Anatomy Physiology SIXTH EDITION Lecture 27 Fluid Electrolyte





















- Slides: 33

Anatomy & Physiology SIXTH EDITION Lecture 27: Fluid, Electrolyte, and Acid-Base Balance Lecturer: Dr. Barjis Room: P 313 Phone: (718) 260 -5285 E-Mail: ibarjis@citytech. cuny. edu Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings Frederic H. Martini Fundamentals of

Learning Objectives • Explain what is meant by “fluid balance, ” “electrolyte balance, ” and “acid-base balance” • Compare the compositions of intracellular and extracellular fluids • Identify the hormones that play important roles in regulating fluid and electrolyte balance • Describe the movement of fluid that takes place within the ECF, between the ECF and the ICF, and between the ECF and the environment

Learning Objectives • Discuss how sodium, potassium, calcium and chloride ions are regulated to maintain electrolyte balance • Explain the buffering systems that balance the p. H of the intracellular and extracellular fluids • Describe the compensatory mechanisms involved in acid-base balance

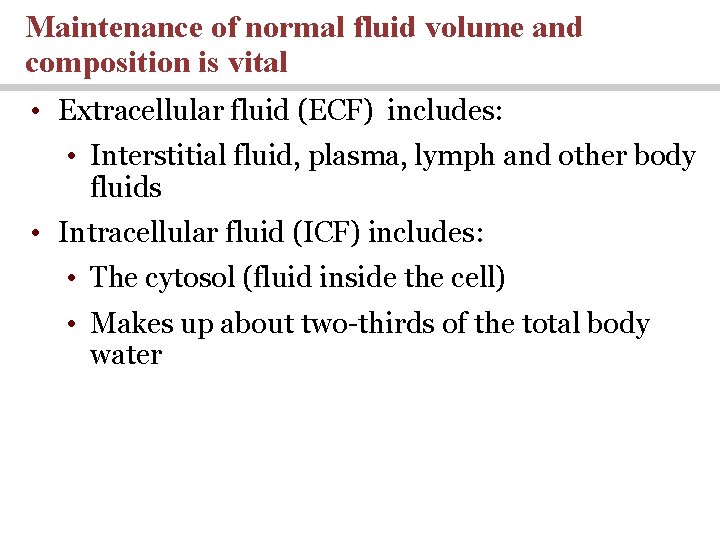
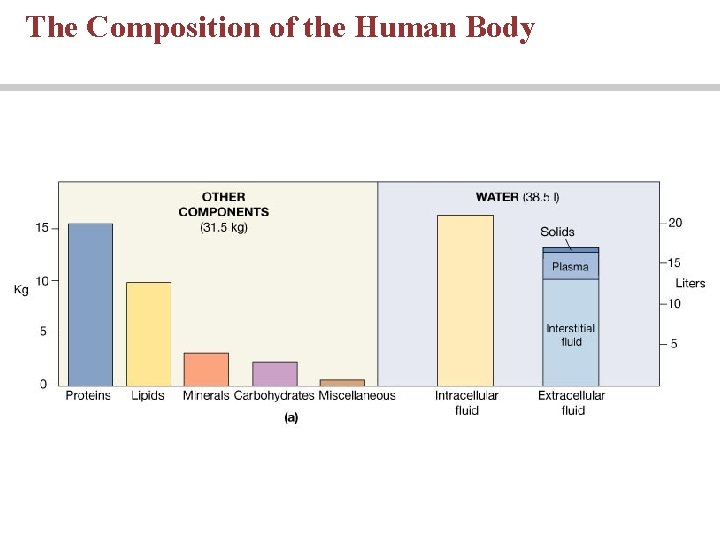
Maintenance of normal fluid volume and composition is vital • Extracellular fluid (ECF) includes: • Interstitial fluid, plasma, lymph and other body fluids • Intracellular fluid (ICF) includes: • The cytosol (fluid inside the cell) • Makes up about two-thirds of the total body water

Fluid and electrolyte balance • Fluid is in a balance when the amount of water gained (e. g. through the digestive system) each day equals the amount of fluid lost (e. g. through urinary system, sweat glands) each day • Electrolyte balance • The ion gain each day equals the ion loss • Electrolytes are ions released through the dissociation of inorganic compounds • Acid-base balance • H+ gain is offset by their loss • When acid–base balance exists, the p. H of body fluids remains within normal limits (7. 35 -7. 45).

The Composition of the Human Body

Regulation of fluids and electrolytes • Homeostatic mechanisms respond to changes in ECF • No receptors directly monitor fluid or electrolyte balance. • However fluid and elctrolyte can be monitored by responding to changes in plasma volume or osmotic concentrations • All water moves passively in response to osmotic gradients • Body content of water or electrolytes rises if intake exceeds outflow

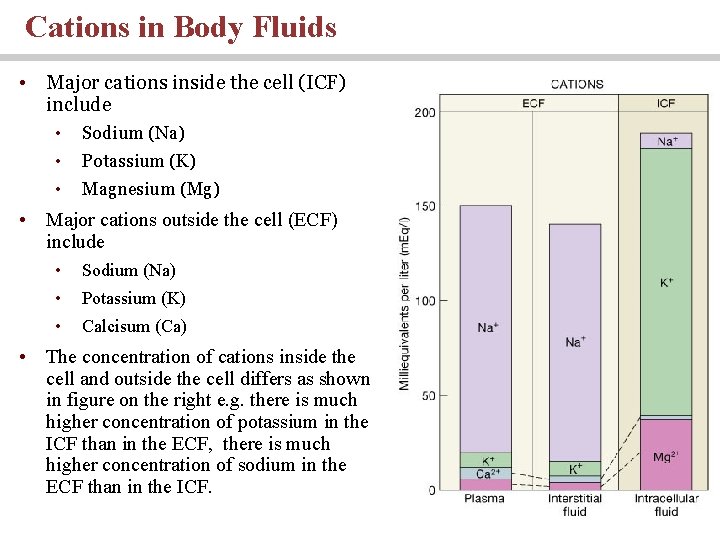
Cations in Body Fluids • Major cations inside the cell (ICF) include • Sodium (Na) • Potassium (K) • Magnesium (Mg) • Major cations outside the cell (ECF) include • Sodium (Na) • Potassium (K) • Calcisum (Ca) • The concentration of cations inside the cell and outside the cell differs as shown in figure on the right e. g. there is much higher concentration of potassium in the ICF than in the ECF, there is much higher concentration of sodium in the ECF than in the ICF.

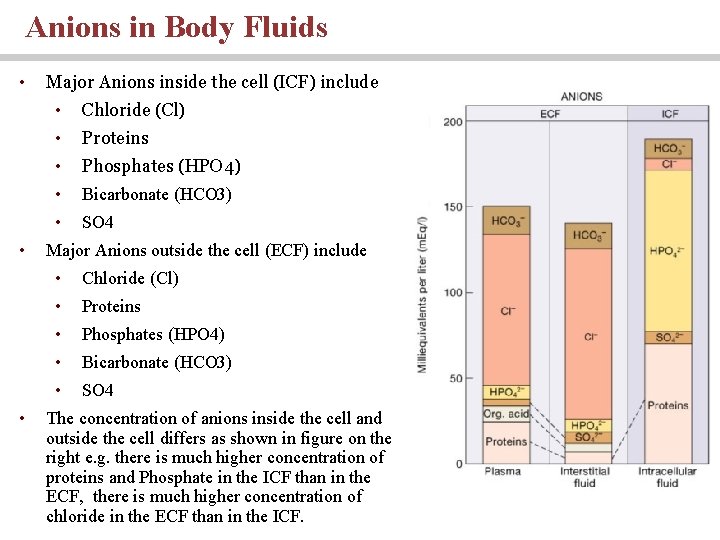
Anions in Body Fluids • • • Major Anions inside the cell (ICF) include • Chloride (Cl) • Proteins • Phosphates (HPO 4) • Bicarbonate (HCO 3) • SO 4 Major Anions outside the cell (ECF) include • Chloride (Cl) • Proteins • Phosphates (HPO 4) • Bicarbonate (HCO 3) • SO 4 The concentration of anions inside the cell and outside the cell differs as shown in figure on the right e. g. there is much higher concentration of proteins and Phosphate in the ICF than in the ECF, there is much higher concentration of chloride in the ECF than in the ICF.

Cations and Anions in Body Fluids • Despite the differences in the concentration of specific substances, the ICF and ECF osmotic concentrations are identical • If the cell membrane were freely permeable, diffusion would continue until these ions were evenly distributed across the membrane

Primary regulatory hormones • Fluid balance and electrolyte balance are mediated by three hormones: • Antidiuretic hormone (ADH) • Stimulates water conservation and the thirst center • Aldosterone • Controls Na+ absorption and K+ loss along the DCT • Natriuretic peptides (ANP and BNP) • Reduce thirst and block the release of ADH and aldosterone

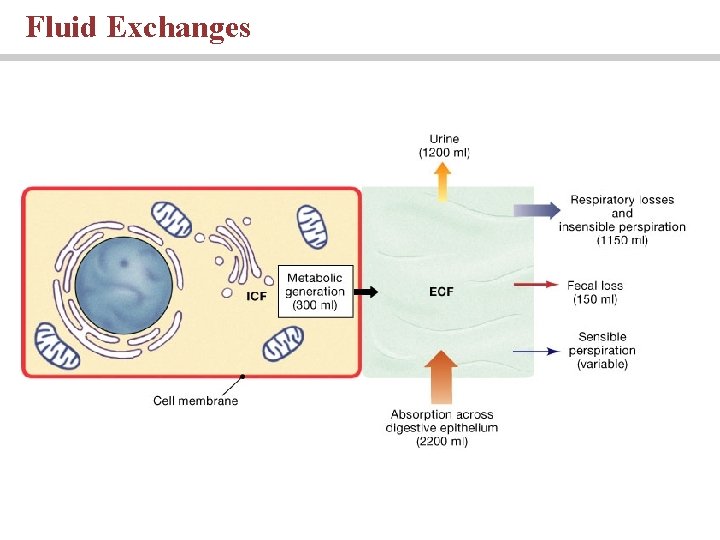
Fluid movement within the ECF • Fluid moves freely within ECF compartment • Water, electrolytes, proteins, fats and other nutrients and molceules are normally balanced by gains via • Eating • Drinking • Metabolic generation • Losses of water and other molcules are by: • Urine • Respiratory losses • Perspiration • Fecal Loss

Fluid Exchanges

Water excess and depletion • Hyponatremia • Na+ concentration in the ECF is reduced (overhydration) • Hypernatremia • Na+ in the ECF is abnormally high • Dehydration • Develops when water loss outpaces water gains

Fluid shifts • Water movement between ECF and ICF • If ECF becomes hypertonic relative to ICF, water moves from ICF to ECF • If ECF becomes hypotonic relative to ICF, mater moves from ECF into cells

Problems with Electrolyte Balance • Usually result from sodium ion imbalances • Potassium imbalances are less common, but more dangerous

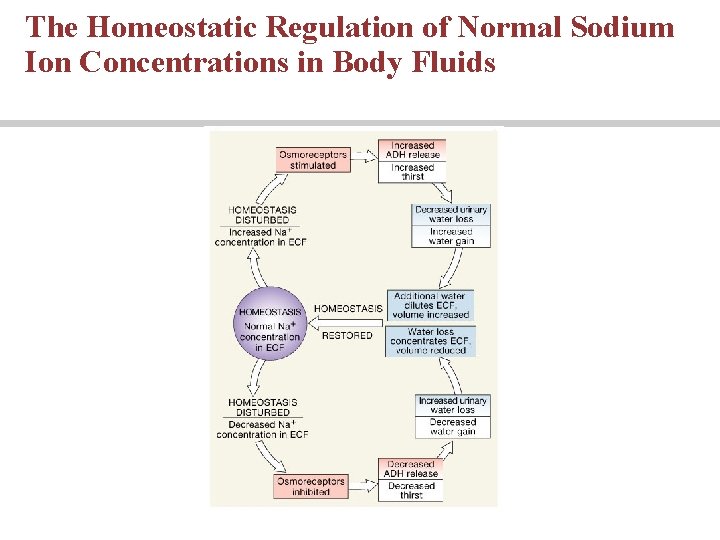
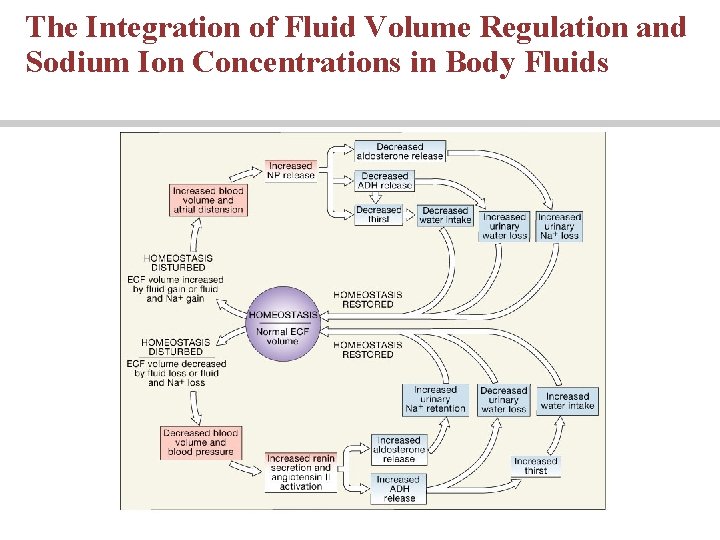
Sodium balance • Rate of sodium uptake across digestive tract directly proportional to dietary intake • Sodium losses occur through urine and perspiration • Shifts in sodium balance result in expansion or contraction of ECF • Large variations corrected by homeostatic mechanisms • Too low, ADH / aldosterone secreted • Too high, ANP secreted

The Homeostatic Regulation of Normal Sodium Ion Concentrations in Body Fluids

The Integration of Fluid Volume Regulation and Sodium Ion Concentrations in Body Fluids

Potassium balance • Potassium ion concentrations in ECF are low • Not as closely regulated as sodium • Potassium ion excretion increases as • ECF concentrations rise • Aldosterone secreted • p. H rises • Potassium retention occurs when p. H falls

ECF Concentrations of other electrolytes • Calcium balance • Bone reserves, absorption in the digestive tract, and loss at kidneys • Magnesium balance • Absorbed by the PCT to keep pace with urinary losses

Acid-base Balance The importance of p. H control • The p. H of the ECF remains between 7. 35 and 7. 45 • If plasma levels fall below 7. 35 (acidemia), acidosis results • If plasma levels rise above 7. 45 (alkalemia), alkalosis results • Alteration outside these boundaries affects all body systems e. g. can result in coma, cardiac failure, and circulatory collapse

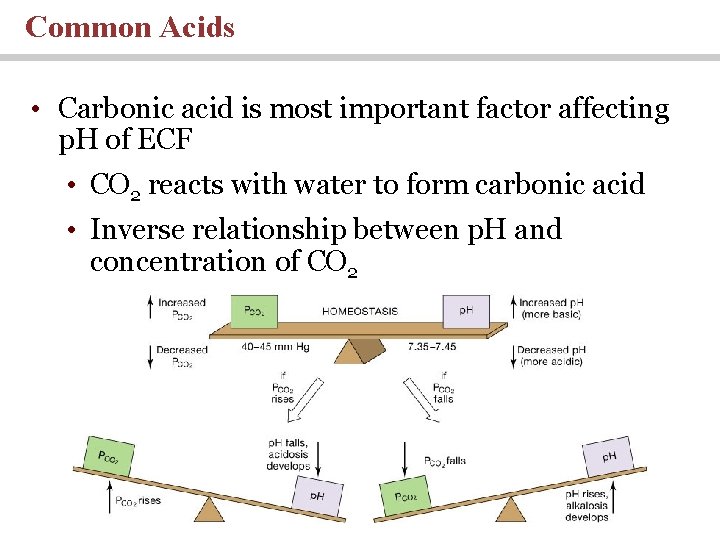
Common Acids • Carbonic acid is most important factor affecting p. H of ECF • CO 2 reacts with water to form carbonic acid • Inverse relationship between p. H and concentration of CO 2

Mechanisms of p. H control • Buffer system consists of a weak acid and its anion • Three major buffering systems: • Protein buffer system • Amino acid • Hemoglobin buffer system • H+ are buffered by hemoglobin • Carbonic acid-bicarbonate • Buffers changes caused by organic and fixed acids

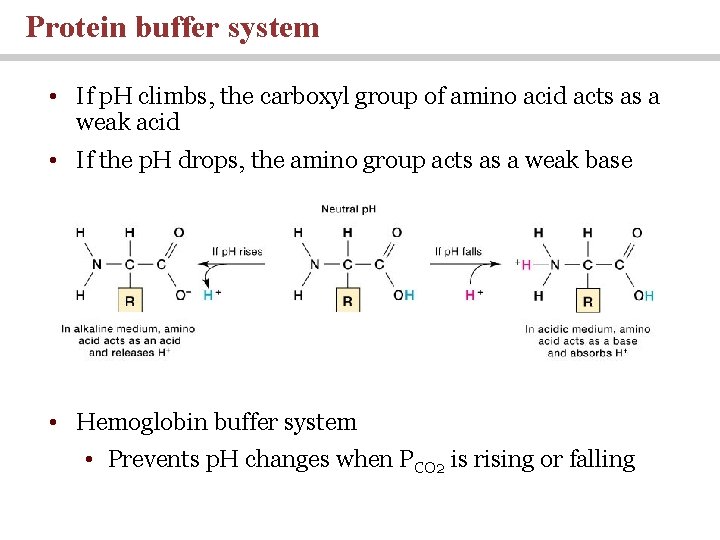
Protein buffer system • If p. H climbs, the carboxyl group of amino acid acts as a weak acid • If the p. H drops, the amino group acts as a weak base • Hemoglobin buffer system • Prevents p. H changes when PCO 2 is rising or falling

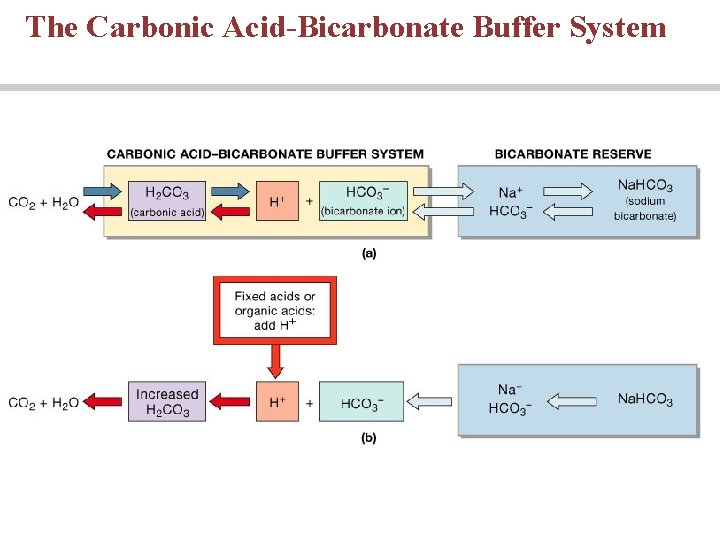
Carbonic Acid-Bicarbonate Buffering System • Carbonic acid-bicarbonate buffer system • CO 2 + H 2 O H 2 CO 3 H+ + CO 3– • Has the following limitations: • Cannot protect the ECF from p. H changes due to increased or depressed CO 2 levels • Only functions when respiratory system and control centers are working normally • It is limited by availability of bicarbonate ions (bicarbonate reserve)

The Carbonic Acid-Bicarbonate Buffer System

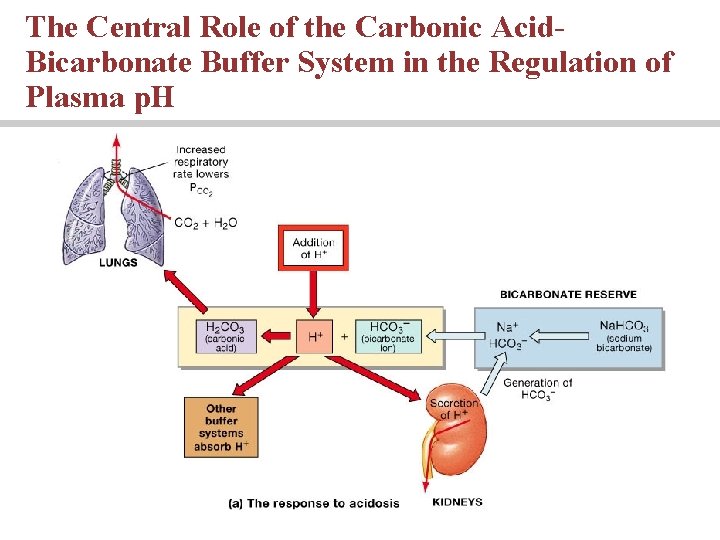
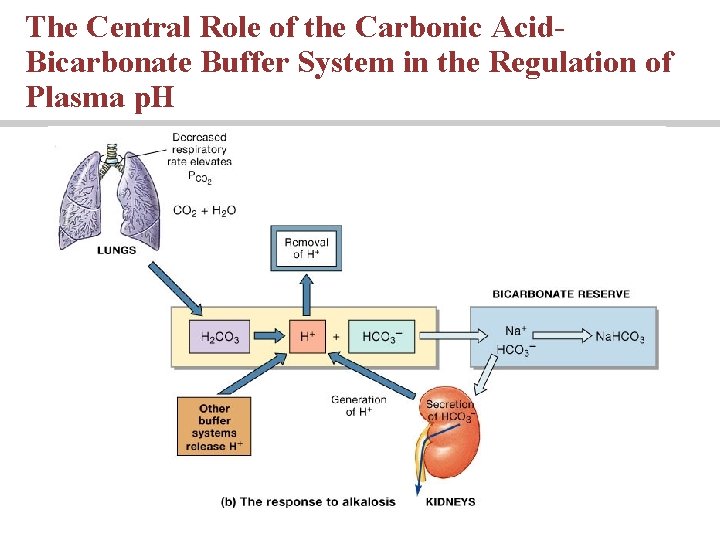
Maintenance of acid-base balance • Lungs help regulate p. H through carbonic acid bicarbonate buffer system • Changing respiratory rates changes PCO 2 • Respiratory compensation • Kidneys help regulate p. H through renal compensation

The Central Role of the Carbonic Acid. Bicarbonate Buffer System in the Regulation of Plasma p. H

The Central Role of the Carbonic Acid. Bicarbonate Buffer System in the Regulation of Plasma p. H

Changes with age include • Reduced total body water content • Impaired ability to perform renal compensation • Increased water demands • Reduced ability to concentrate urine • Reduced sensitivity to ADH/ aldosterone • Net loss of minerals • Inability to perform respiratory compensation • Secondary conditions that affect fluid, electrolyte, acidbase balance

You should now be familiar with: • What is meant by “fluid balance, ” “electrolyte balance, ” and “acid-base balance” • The compositions of intracellular and extracellular fluids • The hormones that play important roles in regulating fluid and electrolyte balance • The movement of fluid that takes place within the ECF, between the ECF and the ICF, and between the ECF and the environment

You should now be familiar with: • How sodium, potassium, calcium and chloride ions are regulated to maintain electrolyte balance • The buffering systems that balance the p. H of the intracellular and extracellular fluids • The compensatory mechanisms involved in acidbase balance