Analyte Ionization Lecture 3 Yuri Kazakevich Seton Hall




![Ionization Equilibria [C 6 H 5 COO-][H+] Ka = [C H COOH] 6 5 Ionization Equilibria [C 6 H 5 COO-][H+] Ka = [C H COOH] 6 5](https://slidetodoc.com/presentation_image_h/993ccf779fda45d1c860f0f8524e8d4d/image-5.jpg)





- Slides: 17

Analyte Ionization Lecture 3 Yuri Kazakevich Seton Hall University 1

Reversed-Phase HPLC Retention The retention of ionizable analytes on the same bonded phase can be varied by: • Type of organic modifier • Concentration of organic modifier • Temperature • p. H of the mobile phase However the p. H of the eluent is dependent upon the type and concentration of the organic modifier and the temperature. 2

Introduction • p. H of the mobile phase affects: • • • analyte ionization and solvation interactions of the analytes with the stationary phase structure and properties bonded phase stability dissolution of silica matrix • p. H and the type of p. H modifier have a major impact on the selectivity alteration. • Recent developments in silica manufacturing and bonded phase chemistry significantly widened applicable p. H range (1. 5 - 10). 3

p. H Definition p. H is the negative logarithm of the proton concentration in the solution p. H = -log[H+] Equilibrium constants for acids are usually written in the following form: 4
![Ionization Equilibria C 6 H 5 COOH Ka C H COOH 6 5 Ionization Equilibria [C 6 H 5 COO-][H+] Ka = [C H COOH] 6 5](https://slidetodoc.com/presentation_image_h/993ccf779fda45d1c860f0f8524e8d4d/image-5.jpg)
Ionization Equilibria [C 6 H 5 COO-][H+] Ka = [C H COOH] 6 5 Ka=6. 4 x 10 -5 , p. Ka=4. 19 Increase of the proton concentration in the HPLC mobile phase shifts equilibrium to the left. p. Kb = 9. 4 p. Ka= 4. 6 p. Ka = 14 – p. Kb 5

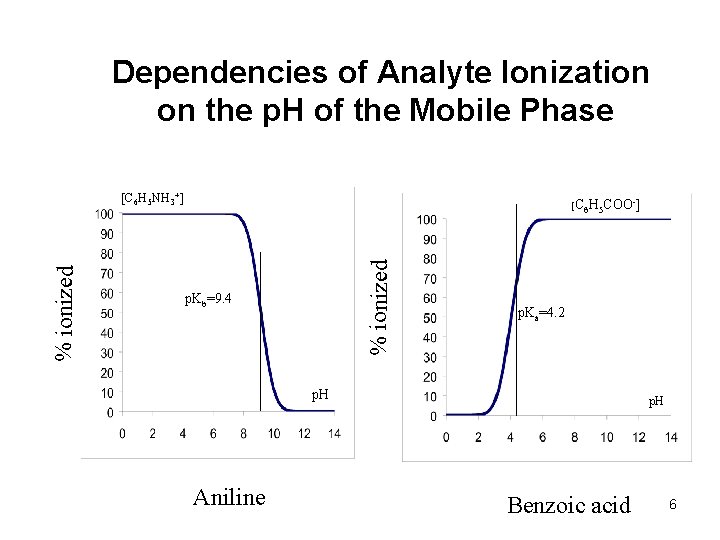
Dependencies of Analyte Ionization on the p. H of the Mobile Phase [C 6 H 5 COO-] % ionized [C 6 H 5 NH 3+] p. Kb=9. 4 p. Ka=4. 2 p. H Aniline p. H Benzoic acid 6

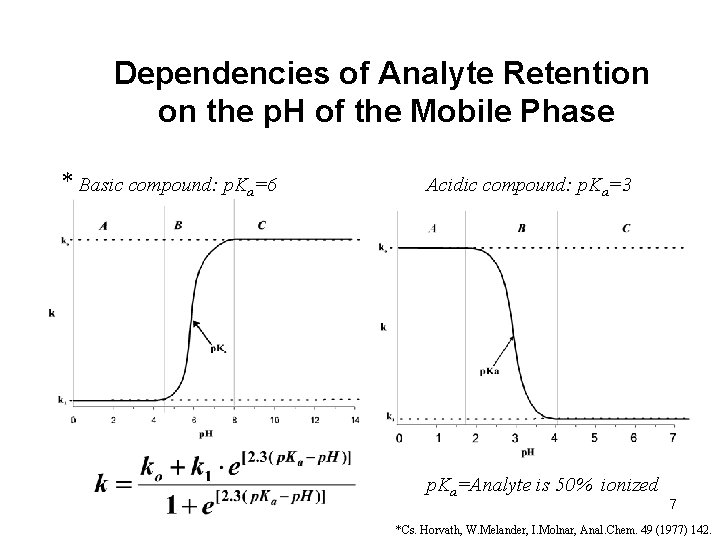
Dependencies of Analyte Retention on the p. H of the Mobile Phase * Basic compound: p. Ka=6 Acidic compound: p. Ka=3 p. Ka=Analyte is 50% ionized 7 *Cs. Horvath, W. Melander, I. Molnar, Anal. Chem. 49 (1977) 142.

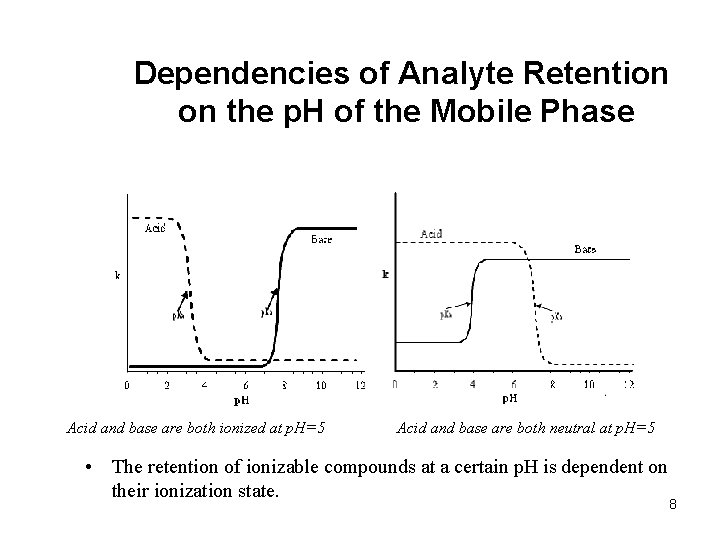
Dependencies of Analyte Retention on the p. H of the Mobile Phase Acid and base are both ionized at p. H=5 Acid and base are both neutral at p. H=5 • The retention of ionizable compounds at a certain p. H is dependent on their ionization state. 8

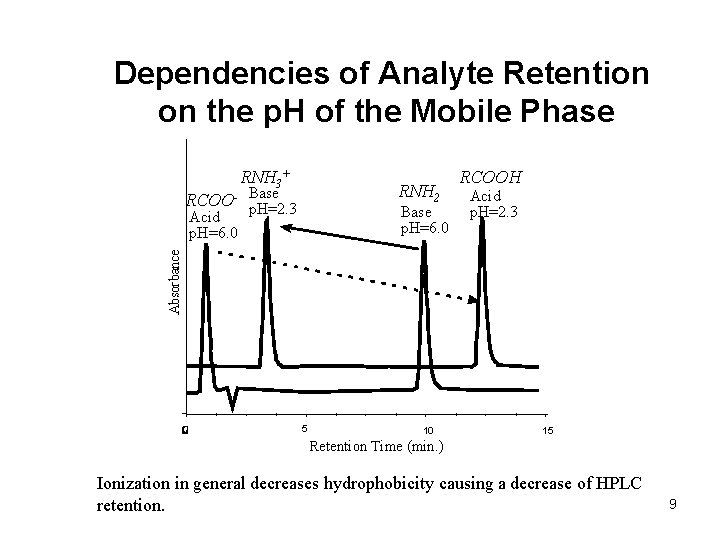
Dependencies of Analyte Retention on the p. H of the Mobile Phase RNH 3+ RNH 2 Base RCOO- p. H=2. 3 Base p. H=6. 0 Acid p. H=2. 3 Absorbance Acid p. H=6. 0 RCOOH 5 10 15 Retention Time (min. ) Ionization in general decreases hydrophobicity causing a decrease of HPLC retention. 9

Effect of p. H on Acidic Analyte Retention HPLC Conditions: Column – Zorbax Eclipse XDB-C 18, 150 mm x 4. 6 mm Flow rate – 1. 0 m. L/min Detection – UV 220 nm Injection volume – 1 L Mobile phase 30% Acetonitrile 70% 20 m. M Na 2 HPO 4 buffer; variable p. H acidic modifier = perchloric acid 10

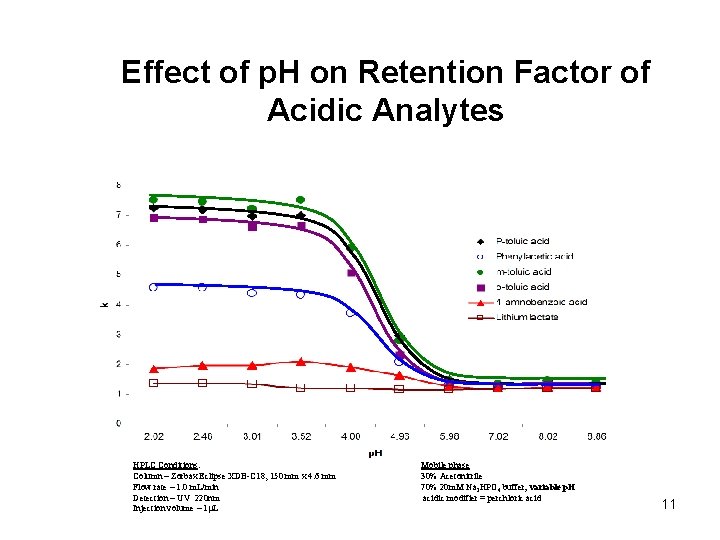
Effect of p. H on Retention Factor of Acidic Analytes HPLC Conditions: Column – Zorbax Eclipse XDB-C 18, 150 mm x 4. 6 mm Flow rate – 1. 0 m. L/min Detection – UV 220 nm Injection volume – 1 L Mobile phase 30% Acetonitrile 70% 20 m. M Na 2 HPO 4 buffer; variable p. H acidic modifier = perchloric acid 11

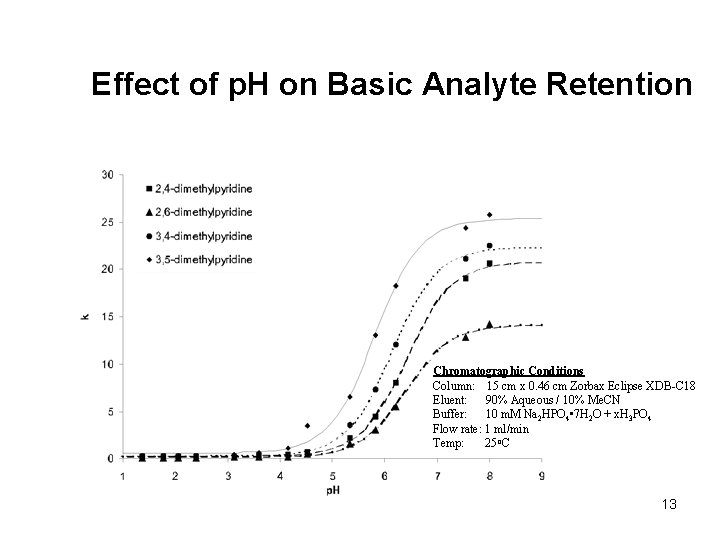
Effect of p. H on Basic Analyte Retention pyridine p. Ka = 5. 17 2, 4 -lutidine p. Ka = 6. 74 CH 3 N H 3 C N CH 2 CH 3 4 -ethylpyridine p. Ka = 5. 87 CH 3 2, 3 -dimethylaniline p. Ka = 4. 70 CH 3 Chromatographic Conditions Column: 15 cm x 0. 46 cm Zorbax Eclipse XDB-C 18 Eluent: 90% Aqueous / 10% Me. CN Buffer: 10 m. M Na 2 HPO 4 • 7 H 2 O + x. H 3 PO 4 Flow rate: 1 ml/min Temp: 25 o. C 2 -picoline p. Ka=5. 96 phenylethylamine p. Ka=9. 83 N NH 2 N CH 3 NH 2 12 CH 3 Time (min. )

Effect of p. H on Basic Analyte Retention Chromatographic Conditions Column: 15 cm x 0. 46 cm Zorbax Eclipse XDB-C 18 Eluent: 90% Aqueous / 10% Me. CN Buffer: 10 m. M Na 2 HPO 4 • 7 H 2 O + x. H 3 PO 4 Flow rate: 1 ml/min Temp: 25 o. C 13

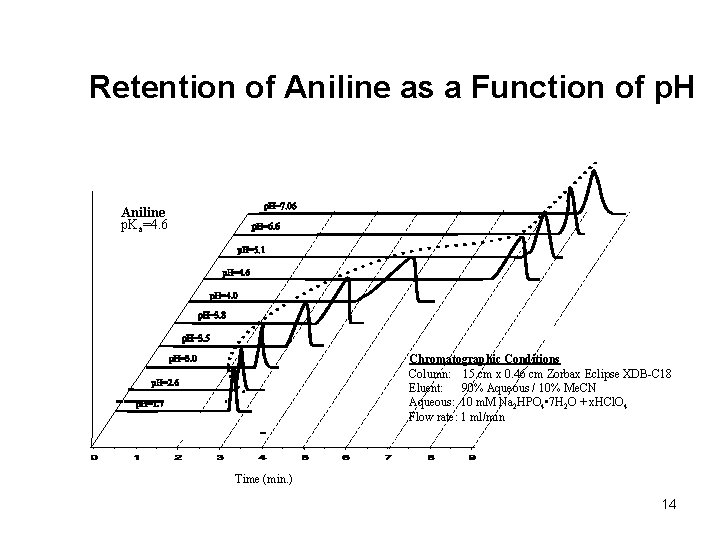
Retention of Aniline as a Function of p. H Aniline p. Ka=4. 6 Chromatographic Conditions Column: 15 cm x 0. 46 cm Zorbax Eclipse XDB-C 18 Eluent: 90% Aqueous / 10% Me. CN Aqueous: 10 m. M Na 2 HPO 4 • 7 H 2 O + x. HCl. O 4 Flow rate: 1 ml/min Time (min. ) 14

Peak Fronting • A condition where the rear of the peak is steeper than the front relative to the baseline. • Related to a secondary chemical equilibrium process -Analyte Ionization p. H of analysis is close to analyte p. Ka -Reaction of analyte with mobile phase components (ex. Aldehydes in presence of water under acidic or basic conditions) 15

Peak Tailing • A condition where the front of the peak is steeper than the rear relative to the baseline. • Appears when the analyte concentration exceed the linear range of adsorption isotherm. • Tailing (Depends upon acidity of silanols, ionization state of basic analyte and mobile phase p. H). 16

Concluding Remarks • p. H is an effective tool for adjustment of selectivity and retention • p. H can be used to optimize the resolution • Reversed phase packings are most stable between p. H’s 2 - 8. 17