Analytical comparability of a human Ig G 1

Analytical comparability of a human Ig. G 1 from different manufacturing sites after cell line switch and process changes: A case study Dr. Margit Jeschke Head Analytical R&D Novartis Biologics www. diahome. org

Disclaimer The views and opinions expressed in the following Power. Point slides are those of the individual presenter and should not be attributed to Drug Information Association, Inc. (“DIA”), its directors, officers, employees, volunteers, members, chapters, councils, Special Interest Area Communities or affiliates, or any organization with which the presenter is employed or affiliated. These Power. Point slides are the intellectual property of the individual presenter and are protected under the copyright laws of the United States of America and other countries. Used by permission. All rights reserved. Drug Information Association, DIA and DIA logo are registered trademarks or trademarks of Drug Information Association Inc. All other trademarks are the property of their respective owners. www. diahome. org

Outline § Introduction § Molecular Challenges with m. Ab 1 NVS § Results • Purification Process Performance • Physicochemical tests • Biological characterization • Accelerated Stabilty Study • Preclinical & PK/PD § Summary § Conclusions & Discussion § Acknowledgements 3 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
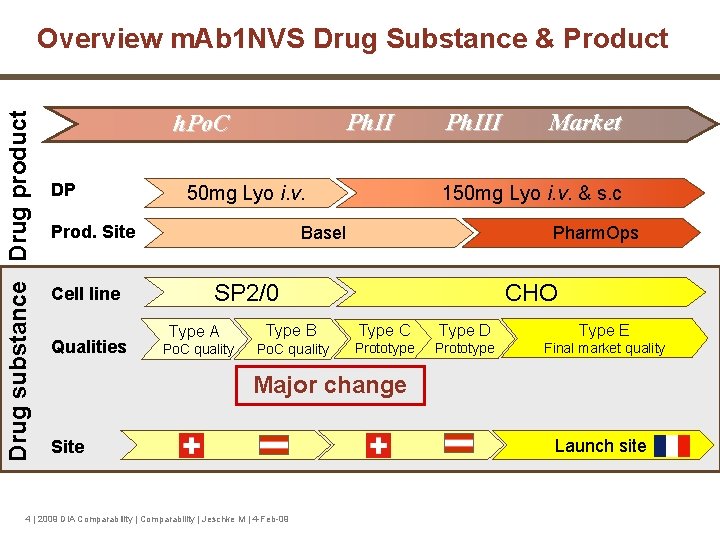
Drug substance Drug product Overview m. Ab 1 NVS Drug Substance & Product Ph. II h. Po. C DP 50 mg Lyo i. v. Prod. Site Cell line Qualities Ph. III Market 150 mg Lyo i. v. & s. c Basel Pharm. Ops SP 2/0 CHO Type A Type B Type C Type D Type E Po. C quality Prototype Final market quality Major change Site 4 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 Launch site

m. Ab 1 NVS Manufacturing Changes § Changes in drug substance manufacturing process for phase I/II clinical development: • production cell line (Sp 2/0 to CHO) • manufacturing process • manufacturing site • scale-up 5 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
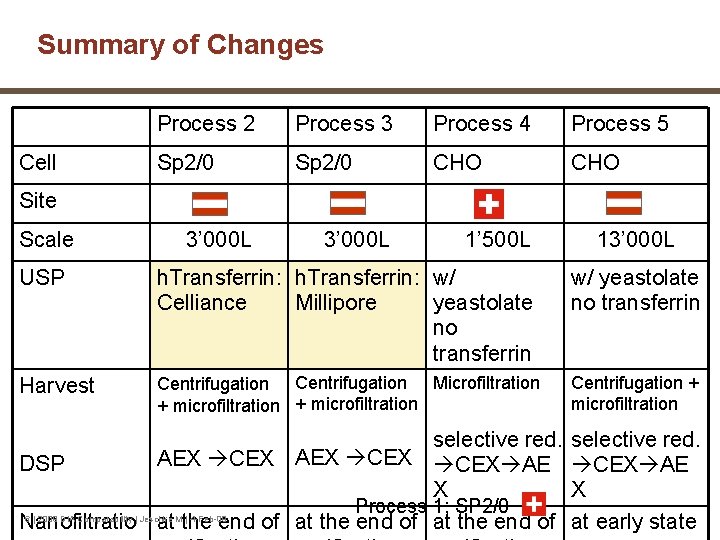
Summary of Changes Cell Process 2 Process 3 Process 4 Process 5 Sp 2/0 CHO Site Scale 3’ 000 L 1’ 500 L 13’ 000 L USP h. Transferrin: w/ Celliance Millipore yeastolate no transferrin w/ yeastolate no transferrin Harvest Centrifugation Microfiltration + microfiltration Centrifugation + microfiltration selective red. AEX CEX AE DSP X X Process 1: SP 2/0 6 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 Nanofiltratio at the end of at early state
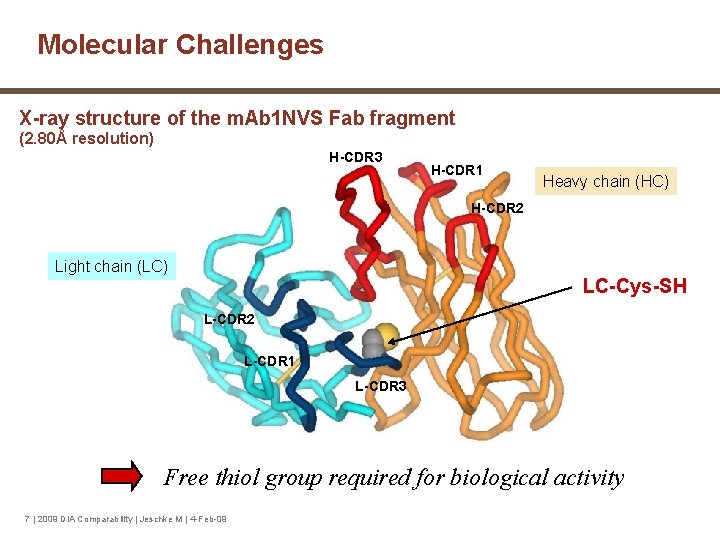
Molecular Challenges X-ray structure of the m. Ab 1 NVS Fab fragment (2. 80Å resolution) H-CDR 3 H-CDR 1 Heavy chain (HC) H-CDR 2 Light chain (LC) LC-Cys-SH L-CDR 2 L-CDR 1 L-CDR 3 Free thiol group required for biological activity 7 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
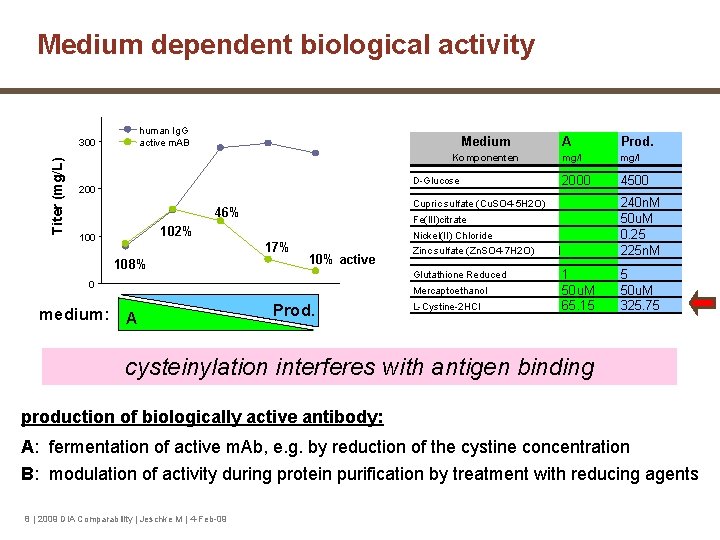
Medium dependent biological activity human Ig. G active m. AB Titer (mg/L) 300 Medium Komponenten D-Glucose 200 17% 108% 10% active medium: A 2055 2000 4500 240 n. M 50 u. M 0. 25 225 n. M Zinc sulfate (Zn. SO 4 -7 H 2 O) Glutathione Reduced 3 mg/l Nickel(II) Chloride 0 2 mg/l Fe(III)citrate 102% 1 Prod. Cupric sulfate (Cu. SO 4 -5 H 2 O) 46% 100 A 4 5 2071 Prod. Mercaptoethanol L-Cystine-2 HCl 1 50 u. M 65. 15 5 50 u. M 325. 75 cysteinylation interferes with antigen binding production of biologically active antibody: A: fermentation of active m. Ab, e. g. by reduction of the cystine concentration B: modulation of activity during protein purification by treatment with reducing agents 8 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
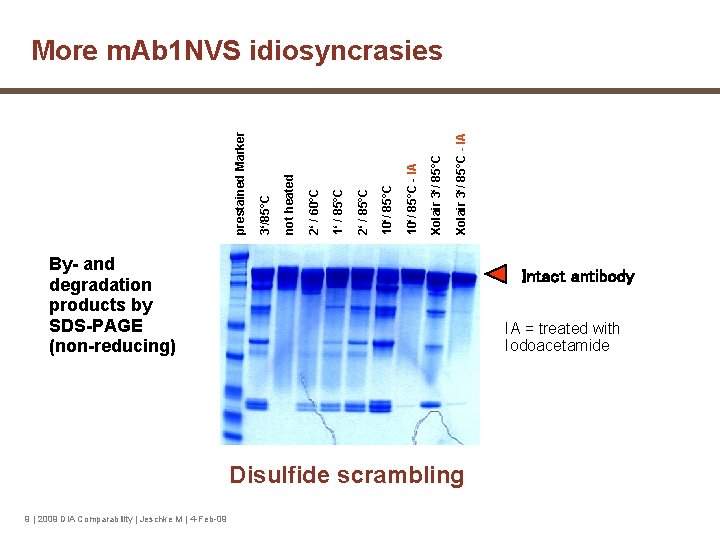
Xolair 3‘/ 85°C - IA Xolair 3‘/ 85°C 10‘/ 85°C - IA 10‘/ 85°C 2‘ / 85°C 1‘ / 85°C 2‘ / 60°C not heated 3‘/85°C prestained Marker More m. Ab 1 NVS idiosyncrasies By- and degradation products by SDS-PAGE (non-reducing) Intact antibody IA = treated with Iodoacetamide Disulfide scrambling 9 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
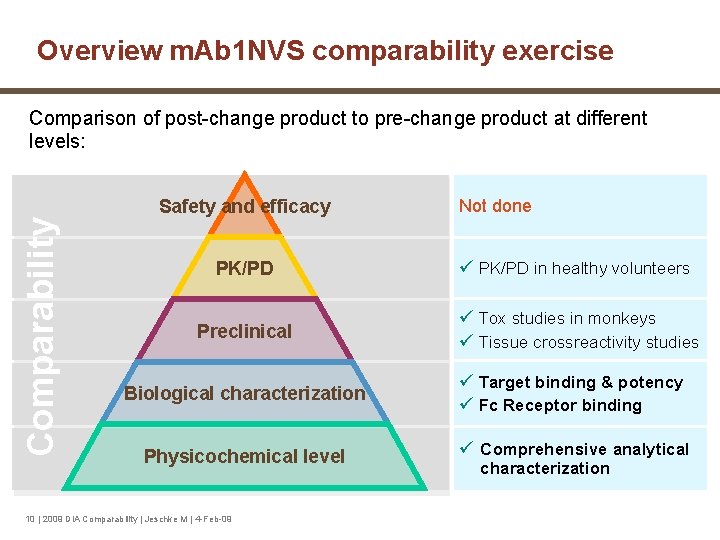
Overview m. Ab 1 NVS comparability exercise Comparability Comparison of post-change product to pre-change product at different levels: Safety and efficacy PK/PD Preclinical Not done ü PK/PD in healthy volunteers ü Tox studies in monkeys ü Tissue crossreactivity studies Biological characterization ü Target binding & potency ü Fc Receptor binding Physicochemical level ü Comprehensive analytical 10 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 characterization
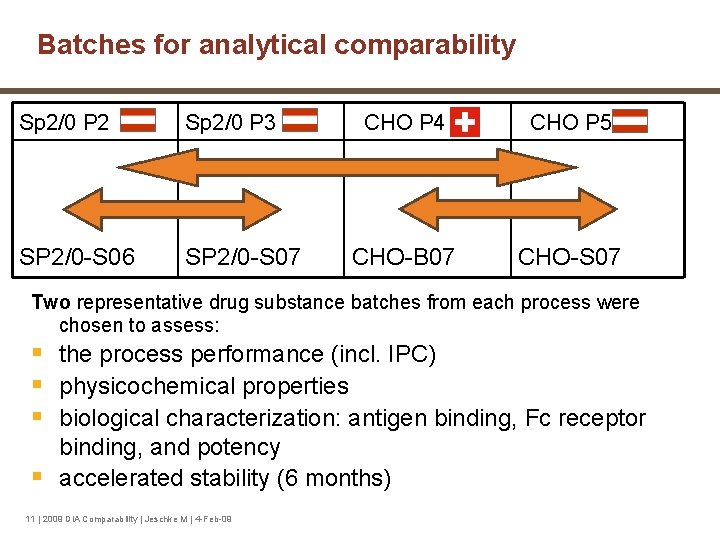
Batches for analytical comparability Sp 2/0 P 2 Sp 2/0 P 3 CHO P 4 CHO P 5 SP 2/0 -S 06 SP 2/0 -S 07 CHO-B 07 CHO-S 07 Two representative drug substance batches from each process were chosen to assess: § the process performance (incl. IPC) § physicochemical properties § biological characterization: antigen binding, Fc receptor binding, and potency § accelerated stability (6 months) 11 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09

Outline § Introduction § Molecular Challenges with m. Ab 1 NVS § Results • Purification Process Performance • Physicochemical tests • Biological characterization • Accelerated Stabilty Study • Preclinical & PK/PD § Summary § Conclusions & Discussion § Acknowledgements 12 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09

Purification Process Performance § Process performance - Prot. A, HCP, DNA and yields: • The single step efficiency is different • Overall removal successful for all process related impurities • Single step yields are different 13 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
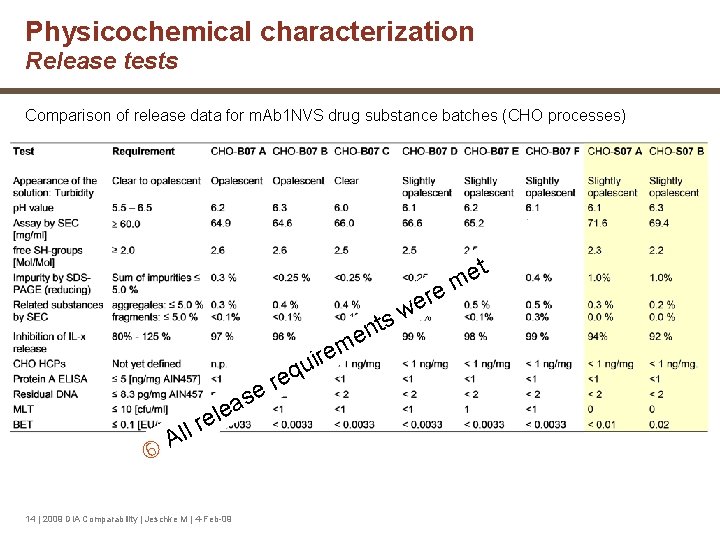
Physicochemical characterization Release tests Comparison of release data for m. Ab 1 NVS drug substance batches (CHO processes) t e m e r e w s t n e m ire u q e r se a e l re l l A 14 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
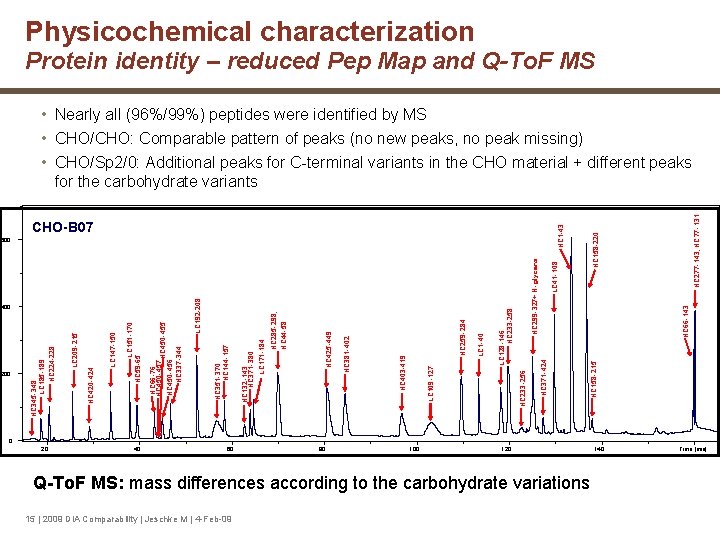
Physicochemical characterization Protein identity – reduced Pep Map and Q-To. F MS • Nearly all (96%/99%) peptides were identified by MS • CHO/CHO: Comparable pattern of peaks (no new peaks, no peak missing) • CHO/Sp 2/0: Additional peaks for C-terminal variants in the CHO material + different peaks for the carbohydrate variants HC 158 -215 HC 66 -143 LC 41 -108 HC 299 -327+ N -glycans HC 233 -256 HC 371 -424 LC 128 -146 HC 233 -258 LC 1 -40 HC 259 -284 LC 109 -127 HC 403 -419 HC 381 -402 HC 425 -449 HC 285 -298, HC 44 -58 LC 171 -184 HC 132 -143 HC 371 -380 HC 351 -370 HC 144 -157 LC 192 -208 HC 450 -455 HC 450 -456 HC 337 -344 HC 66 -76 HC 450 -457 LC 151 -170 HC 59 -65 LC 147 -150 LC 209 -215 HC 420 -424 200 HC 345 -348 LC 185 -189 HC 224 -228 400 HC 158 -220 HC 1 -43 CHO-B 07 600 HC 277 -143, HC 77 -131 ens. 0 20 40 60 80 100 120 140 Q-To. F MS: mass differences according to the carbohydrate variations 15 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 Time [min]
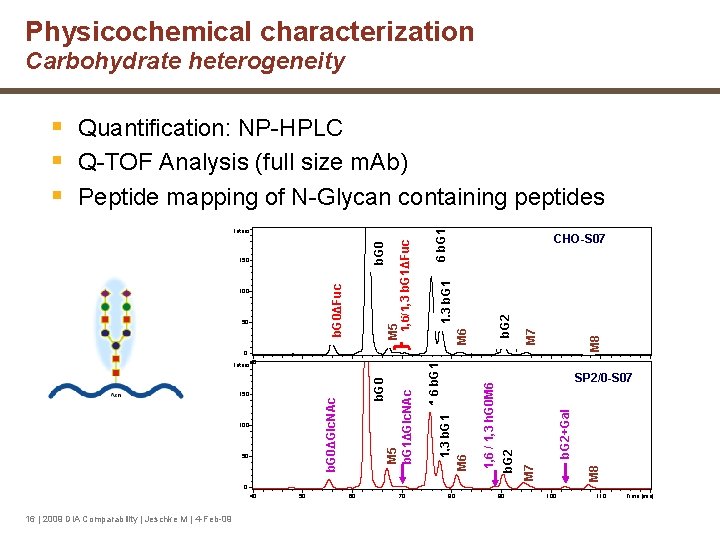
Physicochemical characterization Carbohydrate heterogeneity M 8 b. G 2 M 7 b. G 2+Gal M 8 M 7 50 SP 2/0 -S 07 b. G 2 100 1, 6 / 1, 3 h. G 0 M 6 b. G 0ΔGlc. NAc 150 b. G 0 M 5 b. G 1ΔGlc. NAc 0 Intens. 40 M 6 50 1, 6 b. G 1 100 CHO-S 07 1, 3 b. G 1 M 6 b. G 0ΔFuc 150 M 5 1, 6/1, 3 b. G 1ΔFuc b. G 0 Intens. 1, 3 b. G 1 1, 6 b. G 1 § Quantification: NP-HPLC § Q-TOF Analysis (full size m. Ab) § Peptide mapping of N-Glycan containing peptides 0 40 16 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 50 60 70 80 90 100 110 Time [min]
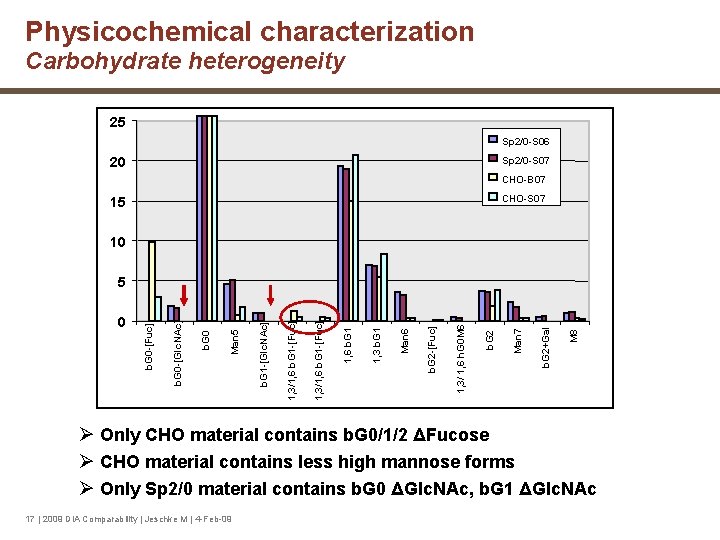
Physicochemical characterization Carbohydrate heterogeneity 25 Sp 2/0 -S 06 20 Sp 2/0 -S 07 CHO-B 07 CHO-S 07 15 10 M 8 b. G 2+Gal Man 7 b. G 2 1, 3/ 1, 6 h. G 0 M 6 b. G 2 -[Fuc] Man 6 1, 3 b. G 1 1, 6 b. G 1 1, 3/1, 6 b. G 1 -[Fuc] b. G 1 -[Glc. NAc] Man 5 b. G 0 -[Glc. NAc] 0 b. G 0 -[Fuc] 5 Ø Only CHO material contains b. G 0/1/2 ΔFucose Ø CHO material contains less high mannose forms Ø Only Sp 2/0 material contains b. G 0 ΔGlc. NAc, b. G 1 ΔGlc. NAc 17 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
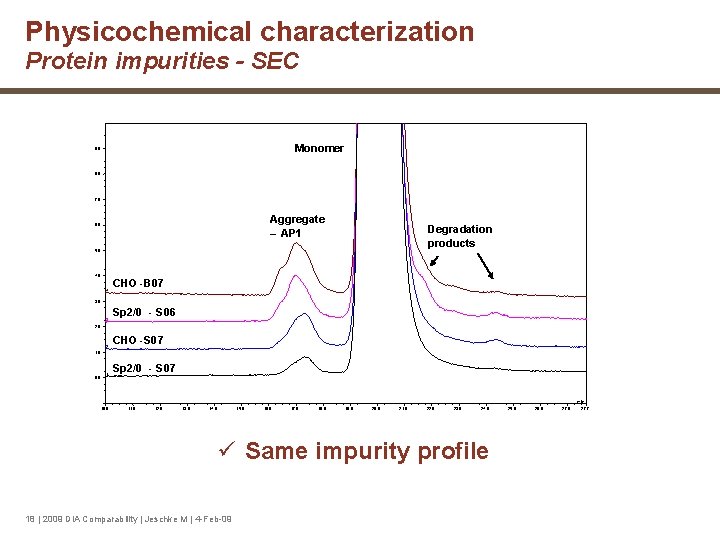
Physicochemical characterization Protein impurities - SEC Monomer 9. 0 8. 0 7. 0 Aggregate – AP 1 6. 0 Degradation products 5. 0 4 CHO --B 07 3. 0 3 Sp 2/0 - S 06 2. 0 2 CHO --S 07 1. 0 0. 0 1 Sp 2/0 - S 07 min 10. 0 11. 0 12. 0 13. 0 14. 0 15. 0 16. 0 17. 0 18. 0 19. 0 20. 0 21. 0 22. 0 23. 0 24. 0 ü Same impurity profile 18 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 25. 0 26. 0 27. 7
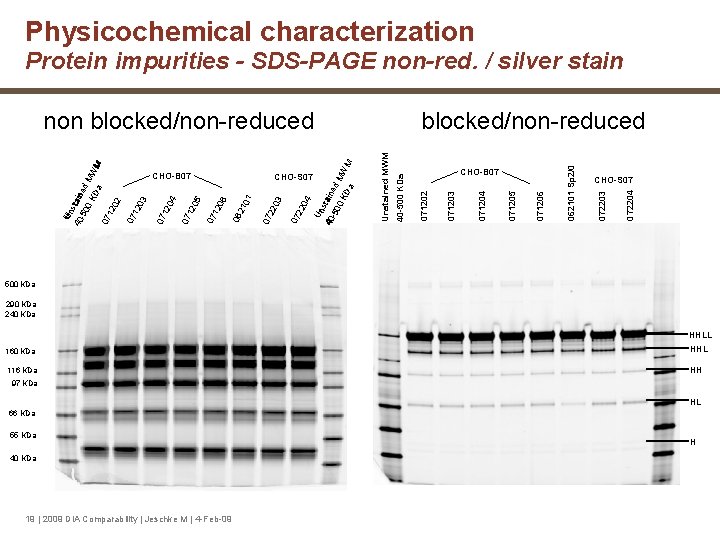
Physicochemical characterization Protein impurities - SDS-PAGE non-red. / silver stain 072204 CHOCHO-S 07 072203 062101 Sp 2/0 071206 071205 071204 071203 CHOCHO-B 07 071202 Unstained MWM blocked/non-reduced 40 -500 KDa 04 Un s 40 - taine 50 d 0 K MW Da M 07 22 CHOCHO-S 07 07 22 03 CHOCHO-B 07 07 12 04 07 12 05 07 12 06 06 21 01 Un s 40 tain -50 e 0 K d MW Da M 07 12 02 07 12 03 non blocked/non-reduced 500 KDa 290 KDa 240 KDa HHLL 160 KDa HHL 116 KDa HH 97 KDa HL 66 KDa 55 KDa 40 KDa 19 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 H
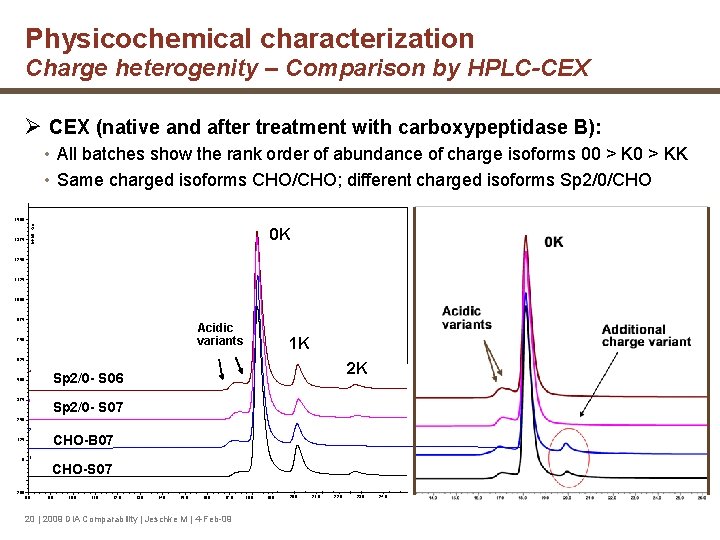
Physicochemical characterization Charge heterogenity – Comparison by HPLC-CEX Ø CEX (native and after treatment with carboxypeptidase B): • All batches show the rank order of abundance of charge isoforms 00 > KK • Same charged isoforms CHO/CHO; different charged isoforms Sp 2/0/CHO 1, 375 Inhbit = Off 1, 500 0 K 1, 250 1, 125 1, 000 875 Acidic variants 750 1 K 625 4 Sp 2/0 - S 06 3 Sp 2/0 - S 07 500 375 2 K 250 2 CHO-B 07 125 0 1 CHO-S 07 -200 8. 0 9. 0 10. 0 11. 0 12. 0 13. 0 14. 0 15. 0 16. 0 17. 0 20 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 18. 0 19. 0 20. 0 21. 0 22. 0 23. 0 24. 0
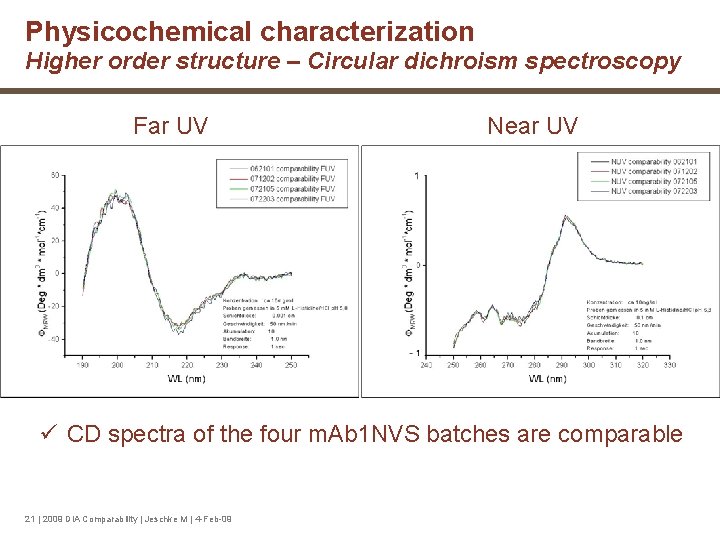
Physicochemical characterization Higher order structure – Circular dichroism spectroscopy Far UV Near UV ü CD spectra of the four m. Ab 1 NVS batches are comparable 21 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
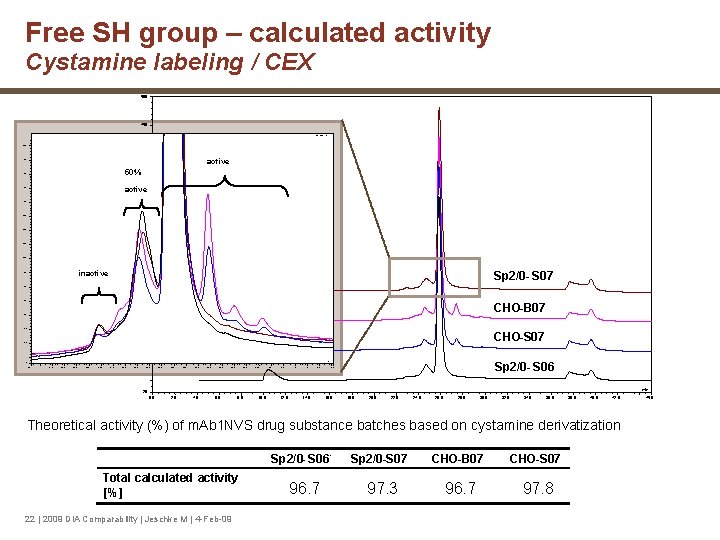
Free SH group – calculated activity Cystamine labeling / CEX 500 450 WVL: 220 nm 40. 0 400 active 37. 5 50% 35. 0 350 active 32. 5 30. 0 300 27. 5 25. 0 250 22. 5 20. 0 200 inactive 17. 5 Sp 2/0 - S 07 15. 0 150 12. 5 CHO-B 07 - 10. 0 100 7. 5 CHO-S 07 - 5. 0 50 4 3 2 1 Sp 2/0 - S 06 min 1. 4 20. 1 21. 0 21. 5 22. 0 22. 5 23. 0 23. 5 24. 0 24. 5 25. 0 25. 5 26. 0 26. 5 27. 0 27. 5 28. 0 28. 5 29. 0 29. 5 30. 0 30. 5 31. 0 31. 5 32. 0 32. 6 min -20 0. 0 2. 0 4. 0 6. 0 8. 0 10. 0 12. 0 14. 0 16. 0 18. 0 20. 0 22. 0 24. 0 26. 0 28. 0 30. 0 32. 0 34. 0 36. 0 38. 0 40. 0 42. 0 Theoretical activity (%) of m. Ab 1 NVS drug substance batches based on cystamine derivatization Total calculated activity [%] 22 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 Sp 2/0 - S 06 96. 7 Sp 2/0 -S 07 CHO-B 07 CHO-S 07 97. 3 96. 7 97. 8 45. 0
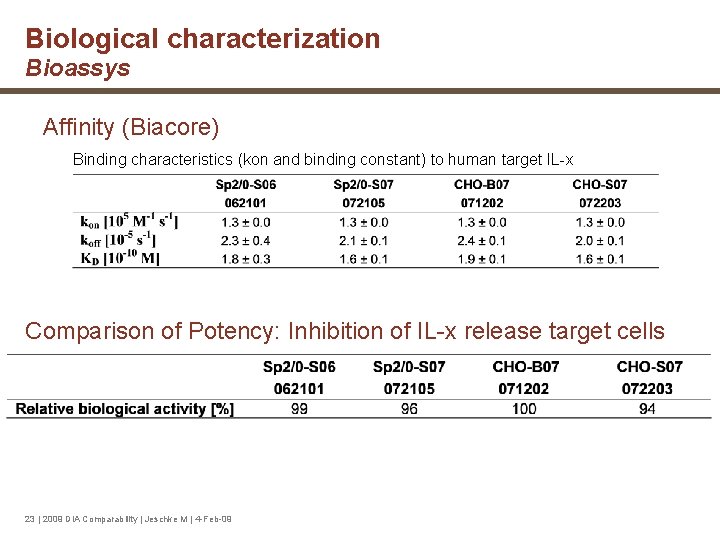
Biological characterization Bioassys Affinity (Biacore) Binding characteristics (kon and binding constant) to human target IL-x Comparison of Potency: Inhibition of IL-x release target cells 23 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
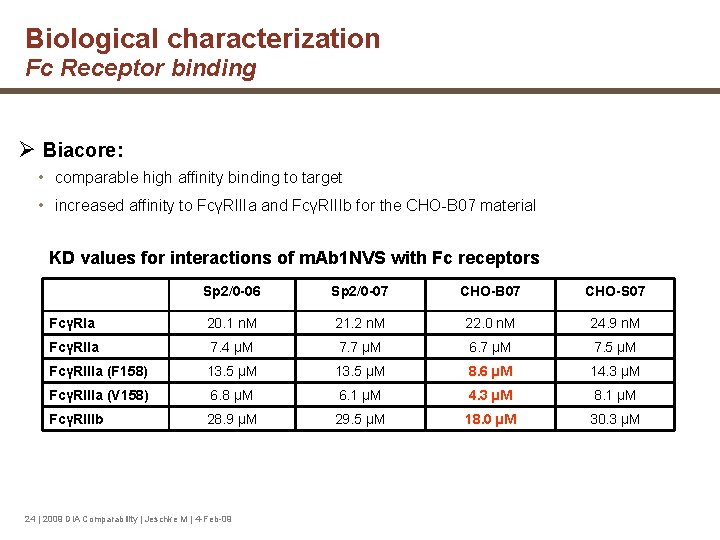
Biological characterization Fc Receptor binding Ø Biacore: • comparable high affinity binding to target • increased affinity to FcγRIIIa and FcγRIIIb for the CHO-B 07 material KD values for interactions of m. Ab 1 NVS with Fc receptors Sp 2/0 -06 Sp 2/0 -07 CHO-B 07 CHO-S 07 FcγRIa 20. 1 n. M 21. 2 n. M 22. 0 n. M 24. 9 n. M FcγRIIa 7. 4 µM 7. 7 µM 6. 7 µM 7. 5 µM FcγRIIIa (F 158) 13. 5 µM 8. 6 µM 14. 3 µM FcγRIIIa (V 158) 6. 8 µM 6. 1 µM 4. 3 µM 8. 1 µM FcγRIIIb 28. 9 µM 29. 5 µM 18. 0 µM 30. 3 µM 24 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
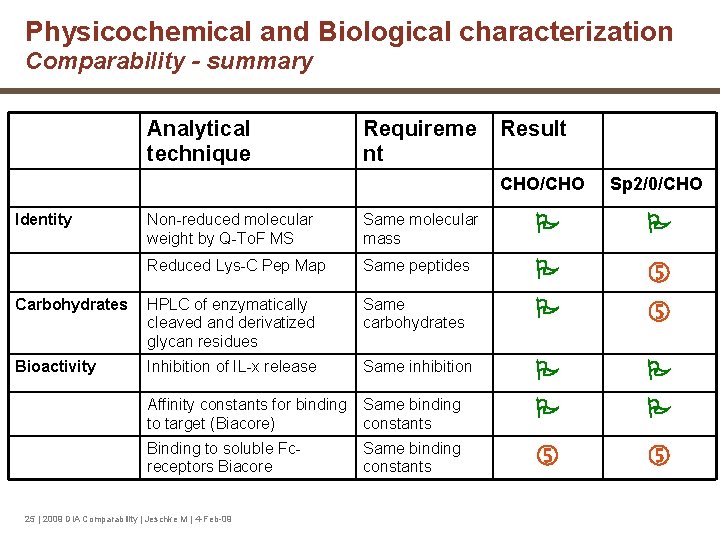
Physicochemical and Biological characterization Comparability - summary Analytical technique Identity Requireme nt Result CHO/CHO Sp 2/0/CHO Non-reduced molecular weight by Q-To. F MS Same molecular mass Reduced Lys-C Pep Map Same peptides Carbohydrates HPLC of enzymatically cleaved and derivatized glycan residues Same carbohydrates Bioactivity Inhibition of IL-x release Same inhibition Affinity constants for binding Same binding to target (Biacore) constants Binding to soluble Fcreceptors Biacore 25 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 Same binding constants
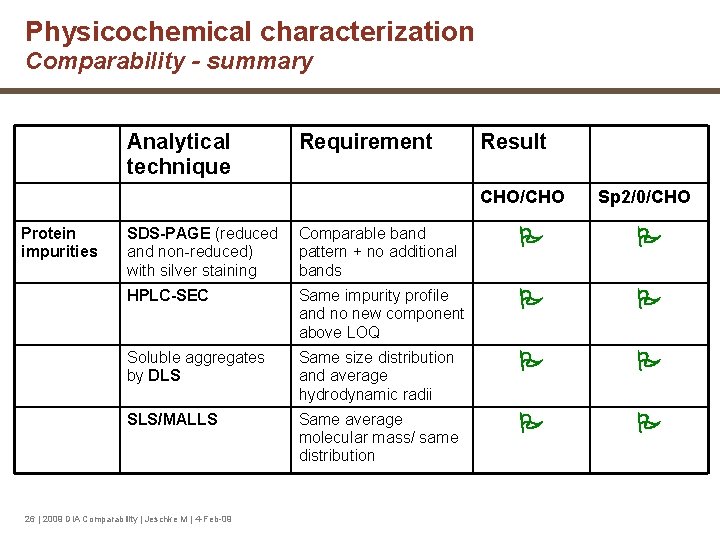
Physicochemical characterization Comparability - summary Analytical technique Requirement Result CHO/CHO Protein impurities Sp 2/0/CHO SDS-PAGE (reduced Comparable band non-reduced) pattern + no additional with silver staining bands HPLC-SEC Same impurity profile and no new component above LOQ Soluble aggregates by DLS Same size distribution and average hydrodynamic radii SLS/MALLS Same average molecular mass/ same distribution 26 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
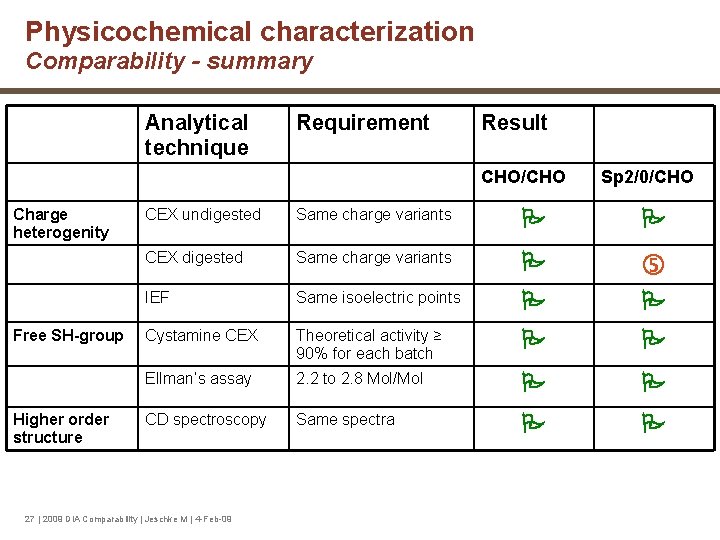
Physicochemical characterization Comparability - summary Analytical technique Requirement Result CHO/CHO Charge heterogenity Free SH-group Higher order structure Sp 2/0/CHO CEX undigested Same charge variants CEX digested Same charge variants IEF Same isoelectric points Cystamine CEX Theoretical activity ≥ 90% for each batch Ellman’s assay 2. 2 to 2. 8 Mol/Mol CD spectroscopy Same spectra 27 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09

Accelerated stability study Program and analytical Tests Short term stability program § Test performed for the head to head stability: • SDS-PAGE • SEC – Assay and Purity • CEX • Free SH groups • Appearance / Color / p. H • Bioassay 28 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
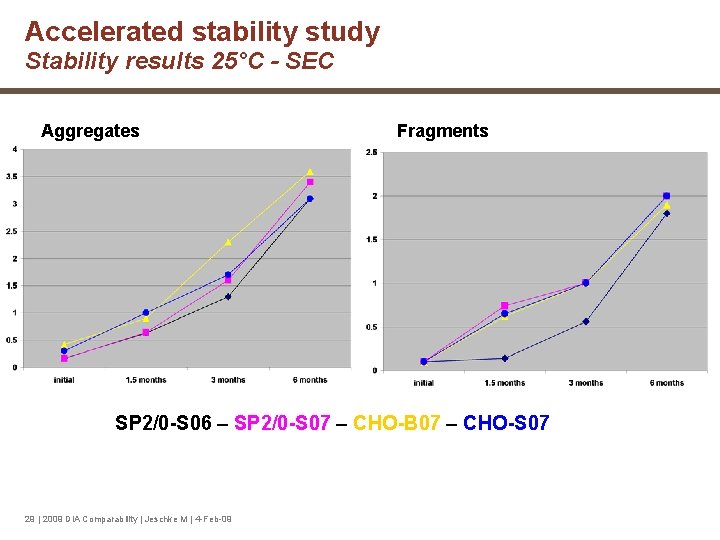
Accelerated stability study Stability results 25°C - SEC Aggregates Fragments SP 2/0 -S 06 – SP 2/0 -S 07 – CHO-B 07 – CHO-S 07 29 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
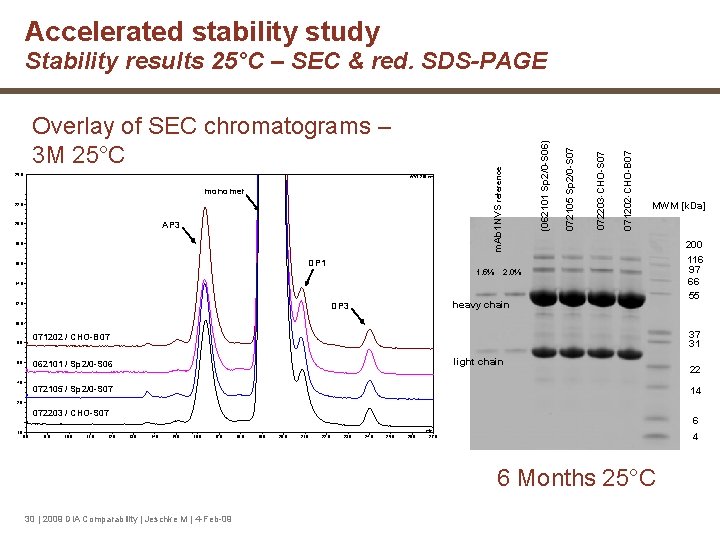
Accelerated stability study AP 3 20. 0 18. 0 DP 1 16. 0 1. 5% 071202 -CHO-B 07 monomer 22. 0 072203 -CHO-S 07 WVL: 210 nm 072105 Sp 2/0 -S 07 25. 0 m. Ab 1 NVS reference Overlay of SEC chromatograms – 3 M 25°C (062101 Sp 2/0 -S 06) Stability results 25°C – SEC & red. SDS-PAGE MWM [k. Da] 2. 0% 14. 0 12. 0 heavy chain DP 3 200 116 97 66 55 10. 0 8. 0 4 6. 0 3 4. 0 37 31 071202 / CHO-B 07 light chain 062101 / Sp 2/0 -S 06 072105 / Sp 2/0 -S 07 22 14 2 2. 0 072203 / CHO-S 07 6 1 min -1. 0 8. 0 9. 0 10. 0 11. 0 12. 0 13. 0 14. 0 15. 0 16. 0 17. 0 18. 0 19. 0 20. 0 21. 0 22. 0 23. 0 24. 0 25. 0 26. 0 4 27. 0 6 Months 25°C 30 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
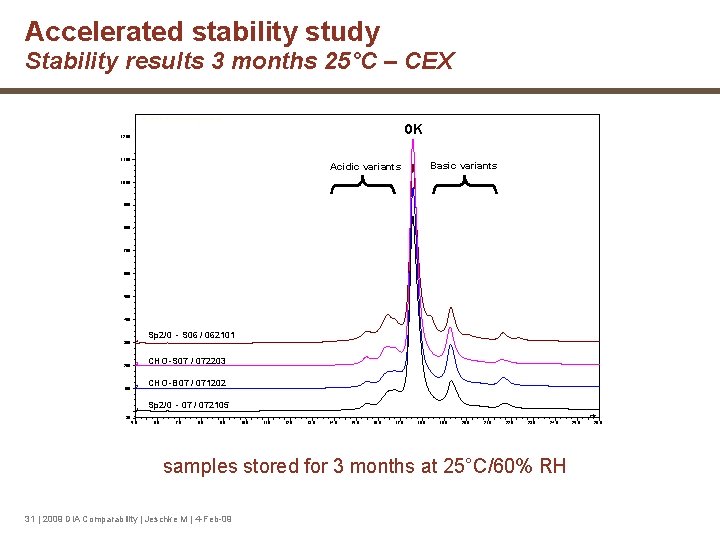
Accelerated stability study Stability results 3 months 25°C – CEX 1 - DM_080318_01 #11 [modified by mezzodo 1] 0 K 1, 200 1, 100 Basic variants Acidic variants 1, 000 900 800 700 600 500 400 Sp 2/0 - S 06 / 062101 300 4 200 3 CHO -S 07 / 072203 100 2 CHO -B 07 / 071202 1 Sp 2/0 - 07 / 072105 min -30 5. 0 6. 0 7. 0 8. 0 9. 0 10. 0 11. 0 12. 0 13. 0 14. 0 15. 0 16. 0 17. 0 18. 0 19. 0 20. 0 21. 0 22. 0 23. 0 24. 0 samples stored for 3 months at 25°C/60% RH 31 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 25. 0 26. 0
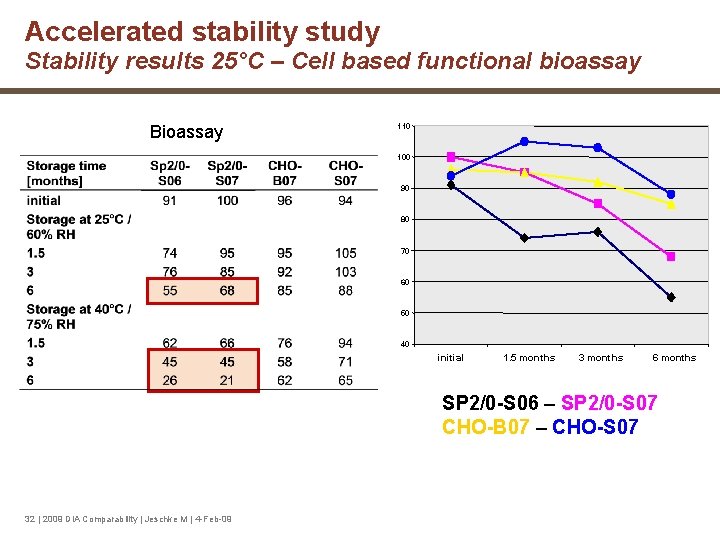
Accelerated stability study Stability results 25°C – Cell based functional bioassay Bioassay 110 100 90 80 70 60 50 40 initial 1. 5 months 3 months 6 months SP 2/0 -S 06 – SP 2/0 -S 07 CHO-B 07 – CHO-S 07 32 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09

Comparability – Summary & Conclusion Ø Process performance - Prot. A, HCP and DNA: • The single step efficiencies are different especially for DNA • Overall removal successful for all processes Ø Release testing: • All release requirements and additional comparability requirements met Ø Stability testing: • no critical observation for any stability parameter tested Ø Additional testing: • CHO-CHO: differences in amount of carbohydrates • CHO-Sp 2/0: • Differences in amount and kind of carbohydrates • Differences in binding strength to soluble Fc receptors • Variations in charge heterogeneity Analysis of the materials (CHO-CHO and CHO-Sp 2/0) shows some differences 33 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
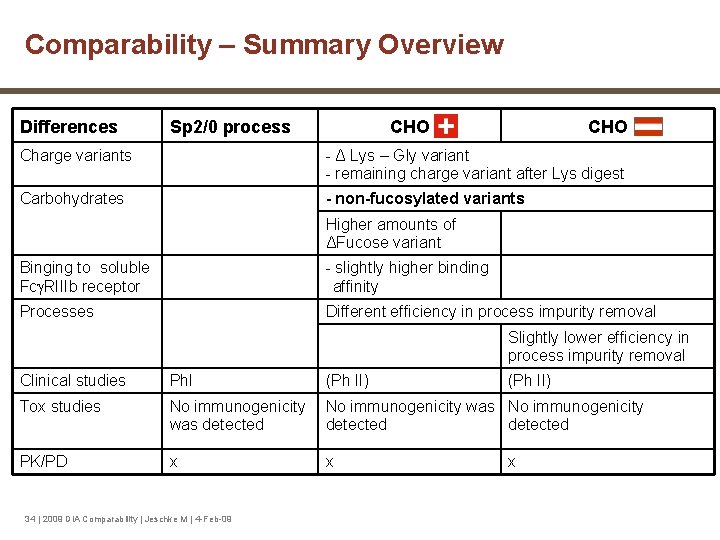
Comparability – Summary Overview Differences Sp 2/0 process CHO Charge variants - Δ Lys – Gly variant - remaining charge variant after Lys digest Carbohydrates - non-fucosylated variants Higher amounts of ΔFucose variant Binging to soluble Fc RIIIb receptor - slightly higher binding affinity Processes Different efficiency in process impurity removal Slightly lower efficiency in process impurity removal Clinical studies Ph. I Tox studies No immunogenicity was detected PK/PD x 34 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 (Ph II) x

Additional studies § Tissue crossreactivity studies • Comparison of positive staining from human tissues treated with SP 2/0 and CHO derived m. AB 1 NVS § In vivo repeated dose toxicity studies • 4 week intravenous toxicity studies in cynomolgus monkeys (incl. 8 or 10 weeks recovery) • All serum samples were analyzed for m. AB 1 NVS by target-based anti-idiotype competitive ELISA § PK and TK in cynomolgus monkey • pharmacokinetic single dose PK and 4 weeks tox studies in cynomolgus monkeys (iv dosing) with SP 2/0 -derived material and CHO-derived material) 35 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
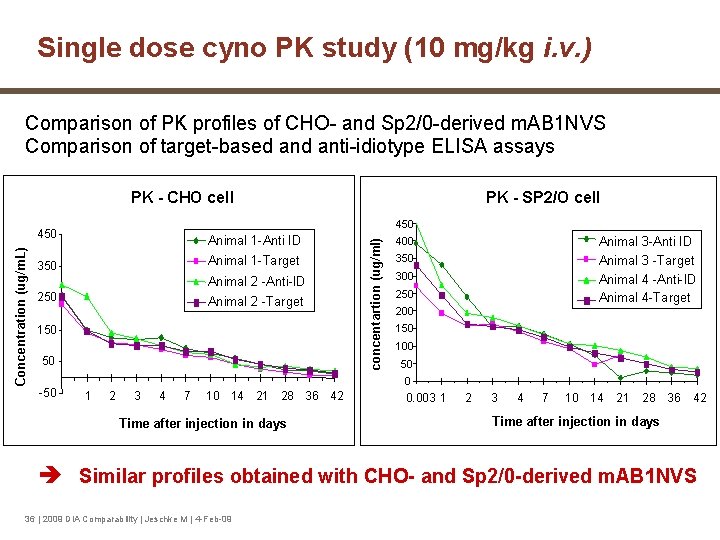
Single dose cyno PK study (10 mg/kg i. v. ) Comparison of PK profiles of CHO- and Sp 2/0 -derived m. AB 1 NVS Comparison of target-based anti-idiotype ELISA assays 450 Animal 1 -Anti ID 350 Animal 1 -Target Animal 2 -Anti-ID 250 Animal 2 -Target 150 50 -50 PK - SP 2/O cell concentartion (ug/ml) Concentration (ug/m. L) PK - CHO cell Animal 3 -Anti ID Animal 3 -Target Animal 4 -Anti-ID Animal 4 -Target 400 350 300 250 200 150 100 50 0 1 2 3 4 7 10 14 21 28 36 42 Time after injection in days 0. 003 1 2 3 4 7 10 14 21 28 36 42 Time after injection in days Similar profiles obtained with CHO- and Sp 2/0 -derived m. AB 1 NVS 36 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
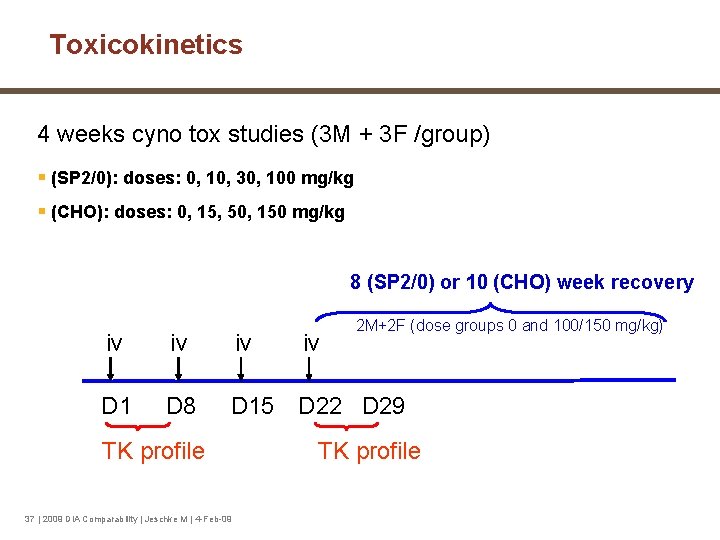
Toxicokinetics 4 weeks cyno tox studies (3 M + 3 F /group) § (SP 2/0): doses: 0, 10, 30, 100 mg/kg § (CHO): doses: 0, 15, 50, 150 mg/kg 8 (SP 2/0) or 10 (CHO) week recovery iv iv iv D 1 D 8 D 15 D 22 D 29 TK profile 37 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 iv 2 M+2 F (dose groups 0 and 100/150 mg/kg) TK profile
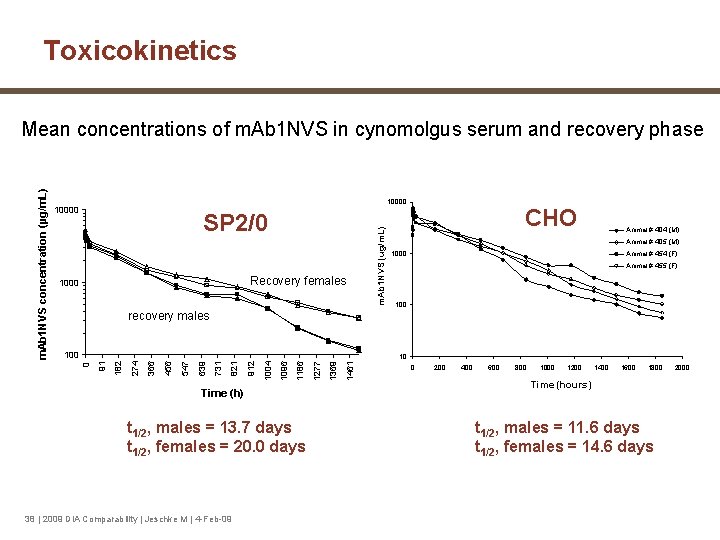
Toxicokinetics SP 2/0 Recovery females 1000 m. Ab 1 NVS (ug/m. L) 10000 CHO Animal # 404 (M) Animal # 405 (M) 1000 Animal # 454 (F) Animal # 455 (F) 100 recovery males 100 Time (h) t 1/2, males = 13. 7 days t 1/2, females = 20. 0 days 38 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09 1461 1369 1277 1186 1096 1004 912 821 731 639 547 456 366 274 182 91 10 0 m. Ab 1 NVS concentration (µg/m. L) Mean concentrations of m. Ab 1 NVS in cynomolgus serum and recovery phase 0 200 400 600 800 1000 1200 1400 1600 1800 Time (hours) t 1/2, males = 11. 6 days t 1/2, females = 14. 6 days 2000

Are they comparable ? ? ? „comparable does not mean identical“ 16, 2004 (Vol 69, # 157, Pages 50386 ff); Scientific Considerations FDA. August Related to Developing Follow-on Protein Products 39 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09

“During characterization of the drug substance a change to the glycosylation pattern was observed during the comparability exercise. Although it was demonstrated, that the difference in glycosylation obviously had no effect on the biological function, one can not exclude that there might be an immunogenic potential of the new glycans, which were identified in the CHO-derived material and therefore glycosylation should currently be part of the release specifications in order to demonstrate batch to batch consistency. ” - Paul-Ehrlich-Institute (2008) 40 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09

Lessons learned · In-depth analytical characterization and understanding of critical quality attributes is key to successful comparability · Perform risk assessment for planned changes and discuss implications upfront · Release Specs alone are inadequate to show no impact of change. Perform side-by-side analysis whenever possible · Keep retain samples of all batches and as long as possible · overload IEF / SDS-PAGE or silver stain to see low amount impurities. · Use exact same samples used for IPC analysis · Same test methods – otherwise describe differences · Explain differences well (incl. variation of methods used) · In the discussion of differences consider intended purpose 41 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09

ACKNOWLEDGEMENTS Manuela Schärpf Christoph Bächler Cornelius Fritsch Georg Hölzl Steffen Pahlich Iso Lengwiler Manuel Diez Marc Hassel Hui Zhao Gerard Bruin Markus Blümel Michaela Dehio Yuan Xu 42 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
Novartis Biologics, Basel, Switzerland Novartis Basel 43 | 2009 DIA Comparability | Jeschke M | 4 -Feb-09
- Slides: 43