Analyzing Differences in Protein Sequences Between Subjects with

Analyzing Differences in Protein Sequences Between Subjects with Varying T Cell Counts J’aime Moehlman Amanda Wavrin Loyola Marymount University March 22, 2010

Outline • • • Introduction Results Discussion Further Research References

Introduction to the HIV-1 Structure • The site on gp 120 that binds to the CD 4 receptor is vulnerable to neutralizing antibodies. • However, most antibodies that interact with the site cannot neutralize HIV-1. • There are many features that help the gp 120 protein escape the immune system of the host such as: – Variable Loops – N- Linked Glycosylation – Confomational Flexibility • The functions of the gp 120 protein are influenced by the structure within the V 3 region.

• Our Proposed Question: Will there be specific differences between the protein sequences in subject 10 and 12 that results in different protein structures, which changes the function of the virus? • Hypothesis: There will be specific amino acids that cause differences in the protein structures between subjects 10 and 12.
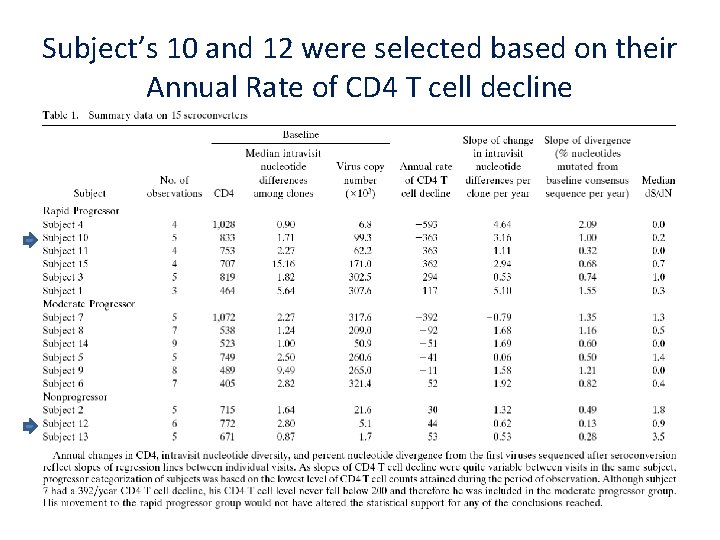
Subject’s 10 and 12 were selected based on their Annual Rate of CD 4 T cell decline
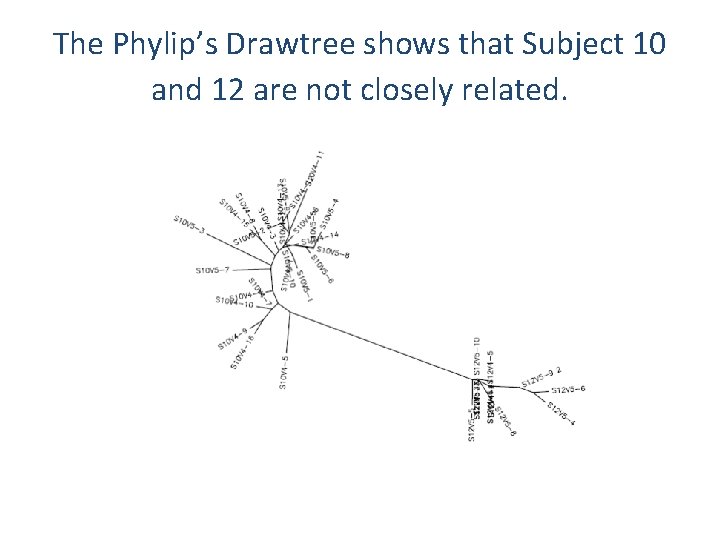
The Phylip’s Drawtree shows that Subject 10 and 12 are not closely related.
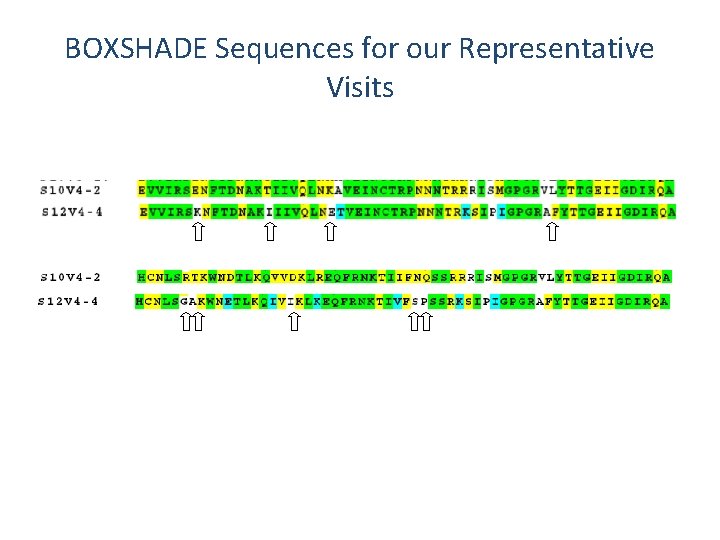
BOXSHADE Sequences for our Representative Visits

Predicting the Secondary Structure of Subject 10’s Protein Sequence Using PSIPRED H: Helical E: Extended C: Random Coil Conf: 947814567777306999858867895368998735651458871002107441055763 Pred: CEEEECCCCEEEEEECCCCCCCCCCCEEEECCCEECHHHHH AA: EVVIRSENFTDNAKTIIVQLNKAVEINCTRPNNNTRRRISMGPGRVLYTTGEIIGDIRQA 10 20 30 40 50 60 Conf: 2054787776579999976189447760589 Pred: HHCCCHHHHHHHHHHCCCEEEEECCC AA: HCNLSRTKWNDTLKQVVDKLREQFRNKTIIFNQSS 70 80 90
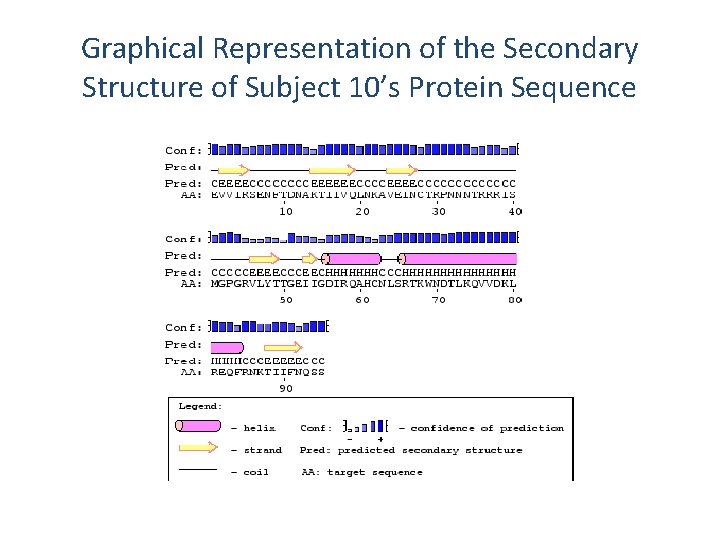
Graphical Representation of the Secondary Structure of Subject 10’s Protein Sequence

Predicting the Secondary Structure of Subject 12’s Protein Sequence Using PSIPRED H: Helical E: Extended C: Random Coil Conf: 94782456777730699985885789636899874565045887100210733105676 Pred: CEEEECCCCEEEEEECCCCCCCCCCCHHEECCCEECHHHH AA: EVVIRSKNFTDNAKIIIVQLNETVEINCTRPNNNTRKSIPIGPGRAFYTTGEIIGDIRQA 10 20 30 40 50 60 Conf: 1055787876579999976189447750689 Pred: HHCCCHHHHHHHHHHCCCEEEECCCC AA: HCNLSGAKWNETLKQIVIKLKEQFRNKTIVFSPSS 70 80 90
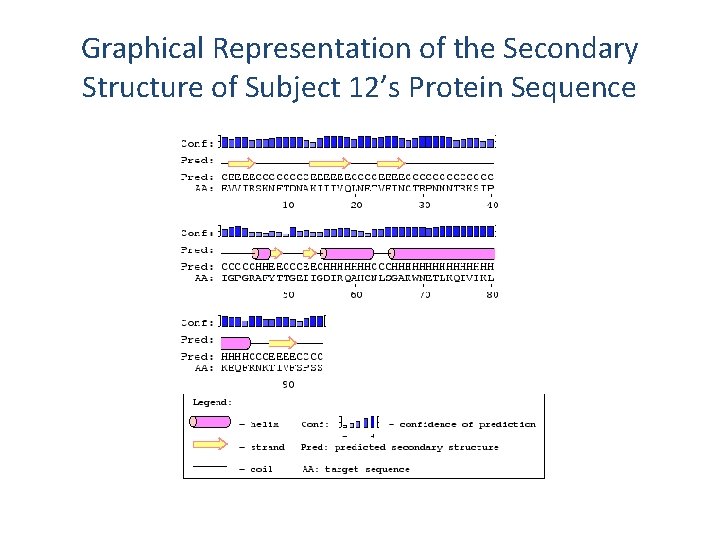
Graphical Representation of the Secondary Structure of Subject 12’s Protein Sequence
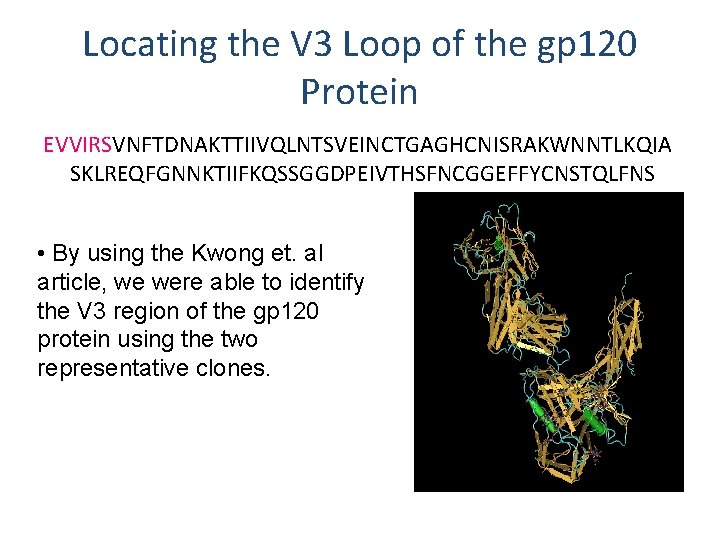
Locating the V 3 Loop of the gp 120 Protein EVVIRSVNFTDNAKTTIIVQLNTSVEINCTGAGHCNISRAKWNNTLKQIA SKLREQFGNNKTIIFKQSSGGDPEIVTHSFNCGGEFFYCNSTQLFNS • By using the Kwong et. al article, we were able to identify the V 3 region of the gp 120 protein using the two representative clones.

The V 3 Loop of the gp 120 Protein
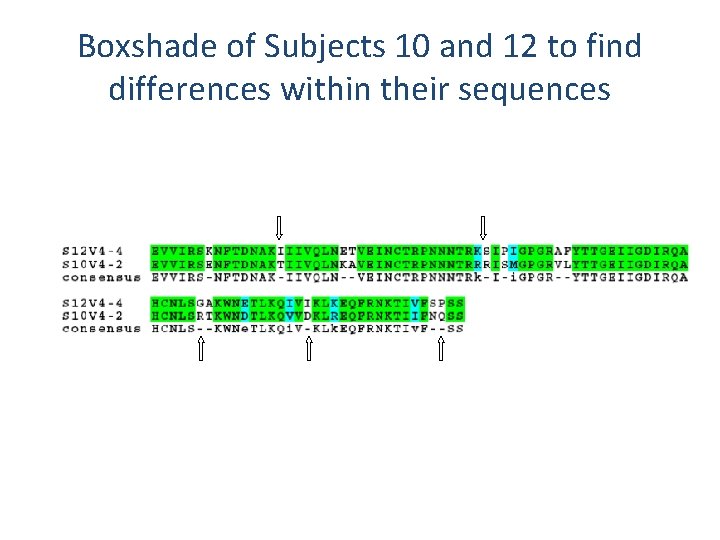
Boxshade of Subjects 10 and 12 to find differences within their sequences

Compared Our Secondary Structure’s with the Kwong et al. 3 -D Structure • In the Kwong Structure: – At location 15 there is a threonine that resulted in an extended sheet. – At location 38 there is an arginine that resulted in a helical structure. – At location 66 there is a glutamine that resulted in an extended sheet. – At location 78 there is a serine that resulted in an extended sheet. – At location 93 there is a leucine that resulted in a random coil.

Comparison Continued with Subject 10 • In our secondary structures we found: – Subject 10: • At amino acid 15, the predicted structure, an extended sheet, was found • At amino acid 38, the predicted structure was a random coil, which is not consistent with the actual structure. • At amino acid 66, the predicted structure was helical, which is not consistent with the actual structure. • At amino acid 78, the predicted structure was helical, which is not consistent with the actual structure. • At amino acid 93, the predicted structure, a random coil, was found.

Comparison Continued with Subject 12 • In our secondary structures we found: – Subject 12: • At amino acid 15, the predicted structure, an extended sheet, was found. • At amino acid 38, the predicted structure was a random coil, which is not consistent with the actual structure. • At amino acid 66, the predicted structure was helical, which is not consistent with the actual structure. • At amino acid 78, the predicted structure was helical, which is not consistent with the actual structure. • At amino acid 93, the predicted structure, a random coil, was found.
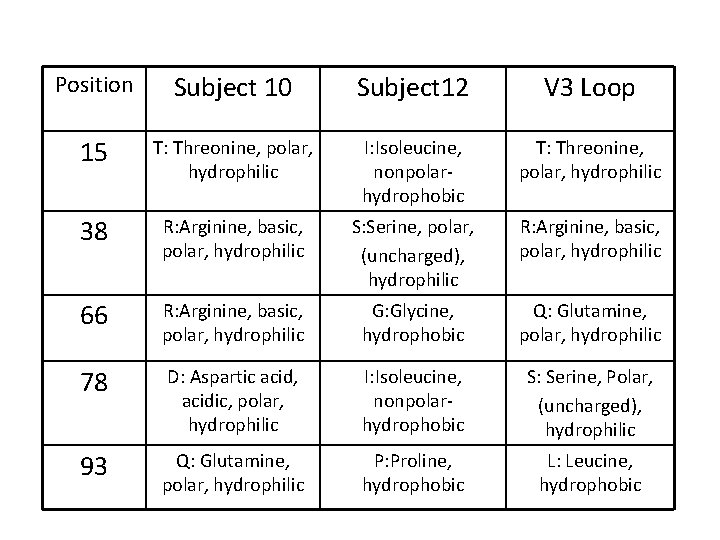
Position Subject 10 Subject 12 V 3 Loop 15 T: Threonine, polar, hydrophilic I: Isoleucine, nonpolarhydrophobic T: Threonine, polar, hydrophilic 38 R: Arginine, basic, polar, hydrophilic S: Serine, polar, (uncharged), hydrophilic R: Arginine, basic, polar, hydrophilic 66 R: Arginine, basic, polar, hydrophilic G: Glycine, hydrophobic Q: Glutamine, polar, hydrophilic 78 D: Aspartic acid, acidic, polar, hydrophilic I: Isoleucine, nonpolarhydrophobic S: Serine, Polar, (uncharged), hydrophilic 93 Q: Glutamine, polar, hydrophilic P: Proline, hydrophobic L: Leucine, hydrophobic
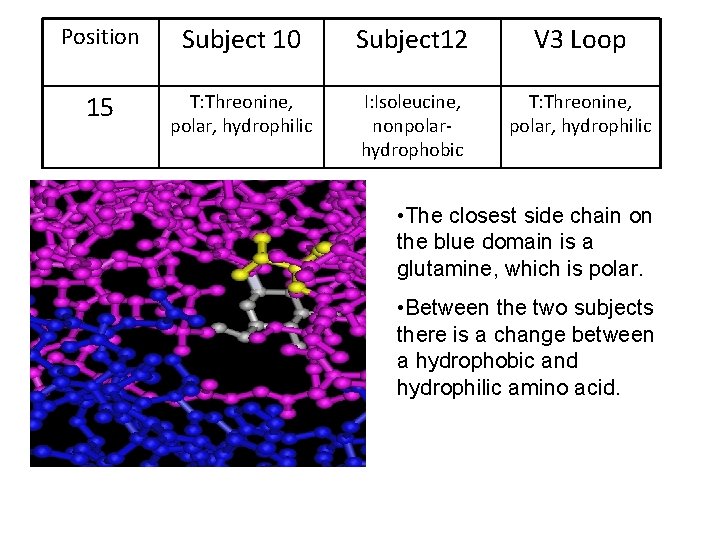
Position Subject 10 Subject 12 V 3 Loop 15 T: Threonine, polar, hydrophilic I: Isoleucine, nonpolarhydrophobic T: Threonine, polar, hydrophilic • The closest side chain on the blue domain is a glutamine, which is polar. • Between the two subjects there is a change between a hydrophobic and hydrophilic amino acid.

Position Subject 10 Subject 12 V 3 Loop 38 R: Arginine, basic, polar, hydrophilic S: Serine, polar, hydrophilic R: Arginine, basic, polar, hydrophilic • Arginine is greater in size than Serine. • They are all hydrophilic amino acids.

Position Subject 10 Subject 12 V 3 Loop 66 R: Arginine, basic, polar, hydrophilic G: Glycine, hydrophobic Q: Glutamine, polar, hydrophilic • In subjects 10 and 12; the amino acid changes from hydrophilic to hydrophobic causing a potential structural difference. • Glutamine is larger in size than Glycine, but is closer in size to Arginine.
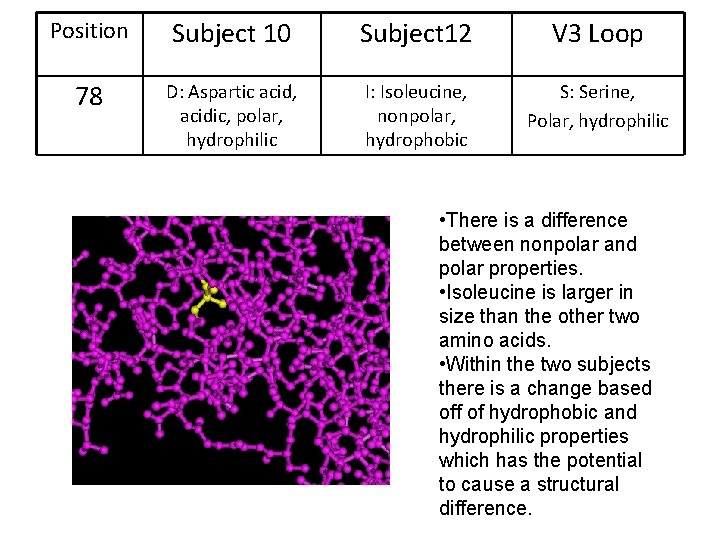
Position Subject 10 Subject 12 V 3 Loop 78 D: Aspartic acid, acidic, polar, hydrophilic I: Isoleucine, nonpolar, hydrophobic S: Serine, Polar, hydrophilic • There is a difference between nonpolar and polar properties. • Isoleucine is larger in size than the other two amino acids. • Within the two subjects there is a change based off of hydrophobic and hydrophilic properties which has the potential to cause a structural difference.
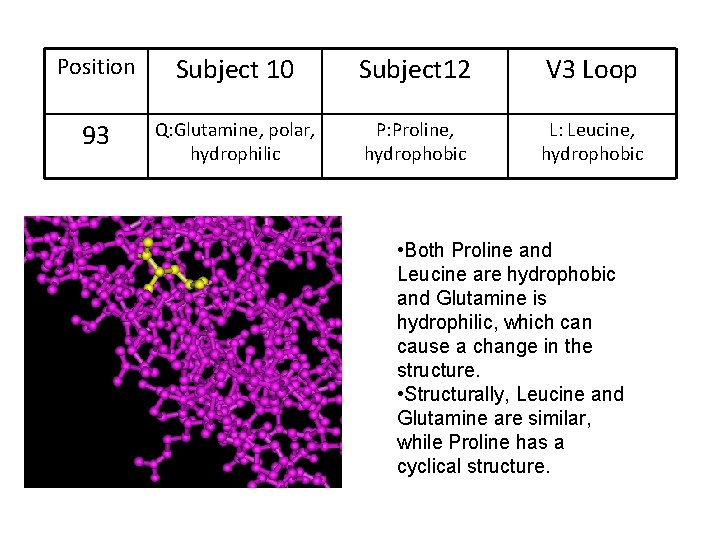
Position Subject 10 Subject 12 V 3 Loop 93 Q: Glutamine, polar, hydrophilic P: Proline, hydrophobic L: Leucine, hydrophobic • Both Proline and Leucine are hydrophobic and Glutamine is hydrophilic, which can cause a change in the structure. • Structurally, Leucine and Glutamine are similar, while Proline has a cyclical structure.

There will be specific amino acids that cause differences in the protein structures of subjects 10 and 12 • Based off of our results, we accept our hypothesis for the tertiary structures of the proteins. • There were specific amino acid changes between the subjects, but they did not result in predicted secondary structural changes. • There were differences in the amino acids between our secondary structure and that of the actual V 3 structure (from Kwong et al). • This resulted in structural differences between them.

Opportunities for Further Research • Analyze an individual subjects amino acid sequence and protein structure to potentially create a neutralizing antibody for the HIV-1 virus. • There are many features that help the gp 120 protein escape the immune system of the host such as: – Variable Loops – N- Linked Glycosylation – Confomational Flexibility

References • Markham RB, Wang WC, Weisstein AE, Wang Z, Munoz A, Faradegan H, and Yu XF. Patterns of HIV-1 evolution in individuals with differing rates of CD 4 T cell decline. Proc Natl Acad Sci U S A 1998 Oct 13; 95(21) 12568 - 73. • Kwong PD, Wyatt R, Robinson J, Sweet RW, Sodroski J, and Hedrickson WA. Structure of an HIV gp 120 envelope glycoprotein in complex with the CD 4 receptor and a neutralizing human antibody. Nature 1998 Jun 18; 393(6686) 648 -59. • Chen L, Kwon YD, Zhou T, Wu X, O’Dell S, Cavacini L, Hessell AJ, Pancera M, Tang M, Xu L, Yang ZY, Zhang MY, Arthos J, Burton DR, Dimitrov DS, Nabel GJ, Posner MR, Sodroski J, Wyatt R, Mascola JR, Kwong PD. Structural basis of immune evasion at the site of CD 4 attachment on HIV-1 gp 120. Science. 2009 Nov 20; 326(5956): 11237.
- Slides: 26