Anatomy Physiology Lecture 2 Chapter 2 The Chemical
































- Slides: 53

Anatomy & Physiology Lecture 2: Chapter 2 The Chemical Level of Organization Pages: 26 - 61 Lecturer: Dr. Barjis Room: P 313/P 307 Phone: (718) 260 -5285 E-Mail: ibarjis@citytech. cuny. edu Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings Frederic H. Martini Fundamentals of

Learning Objectives • Describe an atom and compare the ways atoms combine to form molecules. • Distinguish among the types of chemical reactions that are important to physiology. • Describe the role of enzymes in metabolism. • Distinguish between organic and inorganic compounds. • Explain the importance of water, p. H and buffers to living systems. • Discuss the structures and functions of carbohydrates, lipids, proteins, nucleic acids and high energy compounds.

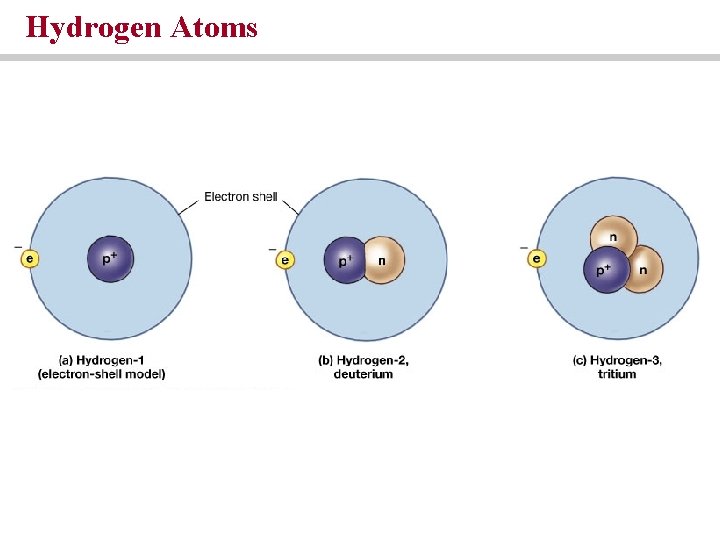
Atoms, Molecules and Bonds Atoms are the smallest stable units of matter • Subatomic particles • Protons = positive charge; weight of approximately 1 Dalton • Neutrons = no charge; weight similar to protons • Electrons = negative charge; weigh 1/1836 th Dalton • Protons and neutrons are found in the nucleus; electrons occupy electron cloud • Atomic number = proton number; atomic mass = protons and neutrons • Isotopes are elements with similar numbers of protons but different numbers of neutron

Hydrogen Atoms

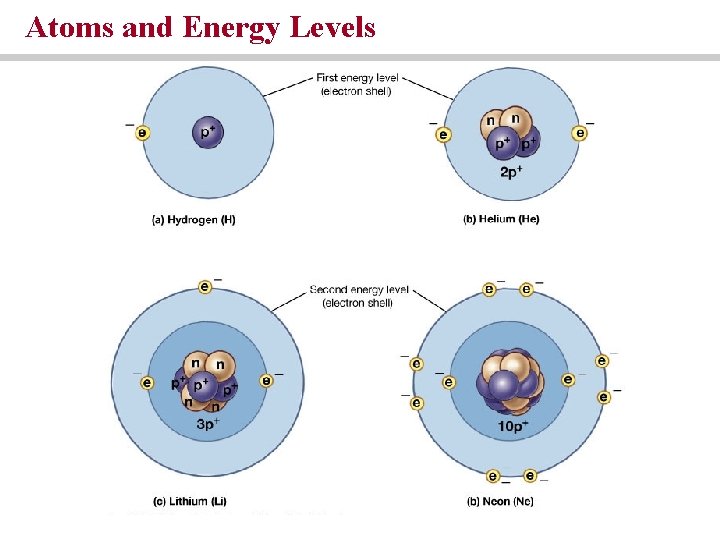
Electrons occupy a series of energy levels or electron shells. • The outermost electron shell determines the reactivity of the element.

Atoms and Energy Levels

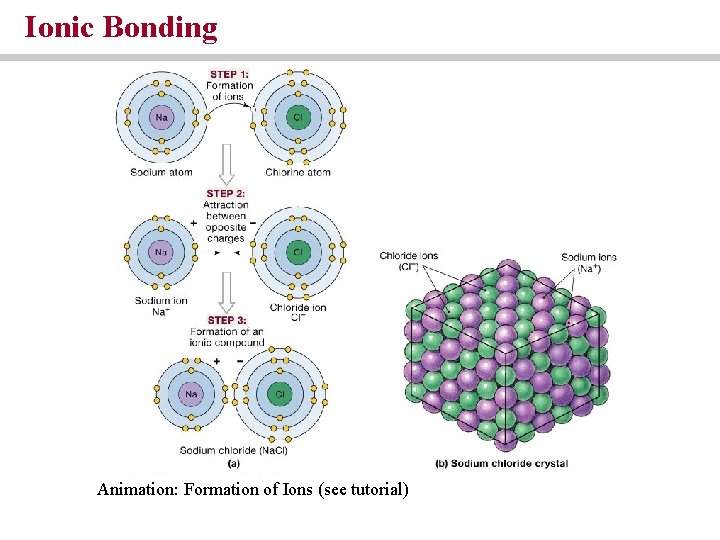
Atoms combine through chemical reactions • Molecule = a chemical structure consisting of molecules held together by covalent bonds • Compound = a chemical substance composed of atoms of two or more elements • There are three types of bond: Ionic, covalent, and hydrogen • Ionic = attraction between positive cations and negative anions

Ionic Bonding Animation: Formation of Ions (see tutorial)

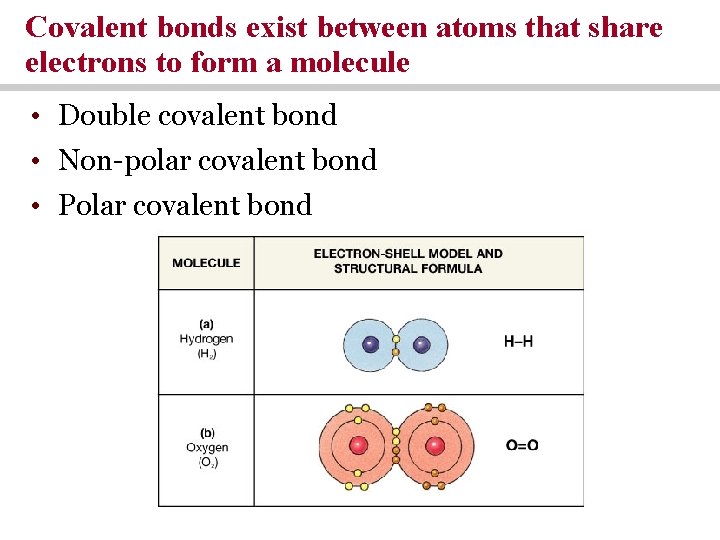
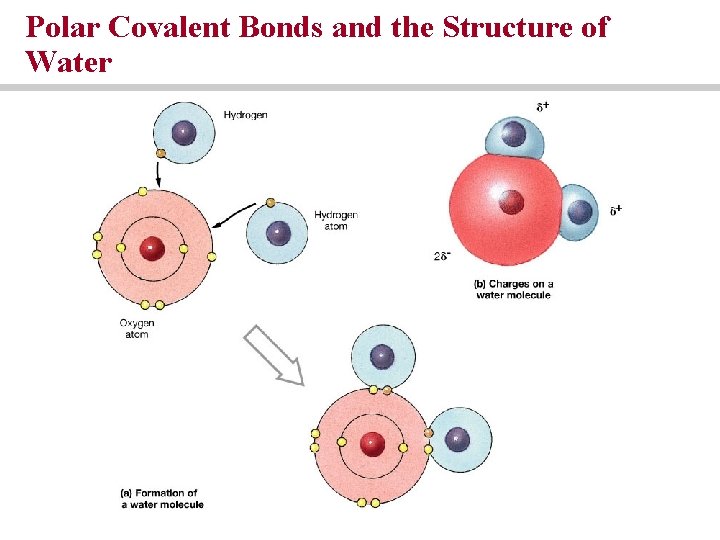
Covalent bonds exist between atoms that share electrons to form a molecule • Double covalent bond • Non-polar covalent bond • Polar covalent bond

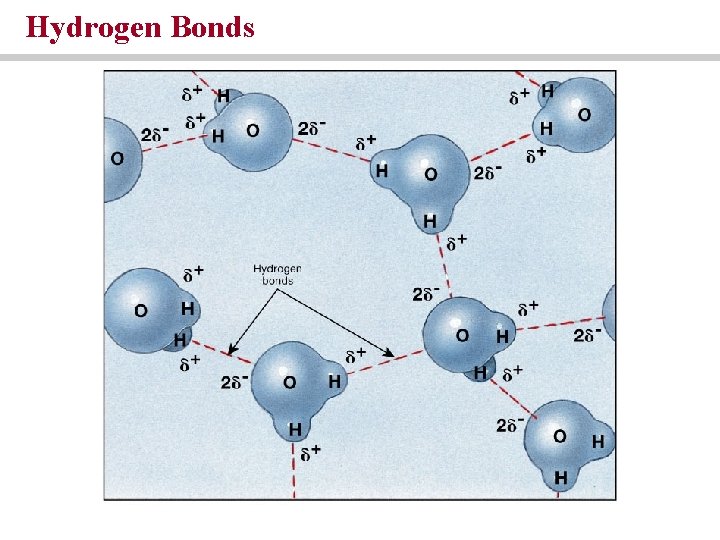
Hydrogen bonds are weak forces that affect the shape and properties of compounds • Polar covalent bonds that occur when hydrogen covalently bonds with another element

Polar Covalent Bonds and the Structure of Water

Hydrogen Bonds

Matter and chemical notation • Matter can exist as a solid, liquid or gas • Depends on the interaction of the component atoms or molecules • Molecular weight is the sum of the atomic weights of the component atoms • Chemical notation • Short-hand that describes chemical compounds and reactions • See table 2. 2 for examples of chemical notation

Chemical Reactions A chemical reaction occurs when reactants combine to generate one or more products • All chemical reactions in the body constitutes metabolism • Metabolism provides for the capture, storage and release of energy

Basic energy concepts • Work = movement of an object or change in its physical structure • Energy = the capacity to perform work • Kinetic energy is energy of motion • Potential energy is stored energy resulting from position or structure • Conversions are not 100% efficient, resulting in release of heat

Metabolism • Types of reaction • Decomposition • Synthesis • Exchange • Metabolism is the sum of all reactions • Through catabolism cells gain energy (break down of complex molecules) • Anabolism uses energy (synthesis of new molecules)

Reversible reactions • All reactions are theoretically reversible • At equilibrium the rates of two opposing reactions are in balance • Anabolism = catabolism

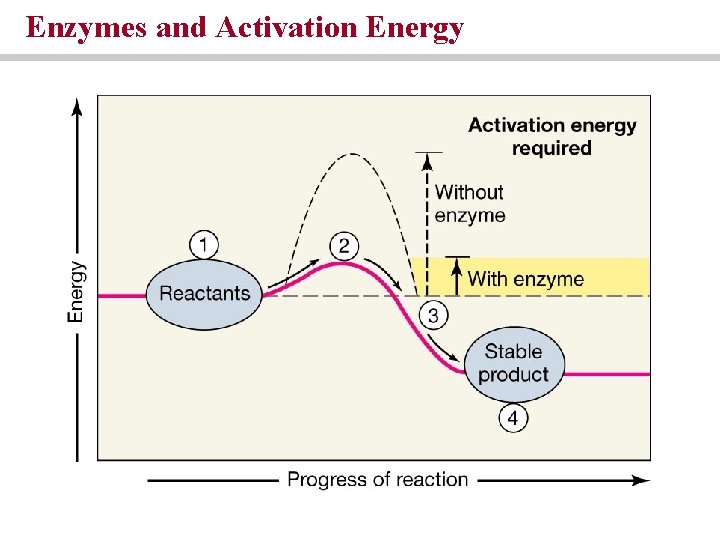
Enzymes, energy and chemical reactions • Activation energy is the amount of energy needed to begin a reaction • Enzymes are catalysts • Reduce energy of activation without being permanently changed or used up • Promote chemical reactions

Enzymes and Activation Energy

Inorganic Compounds Nutrients and Metabolites • Nutrients are essential chemical compounds obtained from the diet • Metabolites are molecules synthesized or broken down inside the body • These can be classified as organic or inorganic compounds • Organic compounds have carbon and hydrogen as their primary structural component • Inorganic compounds are not primarily carbon and hydrogen

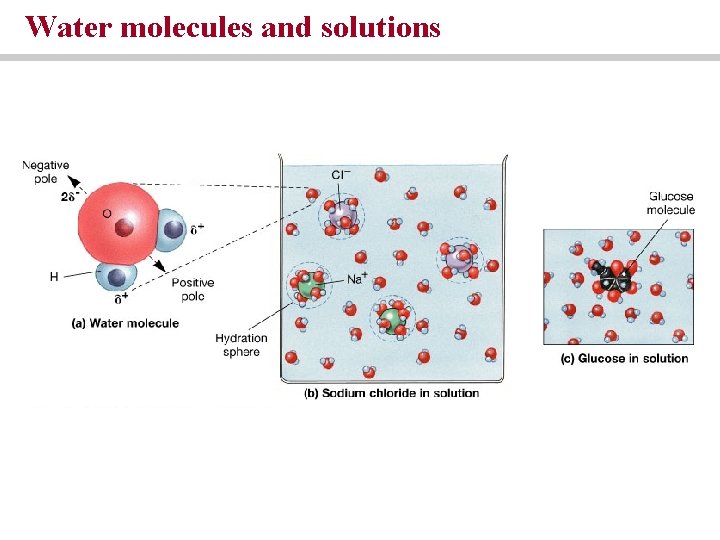
Water and its properties • Water is the most important constituent of the body • Solution is a uniform mixture of two or more substances • Solvent is the medium in which molecules of solute are dispersed • Water is the solvent in aqueous solutions

Water molecules and solutions

Electrolytes undergo ionization • Compounds that interact readily with water are hydrophilic • Compounds that do not interact with water are hydrophobic

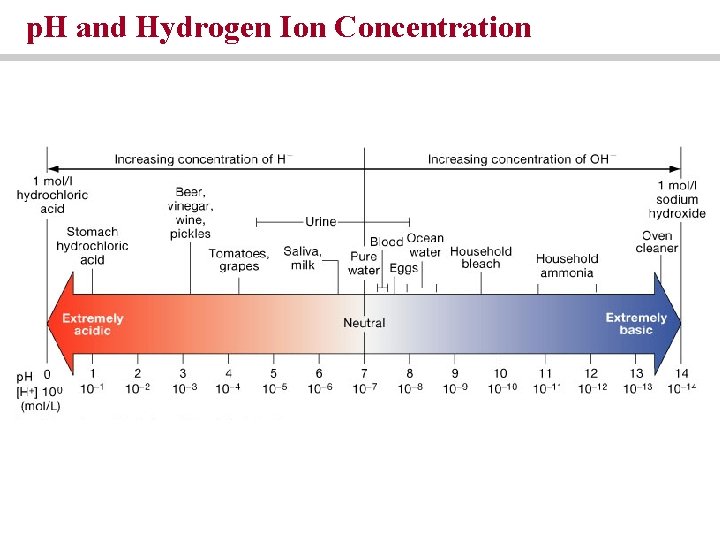
p. H is a measure of the concentration of hydrogen ions solution • Neutral • Acidic • Basic

Acids and Bases • Acids release hydrogen ions into solution • Bases remove hydrogen ions from solution • Strong acids and strong bases ionize completely • Weak acids and weak bases do not ionize

p. H and Hydrogen Ion Concentration

Salts and buffers • Salt = an electrolyte whose cation is not hydrogen and whose anion is not hydroxide • Buffers remove or replace hydrogen ions in solution • Buffer systems maintain the p. H of body fluids

Organic compounds • Organic compounds generally include • Carbon • Hydrogen • and sometimes Oxygen

Organic compounds • Four major classes of organic compounds are • Carbohydrates • Lipids • Proteins • Nucleic acids • High energy compounds are also organic compounds

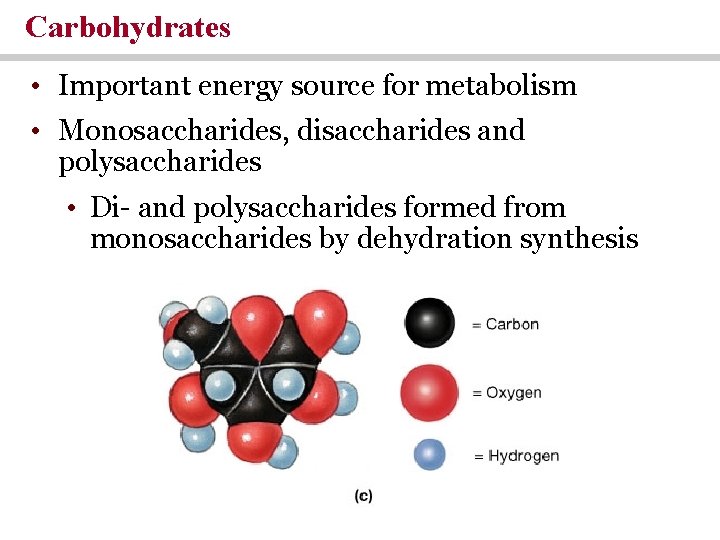
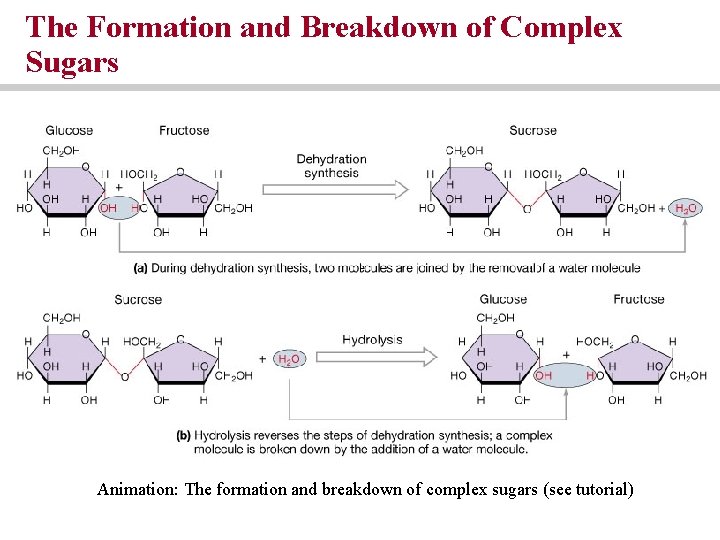
Carbohydrates • Important energy source for metabolism • Monosaccharides, disaccharides and polysaccharides • Di- and polysaccharides formed from monosaccharides by dehydration synthesis

The Formation and Breakdown of Complex Sugars Animation: The formation and breakdown of complex sugars (see tutorial)

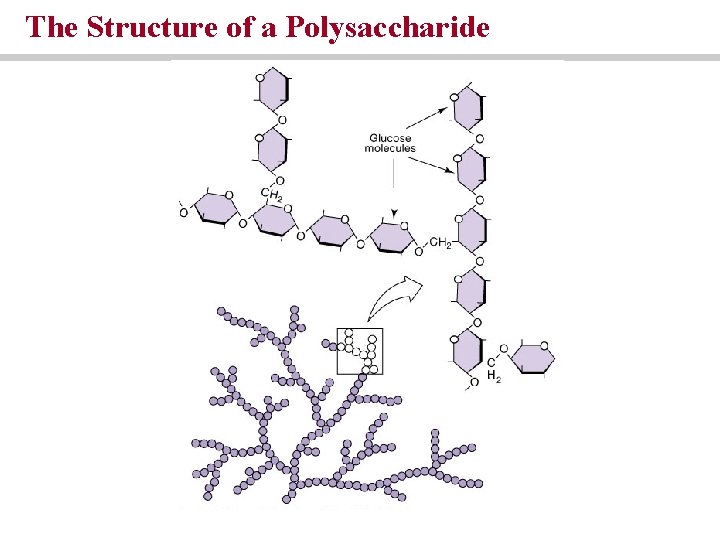
The Structure of a Polysaccharide

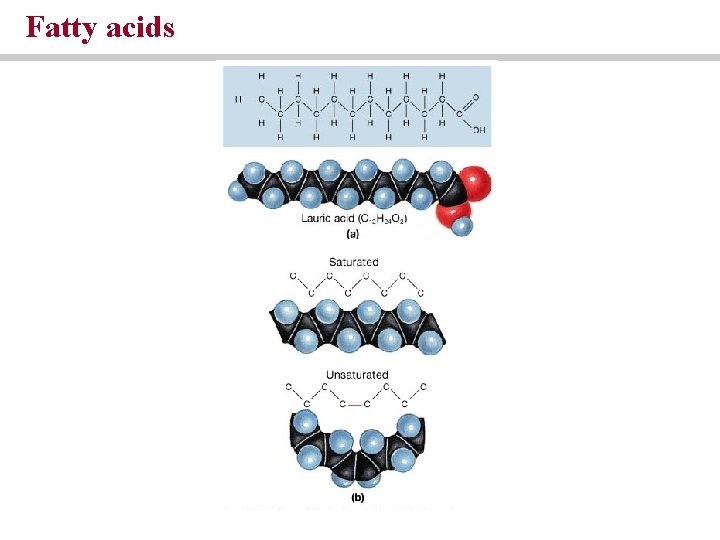
Lipids include fats, oils, and waxes • Five classes: • Fatty acids • Eicosanoids • Glycerides • Steroids • Phospholipids • Glycolipids

Fatty acids

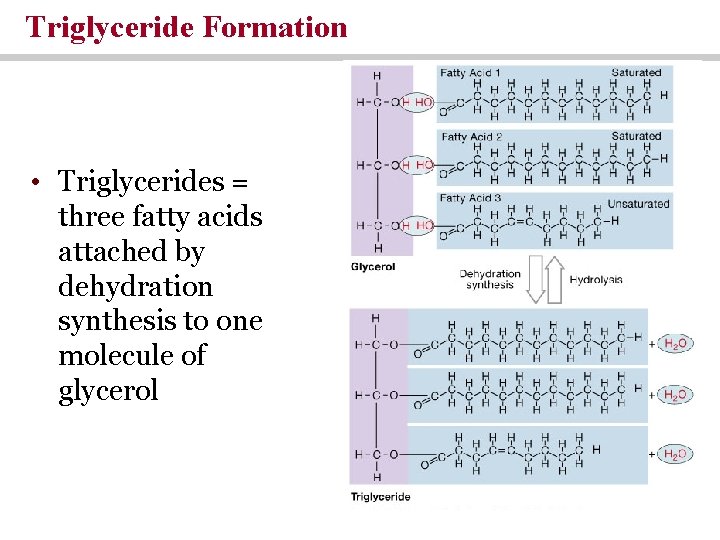
Triglyceride Formation • Triglycerides = three fatty acids attached by dehydration synthesis to one molecule of glycerol

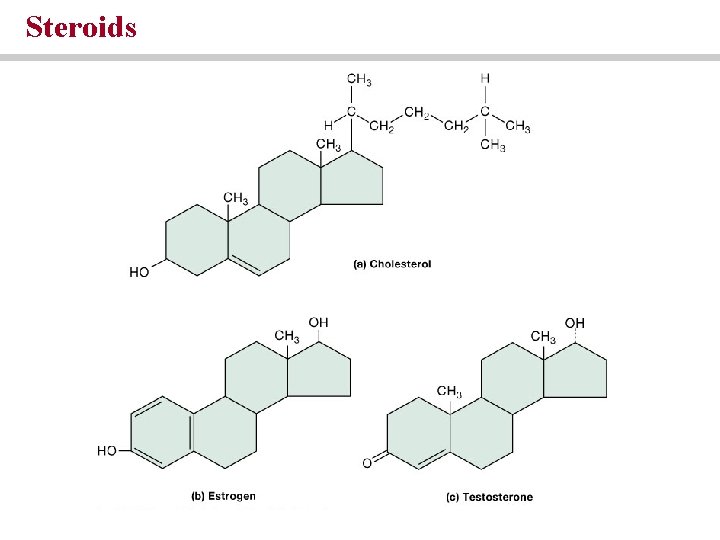
Steroids • Are involved in cell membrane structure • Include sex hormones and hormones regulating metabolism • Are important in lipid digestion

Steroids

Proteins perform many vital functions in the body. The six important types are: • Structural proteins • Contractile proteins • Transport proteins • Enzymes • Buffering proteins • Antibodies

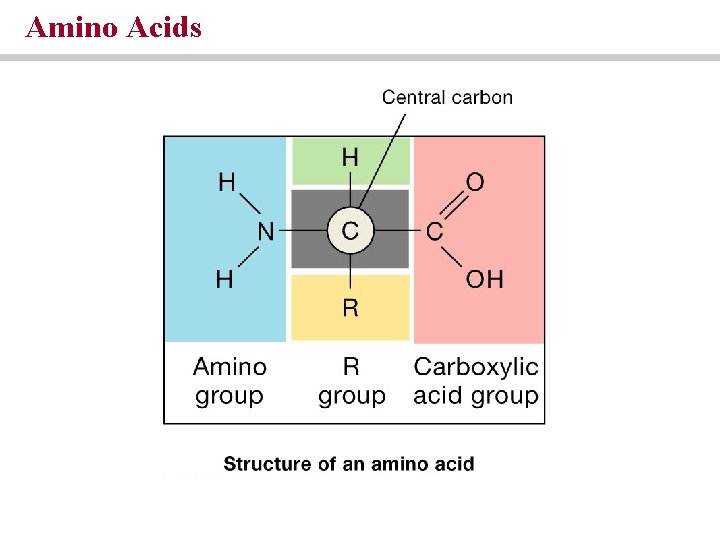
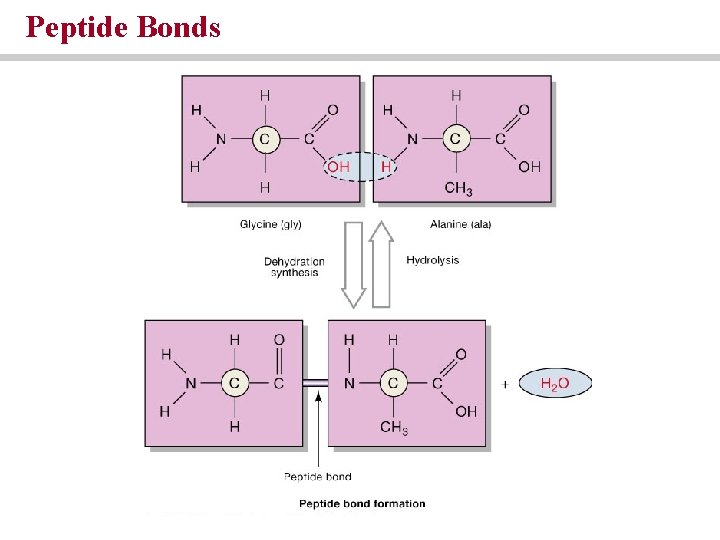
Proteins are chains of amino acids • Amino acids contain an amino group, a carboxylic group and a radical group • Polypeptides are linear sequences of amino acids held together by peptide bonds

Amino Acids

Peptide Bonds

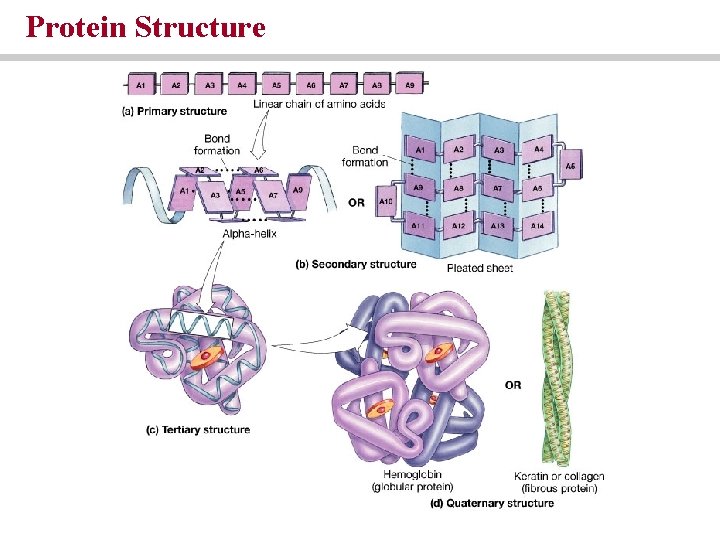
The four levels of protein structure are: • Primary structure (amino acids sequence) • Secondary structure (amino acid interactions) • Tertiary structure (complex folding) • Quaternary structure (protein complexes)

Protein Structure

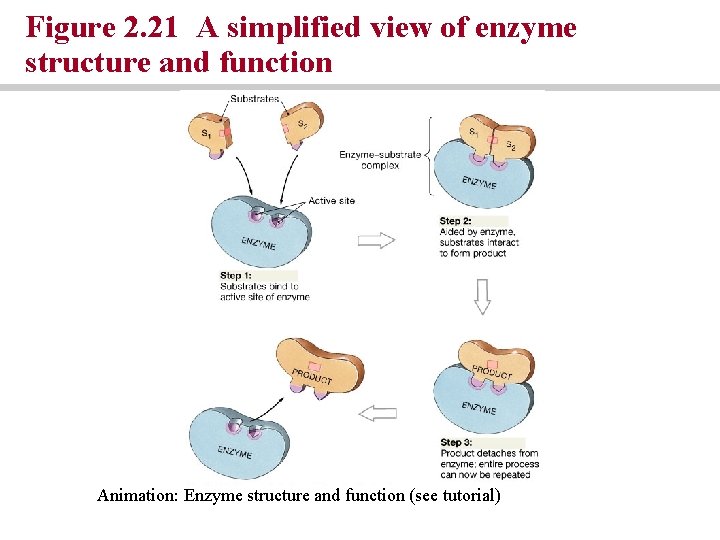
Enzyme reactions • Reactants (substrate) interact to yield a product by binding to the active site of the enzyme • Cofactors must bond to the enzyme before substrate binding can occur • Coenzymes are organic cofactors commonly derived from vitamins

Figure 2. 21 A simplified view of enzyme structure and function Animation: Enzyme structure and function (see tutorial)

The shape of a protein determines its function • Proteins pushed outside their optimal temperature and p. H range become temporarily or permanently denatured and will cease to function

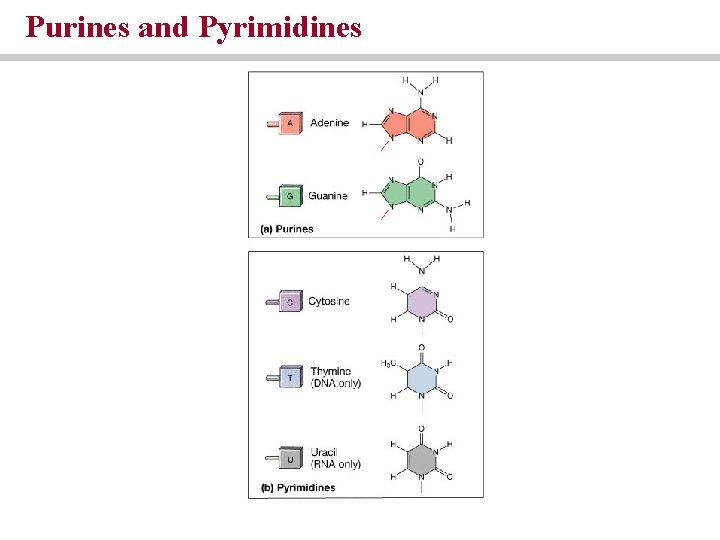
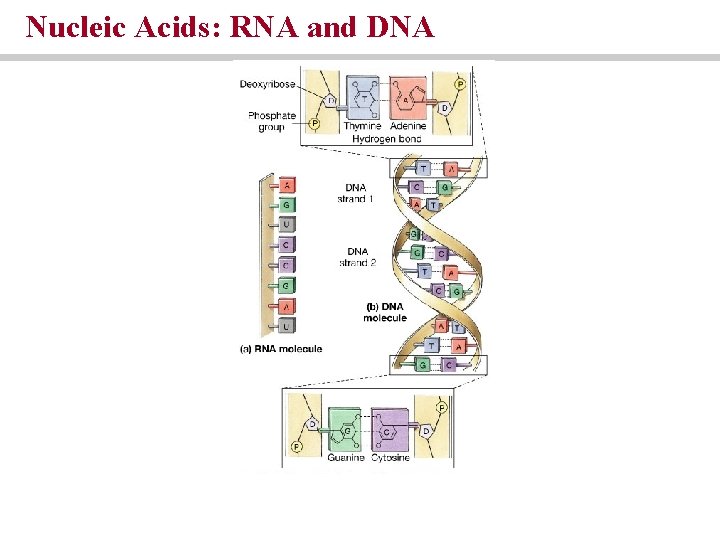
Nucleic acids • Store and process information at the molecular level • Made of purines and pyrimidines • DNA and RNA

Purines and Pyrimidines

Nucleic Acids: RNA and DNA

Nucleic acids are chains of nucleotides • Nucleotides are composed of a sugar, a phosphate and a nitrogenous base • Sugar = deoxyribose (DNA) or ribose (RNA) • DNA Bases = adenine, thymine, cytosine, guanine • RNA bases = adenine, uracil, cytosine, guanine

High energy compounds store cellular energy in high energy bonds • Adenosine triphosphate (ATP) • Made by adding a phosphate group to adenosine diphosphate (ADP) • Process referred to as phosphorylation

Chemicals and Cells Biochemical compounds form functional units called cells • Metabolic turnover allows cells to change and to adapt to changes in their environment

You should now be familiar with: • Atoms and how they combine to form compounds. • Chemical reactions and enzymes. • Organic and inorganic compounds. • Water, p. H, and buffers. • The structure and function of carbohydrates, lipids, proteins, nucleic acids and high energy compounds.