Analytical Chemistry Definition the science of extraction identification





















- Slides: 36

Analytical Chemistry Definition: the science of extraction, identification, and quantitation of an unknown sample. Example Applications: • Human Genome Project • Lab-on-a-Chip (microfluidics) and Nanotechnology • Environmental Analysis • Forensic Science

Course Philosophy l l l develop good lab habits and technique background in classical “wet chemical” methods (titrations, gravimetric analysis, electrochemical techniques) Quantitation using instrumentation (UV-Vis, AAS, GC)

Analyses you will perform l l l l Basic statistical exercises %purity of an acidic sample %purity of iron ore titrations %Cl in seawater Water hardness determination UV-Vis: Amount of caffeine and sodium benzoate in a soft drink AAS: %Cu in pre- and post-1982 pennies GC: Gas phase quantitation using an internal standard

Chapter 1: Chemical Measurements



Example, p. 15: convert 0. 27 p. C to electrons

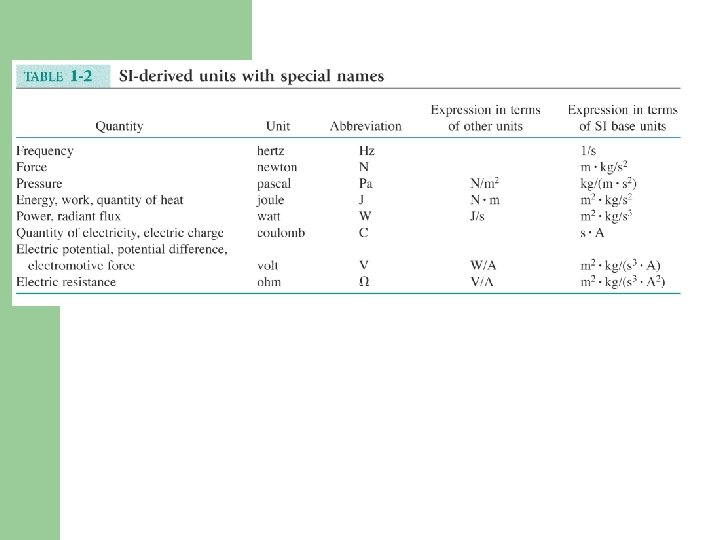
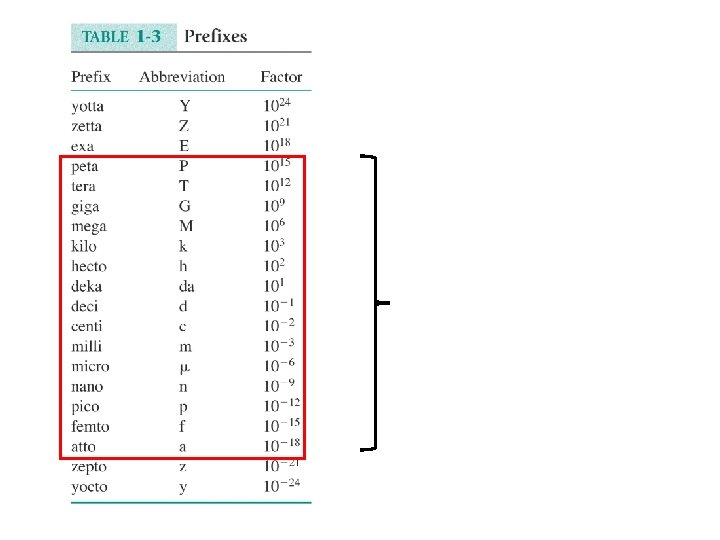
Chemical Concentrations

Example, p. 19: Molarity of Salts in the Sea (a) Calculate molarity of 2. 7 g Na. Cl/d. L (b) [Mg. Cl 2] = 0. 054 M. How many grams in 25 m. L?

Dilution Equation Concentrated HCl is 12. 1 M. How many milliliters should be diluted to 500 m. L to make 0. 100 M HCl? M 1 V 1 = M 2 V 2 (12. 1 M)(x m. L) = (0. 100 M)(500 m. L) x = 4. 13 M

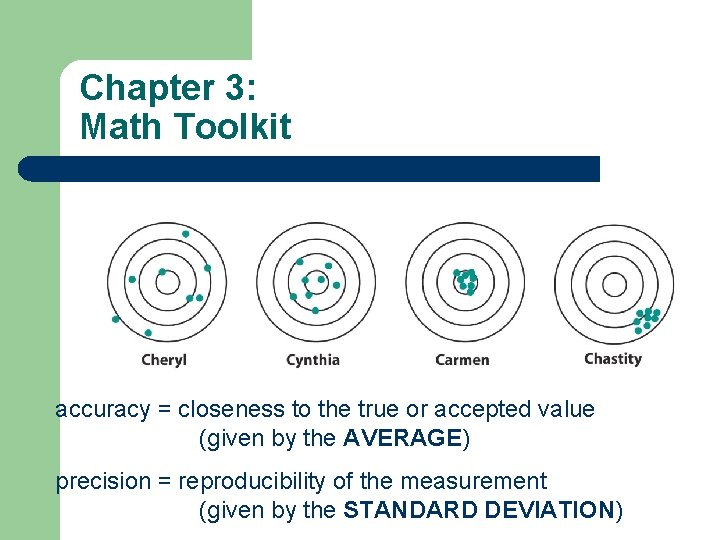
Chapter 3: Math Toolkit accuracy = closeness to the true or accepted value (given by the AVERAGE) precision = reproducibility of the measurement (given by the STANDARD DEVIATION)

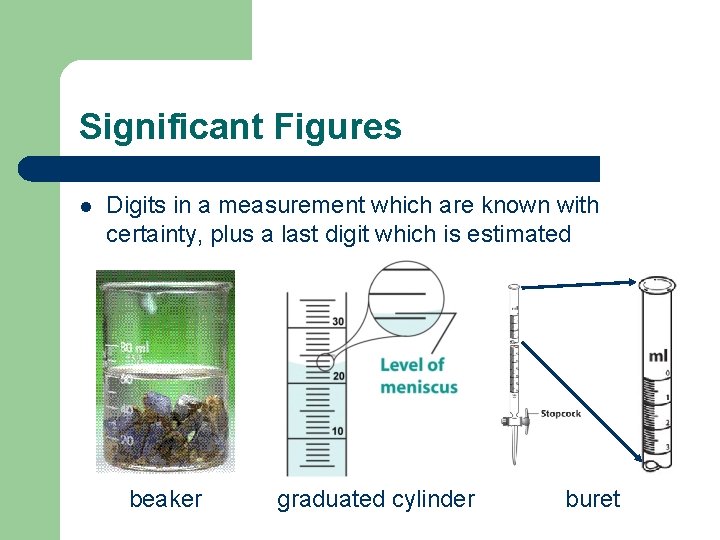
Significant Figures l Digits in a measurement which are known with certainty, plus a last digit which is estimated beaker graduated cylinder buret

Rules for Determining How Many Significant Figures There are in a Number · · · All nonzero digits are significant (4. 006, 12. 012, 10. 070) Interior zeros are significant (4. 006, 12. 012, 10. 070) Trailing zeros FOLLOWING a decimal point are significant (10. 070) Trailing zeros PRECEEDING an assumed decimal point may or may not be significant Leading zeros are not significant. They simply locate the decimal point (0. 00002)

Reporting the Correct # of Sig Fig’s l Multiplication/Division Rule: Round off to the fewest number of sig figs originally present 12. 154 5. 23 36462 24308 60770 63. 56542 ans = 63. 5

Reporting the Correct # of Sig Fig’s l Addition/Subtraction 15. 02 9, 986. 0 3. 518 10004. 538 Rule: Round off to the least certain decimal place

Reporting the Correct # of Sig Fig’s l Addition/Subtraction in Scientific Notation Express all of the numbers with the same exponent first: 1. 632 x 105 + 4. 107 x 103 + 0. 984 x 106

Reporting the Correct # of Sig Fig’s l Logs and anti-logs

Rounding Off Rules l l l digit to be dropped > 5, round UP 158. 7 = 159 digit to be dropped < 5, round DOWN 158. 4 = 158 digit to be dropped = 5, make answer EVEN 158. 5 = 158. 0 l BUT 157. 5 = 158. 0 158. 501 = 159. 000

Wait until the END of a calculation in order to avoid a “rounding error” ? sig figs 5 sig figs (1. 235 - 1. 02) x 15. 239 = 2. 923438 = 1. 12 3 sig figs 1. 235 -1. 02 0. 215 = 0. 22

Propagation of Errors A way to keep track of the error in a calculation based on the errors of the variables used in the calculation error in variable x 1 = e 1 = "standard deviation" (see Ch 4) e. g. 43. 27 0. 12 m. L percent relative error = %e 1 = e 1*100 x 1 e. g. 0. 12*100/43. 27 = 0. 28%

Addition & Subtraction Suppose you're adding three volumes together and you want to know what the total error (et) is: 43. 27 0. 12 42. 98 0. 22 43. 06 0. 15 129. 31 et

Multplication & Division

Combined Example

Chapter 4: Statistics

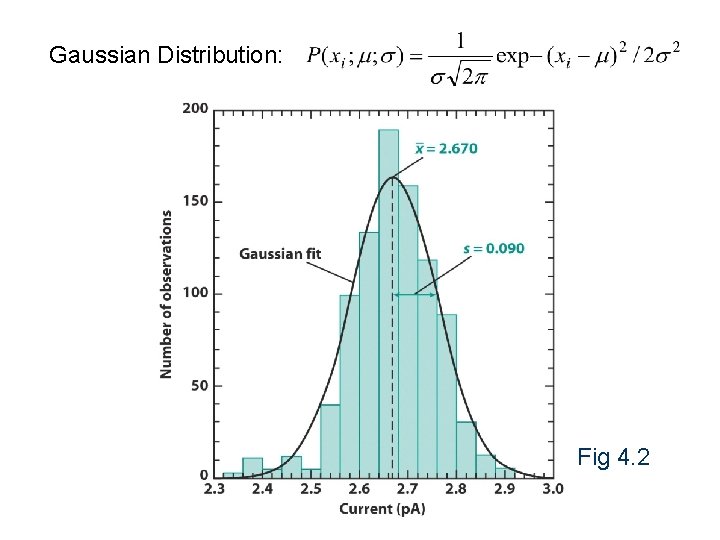
Gaussian Distribution: Fig 4. 2

Mean – measure of the central tendency or average of the data (accuracy) N Infinite population Finite population Standard Deviation – measure of the spread of the data (reproducibility) Infinite population Finite population

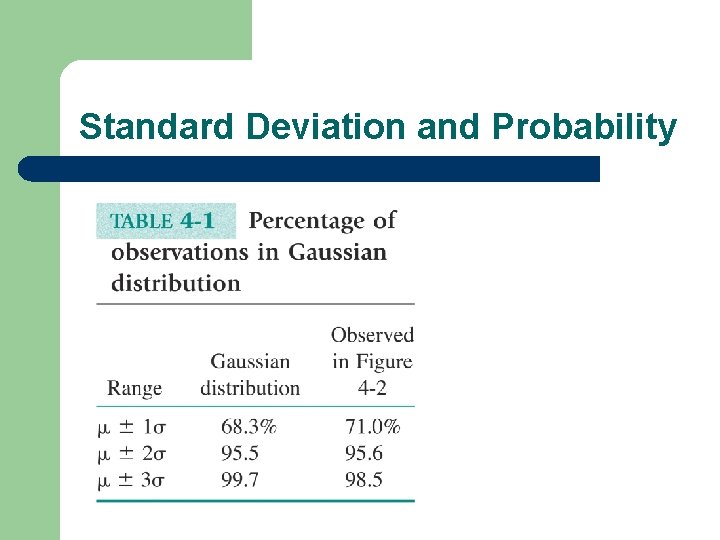
Standard Deviation and Probability

Confidence Intervals

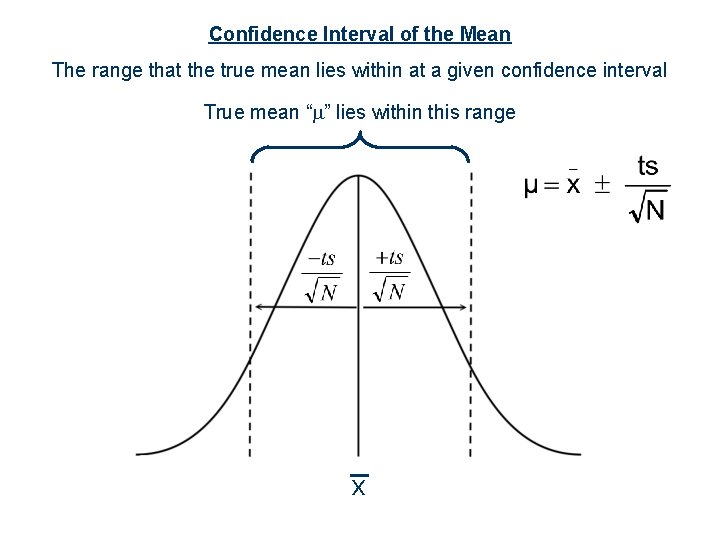
Confidence Interval of the Mean The range that the true mean lies within at a given confidence interval True mean “ ” lies within this range x


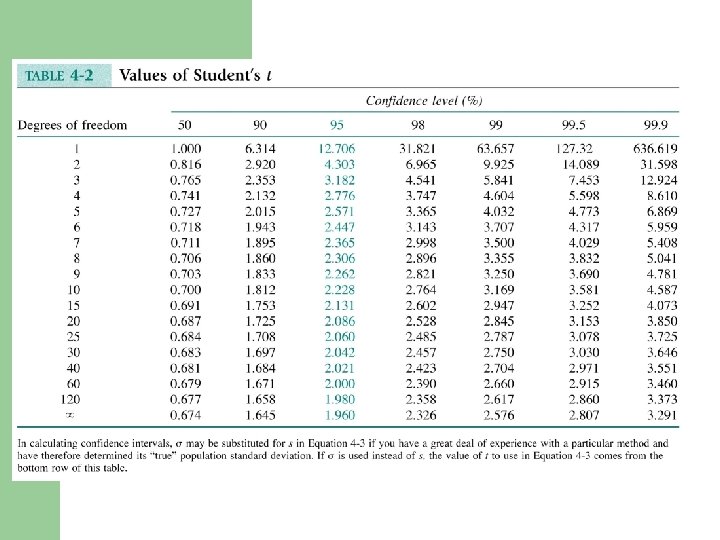
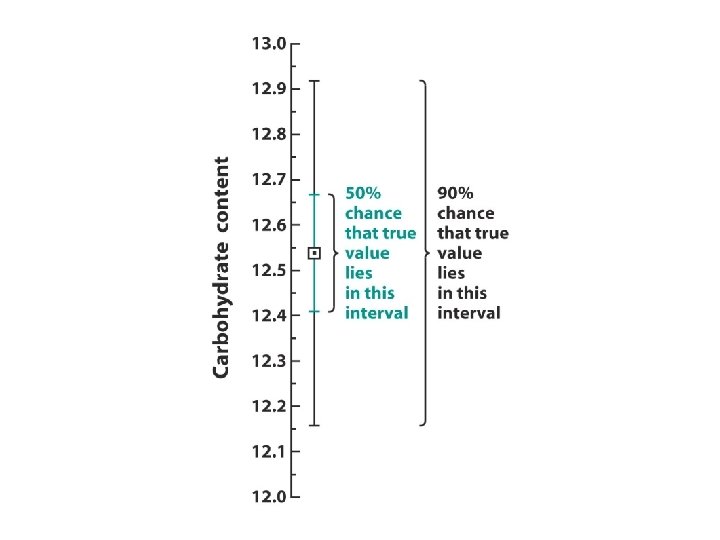
Example - Calculating Confidence Intervals l In replicate analyses, the carbohydrate content of a glycoprotein is found to be 12. 6, 11. 9, 13. 0, 12. 7, and 12. 5 g of carbohydrate per 100 g of protein. Find the 95% confidence interval of the mean. ave = 12. 55, std dev = 0. 465 N = 5, t = 2. 776 (N-1) = 12. 55 ± (0. 465)(2. 776)/sqrt(5) = 12. 55 ± 0. 58


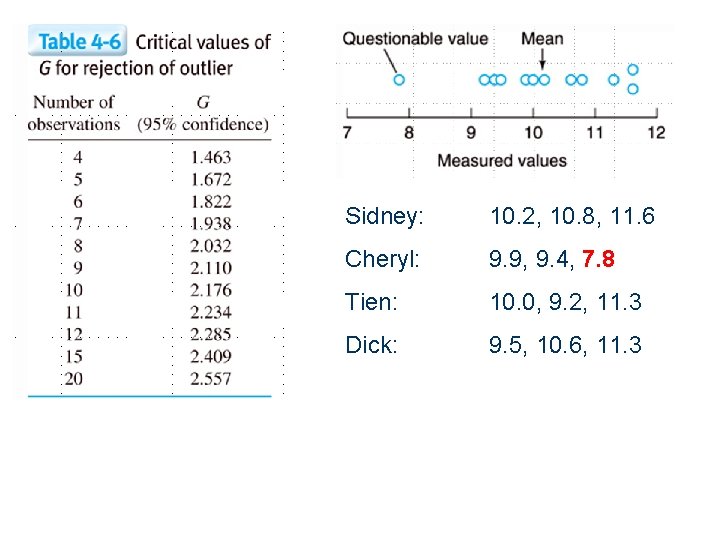
Rejection of Data - the Grubbs Test A way to statistically reject an “outlier” Compare to Gcrit from a table at a given confidence interval. Reject if Gexp > Gcrit

Sidney: 10. 2, 10. 8, 11. 6 Cheryl: 9. 9, 9. 4, 7. 8 Tien: 10. 0, 9. 2, 11. 3 Dick: 9. 5, 10. 6, 11. 3

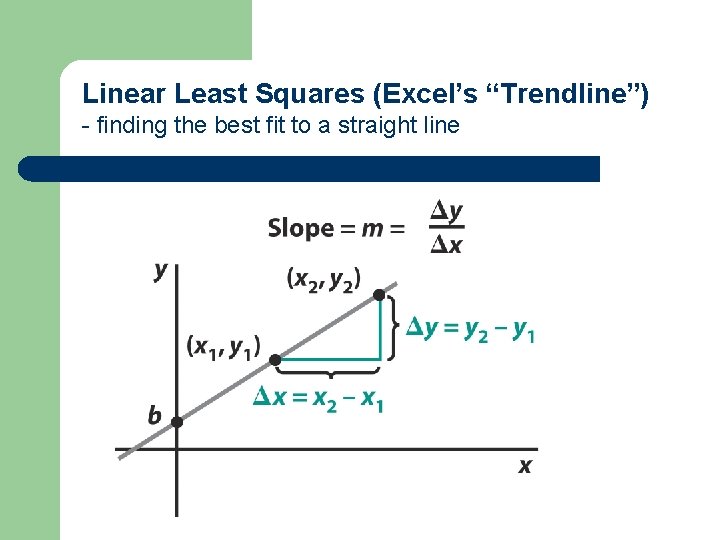
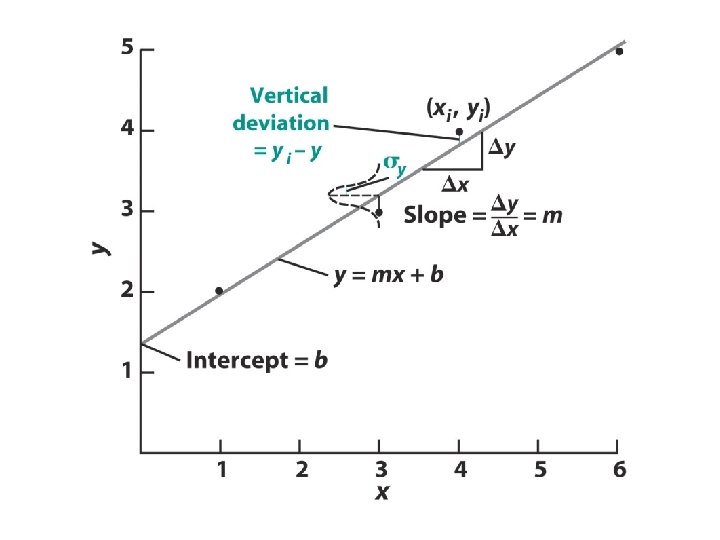
Linear Least Squares (Excel’s “Trendline”) - finding the best fit to a straight line
