Anesthetics In 1846 a Boston dentist William Morton

Anesthetics In 1846, a Boston dentist, William Morton, demonstrated the use of ether as a general anesthetic in a public demonstration Successful operation led to the use of general anesthetics by surgeons and started the “age of anesthesia. ” “Its action is not yet thoroughly understood, and its use should be restricted to responsible persons. ”

General Anesthetics • A clinical definition of general anesthesia is a state where no movement occurs in response to what should be painful. • • Stages of General Anesthesia Analgesia (Stage I): The stage of analgesia lasts from onset of drowsiness to loss of eyelash reflex. The patient is considered unconscious at the end of stage I. Excitement (Stage II): The stage of excitement is characterized by agitation and delirium. During this stage, salivation may be copious. Heart rate and respiration Surgical Anesthesia (Stage III): During this stage, a painful stimuli will not elicit a somatic reflex or deleterious autonomic response. Impending Death (Stage IV): This stage lasts from onset of apnea to failure of circulation and respiration and ends in death. The ideal anesthetic combination will allow the patient to proceed quickly from stage I to stage III and avoid stage IV
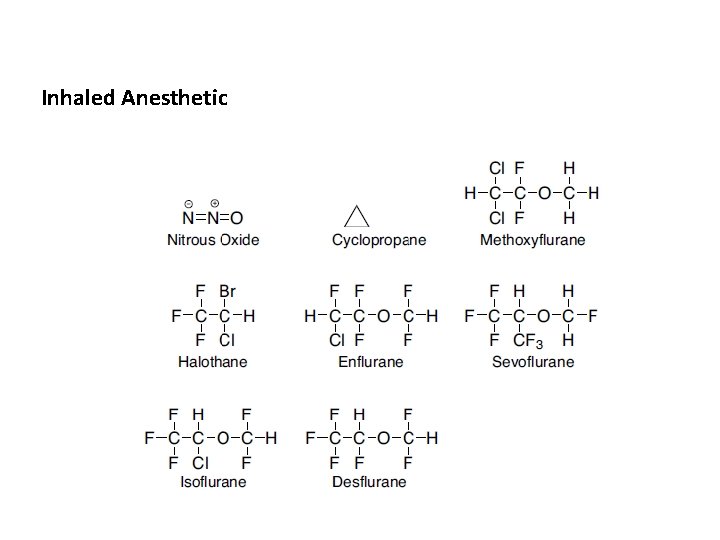
Inhaled Anesthetic

INJECTABLE GENERAL ANESTHETICS Propofol is an injectable sedative–hypnotic used for the induction and maintenance of anesthesia or sedation. positive modulator of the GABAA receptor.
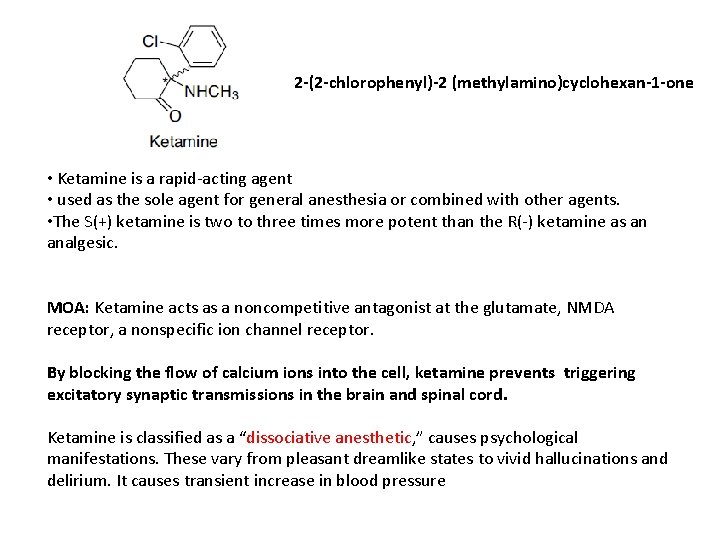
2 -(2 -chlorophenyl)-2 (methylamino)cyclohexan-1 -one • Ketamine is a rapid-acting agent • used as the sole agent for general anesthesia or combined with other agents. • The S(+) ketamine is two to three times more potent than the R(-) ketamine as an analgesic. MOA: Ketamine acts as a noncompetitive antagonist at the glutamate, NMDA receptor, a nonspecific ion channel receptor. By blocking the flow of calcium ions into the cell, ketamine prevents triggering excitatory synaptic transmissions in the brain and spinal cord. Ketamine is classified as a “dissociative anesthetic, ” causes psychological manifestations. These vary from pleasant dreamlike states to vivid hallucinations and delirium. It causes transient increase in blood pressure
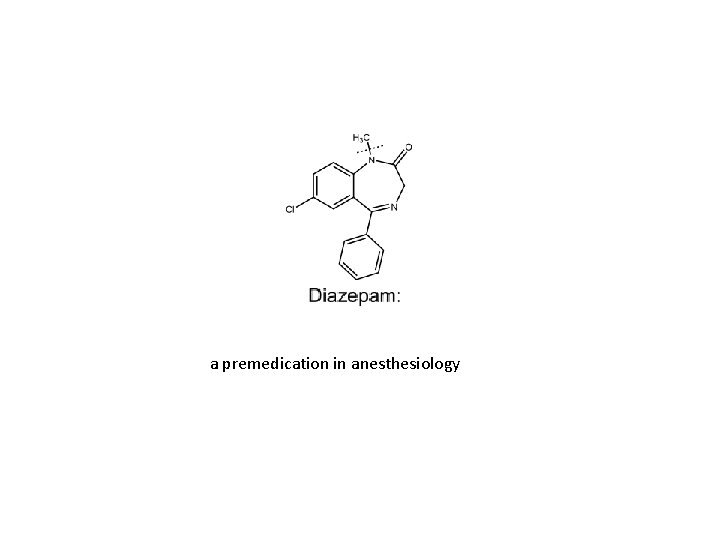
a premedication in anesthesiology

Local Anesthetics • local anesthetics inhibit the conduction of action potentials in all afferent and efferent nerve fibers. • Thus, pain and other sensations are not transmitted effectively to the brain • and motor impulses are not transmitted effectively to muscles. • Local anesthetics have various clinical uses to treat acute or chronic pain or to prevent the sensation of pain during procedures.
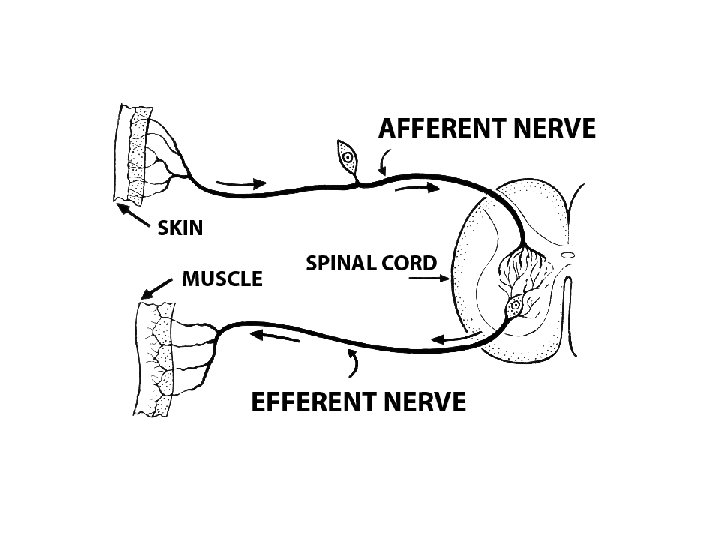
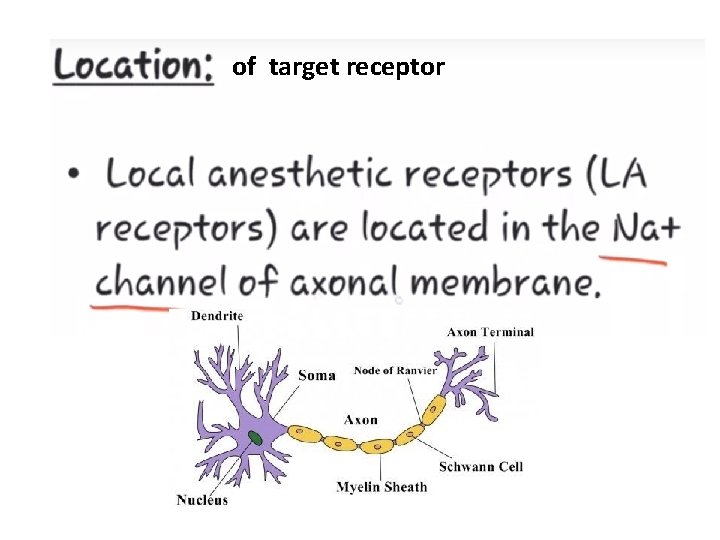
of target receptor

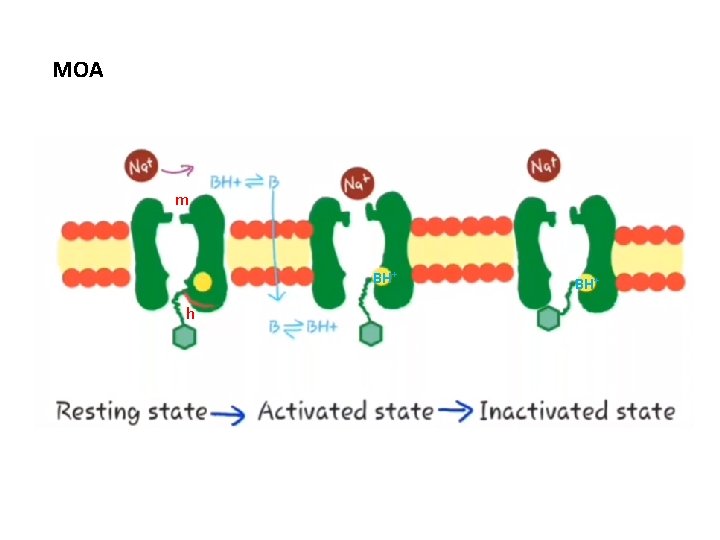
MOA m BH+ h BH+
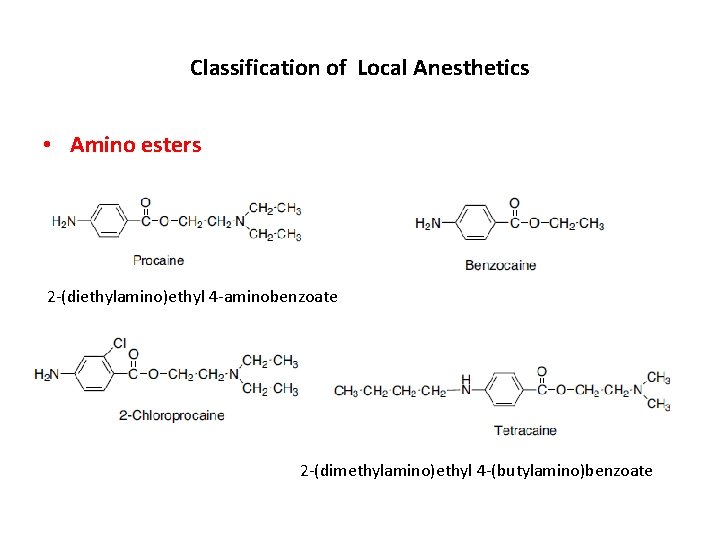
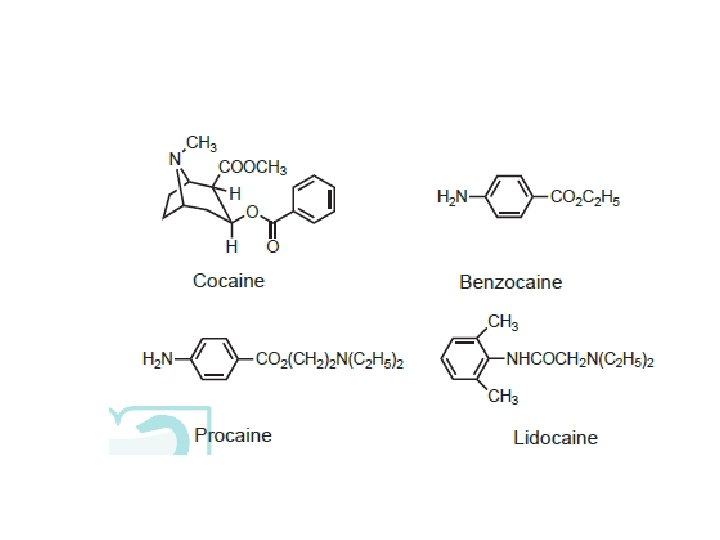
Classification of Local Anesthetics • Amino esters 2 -(diethylamino)ethyl 4 -aminobenzoate 2 -(dimethylamino)ethyl 4 -(butylamino)benzoate
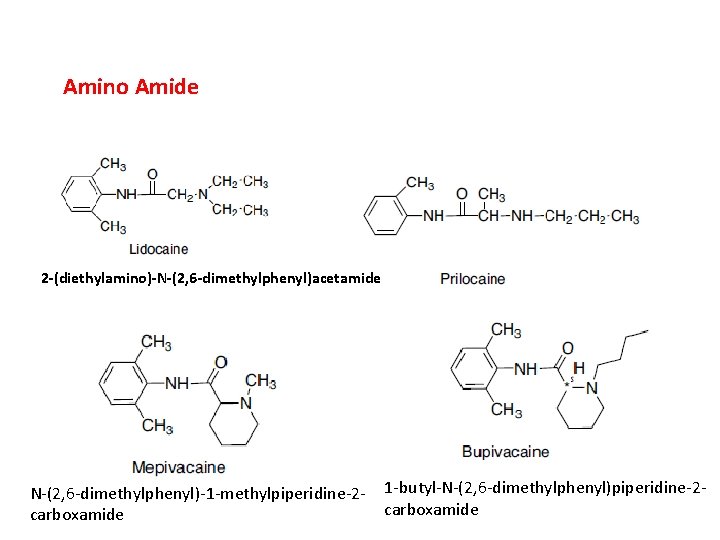
Amino Amide 2 -(diethylamino)-N-(2, 6 -dimethylphenyl)acetamide N-(2, 6 -dimethylphenyl)-1 -methylpiperidine-2 carboxamide 1 -butyl-N-(2, 6 -dimethylphenyl)piperidine-2 carboxamide
![• Amino ethers 4 -[3 -(4 -butoxyphenoxy)propyl]morpholine Alcohols 4 -Allyl-2 -methoxyphenol • Amino ethers 4 -[3 -(4 -butoxyphenoxy)propyl]morpholine Alcohols 4 -Allyl-2 -methoxyphenol](http://slidetodoc.com/presentation_image_h/30d5f720369d98090325aa9d90cd4877/image-14.jpg)
• Amino ethers 4 -[3 -(4 -butoxyphenoxy)propyl]morpholine Alcohols 4 -Allyl-2 -methoxyphenol
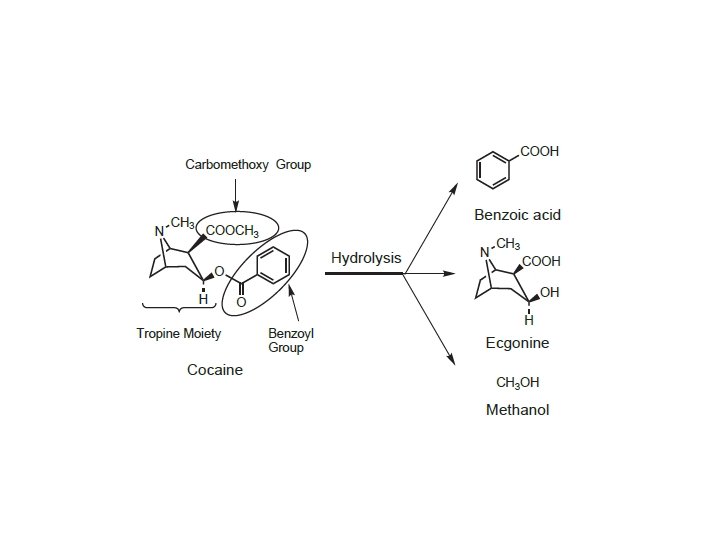

SARs of Local Anesthetics (a) (b) (c) The structure of most local anesthetic agents consists of three parts (a) lipophilic ring that may be substituted (b) linker of various lengths mainly ester or an amide (c) An amine group usually a tertiary amine with a p. Ka between 7. 5 and 9. 5
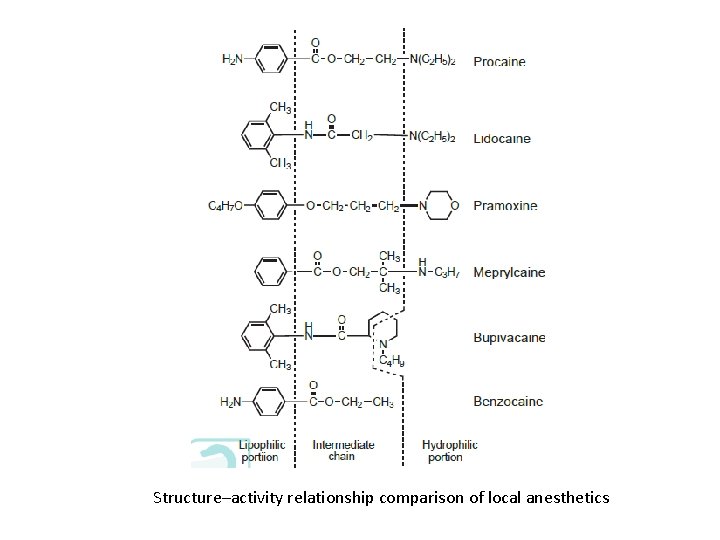
Structure–activity relationship comparison of local anesthetics
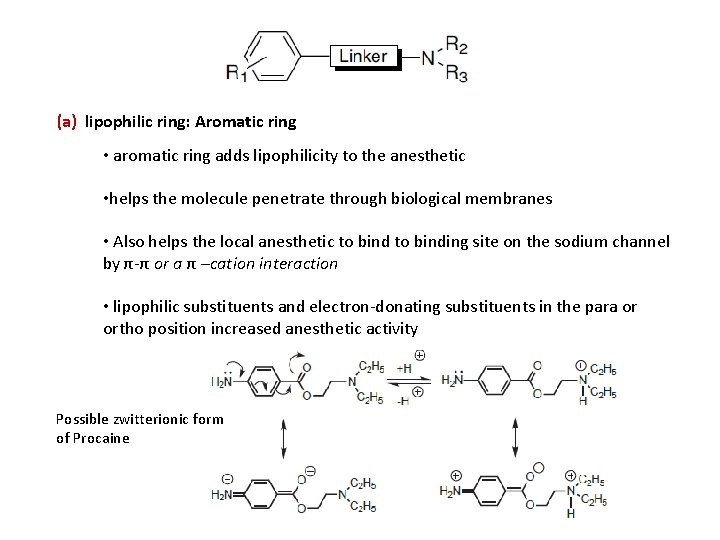
(a) lipophilic ring: Aromatic ring • aromatic ring adds lipophilicity to the anesthetic • helps the molecule penetrate through biological membranes • Also helps the local anesthetic to binding site on the sodium channel by π-π or a π –cation interaction • lipophilic substituents and electron-donating substituents in the para or ortho position increased anesthetic activity Possible zwitterionic form of Procaine
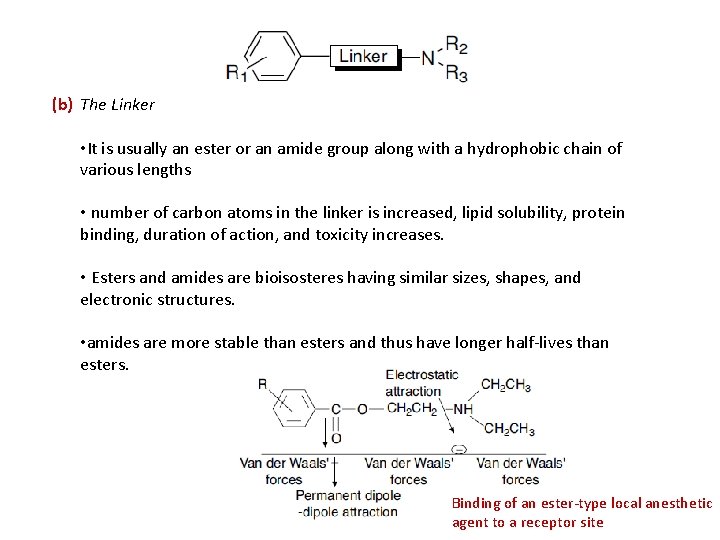
(b) The Linker • It is usually an ester or an amide group along with a hydrophobic chain of various lengths • number of carbon atoms in the linker is increased, lipid solubility, protein binding, duration of action, and toxicity increases. • Esters and amides are bioisosteres having similar sizes, shapes, and electronic structures. • amides are more stable than esters and thus have longer half-lives than esters. Binding of an ester-type local anesthetic agent to a receptor site
(c) An amine group • Most local anesthetics contain a tertiary nitrogen with p. Ka between 7. 5 and 9. 5 • So, at physiological p. H, both the cationic and neutral form of the molecule exists • Anesthetic compounds bind to the anesthetic receptor site in the ionized form. • compound can penetrate the nerve membrane in its neutral form • and then reequilibrate with its cationic form on the internal side of the membrane

• p. Ka of procaine is 8. 9; it has low lipid solubility • the ester group is unstable in basic solutions • Procaine is available inconcentrations ranging from 0. 25% to 10% with p. Hs adjusted to 5. 5 to 6. 0 • very quickly metabolized in the plasma by cholinesterases and in the liver • Ester hydrolysis produces PABA: the compound responsible for the allergic reactions • Procaine is not used topically because of its inability to pass through lipid membranes • for cutaneous or mucous membranes, for short procedures, for peripheral nerve block
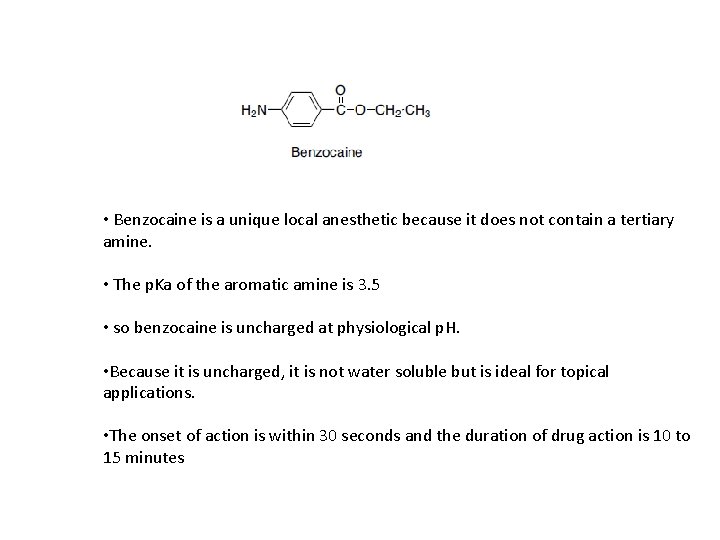
• Benzocaine is a unique local anesthetic because it does not contain a tertiary amine. • The p. Ka of the aromatic amine is 3. 5 • so benzocaine is uncharged at physiological p. H. • Because it is uncharged, it is not water soluble but is ideal for topical applications. • The onset of action is within 30 seconds and the duration of drug action is 10 to 15 minutes
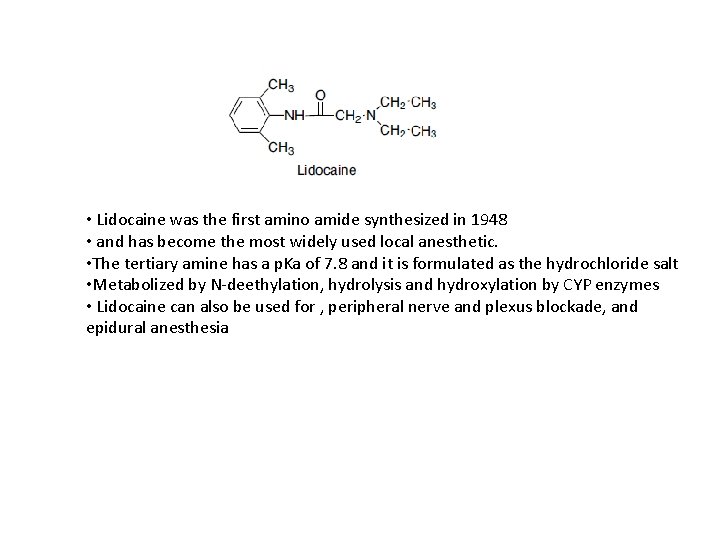
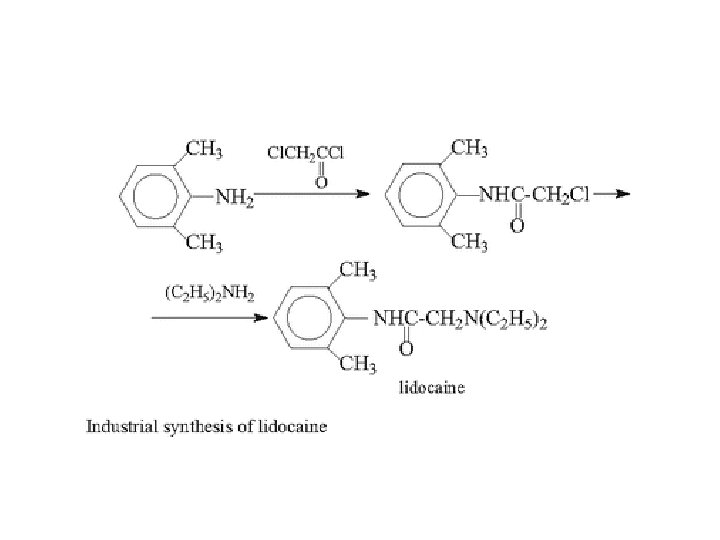
• Lidocaine was the first amino amide synthesized in 1948 • and has become the most widely used local anesthetic. • The tertiary amine has a p. Ka of 7. 8 and it is formulated as the hydrochloride salt • Metabolized by N-deethylation, hydrolysis and hydroxylation by CYP enzymes • Lidocaine can also be used for , peripheral nerve and plexus blockade, and epidural anesthesia
- Slides: 25