Analysis means separating something into its elemental parts












































- Slides: 55

Analysis means separating something into its elemental parts or basic principles. 1

As per IBR Boiler: Boiler meant “any closed vessel exceeding 22. 75 (25) liters in capacity and which is used expressively for generating steam under pressure and includes any mounting or other fitting attached to such vessel which is wholly or partly under pressure when the steam is shut off. ” In new act it is a pressure vessel in which steam is generated for use external to itself by application of heat which is wholly or partly under pressure when steam is shut off but does not include a pressure vessel : i) with capacity less than 25 litres (such capacity measured from the feed check valve to main steam stop valve) ii) with less than one kilogram per centimeter square design gauge pressure and working pressure or iii) in which water is heated below one hundred degree centigrade. Pipe: In new act, it means any pipe through which steam passes. The pressure and diameter limitation will be there. (10 inch and 3. 5 Kg/Cm 2 pressure) 2

Main Circuits: Water Circuit: Feed water & Boiler Circulating water Fuel Circuit: Combustion Air Circuit: Flue Gas Circuit: Steam Circuit: Condensate Circuit: 3

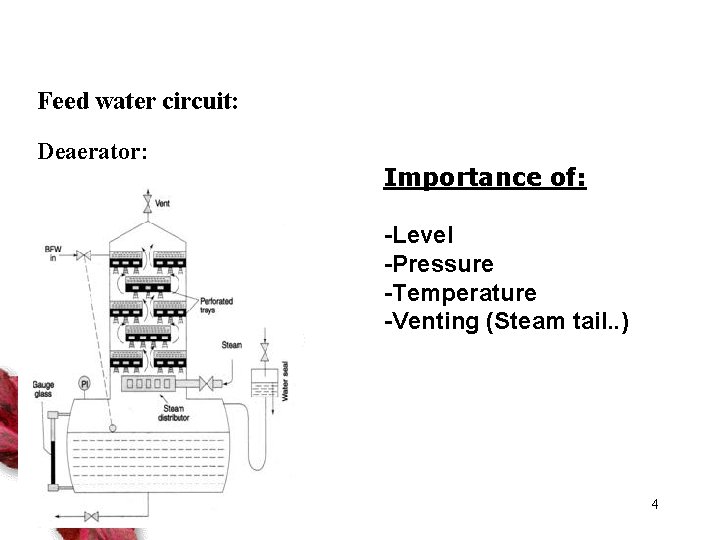
Feed water circuit: Deaerator: Importance of: -Level -Pressure -Temperature -Venting (Steam tail. . ) 4

Pdea + Ph - fh - hsuction > vapor pressure (pump) • Steam coming in Has following functions: – Maintaining level – Maintaining Pressure – Maintaining Temperature 5

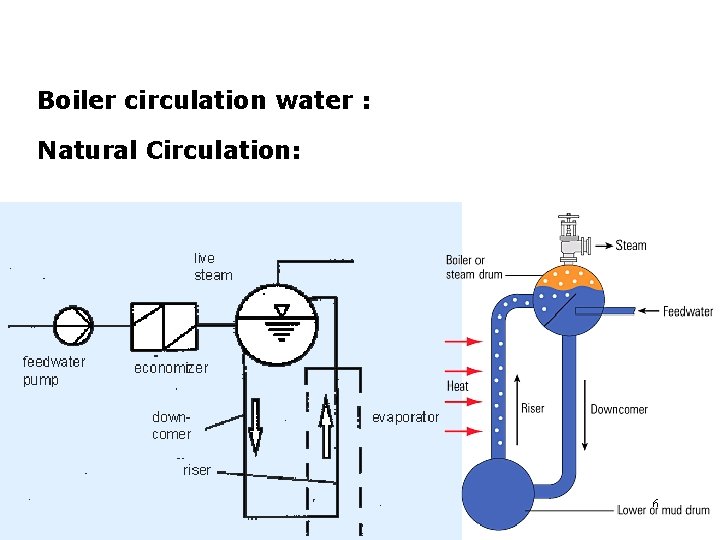
Boiler circulation water : Natural Circulation: 6

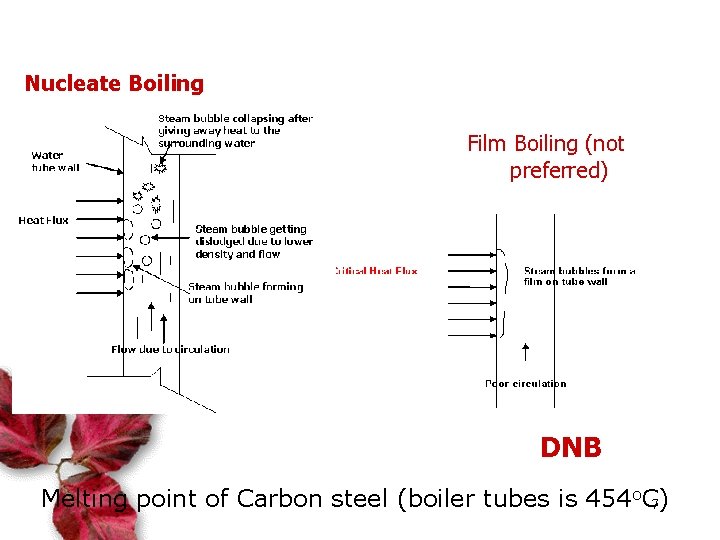
Nucleate Boiling Film Boiling (not preferred) DNB Melting point of Carbon steel (boiler tubes is 454 o. C) 7

Departure from Nucleate Boiling (DNB) leads to Film Boiling – overheating of evaporator tube and rapture Flow = h (density 2 – density 1) Flow = h (constant) Flow is prop to h. That’s why drum level is important Steam density = 1603 x Water density @ 1 Atm 8

Fuel Circuit: IMPORTANT PARAMETERS IN AFBC BOILER 1. Fuel Size 2. Bed Material Size & Specifications 3. Bed Temperature / Furnace Temperature 4. Primary Air Pressure 5. Bed Height / F. D. Air Pressure 9

Bed Material Specifications : Crushed Fire Bricks Castable IS 8 grade Bricks or River Silica Sand Fusion Temperature Shape Bulk Density (Kg/m 3) Silica (Si. O 2) Al 203 Fe 203 Pi. O 2 Mn. O Ca. O Mg. O P 2 O 5 A 2 O K 2 O : : : : 1300 Deg. C. Spherical Angular 1050 68 % 28 % 1. 05% 1. 67% Trace 0. 54% 0. 23% 0. 08% 0. 22% 0. 45% 10

BED TEMP. / FURNACE TEMPERATURE • Depends upon the type of fuel & firing method. • For (Indian Coal) bed temp. in FBC/AFBC/CFBC is to be maintained 850 o. C to 900 o. C. • For Imported Coal bed temp. in FBC/AFBC/CFBC is to be maintained bed 875 o. C to 925 o. C. • For lignite bed temp. in FBC/AFBC/CFBC is to be maintained 800 o. C to 850 o. C. • For Petcock bed temp. in FBC/AFBC/CFBC is to be maintained 875 o. C to 950 o. C. 11

PRIMARY AIR PRESSURE High P. A. Pressure increases the high bed coil erosion and carryover. (due to abrasive nature) Low P. A. pressure cause the blockage of P. A. lines and puffing in boiler and also increases chances of clinkerization. 12

Fouling & Slagging: What is Slagging and fouling? • It is melting or fusing of ash at very high temperature • It is sticky and clings to heating surface in bulk, preventing heat transfer • When molten slag cools and solidifies, becomes rock hard • When ash melts to glassy, sintering, cementing property it forms a hard layer and foul the metal surface 13

Fouling & Slagging: 14

Fouling & Slagging: Why slagging take place? • More the iron, calcium and sodium in ash, more the slagging • Quartz and kaolinite Siliceous matters in ash cause slagging and fouling • Slagging occurs, when ash is subjected to its fusion temperature • Slagging process hastens, when fuel is starving for oxygen Fusing of ash take place, • • If furnace temperature exceeds Initial Deformation range of ash Delay in evacuation of bottom ash, results in build up of slag 15

Low Stack Temperature Corrosion: Ø Stack temperature should be above Acid Dew Point 16

Combustion is a chemical reaction of: C + O 2 = CO 2 + 8084 kcal/ kg 12 Kg + 32 Kg 44 Kg Complete Combustion 2 H 2 + O 2 = 2 H 2 O +28922 kcal/ kg S + O 2 = SO 2 + 2224 kcal/ kg 2 C + O 2 = 2 CO + 2430 kcal/ kg Incomplete Combustion GCV of fuel = [8084 C + 28922 (H – O/8) + 2224 S] kcal/ kg Minimum Theoretical Air required for complete combustion = [2. 67 C + 8(H - O/8) + S] x 100/23 % Excess air supplied (on O 2% basis) = (O 2% x 100) / (21 - O 2%) 17

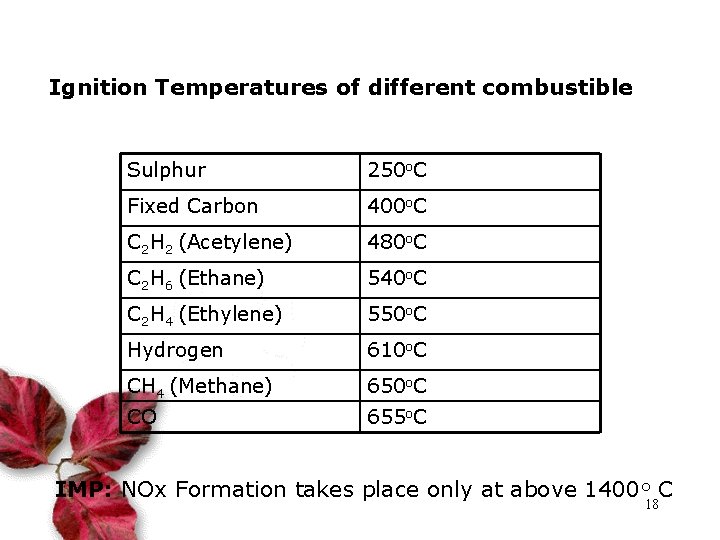
Ignition Temperatures of different combustible Sulphur 250 o. C Fixed Carbon 400 o. C C 2 H 2 (Acetylene) 480 o. C C 2 H 6 (Ethane) 540 o. C C 2 H 4 (Ethylene) 550 o. C Hydrogen 610 o. C CH 4 (Methane) 650 o. C CO 655 o. C IMP: NOx Formation takes place only at above 1400 o C 18

Steam Generator efficiency or thermal efficiency: Ø Direct Method Ø Indirect Losses Method 19

Thermal efficiency by Direct Method: η boiler = Where, ma ( H - h ) -------- x 100 mf C ma = Actual evaporation (steam) mf = Mass of fuel H = Enthalpy of steam h = Enthalpy of Feed Water C = Calorific value of fuel in kcal/ kg Indirect Losses Method η boiler = 100 (GCV) - Heat Losses as % of GCV 20 We can say Indirect method is for finding the percentage of Non losses.

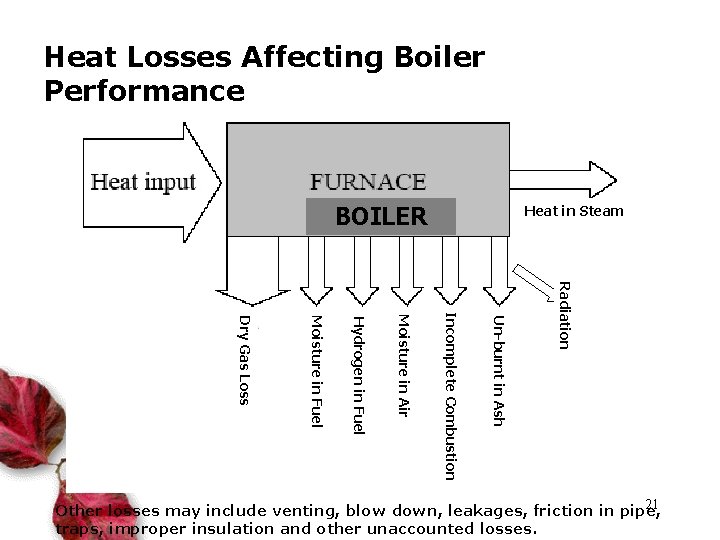
Heat Losses Affecting Boiler Performance BOILER Heat in Steam Radiation Un-burnt in Ash Incomplete Combustion Moisture in Air Hydrogen in Fuel Moisture in Fuel Dry Gas Loss 21 Other losses may include venting, blow down, leakages, friction in pipe, traps, improper insulation and other unaccounted losses.

1. Heat lost with dry flue gas through chimney = mg x Cg x (Tg - Ta) 2. Heat loss due to moisture formed by combustion of Hydrogen in fuel = 9 H x {584 + 0. 45 x (Tg - Ta)} kcal/ kg of fuel 3. Heat loss due to moisture present in fuel = Mw x {584 + 0. 45 x (Tg - Ta)} kcal/ kg of fuel 4. Heat loss due to moisture present in combustion air = Ma x FH x 0. 45 x (Tg - Ta)} kcal/ kg of fuel 5. Heat Lost due to incomplete combustion = MCO x 5744 kcal/ kg of fuel [MCO = {CO% / (CO% + CO 2%)} x C] 6. Heat lost due to un-burnt = Mash x GCV of ash 7. Heat Lost due to radiation and convection = 0. 548 x [(Ts / 55. 55)4 - (Ta / 55. 55)4] + 1. 957 x (Ts - Ta)1. 25 x √[(196. 85 Va + 68. 9) / 68. 9] W/ m 2 22

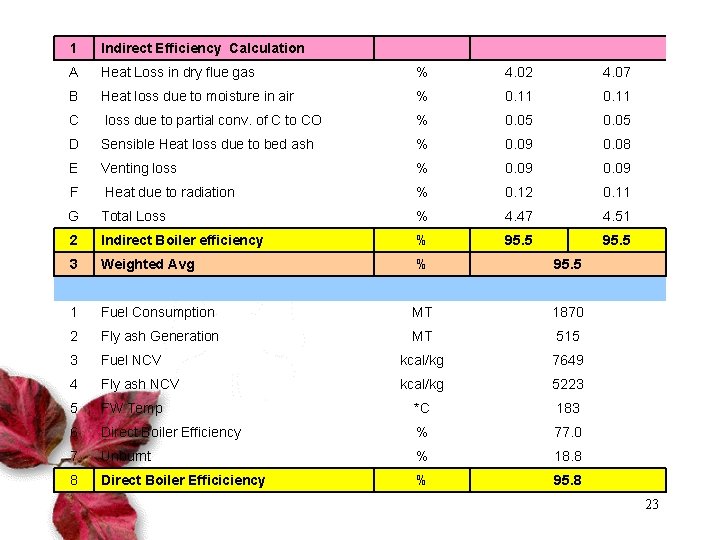
1 Indirect Efficiency Calculation A Heat Loss in dry flue gas % 4. 02 4. 07 B Heat loss due to moisture in air % 0. 11 C loss due to partial conv. of C to CO % 0. 05 D Sensible Heat loss due to bed ash % 0. 09 0. 08 E Venting loss % 0. 09 F Heat due to radiation % 0. 12 0. 11 G Total Loss % 4. 47 4. 51 2 Indirect Boiler efficiency % 95. 5 3 Weighted Avg % 95. 5 1 Fuel Consumption MT 1870 2 Fly ash Generation MT 515 3 Fuel NCV kcal/kg 7649 4 Fly ash NCV kcal/kg 5223 5 FW Temp *C 183 6 Direct Boiler Efficiency % 77. 0 7 Unburnt % 18. 8 8 Direct Boiler Efficiciency % 95. 8 23

Water Analysis: Q. Why is it necessary? ? Ans. As water is the blood running in veins of boiler. What do we check in the Boiler Water? p. H Conductivity or TDS Silica Phosphate Residual Hydrazine Chloride Iron & Copper (occasionally) We keep an eye on these parameters because they play vital role in the boiler chemistry. 24

Water Analysis: Feed Water Analysis at Shree p. H 8. 5 to 9. 5 Conductivity < 10 micro mho/ cm Si. O 2 < 0. 05 ppm N 2 H 4 < 0. 1 ppm Hardness Nil Boiler Water Analysis at Shree p. H of steam is 8. 5 to 9. 5 p. H 9. 5 to 10. 5 Conductivity < 200 micro mho/ cm Si. O 2 < 5 ppm ? ? N 2 H 4 -- Phosphate 5 to 15 ppm Dosing is done to maintain p. H. 25

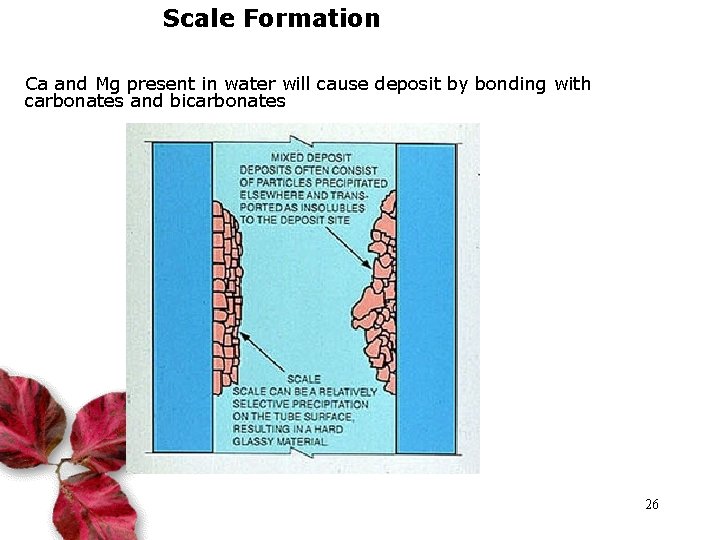
Scale Formation Ca and Mg present in water will cause deposit by bonding with carbonates and bicarbonates 26

Silica can vaporize into the steam at operating pressures as low as 28 bars. Its solubility in steam increases with increased temperature but decreases with increased p. H IMP!!!!!!!: Between 9 to 10 p. H solubility of silica is less Corrosion Mainly due to Dissolved Oxygen The degree of oxygen attack depends on • The concentration of dissolved oxygen • The p. H (lesser p. H more corrosion) • The temperature of the water (more temp more corrosion) But for Copper, increasing p. H inc corrosion, that’s why cooling tower 27 has p. H 7. 5

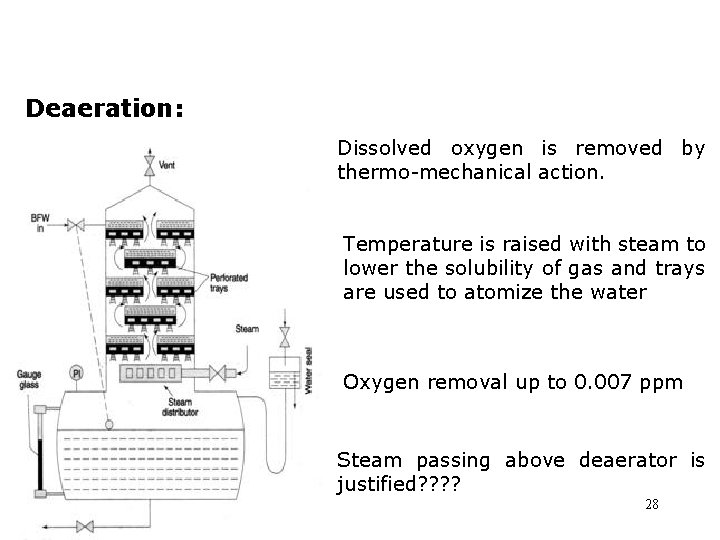
Deaeration: Dissolved oxygen is removed by thermo-mechanical action. Temperature is raised with steam to lower the solubility of gas and trays are used to atomize the water Oxygen removal up to 0. 007 ppm Steam passing above deaerator is justified? ? 28

Hydrazine dosing is done either at condensate line or at feed water suction line where the temperature is well below 270°C & Above 75 o C as Hydrazine is reactive only above 75 o. C At 270°C, hydrazine undergoes rapid thermal degradation 3 N 2 H 4 + Heat → N 2 + 4 NH 3 • Ammonia further formed is from amide group and causes rusting. • Residual hydrazine should be less than 0. 05% of dozed. 29

p. H correction In high pressure Boiler, the pre-boiler conditions are having a high temperature Ammonia and/or Morpholine or Cyclo-hexylamine is sometimes added before the Deaerator for p. H control In case Copper alloys are used in the pre-boiler section like HP heater, p. H is to be kept lower to avoid oxidation 30

The boiler water p. H is maintained by dosing of TSP or Tri Sodium Phosphate in the boiler section Na 3 PO 4 + H 2 O ↔ Na 2 HPO 4 + Na. OH • 1 TSP produces 1 phosphate & 3 Na. OH (by molar ratio) Extra Na. OH is free Caustic. • Phosphate reacts with Na. OH so as to reduce free caustic. That’s why phosphate should be present in water (5 -6 ppm) • If Ca & Mg are not present, then we can use Na. OH so as to avoid phosphate. Disodium phosphate and Monosodium phosphate is also used for dozing. 31

Blow Down control: CBD (Continuous Blow Down) (from in between boiler, but many times done from lower part of boiler) Used for removing dissolved salts (decreasing TDS) as maximum concentration is on surface only and keeps on decreasinh with depth. ) IBD (Intermittent Blow Down) (from lower part of boiler) Used for removing Scales formed at the lower surface. Done with huge flow, to let momentum come in play and carry more solid crystals) 32

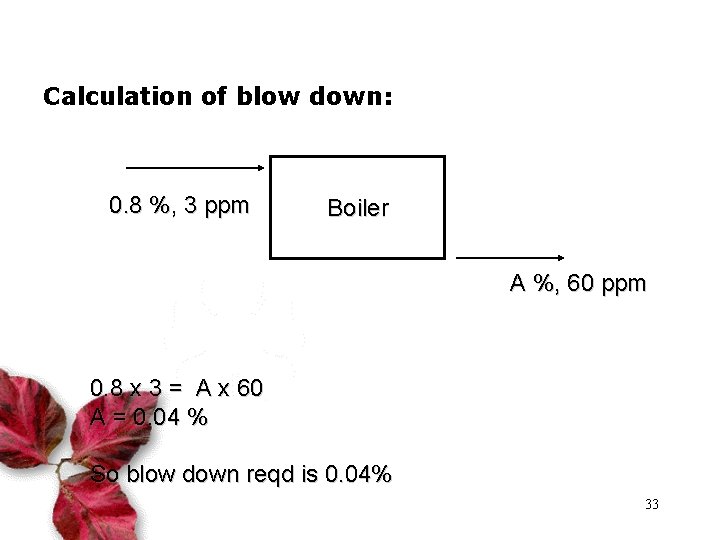
Calculation of blow down: 0. 8 %, 3 ppm Boiler A %, 60 ppm 0. 8 x 3 = A x 60 A = 0. 04 % So blow down reqd is 0. 04% 33

Protective Layer Gray colour Protective Layer inside the boiler and boiler tube Magnetite Layer (Fe 3 O 4) Created through presence of insufficient oxygen. SO Oxygen is must for Boiler water. Formed by Hydrazene Fe. O Fe 2 O 3 Fe 3 O 4 (presence of insufficient oxygen) 34

What causes Boiler tube failures? Corrosion Scale Erosion Unwanted Stress Fatigue Creep 35

Failure Mechanisms Various reasons for pressure parts failures are: Corrosion Erosion Over pressure Over heating Creep Caustic embrittlement Hydrogen attack Fatigue Stress corrosion 36

CORROSION WATER SIDE CORROSION: Low p. H Corrosion Oxygen pitting Caustic / Acid Attack Caustic Gouging FIRE SIDE CORROSION: Low temperature / cold end corrosion Harmful metals, Sulphur and Chlorine Corrosive characteristics of ash Slag deposits 37

REASONS FOR OCCURRENCE OF EROSION DUE TO SOLID PARTICLES: Abrasive nature of ash Unburned solids in flue gas Irregular Bed material ( in case of FBC) DUE TO STEAM IMPINGEMENT: Steam from soot Blowing Steam leak impinging adjacent tubes 38

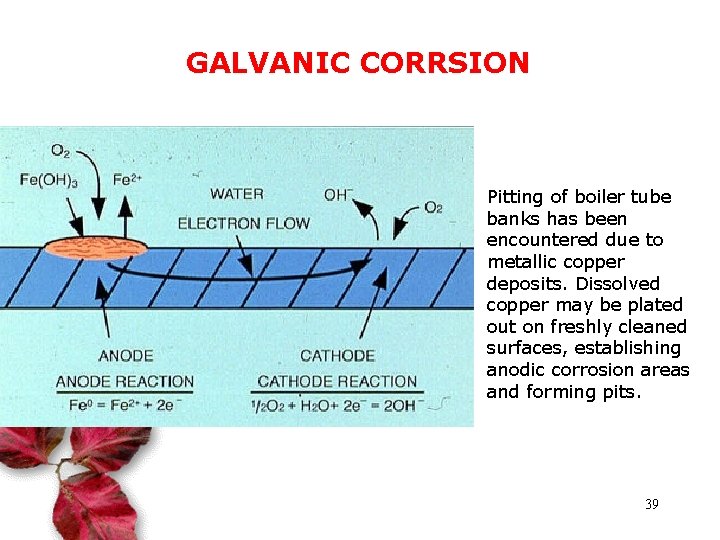
GALVANIC CORRSION Pitting of boiler tube banks has been encountered due to metallic copper deposits. Dissolved copper may be plated out on freshly cleaned surfaces, establishing anodic corrosion areas and forming pits. 39

LOW p. H CORROSION AND OXYGEN PITTINGS 40

LOW p. H CORROSION AND OXYGEN PITTINGS REASONS: High level of dissolved oxygen in feed water and boiler water Ineffective p. H control REMEDIAL ACTIONS: Control oxygen level in feed water Maintain proper feed and drum water p. H Use proper lay up procedures 41

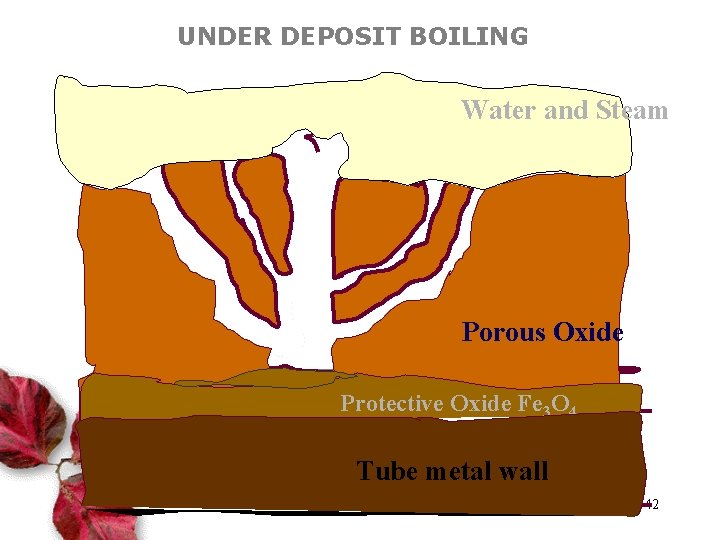
UNDER DEPOSIT BOILING Water and Steam Porous Oxide Protective Oxide Fe 3 O 4 Tube metal wall 42

ACID CHLORIDE CORROSION Mg. Cl 2 + H 2 O ---> Mg. O + 2 HCl Fe 3 O 4 +HCl ---> Fe. Cl 2 + Fe. Cl +H 2 O Fe + 2 HCl ---> Fe. Cl 2 + H 2 43

FIRE SIDE CORROSION Low Temperature / Cold End Corrosion REASONS: Sulphur in fuel Sodium or Vanadium Pentoxide and ferric oxide in fuel Excess Oxygen Low Economizer inlet temperature Low gas/ air temperature at APH inlet 44

EROSION REASONS: Moving fly ash particles Abrasive particles (coal etc. ) Steam impingement by leaking steam of adjacent tube 45

REMEDIAL ACTION FLY ASH EROSION Install baffles / shields in flow path Use ceramic / hard metal pads Reduce flow velocity of gas by modifications COAL PARTICLE EROSION Change flow pattern Use wear resistant liners Ensure proper sieve sizing of coal Optimum coal / air mixer velocity 46

STRESS FAILURES REASONS: OVERPRESSURE Blockage in tubes Operating Pressure Excursion Reduction in tube thickness due to metal loss 47

STRESS RUPTURE FAILURES OVERHEATING REASONS Insufficient circulation of fluid inside tube - DNB Operating Temperature Excursion Flame Impingement 48

SHORT TERM OVERHEATING TYPICAL CAUSES: Low water level Partial or complete plugging of tubes Rapid start-ups Excessive load swings Excessive heat input 49

LONG TERM OVERHEATING TYPICAL CLAUSES: Gradual accumulation of deposits or scale Partially restricted steam or water flow Excessive heat input from burners Undesired channeling of fireside gases Operation slightly above oxidation limits of given tube steel (850 ºF for carbon steel) 50

REMEDIAL ACTIONS SHORT TERM OVERHEATING Prevent tube blockages Control drum water level Ensure coolant circulation Avoid over firing / flame impingement 51

CAUSTIC EMBRITTLEMENT REASONS: For caustic embrittlement to occur, three conditions must exist: The boiler metal must have a high level of stress A mechanism for the concentration of boiler water must be present The boiler water must have embrittlement-producing characteristics 52

CAUSTIC EMBRITTLEMENT REMEDIAL ACTIONS: Proper stress relieving of all welded or rolled section Application of a coordinated p. H/phosphate control Using Sodium Nitrate treatment (removes excess Na. OH) 53

Failure Investigation Methods for Boiler Tubes We need to find out the answers to: How? [Mode of failure] Where? When? Why? [Cause of failure] 54

Two Golden Rules for Failure Determination Failures do not always exhibit a single classic mechanism Careful coordination required between laboratory examination, field inspection, and operating records 55