Analogs as a Focus Bruce W Bode MD

Analogs as a Focus Bruce W. Bode, MD, FACE Atlanta Diabetes Associates Atlanta, Georgia

Goals of Intensive Diabetes Management • Near-normal glycemia — Hb. A 1 c <6. 5% to 7. 0% • Avoid short-term crisis — Hypoglycemia — Hyperglycemia — DKA • Minimize long-term complications • Improve QOL ADA. Clinical Practice Recommendations. 2001.

ACE/AACE Targets for Glycemic Control Hb. A 1 c <6. 5% Fasting/preprandial glucose <110 mg/d. L Postprandial glucose <140 mg/d. L ACE/AACE Consensus Conference. Washington, DC. August 2001.

Insulin The Most Powerful Agent We Have to Control Glucose

The Discovery of Insulin (Toronto 1921) Fred Banting (1891 -1941) James B. Collip (1892 -1965) Charles H. Best (1899 -1978) John J. R. Mc. Leod (1876 -1935) Marjorie (? -? )
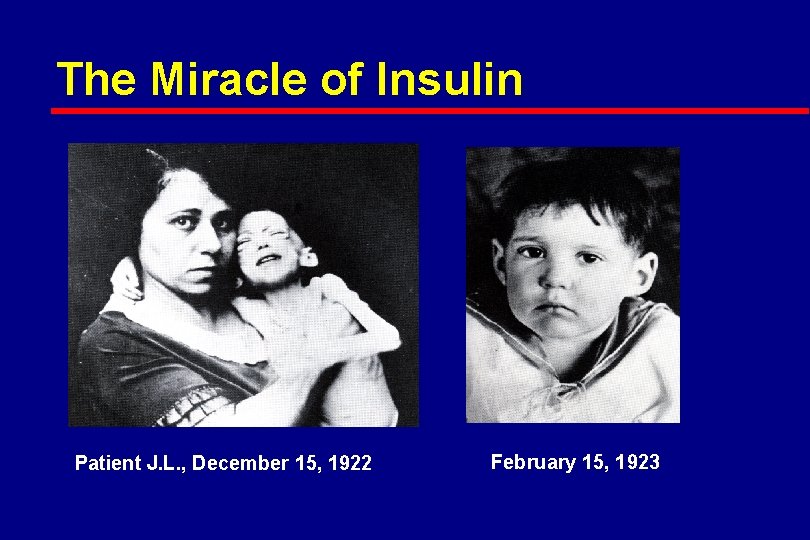
The Miracle of Insulin Patient J. L. , December 15, 1922 February 15, 1923
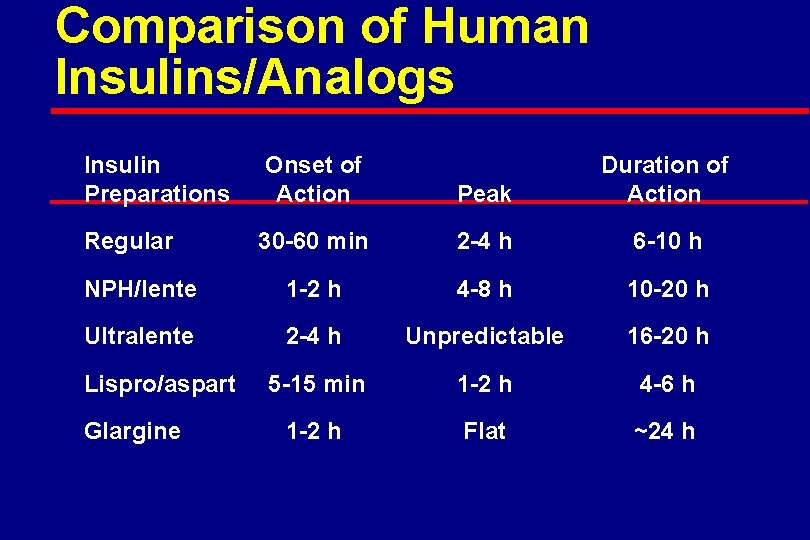
Comparison of Human Insulins/Analogs Insulin Preparations Onset of Action Peak Duration of Action Regular 30 -60 min 2 -4 h 6 -10 h NPH/lente 1 -2 h 4 -8 h 10 -20 h Ultralente 2 -4 h Unpredictable 16 -20 h 5 -15 min 1 -2 h 4 -6 h 1 -2 h Flat ~24 h Lispro/aspart Glargine
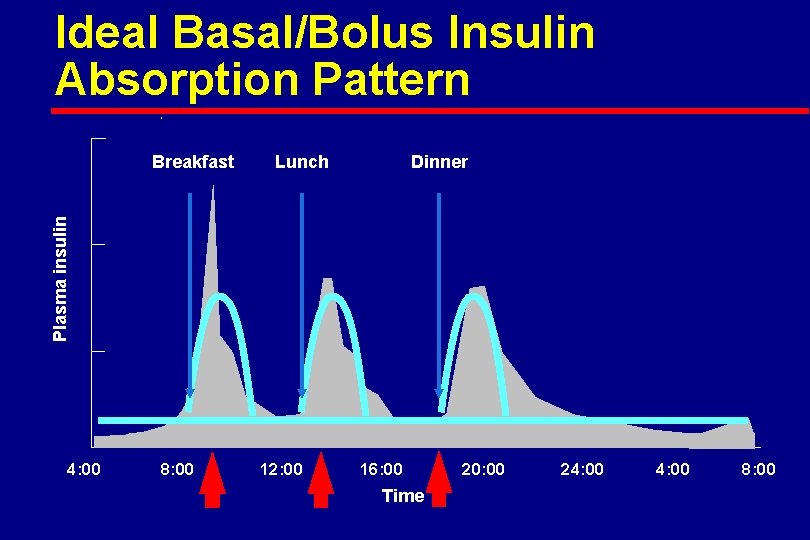
Ideal Basal/Bolus Insulin Absorption Pattern Lunch Dinner Plasma insulin Breakfast 4: 00 8: 00 12: 00 16: 00 Time 20: 00 24: 00 8: 00

Rapid-acting Insulin Analogs: Medical Rationale • Administration at mealtime • Mimic physiologic insulin profile • Improved postprandial glycemic control • Lower risk of late hypoglycemia
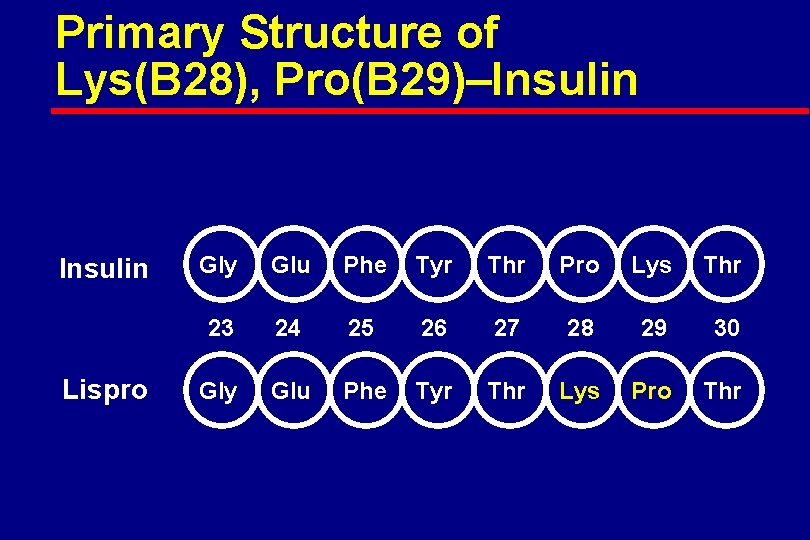
Primary Structure of Lys(B 28), Pro(B 29)–Insulin Lispro Gly Glu Phe Tyr Thr Pro Lys Thr 23 24 25 26 27 28 29 30 Gly Glu Phe Tyr Thr Lys Pro Thr
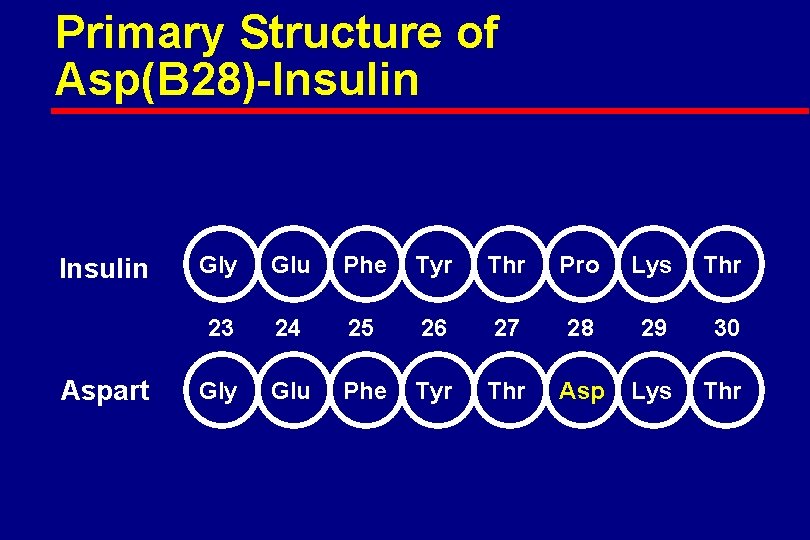
Primary Structure of Asp(B 28)-Insulin Aspart Gly Glu Phe Tyr Thr Pro Lys Thr 23 24 25 26 27 28 29 30 Gly Glu Phe Tyr Thr Asp Lys Thr
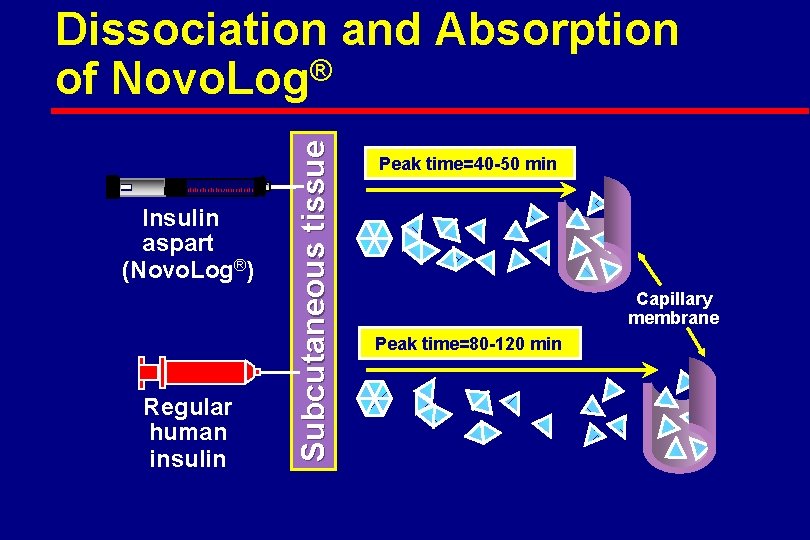
Insulin aspart (Novo. Log®) Regular human insulin Subcutaneous tissue Dissociation and Absorption of Novo. Log® Peak time=40 -50 min Capillary membrane Peak time=80 -120 min
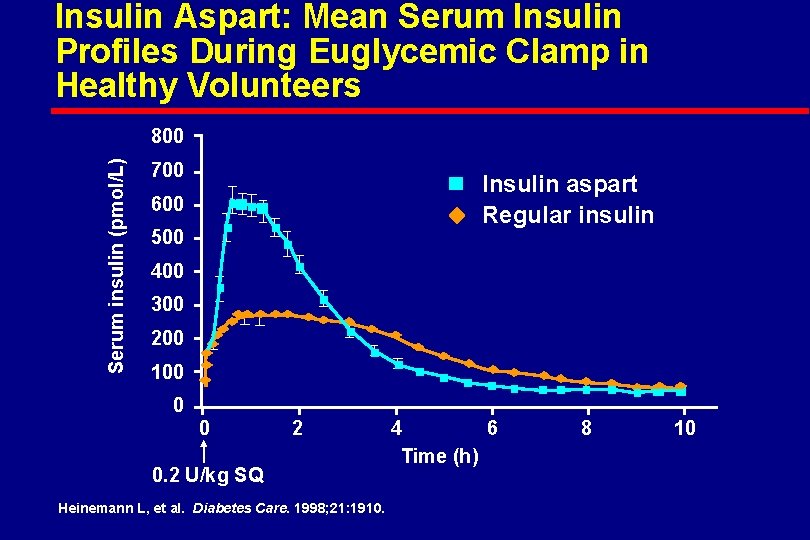
Insulin Aspart: Mean Serum Insulin Profiles During Euglycemic Clamp in Healthy Volunteers Serum insulin (pmol/L) 800 700 Insulin aspart Regular insulin 600 500 400 300 200 100 0 0 2 0. 2 U/kg SQ Heinemann L, et al. Diabetes Care. 1998; 21: 1910. 4 6 Time (h) 8 10

Glucose Area Under the Curve None Regular Aspart
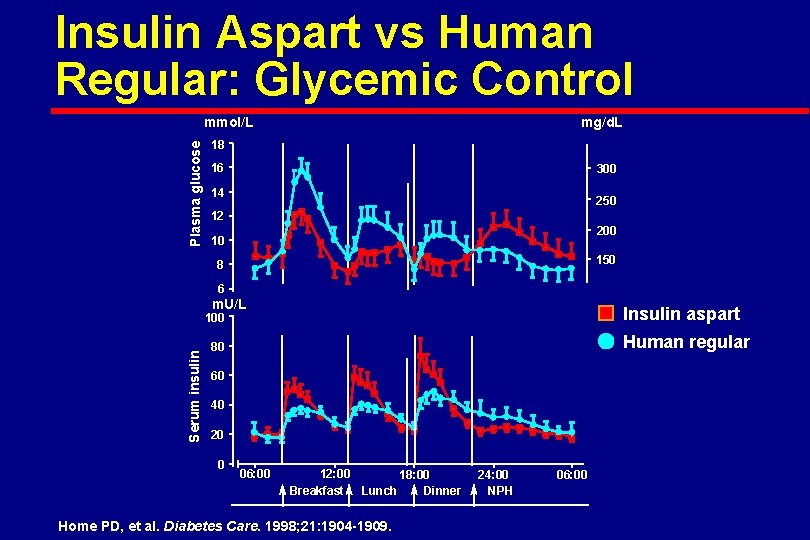
Insulin Aspart vs Human Regular: Glycemic Control Plasma glucose mmol/L mg/d. L 18 16 300 14 250 12 200 10 150 8 6 m. U/L Insulin aspart Serum insulin 100 Human regular 80 60 40 20 0 06: 00 12: 00 18: 00 Breakfast Lunch Dinner Home PD, et al. Diabetes Care. 1998; 21: 1904 -1909. 24: 00 NPH 06: 00
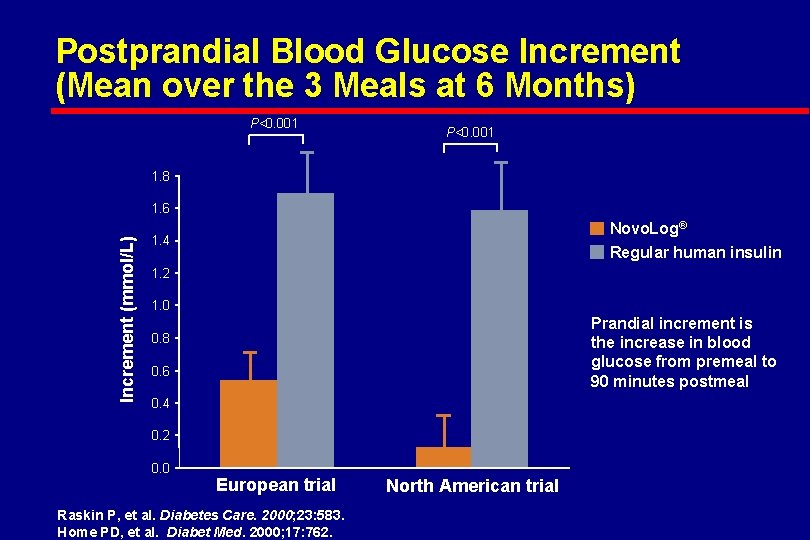
Postprandial Blood Glucose Increment (Mean over the 3 Meals at 6 Months) P<0. 001 1. 8 Increment (mmol/L) 1. 6 Novo. Log® Regular human insulin 1. 4 1. 2 1. 0 Prandial increment is the increase in blood glucose from premeal to 90 minutes postmeal 0. 8 0. 6 0. 4 0. 2 0. 0 European trial Raskin P, et al. Diabetes Care. 2000; 23: 583. Home PD, et al. Diabet Med. 2000; 17: 762. North American trial
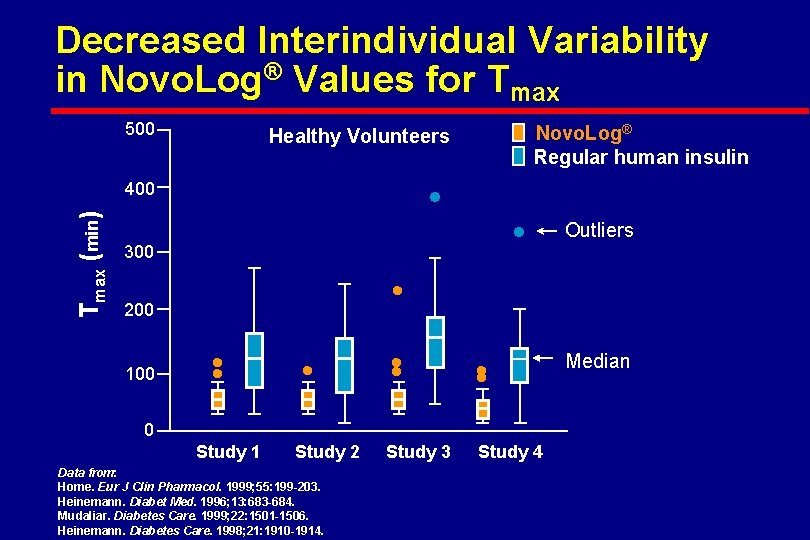
Decreased Interindividual Variability in Novo. Log® Values for Tmax 500 · 400 Tmax (min) Novo. Log® Regular human insulin Healthy Volunteers · 300 · 200 100 Outliers ·· ·· Study 3 Study 4 0 Study 1 Study 2 Data from: Home. Eur J Clin Pharmacol. 1999; 55: 199 -203. Heinemann. Diabet Med. 1996; 13: 683 -684. Mudaliar. Diabetes Care. 1999; 22: 1501 -1506. Heinemann. Diabetes Care. 1998; 21: 1910 -1914. Median
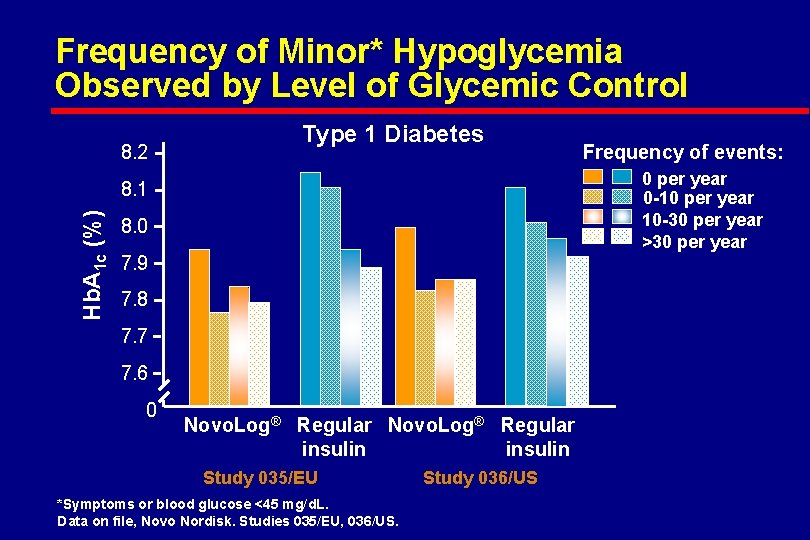
Frequency of Minor* Hypoglycemia Observed by Level of Glycemic Control 8. 2 Type 1 Diabetes 0 per year 0 -10 per year 10 -30 per year >30 per year Hb. A 1 c (%) 8. 1 8. 0 7. 9 7. 8 7. 7 7. 6 0 Frequency of events: Novo. Log® Regular insulin Study 035/EU *Symptoms or blood glucose <45 mg/d. L. Data on file, Novo Nordisk. Studies 035/EU, 036/US. Study 036/US
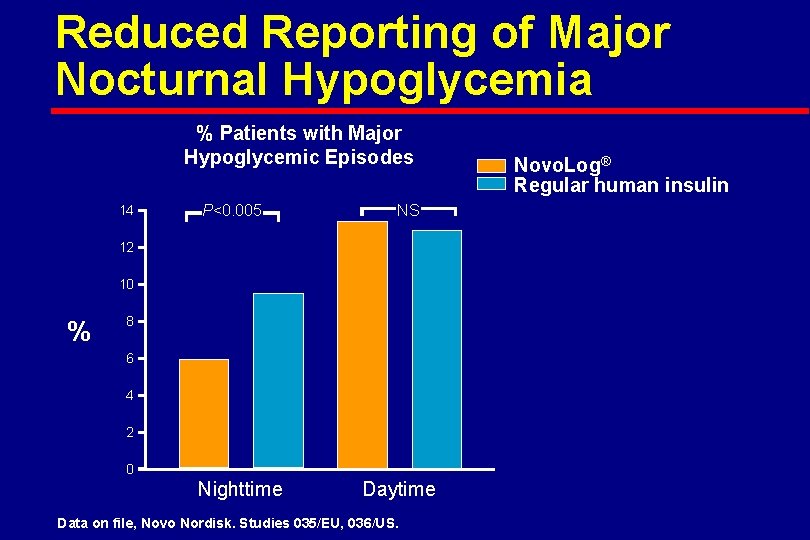
Reduced Reporting of Major Nocturnal Hypoglycemia % Patients with Major Hypoglycemic Episodes 14 P<0. 005 NS 12 10 % 8 6 4 2 0 Nighttime Daytime Data on file, Novo Nordisk. Studies 035/EU, 036/US. Novo. Log® Regular human insulin
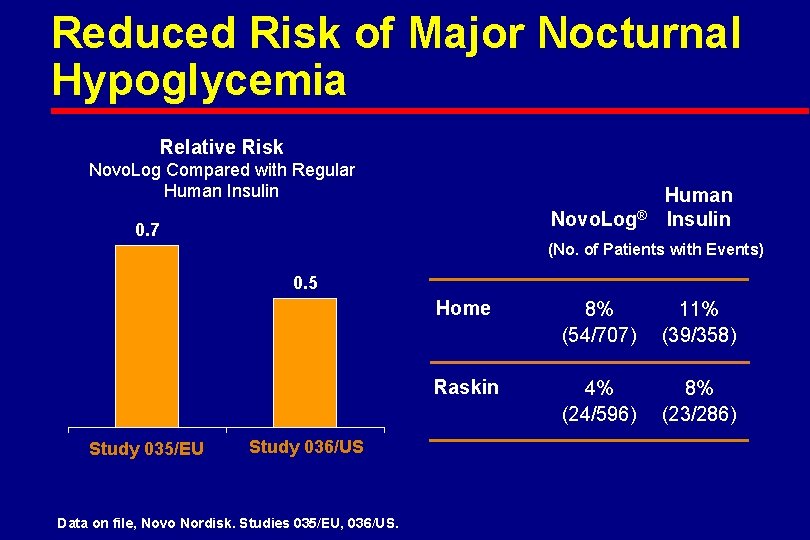
Reduced Risk of Major Nocturnal Hypoglycemia Relative Risk Novo. Log Compared with Regular Human Insulin Novo. Log® 0. 7 Human Insulin (No. of Patients with Events) 0. 5 Study 035/EU Study 036/US Data on file, Novo Nordisk. Studies 035/EU, 036/US. Home 8% (54/707) 11% (39/358) Raskin 4% (24/596) 8% (23/286)
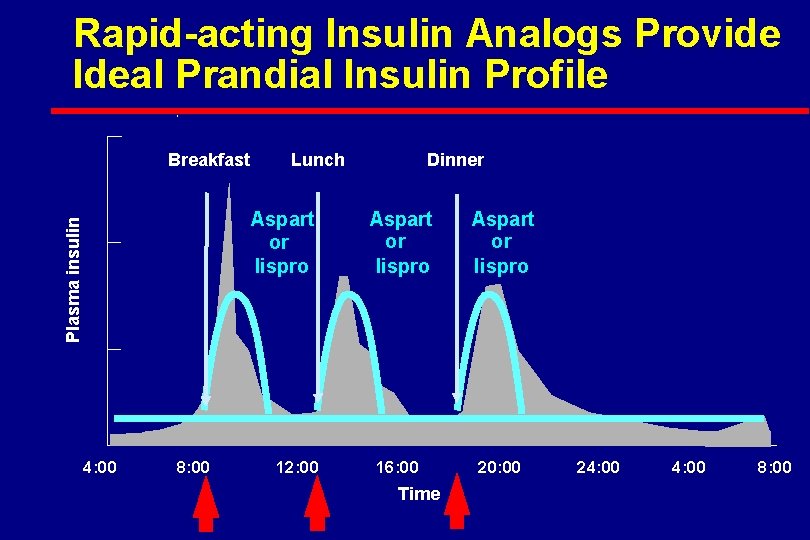
Rapid-acting Insulin Analogs Provide Ideal Prandial Insulin Profile Breakfast Lunch Plasma insulin Aspart or lispro 4: 00 8: 00 12: 00 Dinner Aspart or lispro 16: 00 20: 00 Time 24: 00 8: 00
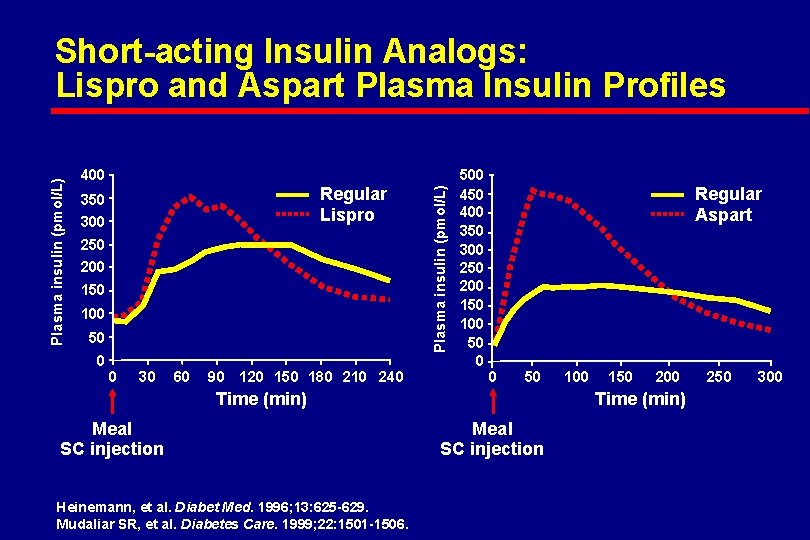
400 Regular Lispro 350 300 250 200 150 100 50 0 0 30 60 90 120 150 180 210 240 Plasma insulin (pmol/L) Short-acting Insulin Analogs: Lispro and Aspart Plasma Insulin Profiles 500 450 400 350 300 250 200 150 100 50 0 Regular Aspart 0 50 Time (min) Meal SC injection Heinemann, et al. Diabet Med. 1996; 13: 625 -629. Mudaliar SR, et al. Diabetes Care. 1999; 22: 1501 -1506. 100 150 200 Time (min) Meal SC injection 250 300
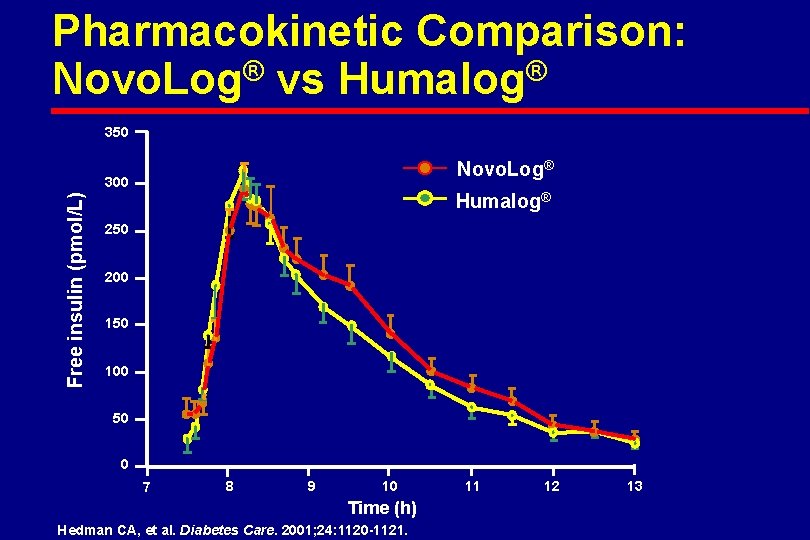
Pharmacokinetic Comparison: Novo. Log® vs Humalog® 350 Novo. Log® Free insulin (pmol/L) 300 Humalog® 250 200 150 100 50 0 7 8 9 10 Time (h) Hedman CA, et al. Diabetes Care. 2001; 24: 1120 -1121. 11 12 13
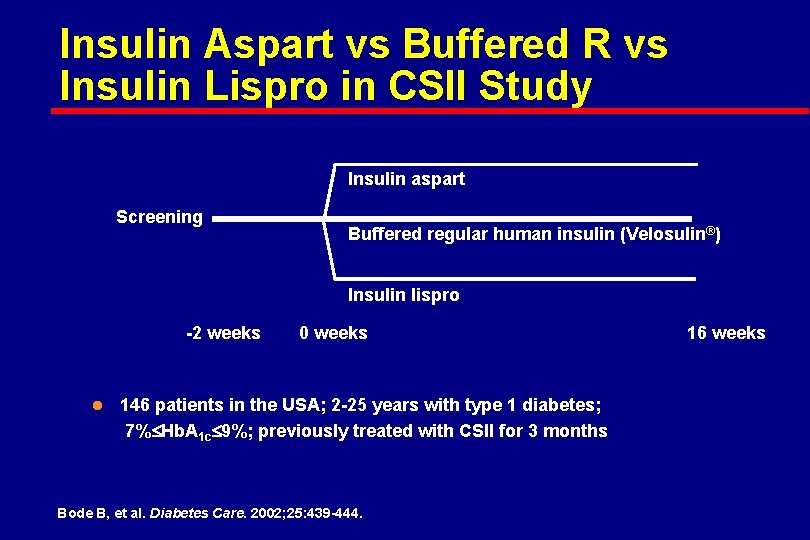
Insulin Aspart vs Buffered R vs Insulin Lispro in CSII Study Insulin aspart Screening Buffered regular human insulin (Velosulin®) Insulin lispro -2 weeks l 0 weeks 146 patients in the USA; 2 -25 years with type 1 diabetes; 7% Hb. A 1 c 9%; previously treated with CSII for 3 months Bode B, et al. Diabetes Care. 2002; 25: 439 -444. 16 weeks
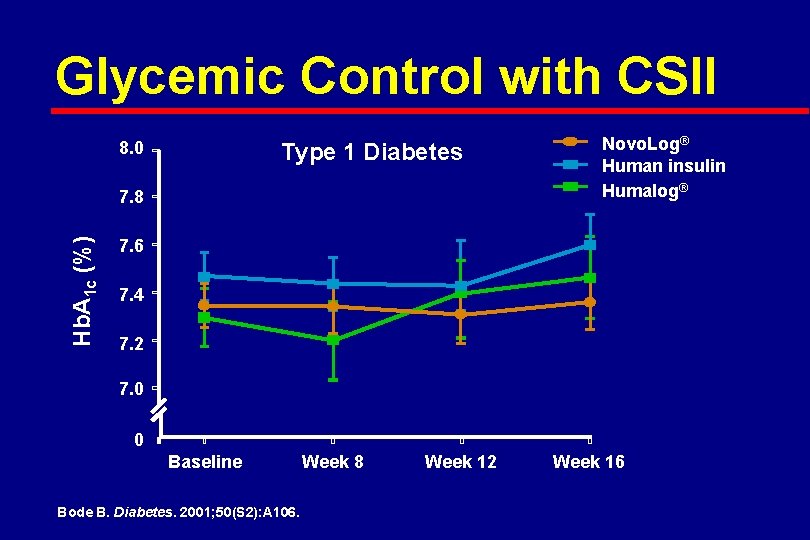
Glycemic Control with CSII 8. 0 Type 1 Diabetes Hb. A 1 c (%) 7. 8 Novo. Log® Human insulin Humalog® 7. 6 7. 4 7. 2 7. 0 0 Baseline Bode B. Diabetes. 2001; 50(S 2): A 106. Week 8 Week 12 Week 16
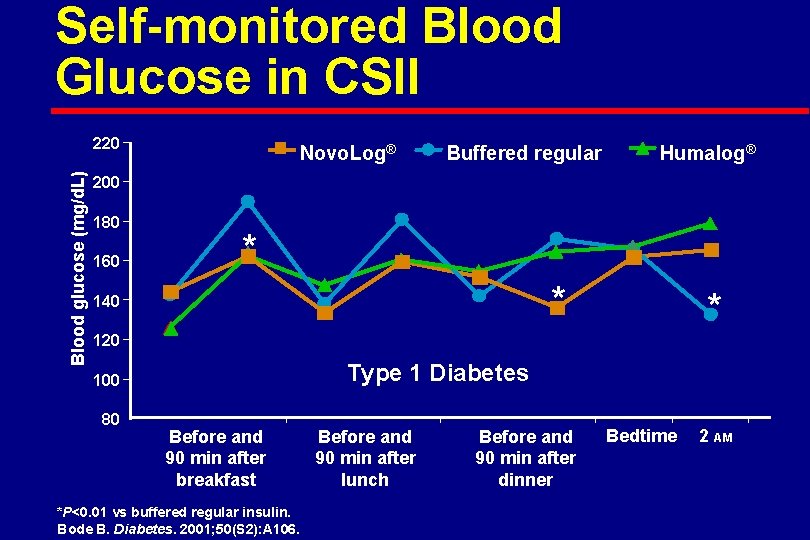
Self-monitored Blood Glucose in CSII Blood glucose (mg/d. L) 220 Novo. Log® Buffered regular Humalog® 200 180 160 * * 140 * 120 Type 1 Diabetes 100 80 Before and 90 min after breakfast *P<0. 01 vs buffered regular insulin. Bode B. Diabetes. 2001; 50(S 2): A 106. Before and 90 min after lunch Before and 90 min after dinner Bedtime 2 AM
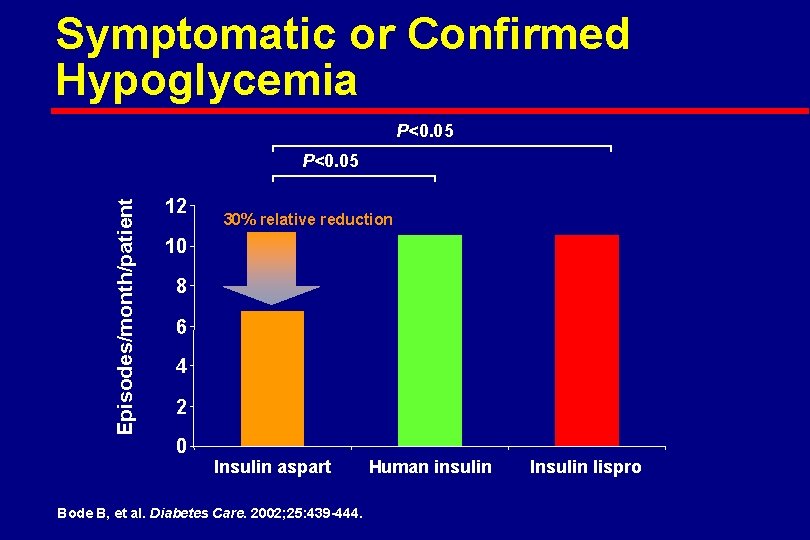
Symptomatic or Confirmed Hypoglycemia P<0. 05 Episodes/month/patient P<0. 05 12 30% relative reduction 10 8 6 4 2 0 Insulin aspart Bode B, et al. Diabetes Care. 2002; 25: 439 -444. Human insulin Insulin lispro
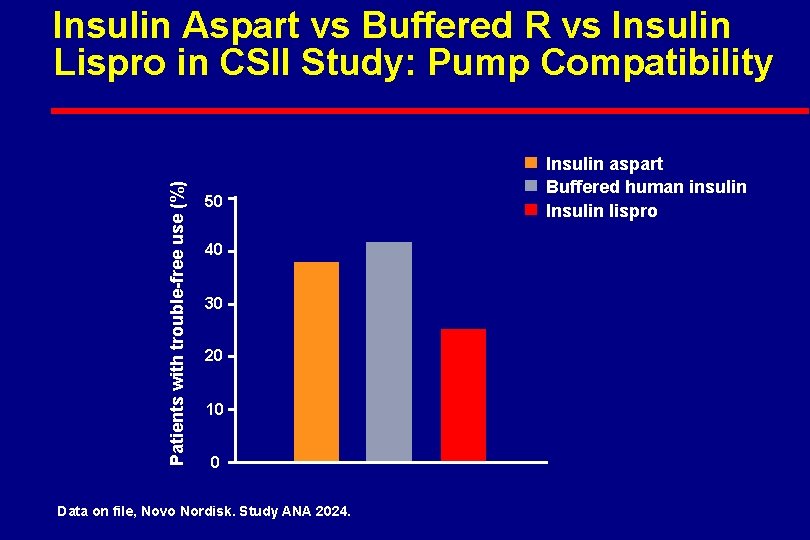
Patients with trouble-free use (%) Insulin Aspart vs Buffered R vs Insulin Lispro in CSII Study: Pump Compatibility 50 40 30 20 10 0 Data on file, Novo Nordisk. Study ANA 2024. Insulin aspart Buffered human insulin Insulin lispro

Long-acting Soluble Insulin Analogs: Medical Rationale • Mimic basal physiological insulin profile • Improved glycemic control • More reproducible insulin delivery • May be used in insulin pens

Limitations of NPH, Lente, and Ultralente • Do not mimic basal insulin profile — Variable absorption — Pronounced peaks — Less than 24 -hour duration of action • Cause unpredictable hypoglycemia — Major factor limiting insulin adjustments
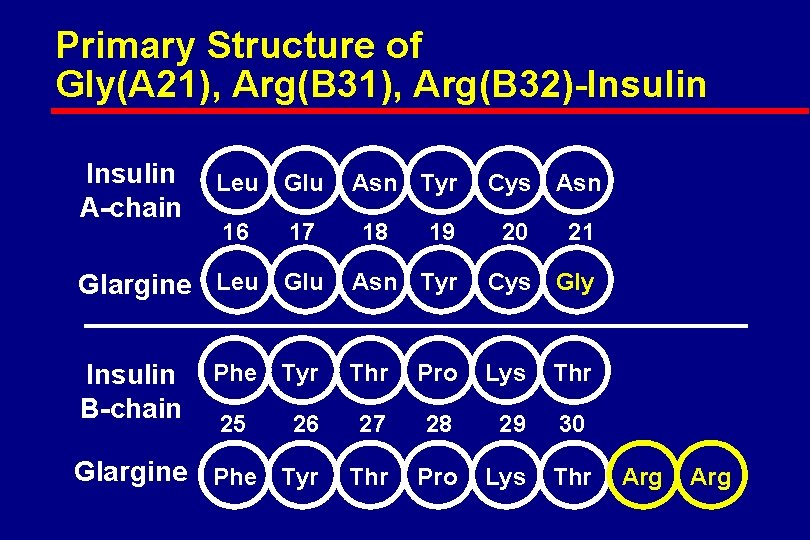
Primary Structure of Gly(A 21), Arg(B 32)-Insulin A-chain Leu Glu 16 17 Asn Tyr Cys Asn 19 20 21 Glargine Leu Glu Asn Tyr Cys Gly Insulin B-chain 18 Phe Tyr Thr Pro Lys Thr 25 26 27 28 29 30 Glargine Phe Tyr Thr Pro Lys Thr Arg
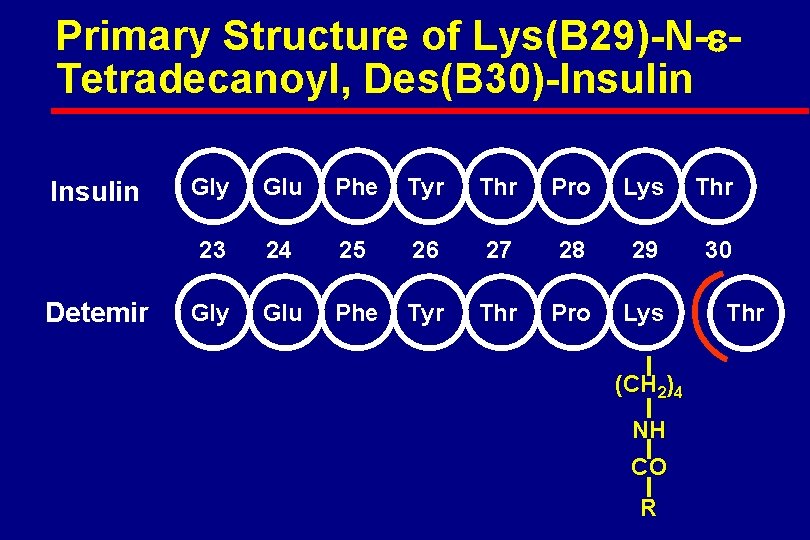
Primary Structure of Lys(B 29)-N- Tetradecanoyl, Des(B 30)-Insulin Detemir Gly Glu Phe Tyr Thr Pro Lys Thr 23 24 25 26 27 28 29 30 Gly Glu Phe Tyr Thr Pro Lys (CH 2)4 NH CO R Thr

Basis of Effect of Insulin Glargine • Isoelectric point change • Precipitates at neutral tissue p. H — Acid in solution; cannot be mixed with other insulins • Retarded absorption rate • Corresponding longer duration of action
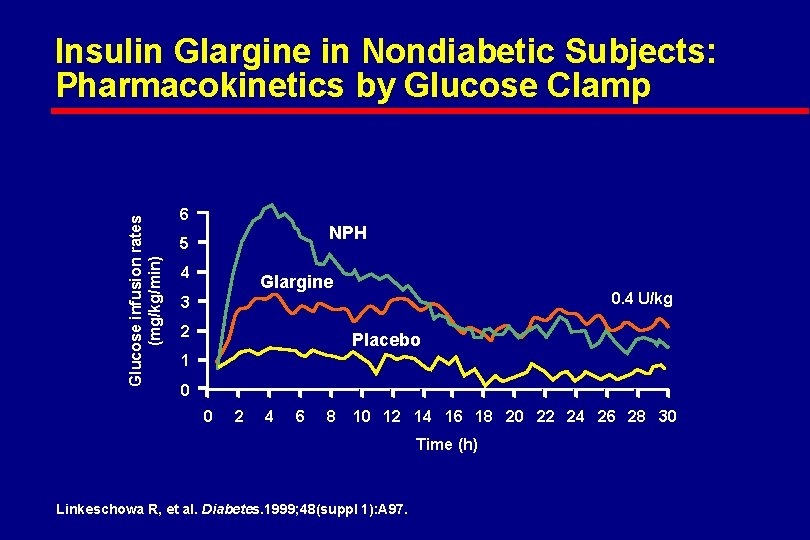
Glucose infusion rates (mg/kg/min) Insulin Glargine in Nondiabetic Subjects: Pharmacokinetics by Glucose Clamp 6 NPH 5 4 Glargine 0. 4 U/kg 3 2 Placebo 1 0 0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30 Time (h) Linkeschowa R, et al. Diabetes. 1999; 48(suppl 1): A 97.

Overall Summary: Glargine • Insulin glargine has the following clinical benefits: — Once-daily dosing because of its prolonged duration of action and smooth, peakless timeaction profile — Comparable or better glycemic control (FBG) — Lower risk of nocturnal hypoglycemic events — Safety profile similar to that of human insulin

Basis of Effect of Acylated Insulin Analogs (Detemir) • Bind to serum albumin • Prolonged time in circulation • Longer duration of action
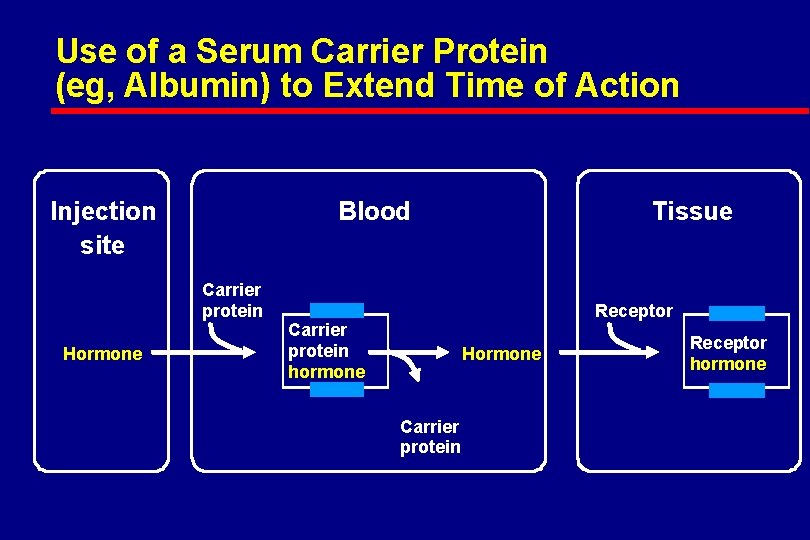
Use of a Serum Carrier Protein (eg, Albumin) to Extend Time of Action Injection site Blood Carrier protein Hormone Tissue Receptor Carrier protein hormone Hormone Carrier protein Receptor hormone
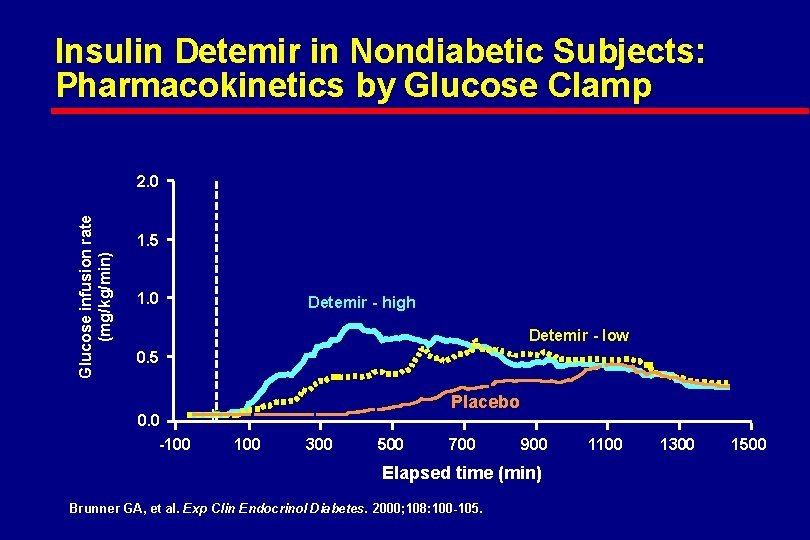
Insulin Detemir in Nondiabetic Subjects: Pharmacokinetics by Glucose Clamp Glucose infusion rate (mg/kg/min) 2. 0 1. 5 1. 0 Detemir - high Detemir - low 0. 5 0. 0 -100 Placebo 100 300 500 700 900 Elapsed time (min) Brunner GA, et al. Exp Clin Endocrinol Diabetes. 2000; 108: 100 -105. 1100 1300 1500
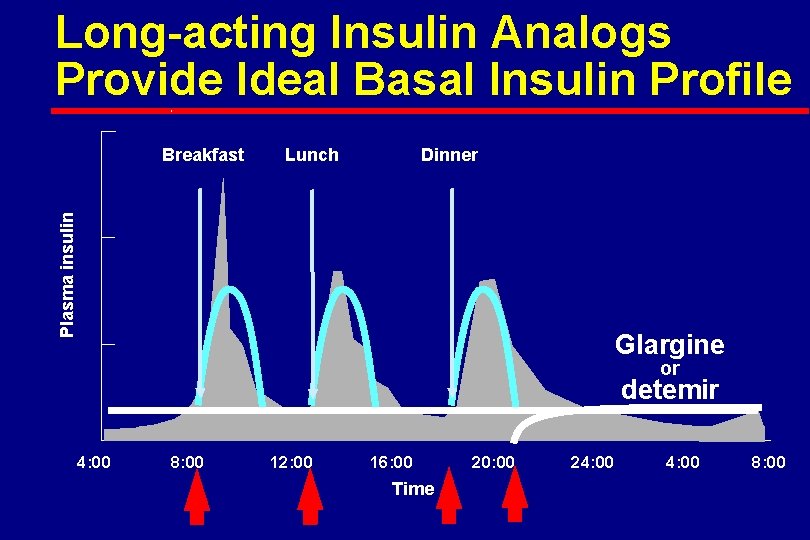
Long-acting Insulin Analogs Provide Ideal Basal Insulin Profile Lunch Dinner Plasma insulin Breakfast Glargine or detemir 4: 00 8: 00 12: 00 16: 00 Time 20: 00 24: 00 8: 00
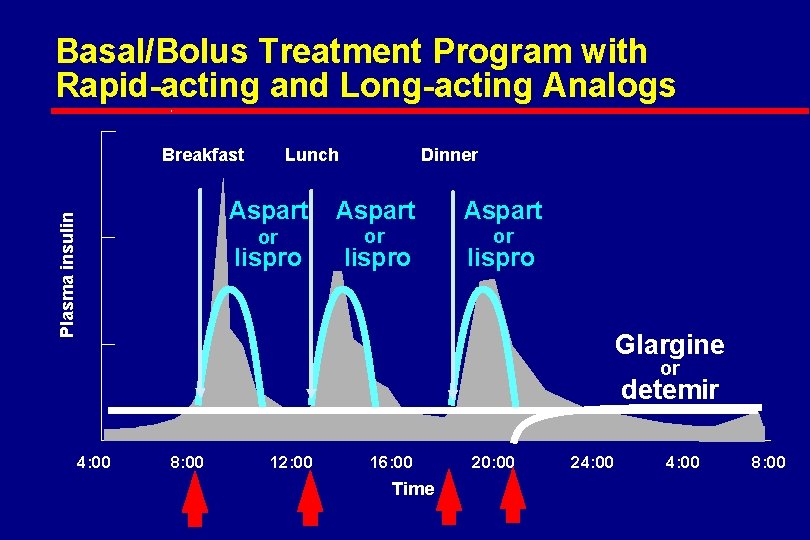
Basal/Bolus Treatment Program with Rapid-acting and Long-acting Analogs Plasma insulin Breakfast Lunch Dinner Aspart or or or lispro Glargine or detemir 4: 00 8: 00 12: 00 16: 00 Time 20: 00 24: 00 8: 00

Receptor Binding Affinities Insulin Receptor Affinity (%) Cells Solubilized Receptors Relative to human insulin IGF-1 Receptor Affinity (%) Cells Solubilized Receptors Relative to human insulin Human insulin 100 100 Insulin aspart 92 92 69 81 Insulin lispro 102 ND 142 156 Insulin glargine ND 86 ND 641 ND = not determined.

Insulin Analogs Fulfilling the Promise of Recombinant DNA Technology: Better Basal Better Bolus Better Blood Glucose
- Slides: 42