Anaemia treatment in CKD ESRD and kidney transplant
































- Slides: 67

Anaemia treatment in CKD, ESRD, and kidney transplant recipients Iain C Macdougall BSc, MD, FRCP Consultant Nephrologist and Honorary Senior Lecturer Renal Unit, King’s College Hospital, London, UK

Outline of presentation l Erythropoiesis in 2009 l ESA therapy l Target Hb l Iron management l Anaemia management in kidney transplantation l The future

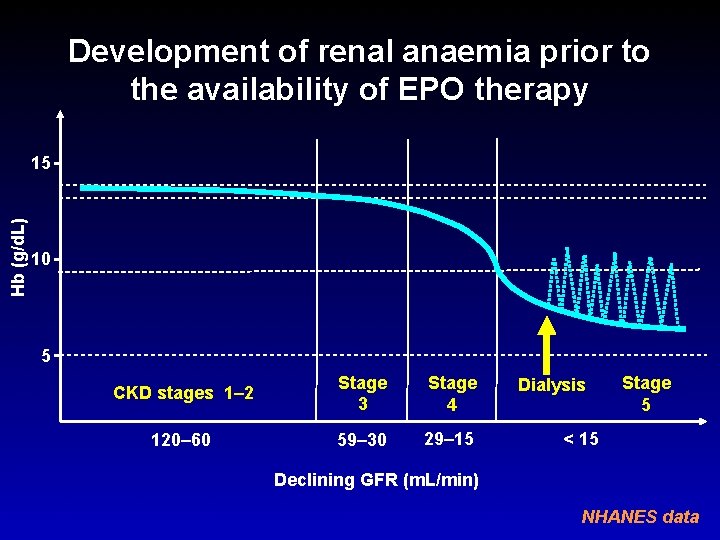
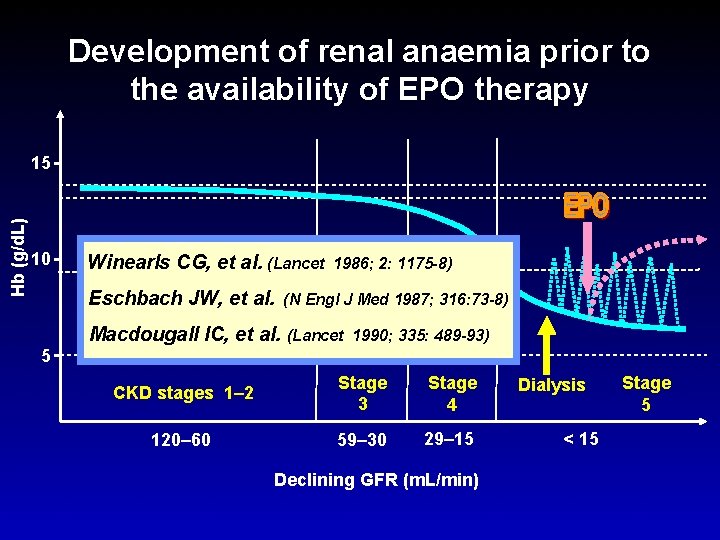
Development of renal anaemia prior to the availability of EPO therapy Hb (g/d. L) 15 10 5 CKD stages 1– 2 Stage 3 Stage 4 120– 60 59– 30 29– 15 Dialysis Stage 5 < 15 Declining GFR (m. L/min) NHANES data

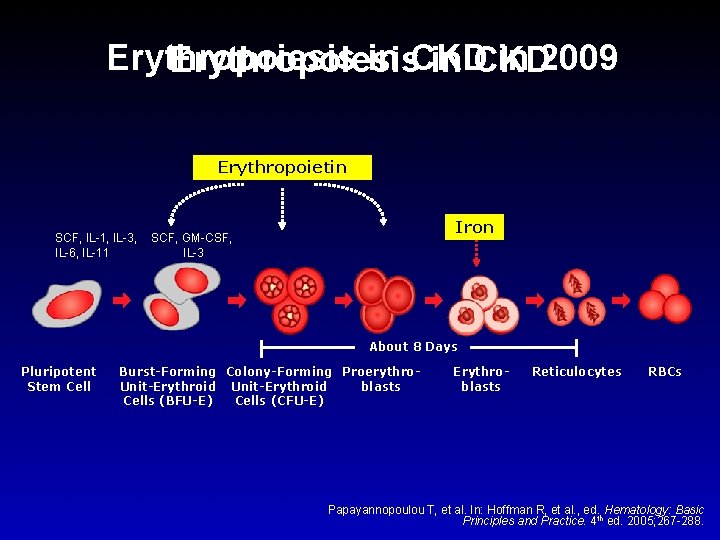
Erythropoiesis in CKD in 2009 Erythropoiesis in CKD Erythropoietin SCF, IL-1, IL-3, IL-6, IL-11 Iron SCF, GM-CSF, IL-3 About 8 Days Pluripotent Stem Cell Burst-Forming Colony-Forming Proerythro. Unit-Erythroid blasts Cells (BFU-E) Cells (CFU-E) Erythroblasts Reticulocytes RBCs Papayannopoulou T, et al. In: Hoffman R, et al. , ed. Hematology: Basic Principles and Practice. 4 th ed. 2005; 267 -288.

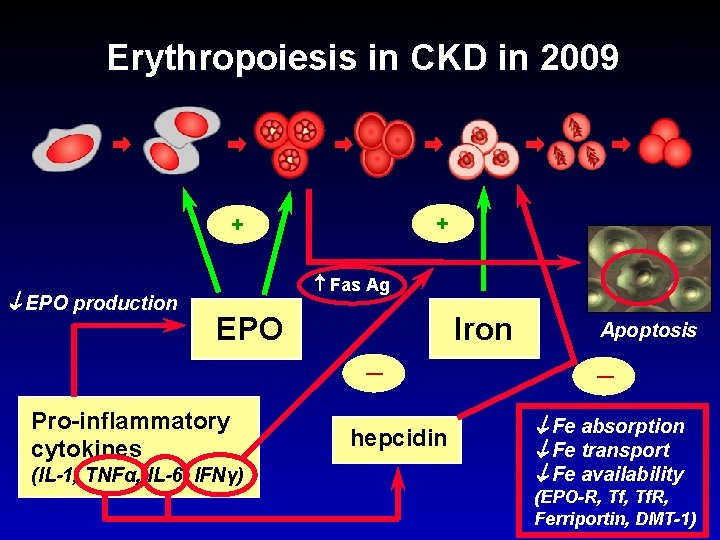
Erythropoiesis in CKD in 2009 + + EPO production Fas Ag Iron EPO ─ Pro-inflammatory cytokines (IL-1, TNFα, IL-6, IFNγ) hepcidin Apoptosis ─ Fe absorption Fe transport Fe availability (EPO-R, Tf. R, Ferriportin, DMT-1)

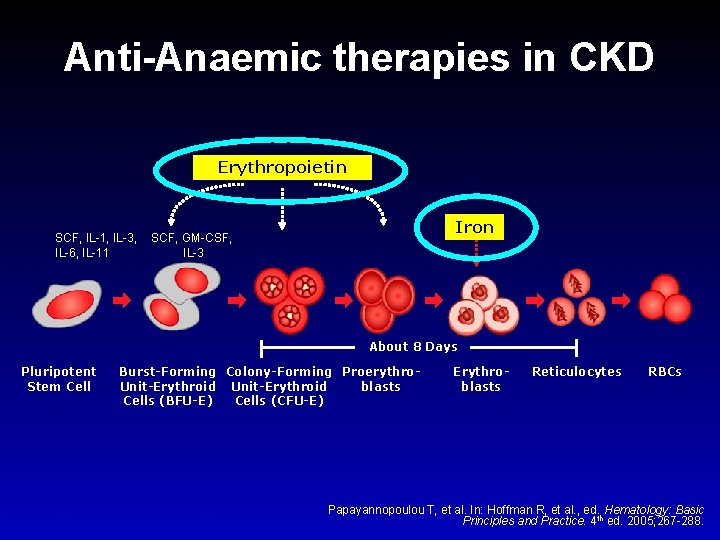
Anti-Anaemic therapies in CKD Erythropoietin SCF, IL-1, IL-3, IL-6, IL-11 Iron SCF, GM-CSF, IL-3 About 8 Days Pluripotent Stem Cell Burst-Forming Colony-Forming Proerythro. Unit-Erythroid blasts Cells (BFU-E) Cells (CFU-E) Erythroblasts Reticulocytes RBCs Papayannopoulou T, et al. In: Hoffman R, et al. , ed. Hematology: Basic Principles and Practice. 4 th ed. 2005; 267 -288.

Outline of presentation l Erythropoiesis in 2009 l ESA therapy l Target Hb l Iron management l Anaemia management in kidney transplantation l The future

Development of recombinant human EPO 1977 - human EPO isolated from 2, 500 litres of urine (Miyake et al) 1983 - gene for human EPO isolated and cloned (FK Lin et al) 1986 - first clinical report in dialysis patients 1990 - r-Hu. EPO licensed for use in Europe

Epoetin alfa (Eprex) Epoetin beta (Neo. Recormon)

Development of renal anaemia prior to the availability of EPO therapy Hb (g/d. L) 15 10 Winearls CG, et al. (Lancet Eschbach JW, et al. 1986; 2: 1175 -8) (N Engl J Med 1987; 316: 73 -8) Macdougall IC, et al. (Lancet 1990; 335: 489 -93) 5 CKD stages 1– 2 Stage 3 Stage 4 120– 60 59– 30 29– 15 Declining GFR (m. L/min) Dialysis < 15 Stage 5

Macdougall et al. , Lancet 1990; 335: 489 -493.

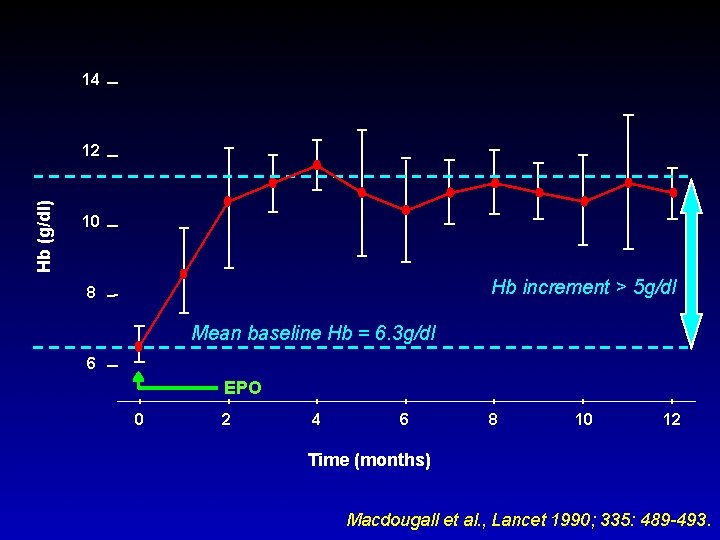
14 Hb (g/dl) 12 10 Hb increment > 5 g/dl 8 Mean baseline Hb = 6. 3 g/dl 6 EPO 0 2 4 6 8 10 12 Time (months) Macdougall et al. , Lancet 1990; 335: 489 -493.

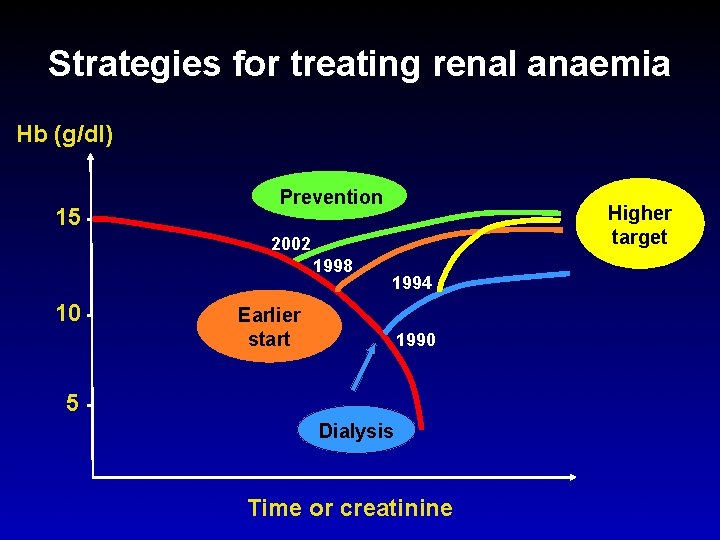
Strategies for treating renal anaemia Hb (g/dl) 15 Prevention Higher target 2002 1998 10 1994 Earlier start 1990 5 Dialysis Time or creatinine

Anaemia therapy in CKD l Initially, Epoetin alfa (Eprex, Erypo) – 1990 Epoetin beta (Neo. Recormon) – 1990 Epoetin alfa Epoetin beta

Anaemia therapy in CKD l Initially, Epoetin alfa (Eprex, Erypo) – 1990 Epoetin beta (Neo. Recormon) – 1990 l 2 nd generation ESA: Darbepoetin alfa (Aranesp) – 2001 Epoetin alfa Epoetin beta

Darbepoetin alfa: a molecule with two more N-linked glycosylation chains than r-Hu. EPO First extra N-linked chain Second extra N-linked chain

Third-generation ESAs C. E. R. A. (MIRCERA) Methoxy polyethylene glycol epoetin beta – licensed 2007

C. E. R. A. l Continuous Erythropoietin Receptor Activator CERA l PEGylated Epoetin beta EPO

Epoetin delta (DYNEPOTM)

Biosimilar EPOs l First biosimilar epoetins licensed in Europe – Binocrit. TM (Sandoz) – Retacrit. TM (Hospira)

Current licensed ESAs in Europe l Epoetin alfa (Eprex) l Epoetin beta (Neo. Recormon) l Darbepoetin alfa (Aranesp) l C. E. R. A. (MIRCERA) l Epoetin delta (Dynepo) l Biosimilar Epoetin alfa (Binocrit) l Biosimilar Epoetin zeta (Retacrit)

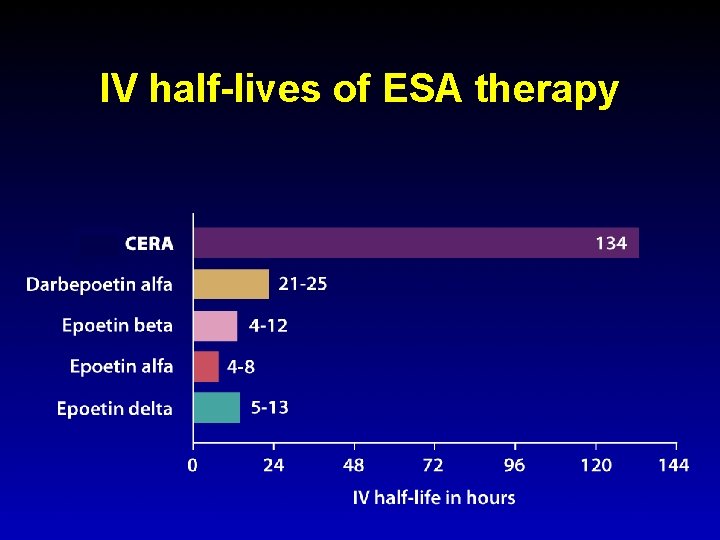
IV half-lives of ESA therapy

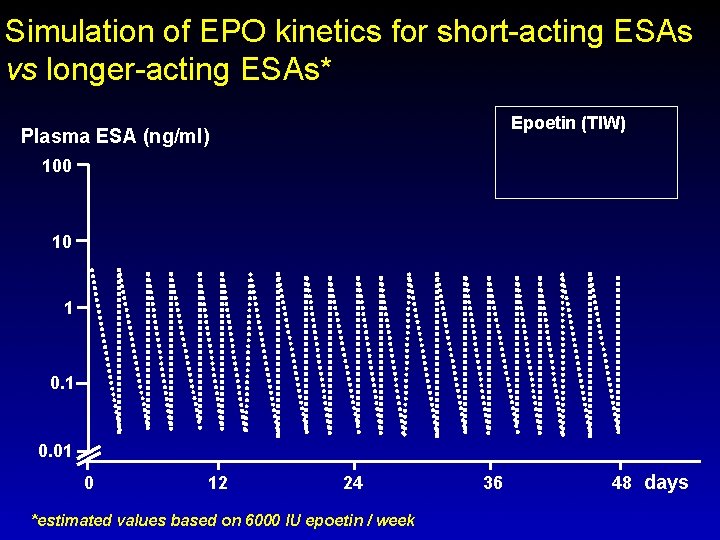
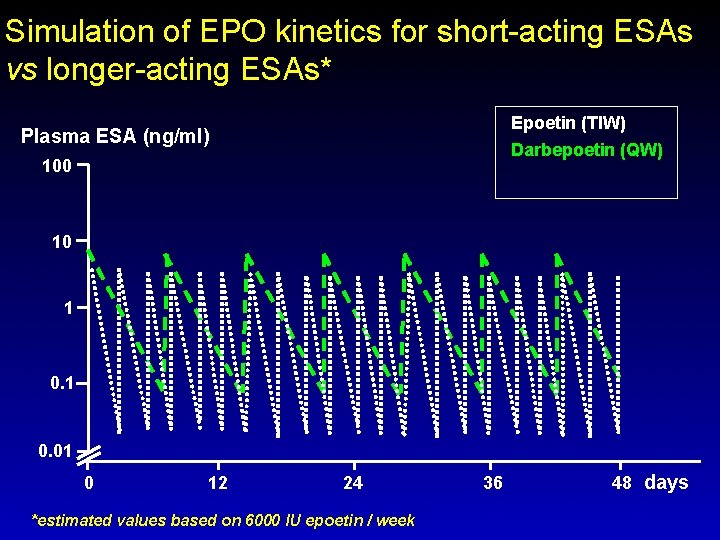
Simulation of EPO kinetics for short-acting ESAs vs longer-acting ESAs* Plasma ESA (ng/ml) Epoetin (TIW) 100 1 0. 01 0 12 24 36 48 days *estimated values based on 6000 IU epoetin / week

Simulation of EPO kinetics for short-acting ESAs vs longer-acting ESAs* Plasma ESA (ng/ml) 100 Epoetin (TIW) Darbepoetin (QW) 10 1 0. 01 0 12 24 36 48 days *estimated values based on 6000 IU epoetin / week

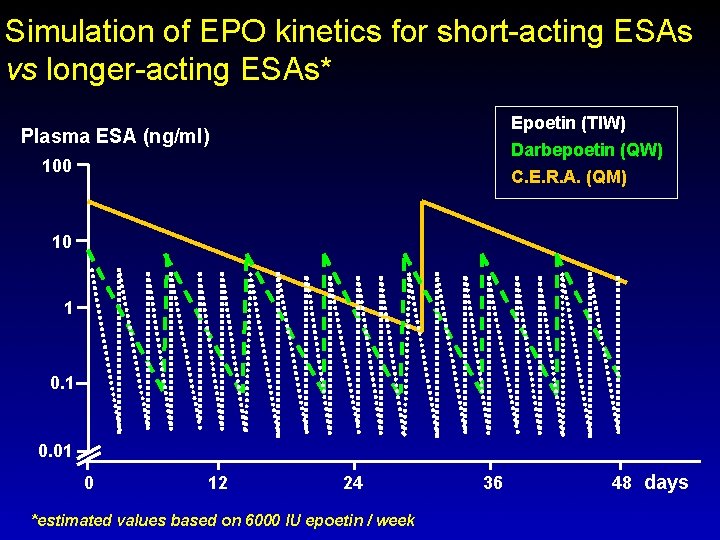
Simulation of EPO kinetics for short-acting ESAs vs longer-acting ESAs* Plasma ESA (ng/ml) 100 Epoetin (TIW) Darbepoetin (QW) C. E. R. A. (QM) 10 1 0. 01 0 12 24 36 48 days *estimated values based on 6000 IU epoetin / week

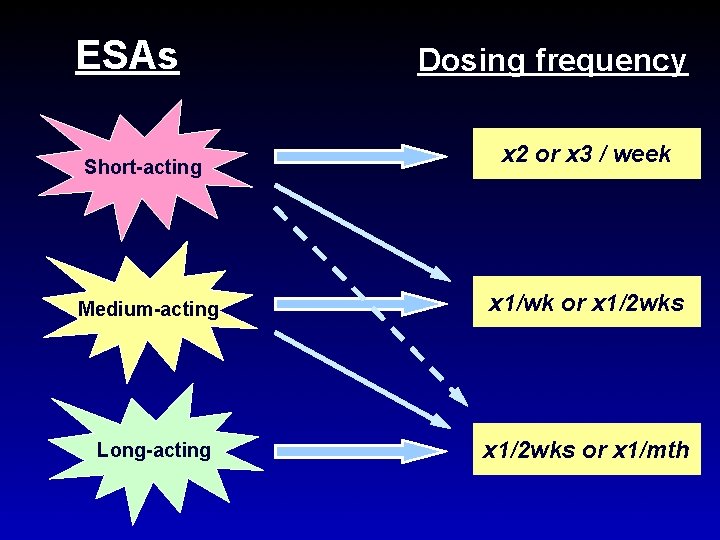
ESAs Short-acting Dosing frequency x 2 or x 3 / week Medium-acting x 1/wk or x 1/2 wks Long-acting x 1/2 wks or x 1/mth

Outline of presentation l Erythropoiesis in 2009 l ESA therapy l Target Hb l Iron management l Anaemia management in kidney transplantation l The future

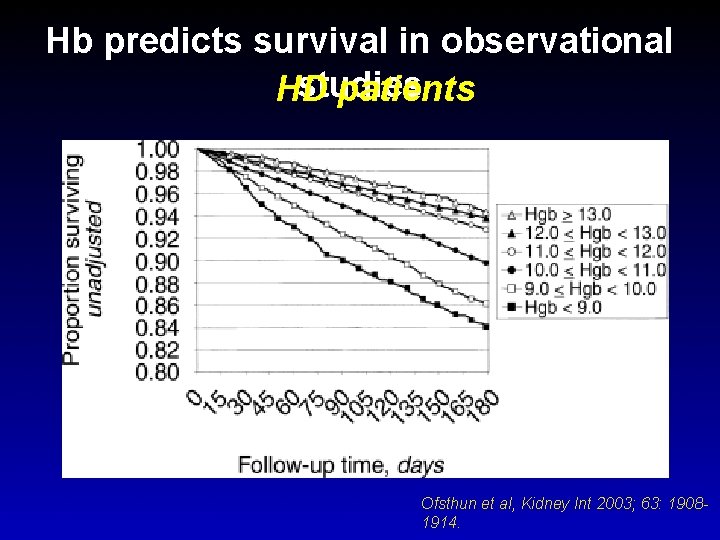
Hb predicts survival in observational studies HD patients Ofsthun et al, Kidney Int 2003; 63: 19081914.

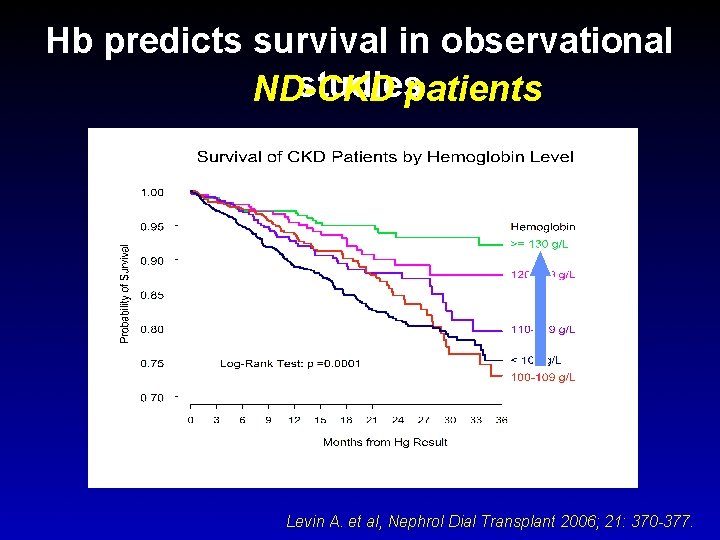
Hb predicts survival in observational studies ND-CKD patients Levin A. et al, Nephrol Dial Transplant 2006; 21: 370 -377.

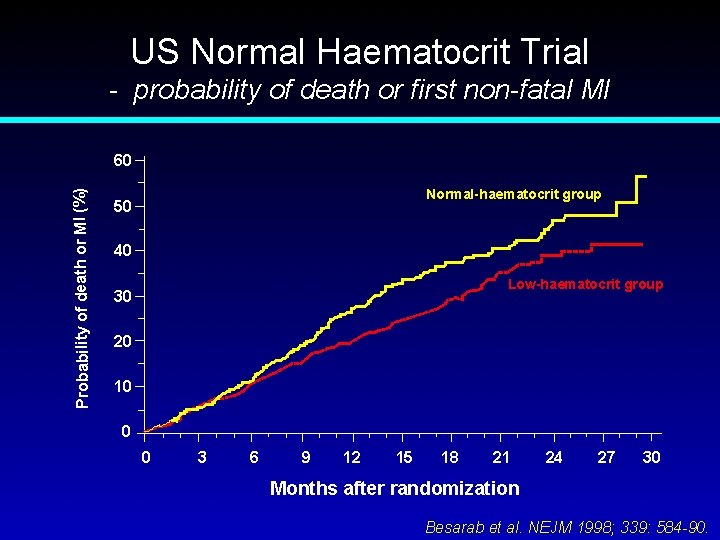
US Normal Haematocrit Trial Besarab A et al. N Engl J Med 339: 584 -590, 1998.

US Normal Haematocrit Trial - probability of death or first non-fatal MI Probability of death or MI (%) 60 Normal-haematocrit group 50 40 Low-haematocrit group 30 20 10 0 0 3 6 9 12 15 18 21 24 27 30 Months after randomization Besarab et al. NEJM 1998; 339: 584 -90.

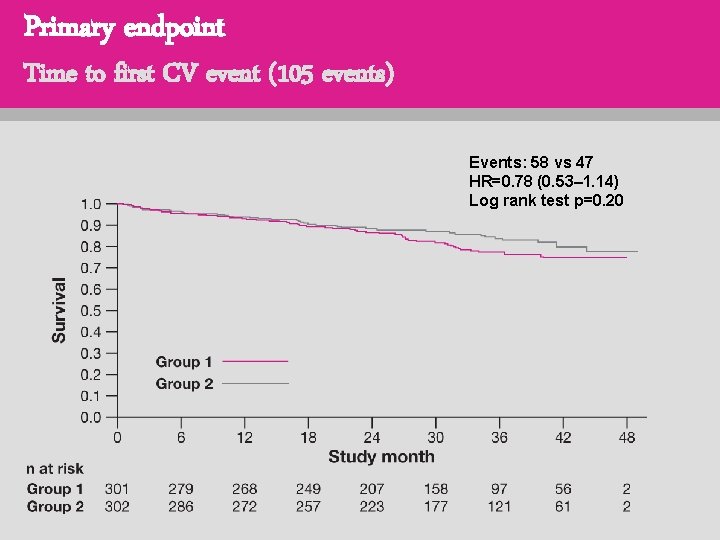
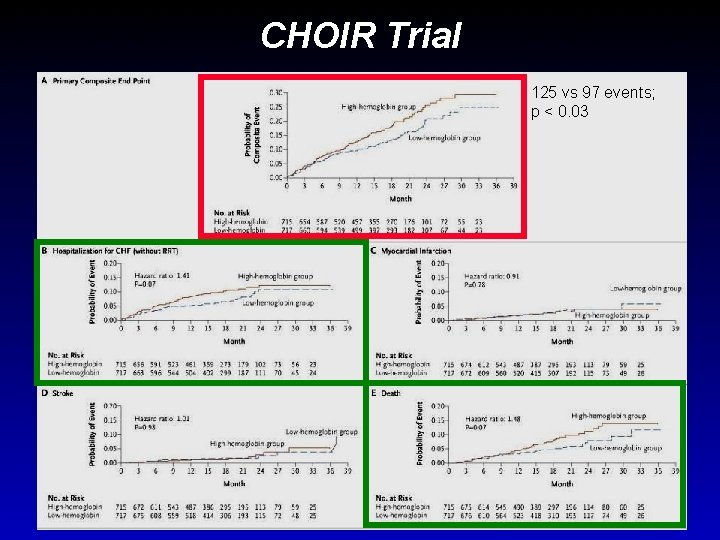
CREATE CHOIR


Primary endpoint Time to first CV event (105 events) Events: 58 vs 47 HR=0. 78 (0. 53– 1. 14) Log rank test p=0. 20


CHOIR Trial 125 vs 97 events; p < 0. 03


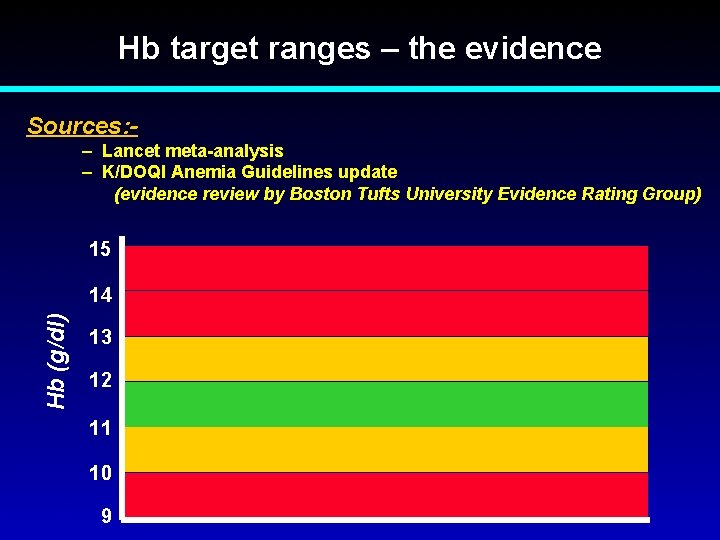
Hb target ranges – the evidence Sources: – Lancet meta-analysis – K/DOQI Anemia Guidelines update (evidence review by Boston Tufts University Evidence Rating Group) 15 Hb (g/dl) 14 13 12 11 10 9

Outline of presentation l Erythropoiesis in 2009 l ESA therapy l Target Hb l Iron management l Anaemia management in kidney transplantation l The future

Why are CKD patients prone to develop iron deficiency? REDUCED INTAKE INCREASED LOSSES l Poor appetite l Occult G-I losses l Poor G-I absorption l Peptic ulceration l Concurrent medication l Blood sampling – e. g. omeprazole l Dialyser losses l Food interactions l Concurrent meds. – e. g. aspirin l Heparin on dialysis

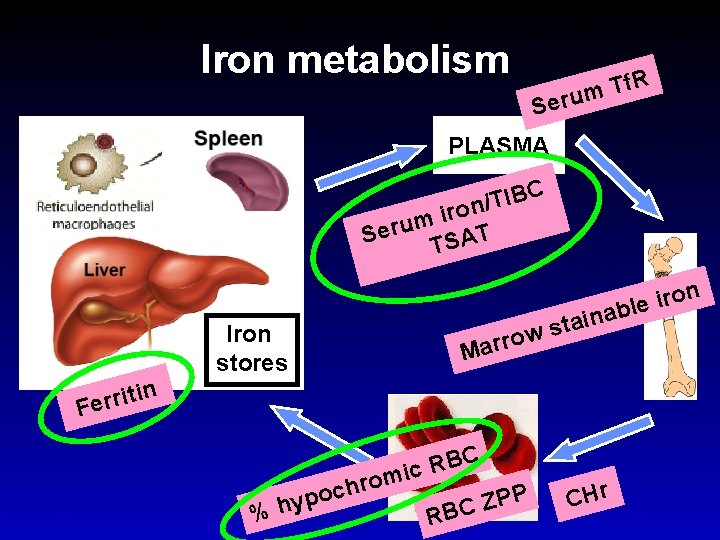
Iron metabolism R f T m eru S PLASMA C B I T / n o r i m Seru TSAT n Iron stores Mar iro e l b a n i a t s row in t i r r Fe C RB c i om r % ch o p y h R P P Z BC CHr

Monitoring iron status Minimum ranges: Serum ferritin > 100 g/l Hypochromic RBC < 10% TSAT > 20% Aim for : Serum ferritin 200 -500 g/l Hypochromic RBC < 2. 5% TSAT 30 -40%

IV Iron Agents are Spheroid Particles with an Iron Core and a Carbohydrate Shell iron oxyhydroxide core carbohydrate shell

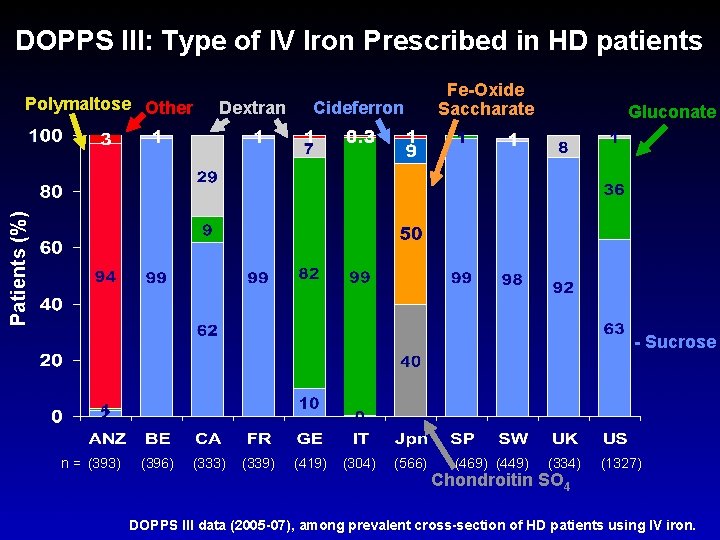
DOPPS III: Type of IV Iron Prescribed in HD patients Dextran Cideferron Fe-Oxide Saccharate Gluconate Patients (%) Polymaltose Other - Sucrose n = (393) (396) (333) (339) (419) (304) (566) (469) (449) (334) Chondroitin SO 4 (1327) DOPPS III data (2005 -07), among prevalent cross-section of HD patients using IV iron.

Benefits of IV iron in CKD patients IV iron can improve the anaemia of CKD even in the absence of ESA therapy IV iron can significantly enhance the response to ESA therapy, even in iron-replete patients

Potential dangers of IV iron ? Short-term Anaphylactic reactions (iron dextran only; dextran Abs) “Free iron” reactions (all IV iron preparations) Long-term Increased susceptibility to infection Increased oxidative stress Iron overload

Balance of benefits vs. risks of IV iron An ax l y h ap is on i t c e Inf risk e v i t a Oxid ss stre k s i r y alit t r o M B s t i f e en on r i V of I

Outline of presentation l Erythropoiesis in 2009 l ESA therapy l Target Hb l Iron management l Anaemia management in kidney transplantation l The future

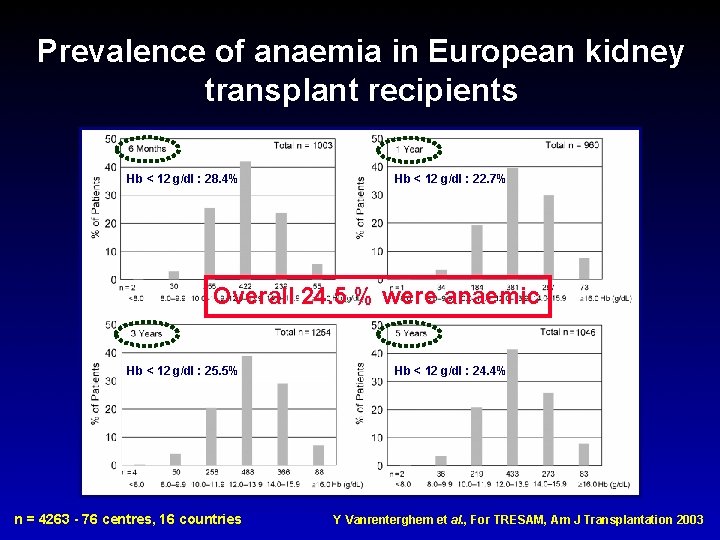
Prevalence of anaemia in European kidney transplant recipients Hb < 12 g/dl : 28. 4% Hb < 12 g/dl : 22. 7% Overall 24. 5 % were anaemic Hb < 12 g/dl : 25. 5% n = 4263 - 76 centres, 16 countries Hb < 12 g/dl : 24. 4% Y Vanrenterghem et al. , For TRESAM, Am J Transplantation 2003

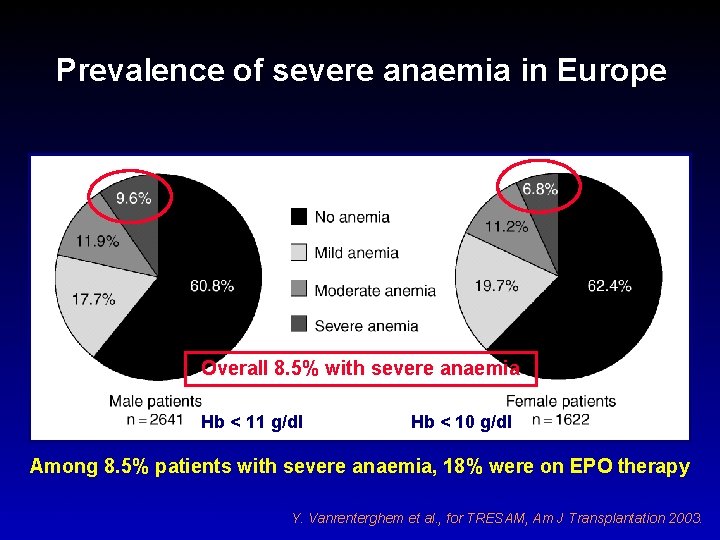
Prevalence of severe anaemia in Europe Overall 8. 5% with severe anaemia Hb < 11 g/dl Hb < 10 g/dl Among 8. 5% patients with severe anaemia, 18% were on EPO therapy Y. Vanrenterghem et al. , for TRESAM, Am J Transplantation 2003.

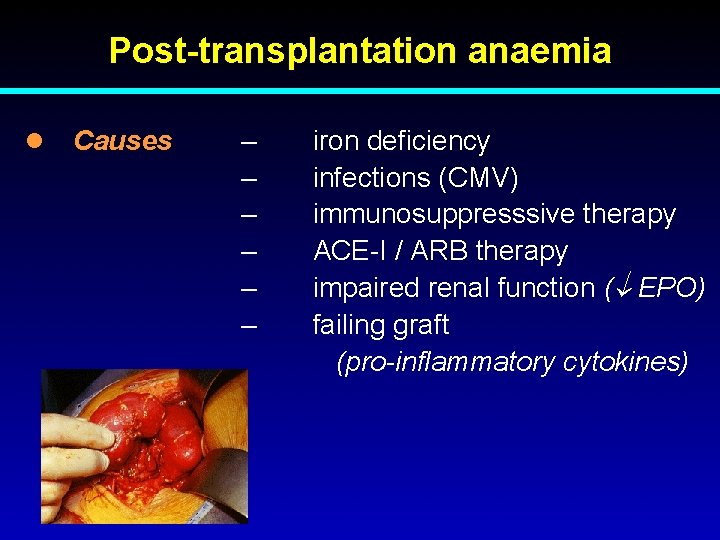
Post-transplantation anaemia l Causes – – – iron deficiency infections (CMV) immunosuppresssive therapy ACE-I / ARB therapy impaired renal function ( EPO) failing graft (pro-inflammatory cytokines)

ESA hyporesponsiveness in renal transplantation Iron deficiency Infection/ inflammation Underdialysis Hyperparathyroidism Aluminium toxicity Carnitine deficiency PRCA Blood loss Haemolysis B 12/folate deficiency Marrow disorders Haemoglobinopathies ACE inhibitors Viral (CMV, EBV, Parvovirus) Malignancy (e. g. lymphoma) Immunosuppression (Aza, MMF, SRL)

Outline of presentation l Erythropoiesis in 2009 l ESA therapy l Target Hb l Iron management l Anaemia management in kidney transplantation l The future

Clin J Am Soc Nephrol, 2008

Hematide l EPO-mimetic peptide, now in Phase III clinical trials l Amino acid sequences completely unrelated to native EPO l Shows same functional / biological properties as EPO

What is different about Hematide? l Peptide-based (epoetin, darbepoetin, CERA – all protein-based) l Not genetically-engineered in cells (unlike epoetin, darbepoetin, CERA) l Manufactured by synthetic peptide chemical techniques l ? More stable at room temperature l ? less immunogenic l Does not cross-react with antibodies against EPO – should not cause PRCA; can be used to treat Ab+PRCA l First ESA to be tested de novo once-monthly in CKD patients

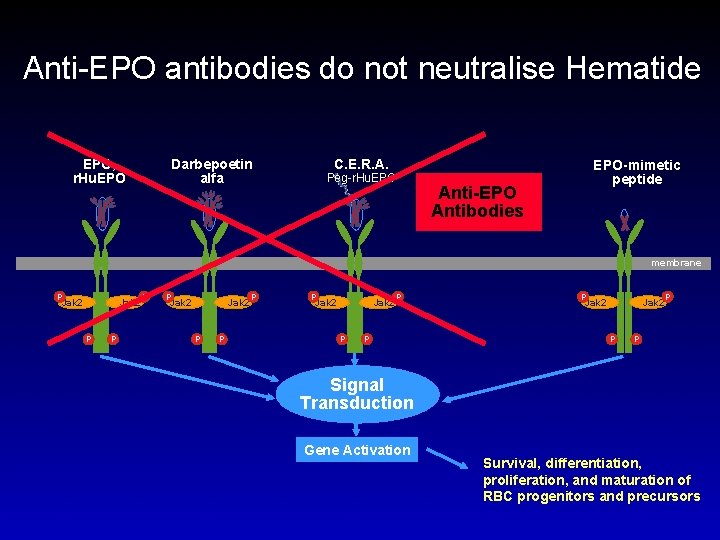
Anti-EPO antibodies do not neutralise Hematide EPO, r. Hu. EPO Darbepoetin alfa C. E. R. A. Peg-r. Hu. EPO-mimetic peptide Anti-EPO Antibodies membrane P Jak 2 Jak 2 P P P Signal Transduction Gene Activation Survival, differentiation, proliferation, and maturation of RBC progenitors and precursors

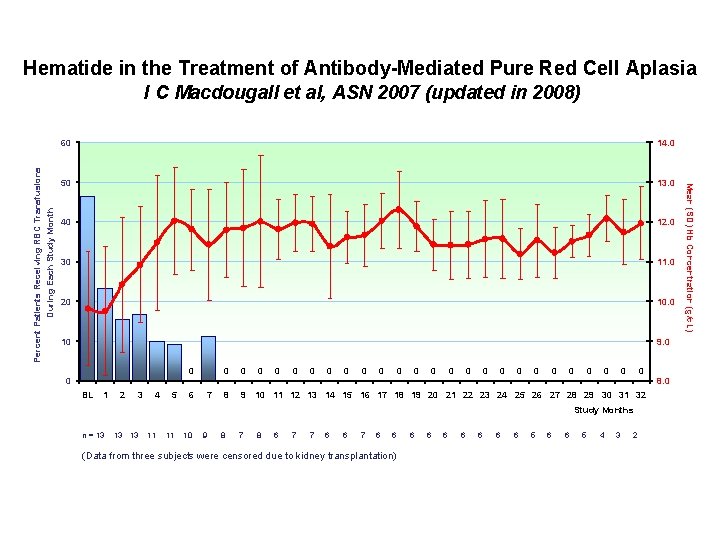
Hematide in the Treatment of Antibody-Mediated Pure Red Cell Aplasia I C Macdougall et al, ASN 2007 (updated in 2008) 60 14. 0 50 13. 0 40 12. 0 30 11. 0 20 10 9. 0 0 0 BL 1 2 3 4 5 6 7 0 0 8 9 0 0 0 0 0 0 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 Study Months n = 13 13 13 11 11 10 9 8 7 8 6 7 7 6 6 (Data from three subjects were censored due to kidney transplantation) 58 6 6 6 6 5 4 3 2 8. 0 Mean (SD) Hb Concentration (g/d. L) Percent Patients Receiving RBC Transfusions During Each Study Month

HIF stabilisers l HIF is the hypoxic sensor that upregulates EPO gene expression l HIF is broken down by a prolyl hydroxylase enzyme l An inhibitor of HIF hydroxylase has been synthesised (Fibro. Gen) l It causes an increase in EPO levels, even in CKD patients Upside l This enzyme inhibitor is orally-active Downside l >100 other genes (e. g. VEGF) also turned on l Rare development of severe liver toxicity (may be fatal)

New IV irons pending……. l 2 new IV irons forthcoming: – Ferumoxytol (US) – Ferric carboxymaltose – Ferinject. TM (Europe) l Advantages – ? safer – no need for test dose – more rapid high-dose bolus injection – main benefits in the pre-ESRD population

Ferric carboxymaltose (Ferinject) Ferric hydroxide molecules Ribbon-like carboxymaltose l Licensed in Europe l Stable iron complex l Low immunogenic potential – dextran-free l Minimal detectable and releasable free iron l No test dose required l Rapid administration – 200 mg push – 500 mg in 6 mins – 1000 mg infusion in 15 mins

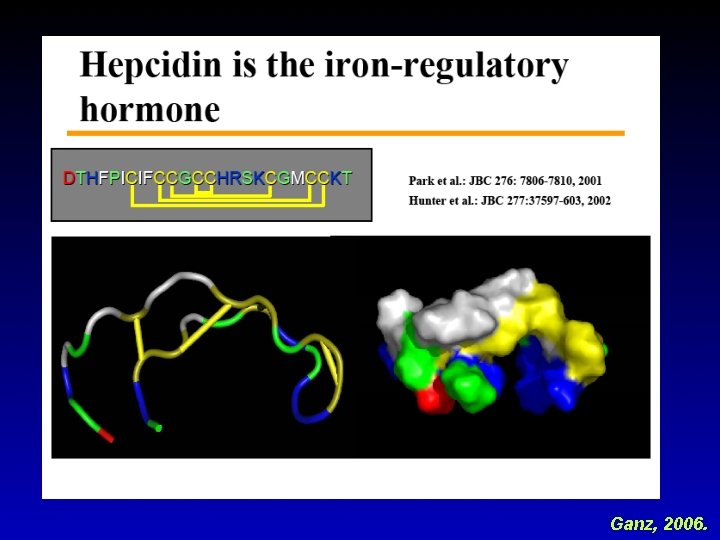
Ganz, 2006.

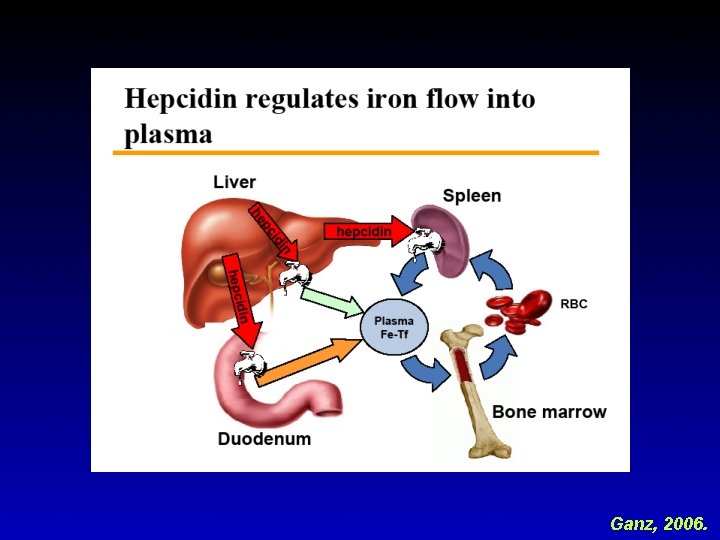
Ganz, 2006.

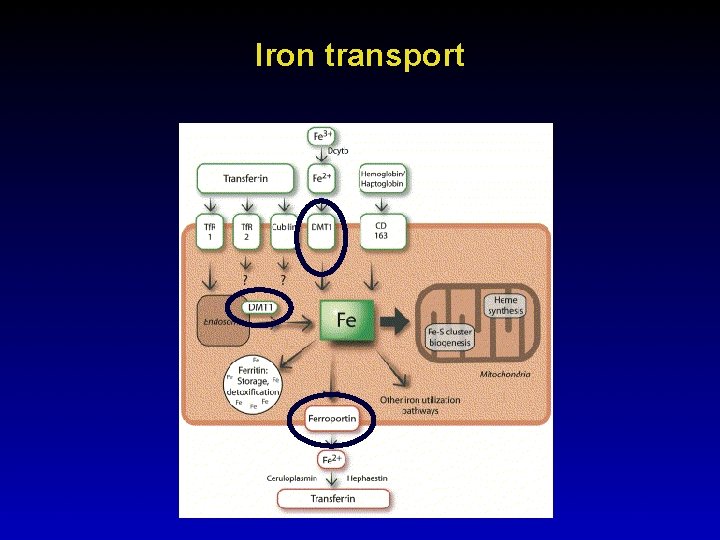
Iron transport

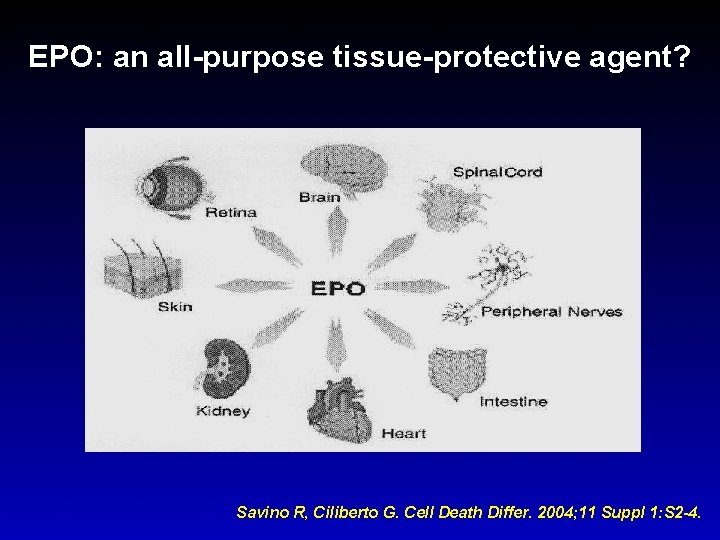
EPO: an all-purpose tissue-protective agent? Savino R, Ciliberto G. Cell Death Differ. 2004; 11 Suppl 1: S 2 -4.

EPO therapy: beyond Hb l Mediated via the anti-apoptotic action of EPO on nonerythroid cells l Relevant for acute cardiac, renal, and cerebral ischaemia l ? Therapeutic benefit in : – Acute MI – Acute stroke – Reperfusion injury – Post-transplantation

Conclusions l Our understanding of erythropoiesis in 2009 has advanced to include the role of hepcidin and pro-inflammatory cytokines l Until further evidence is forthcoming, we should generally target an Hb of 11– 12 g/dl l Even in 2009, there is still a need for additional grade A level evidence in the management of anaemia in CKD l Several new ESAs and IV iron preparations are appearing, and the non-erythropoietic effects of ESAs are being explored