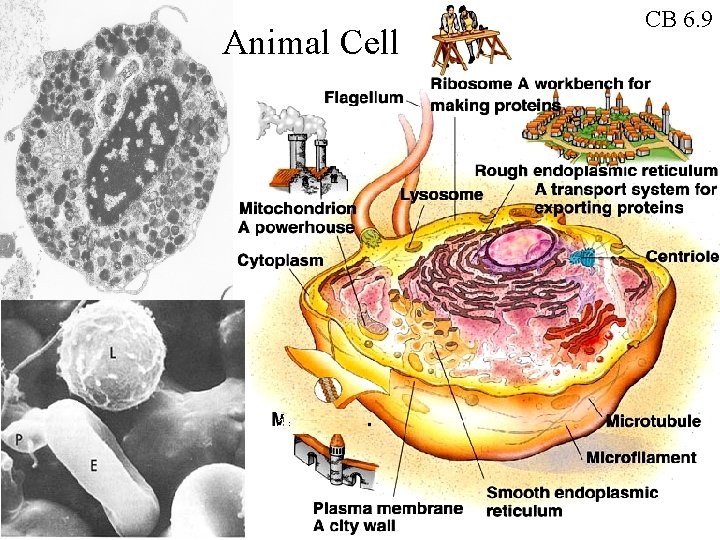
Animal Cell CB 6. 9

Bonds holding atoms together to make molecules: • Covalent – Strong – Sharing Electrons – When broken = chemical reaction • energy consumed or released • Ionic – Weak – Positive and Negative atoms attracted to each other
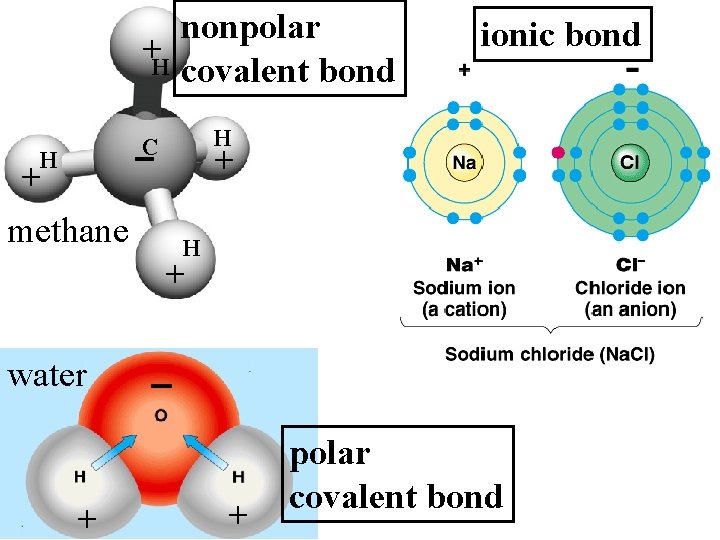
nonpolar +H covalent bond H C H + methane ionic bond + H + water + + polar covalent bond
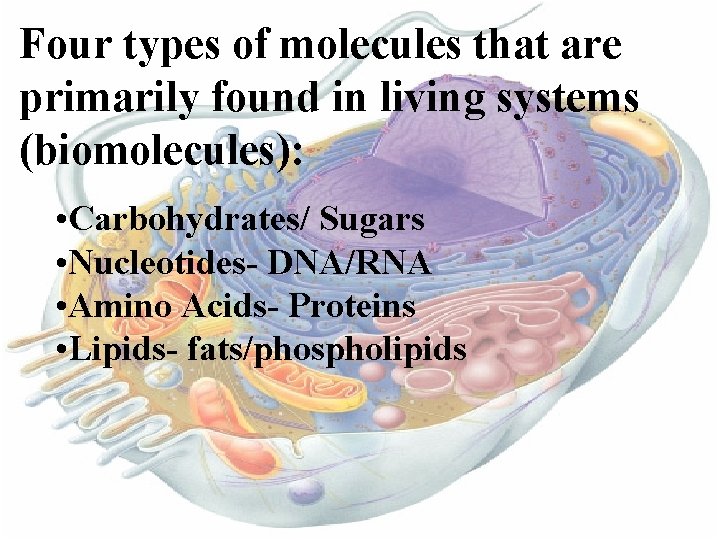
Four types of molecules that are primarily found in living systems (biomolecules): • Carbohydrates/ Sugars • Nucleotides- DNA/RNA • Amino Acids- Proteins • Lipids- fats/phospholipids
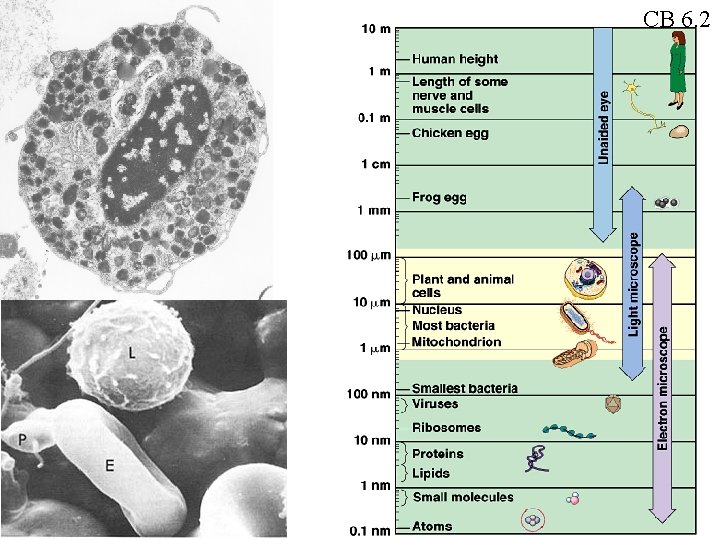
CB 6. 2
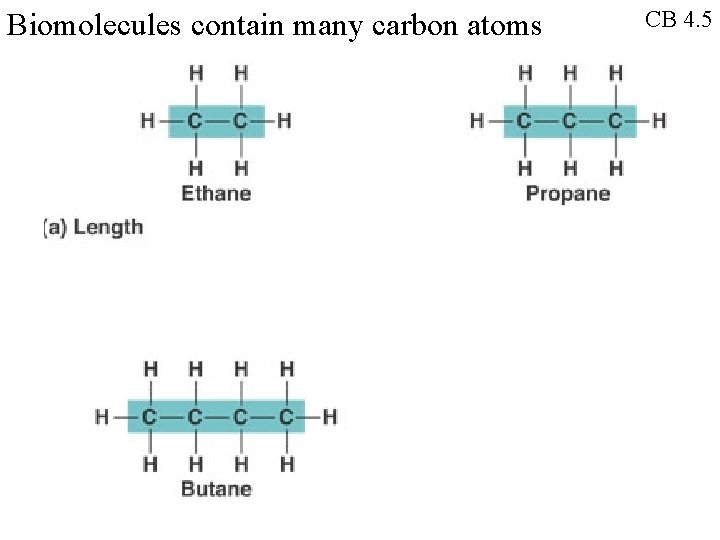
Biomolecules contain many carbon atoms CB 4. 5
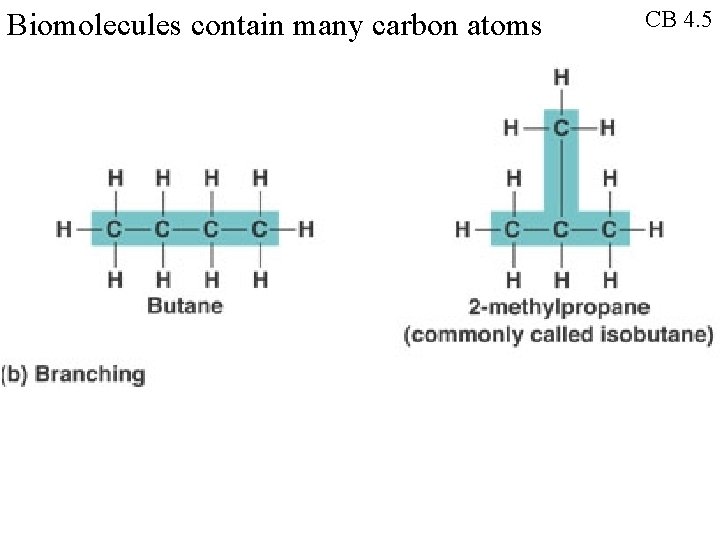
Biomolecules contain many carbon atoms CB 4. 5
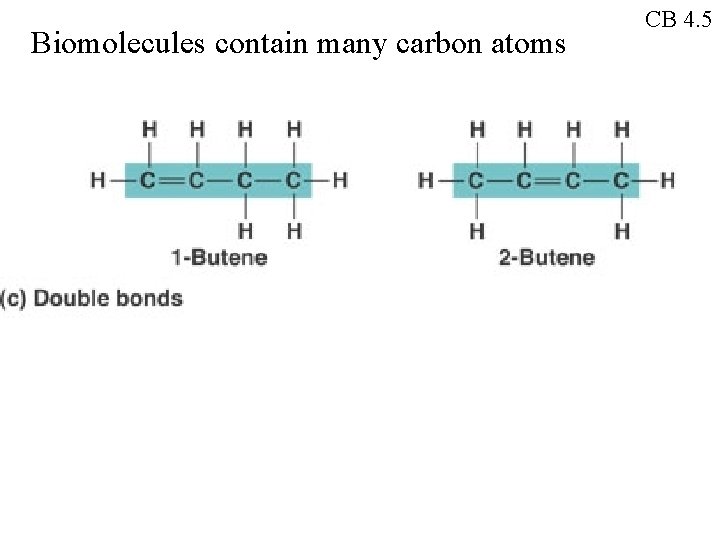
Biomolecules contain many carbon atoms CB 4. 5
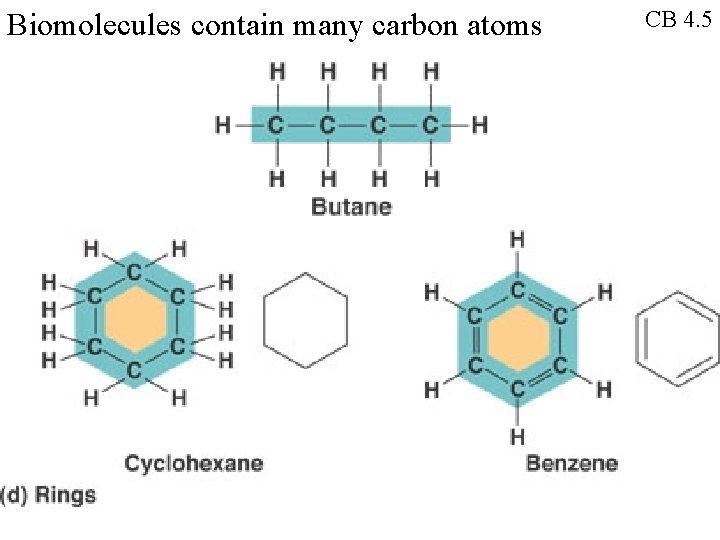
Biomolecules contain many carbon atoms CB 4. 5
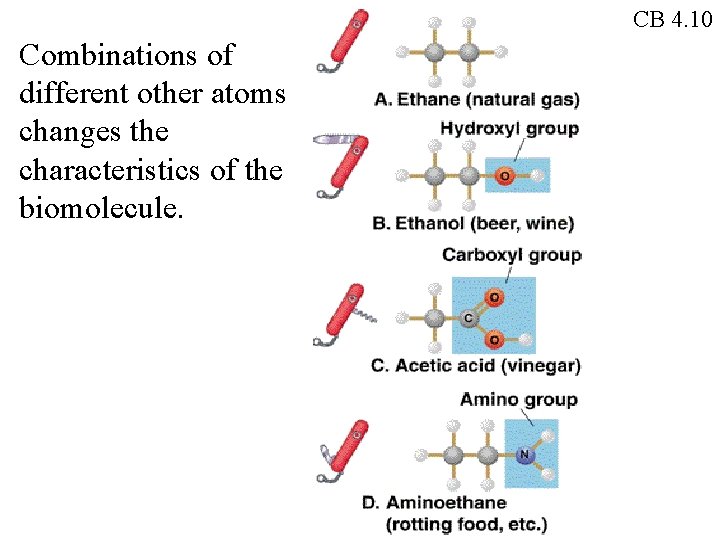
CB 4. 10 Combinations of different other atoms changes the characteristics of the biomolecule.
Four types of molecules that are primarily found in living systems (biomolecules): • Carbohydrates/ Sugars • Nucleotides- DNA/RNA • Amino Acids- Proteins • Lipids- fats/phospholipids
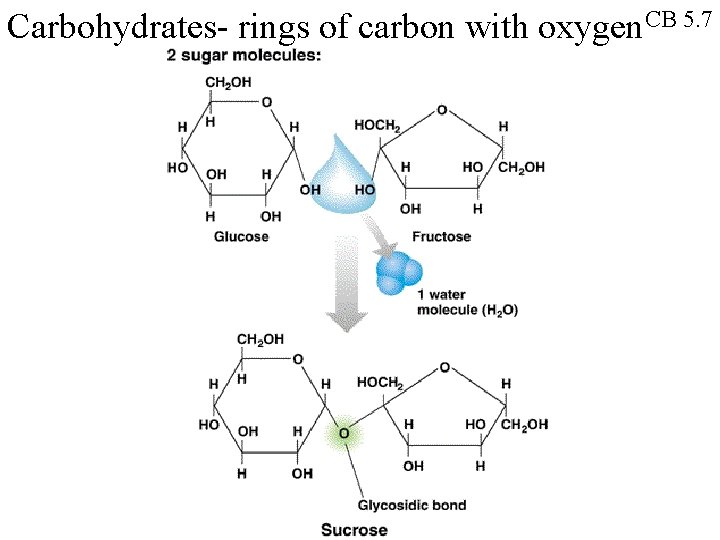
Carbohydrates- rings of carbon with oxygen. CB 5. 7
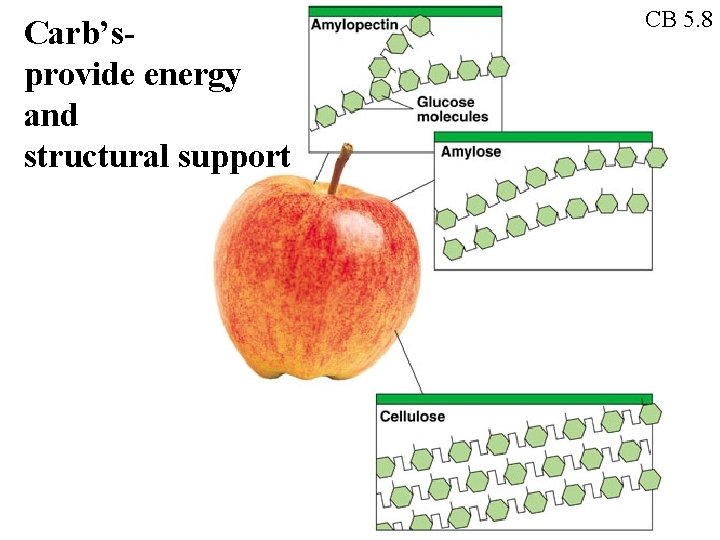
Carb’sprovide energy and structural support CB 5. 8
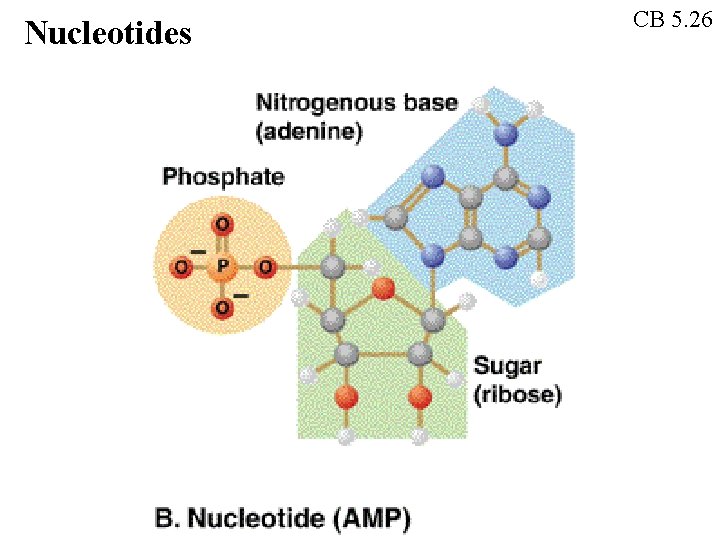
Nucleotides CB 5. 26
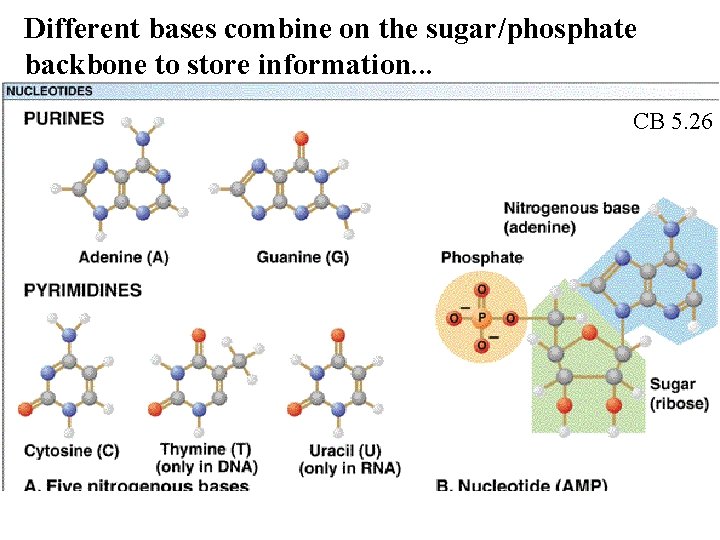
Different bases combine on the sugar/phosphate backbone to store information. . . CB 5. 26
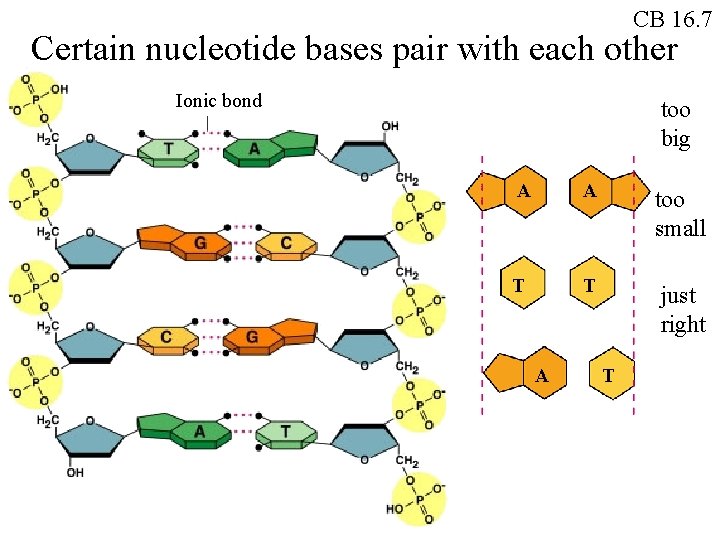
CB 16. 7 Certain nucleotide bases pair with each other Ionic bond too big A A too small T T just right A T
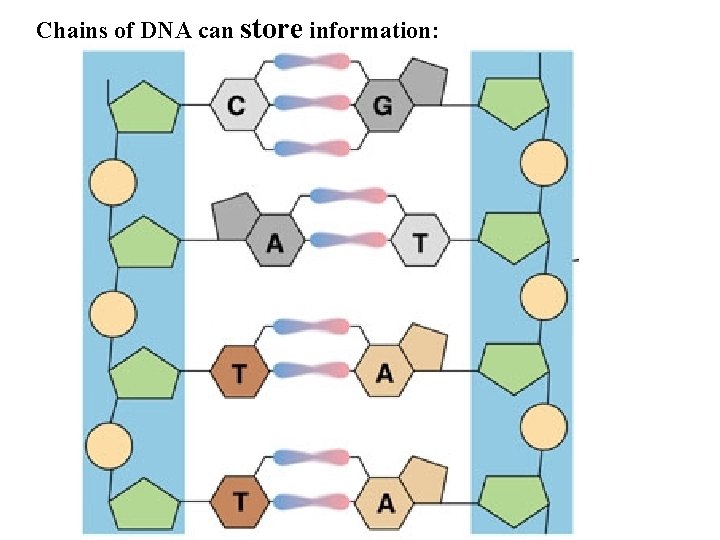
Chains of DNA can store information:
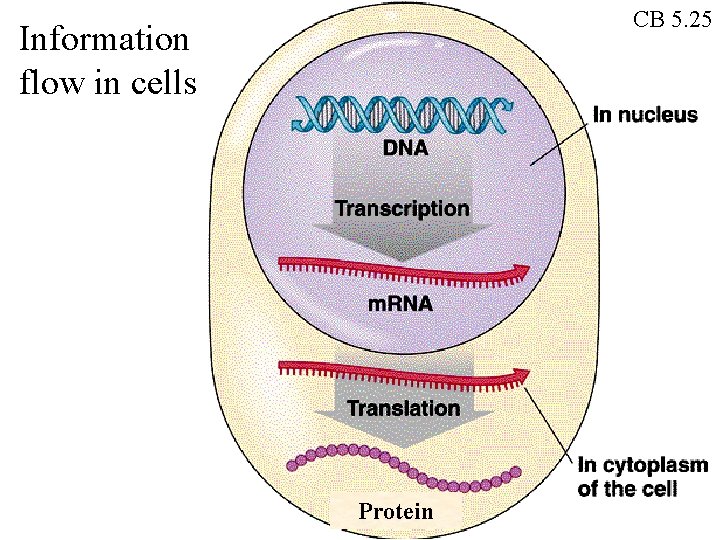
CB 5. 25 Information flow in cells Protein
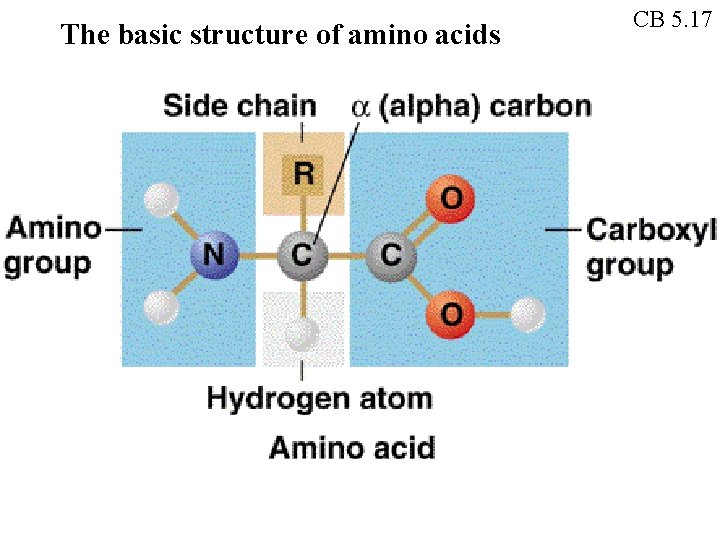
The basic structure of amino acids CB 5. 17
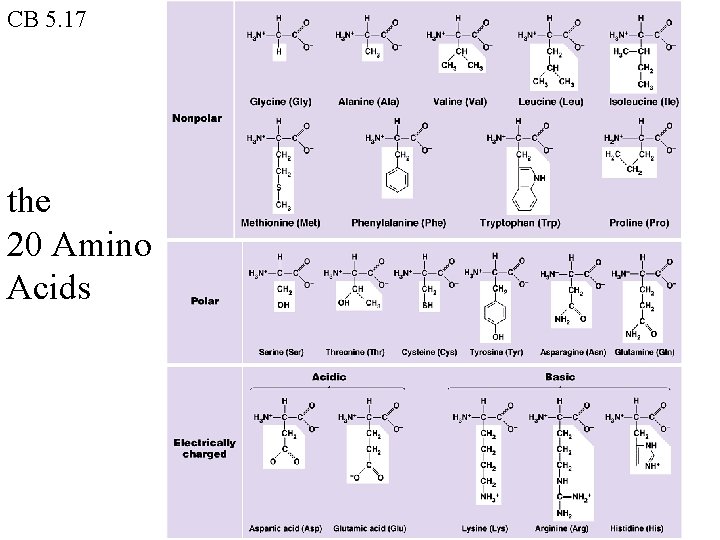
CB 5. 17 the 20 Amino Acids
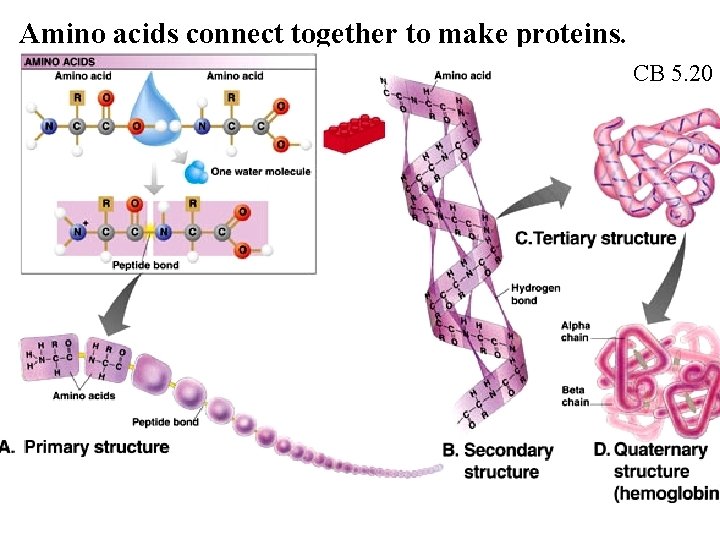
Amino acids connect together to make proteins. CB 5. 20
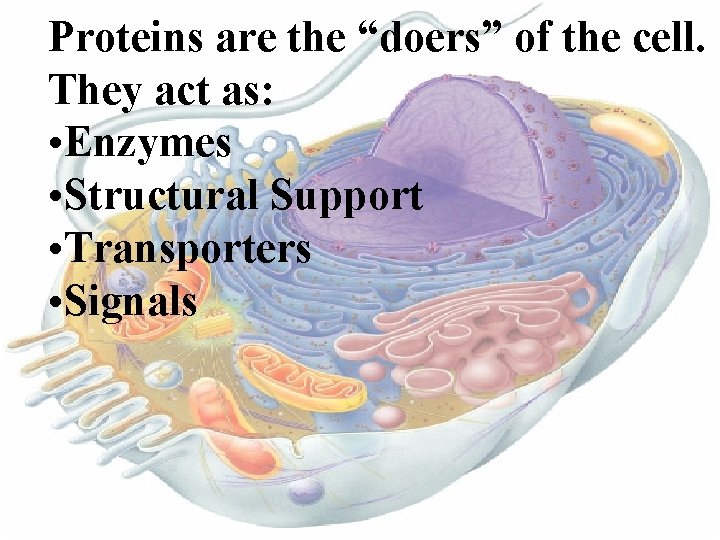
Proteins are the “doers” of the cell. They act as: • Enzymes • Structural Support • Transporters • Signals
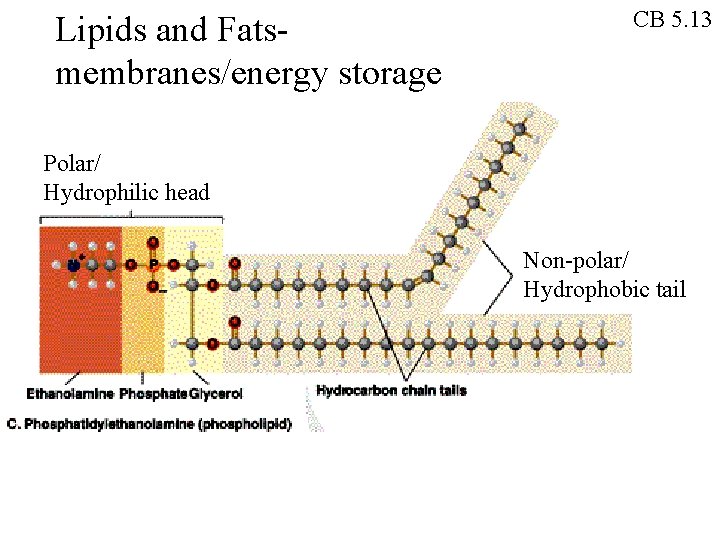
Lipids and Fatsmembranes/energy storage CB 5. 13 Polar/ Hydrophilic head Non-polar/ Hydrophobic tail
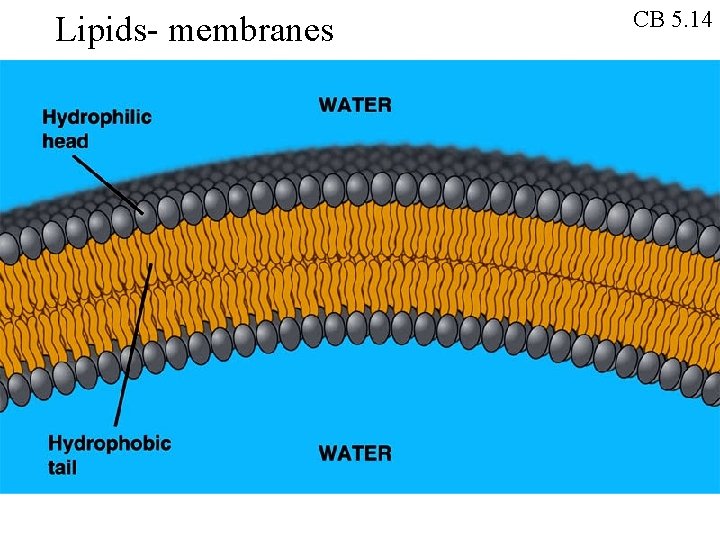
Lipids- membranes CB 5. 14
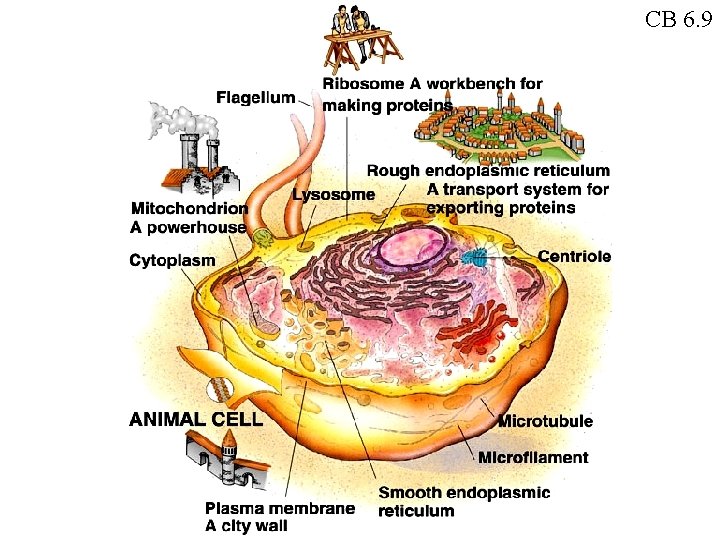
CB 6. 9
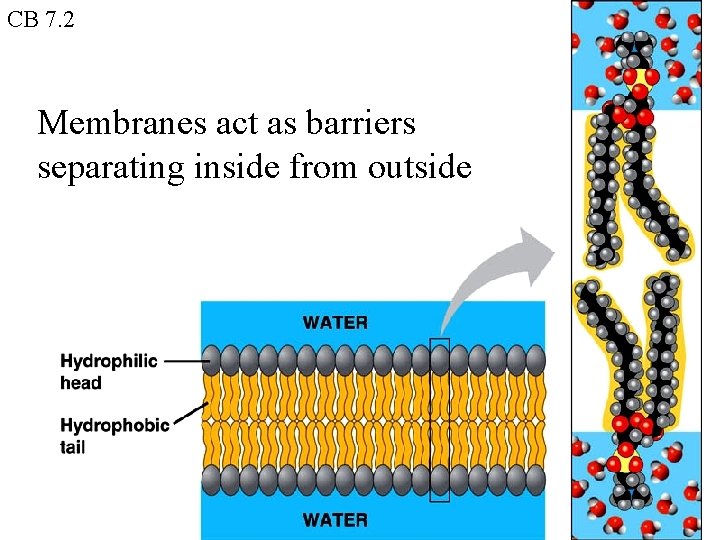
CB 7. 2 Membranes act as barriers separating inside from outside
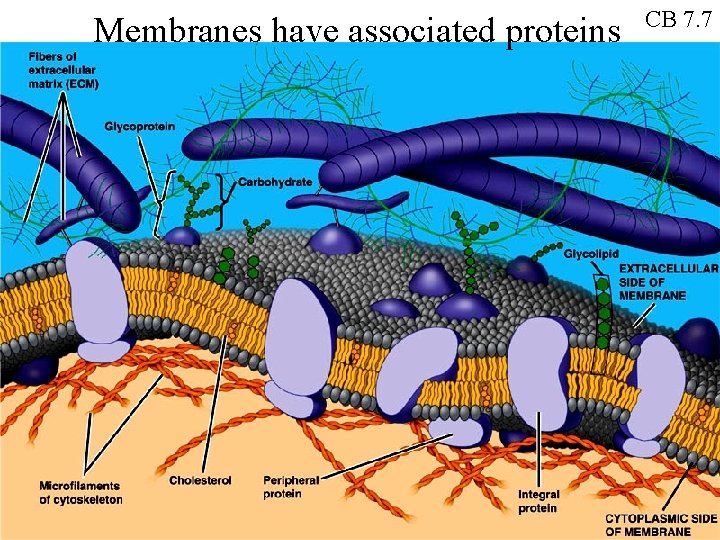
Membranes have associated proteins CB 7. 7
- Slides: 27