An overview of Future Cements An overview of














- Slides: 28

An overview of Future Cements An overview of the alternative mineral binder systems including novel concrete technologies addressing practical supply chain and economic issues including energy 9/03/2021 www. tececo. com www. propubs. com 1

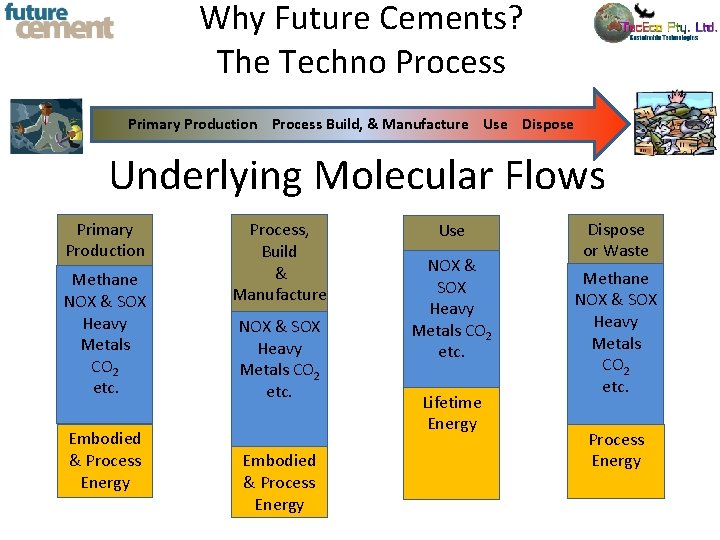
Why Future Cements? The Techno Process Primary Production Process Build, & Manufacture Use Dispose Underlying Molecular Flows Primary Production Methane NOX & SOX Heavy Metals CO 2 etc. Embodied & Process Energy Process, Build & Manufacture NOX & SOX Heavy Metals CO 2 etc. Embodied & Process Energy Use NOX & SOX Heavy Metals CO 2 etc. Lifetime Energy Dispose or Waste Methane NOX & SOX Heavy Metals CO 2 etc. Process Energy

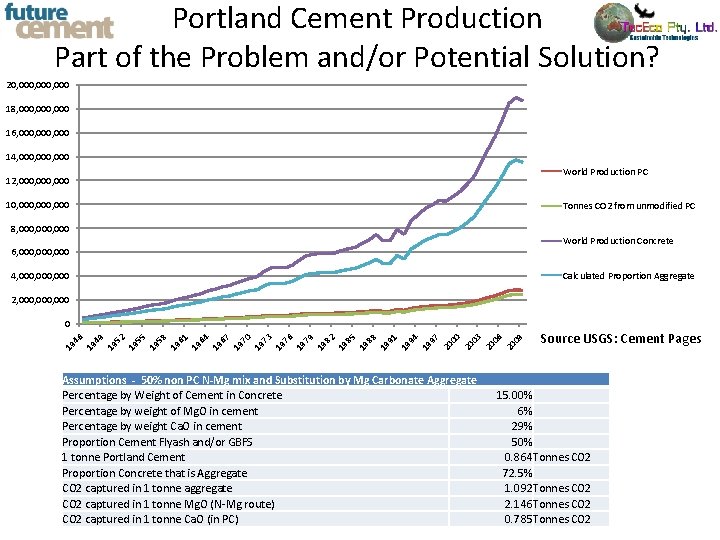
Portland Cement Production Part of the Problem and/or Potential Solution? 20, 000, 000 18, 000, 000 16, 000, 000 14, 000, 000 World Production PC 12, 000, 000 10, 000, 000 Tonnes CO 2 from unmodified PC 8, 000, 000 World Production Concrete 6, 000, 000 Calculated Proportion Aggregate 4, 000, 000 2, 000, 000 19 46 19 49 19 52 19 55 19 58 19 61 19 64 19 67 19 70 19 73 19 76 19 79 19 82 19 85 19 88 19 91 19 94 19 97 20 00 20 03 20 06 20 09 0 Assumptions - 50% non PC N-Mg mix and Substitution by Mg Carbonate Aggregate Percentage by Weight of Cement in Concrete Percentage by weight of Mg. O in cement Percentage by weight Ca. O in cement Proportion Cement Flyash and/or GBFS 1 tonne Portland Cement Proportion Concrete that is Aggregate CO 2 captured in 1 tonne aggregate CO 2 captured in 1 tonne Mg. O (N-Mg route) CO 2 captured in 1 tonne Ca. O (in PC) Source USGS: Cement Pages 15. 00% 6% 29% 50% 0. 864 Tonnes CO 2 72. 5% 1. 092 Tonnes CO 2 2. 146 Tonnes CO 2 0. 785 Tonnes CO 2

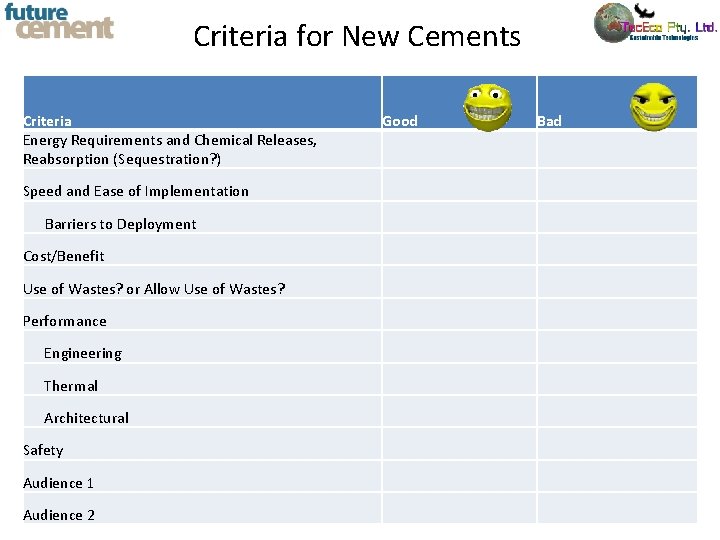
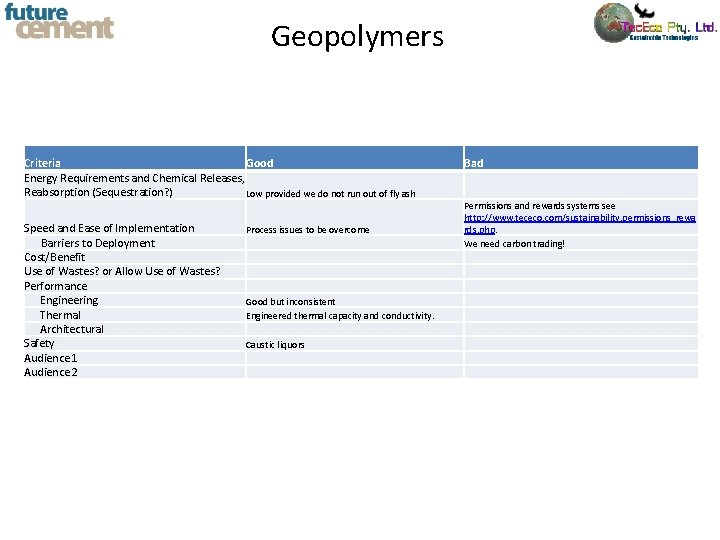
Criteria for New Cements Criteria Energy Requirements and Chemical Releases, Reabsorption (Sequestration? ) Speed and Ease of Implementation Barriers to Deployment Cost/Benefit Use of Wastes? or Allow Use of Wastes? Performance Engineering Thermal Architectural Safety Audience 1 Audience 2 Good Bad

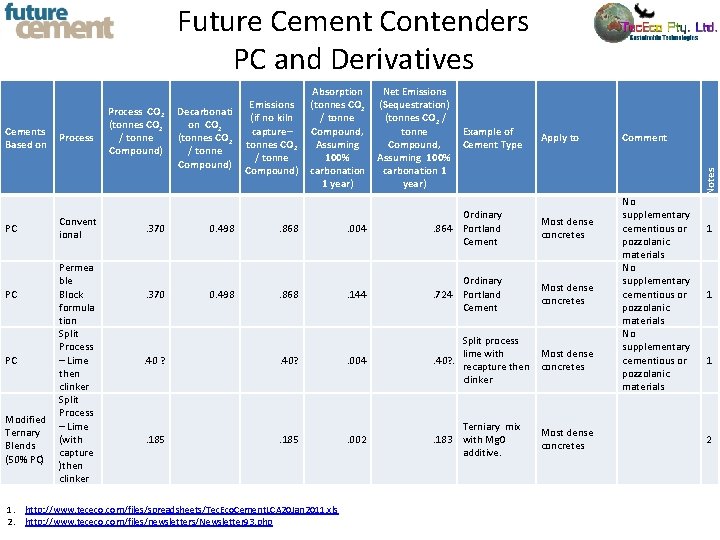
Future Cement Contenders PC and Derivatives PC Process Convent ional Permea ble PC Block formula tion Split Process PC – Lime then clinker Split Process Modified – Lime Ternary (with Blends capture (50% PC) )then clinker Decarbonati on CO 2 (tonnes CO 2 / tonne Compound) Emissions (if no kiln capture– tonnes CO 2 / tonne Compound) Net Emissions (Sequestration) (tonnes CO 2 / tonne Compound, Assuming 100% carbonation 1 year) Example of Cement Type . 370 0. 498 . 868 . 004 Ordinary . 864 Portland Cement . 370 0. 498 . 868 . 144 Ordinary . 724 Portland Cement Apply to Most dense concretes . 40 ? . 40? . 004 Split process lime with Most dense . 40? . recapture then concretes clinker . 185 . 002 Terniary mix . 183 with Mg. O additive. 1. http: //www. tececo. com/files/spreadsheets/Tec. Eco. Cement. LCA 20 Jan 2011. xls 2. http: //www. tececo. com/files/newsletters/Newsletter 93. php Most dense concretes Comment No supplementary cementious or pozzolanic materials Notes Cements Based on Process CO 2 (tonnes CO 2 / tonne Compound) Absorption (tonnes CO 2 / tonne Compound, Assuming 100% carbonation 1 year) 1 1 1 2

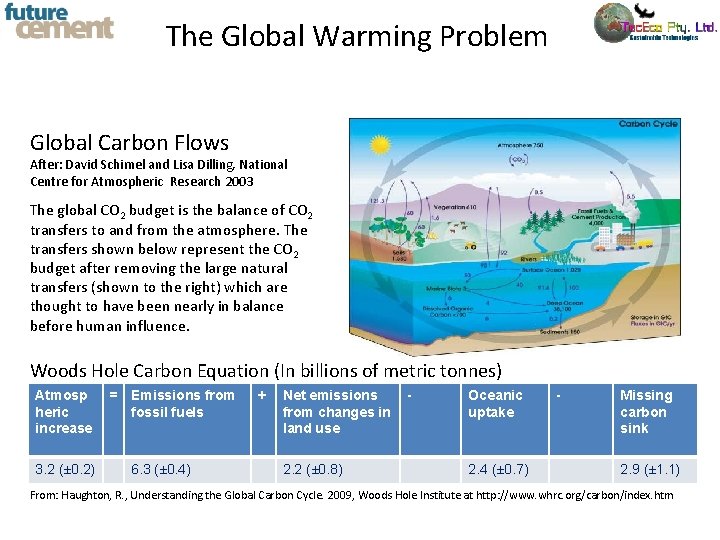
The Global Warming Problem Global Carbon Flows After: David Schimel and Lisa Dilling, National Centre for Atmospheric Research 2003 The global CO 2 budget is the balance of CO 2 transfers to and from the atmosphere. The transfers shown below represent the CO 2 budget after removing the large natural transfers (shown to the right) which are thought to have been nearly in balance before human influence. Woods Hole Carbon Equation (In billions of metric tonnes) Atmosp heric increase 3. 2 (± 0. 2) = Emissions from fossil fuels 6. 3 (± 0. 4) + Net emissions from changes in land use 2. 2 (± 0. 8) - Oceanic uptake 2. 4 (± 0. 7) - Missing carbon sink 2. 9 (± 1. 1) From: Haughton, R. , Understanding the Global Carbon Cycle. 2009, Woods Hole Institute at http: //www. whrc. org/carbon/index. htm

Net Atmospheric Increase in Terms of Billions of Tonnes CO 2 Using the Figures from Woods Hole on the Previous Slide Atmospheric increase = 3. 2 (± 0. 2) Emissions from fossil fuels + 6. 3 (± 0. 4) Net emissions from changes in land use - 2. 2 (± 0. 8) Oceanic uptake - 2. 4 (± 0. 7) Missing carbon sink 2. 9 (± 1. 1) Converting to tonnes CO 2 in the same units by multiplying by 44. 01/12. 01, the ratio of the respective molecular weights. Atmospheric increase 11. 72 (± 0. 2) = Emissions from fossil fuels 23. 08 (± 0. 4) + Net emissions from changes in land use 8. 016 (± 0. 8) - Oceanic uptake 8. 79 (± 0. 7) - Missing carbon sink 10. 62 (± 1. 1) From the above the annual atmospheric increase of CO 2 is in the order of 12 billion metric tonnes.

How Much Man Made Carbonate to Solve Global Warming? If a proportion of the built environment were man made carbonate, how much would we need to reverse global warming? Mg. O + H 2 O => Mg(OH)2 + CO 2 + 2 H 2 O => Mg. CO 3. 3 H 2 O 40. 31 + 18(l) => 58. 31 + 44. 01(g) + 2 X 18(l) => 138. 368 molar masses. 44. 01 parts by mass of CO 2 ~= 138. 368 parts by mass Mg. CO 3. 3 H 2 O 1 ~= 138. 368/44. 01= 3. 144 12 billion tonnes CO 2 ~= 37. 728 billion tonnes of nesquehonite or Mg. O + H 2 O => Mg(OH)2 + CO 2 + 2 H 2 O => Mg. CO 3 40. 31 + 18(l) => 58. 31 + 44. 01(g) + 2 X 18(l) => 84. 32 molar masses. CO 2 ~= Mg. CO 3 44. 01 parts by mass of CO 2 ~= 84. 32 parts by mass Mg. CO 3 1 ~= 84. 32/44. 01= 1. 9159 12 billion tonnes CO 2 ~= 22. 99 billion tonnes magnesite Ca. O + H 2 O => Ca(OH)2 + CO 2 + 2 H 2 O => Ca. CO 3 56. 08 + 18(l) => 74. 08 + 44. 01(g) + 2 X 18(l) => 100. 09 molar masses. CO 2 ~= Ca. CO 3 44. 01 parts by mass of CO 2 ~= 100. 09 parts by mass Mg. CO 3 1 ~= 100. 09/44. 01= 2. 274 12 billion tonnes CO 2 ~= 27. 29 billion tonnes calcite (limestone)

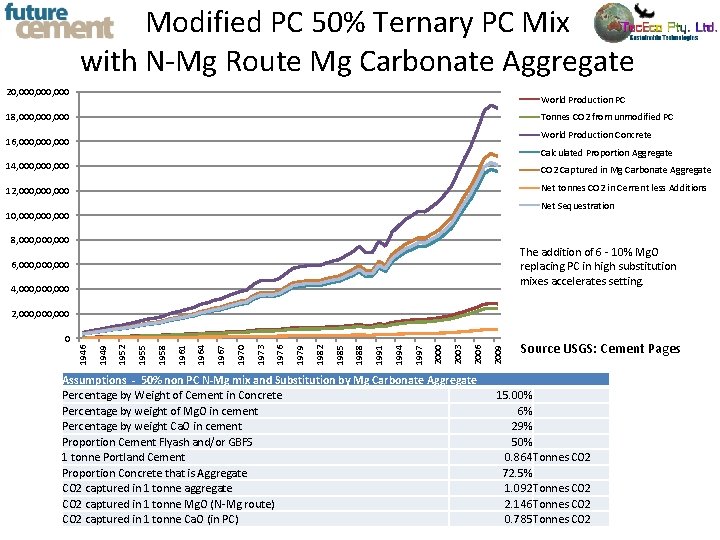
Modified PC 50% Ternary PC Mix with N-Mg Route Mg Carbonate Aggregate 20, 000, 000 World Production PC 18, 000, 000 Tonnes CO 2 from unmodified PC World Production Concrete 16, 000, 000 Calculated Proportion Aggregate 14, 000, 000 CO 2 Captured in Mg Carbonate Aggregate 12, 000, 000 Net tonnes CO 2 in Cement less Additions Net Sequestration 10, 000, 000 8, 000, 000 The addition of 6 - 10% Mg. O replacing PC in high substitution mixes accelerates setting. 6, 000, 000 4, 000, 000 2, 000, 000 Assumptions - 50% non PC N-Mg mix and Substitution by Mg Carbonate Aggregate Percentage by Weight of Cement in Concrete Percentage by weight of Mg. O in cement Percentage by weight Ca. O in cement Proportion Cement Flyash and/or GBFS 1 tonne Portland Cement Proportion Concrete that is Aggregate CO 2 captured in 1 tonne aggregate CO 2 captured in 1 tonne Mg. O (N-Mg route) CO 2 captured in 1 tonne Ca. O (in PC) 2009 2006 2003 2000 1997 1994 1991 1988 1985 1982 1979 1976 1973 1970 1967 1964 1961 1958 1955 1952 1949 1946 0 Source USGS: Cement Pages 15. 00% 6% 29% 50% 0. 864 Tonnes CO 2 72. 5% 1. 092 Tonnes CO 2 2. 146 Tonnes CO 2 0. 785 Tonnes CO 2

Modified PC 50% Ternary Mix with N-Mg Route Mg Carbonate Aggregate • • 25 -30% improvement in strength Fast first set Better Rheology Less shrinkage – less cracking Less bleeding Long term durability Solve autogenous shrinkage? Criteria Good Energy Requirements and Chemical Releases, Use >50% replacements and still set like “normal” Reabsorption (Sequestration? ) concrete! Speed and Ease of Implementation Rapid adoption possible Barriers to Deployment Cost/Benefit Use of Wastes? or Allow Use of Wastes? Performance Engineering Thermal Architectural Safety Audience 1 Audience 2 Excellent until fly ash runs out! Uses GBFS and fly ash and nanufactured nesquehonite based aggregate Excellent all round High thermal capacity Excellent No issues Bad Permissions and rewards systems see http: //www. tececo. com/sustainability. permissions_rewa rds. php

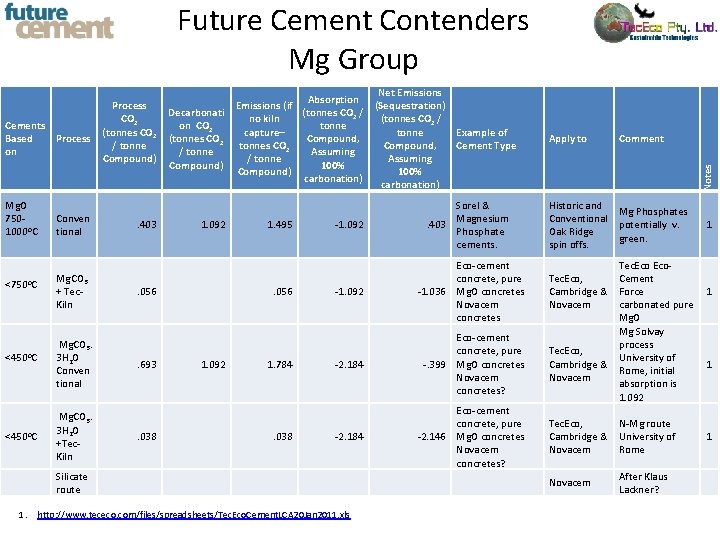
Future Cement Contenders Mg Group Mg. O 7501000 o. C <750 o. C <450 o. C Process Conven tional Mg. CO 3 + Tec. Kiln Mg. CO 3. 3 H 2 O Conven tional Mg. CO 3. 3 H 2 O +Tec. Kiln . 403 Decarbonati on CO 2 (tonnes CO 2 / tonne Compound) 1. 092 . 056 . 693 . 038 Net Emissions Absorption Emissions (if (Sequestration) (tonnes CO 2 / no kiln (tonnes CO 2 / tonne capture– tonne Compound, tonnes CO 2 Compound, Assuming / tonne Assuming 100% Compound) 100% carbonation) 1. 495 . 056 1. 092 1. 784 . 038 Example of Cement Type -1. 092 Sorel & Magnesium . 403 Phosphate cements. -1. 092 Eco-cement concrete, pure -1. 036 Mg. O concretes Novacem concretes -2. 184 Eco-cement concrete, pure -. 399 Mg. O concretes Novacem concretes? -2. 184 Eco-cement concrete, pure -2. 146 Mg. O concretes Novacem concretes? Silicate route 1. http: //www. tececo. com/files/spreadsheets/Tec. Eco. Cement. LCA 20 Jan 2011. xls Apply to Comment Historic and Conventional Oak Ridge spin offs. Mg Phosphates potentially v. green. Notes Cements Based on Process CO 2 (tonnes CO 2 / tonne Compound) 1 Tec. Eco, Cement Cambridge & Force 1 Novacem carbonated pure Mg. O Mg Solvay process Tec. Eco, University of Cambridge & 1 Rome, initial Novacem absorption is 1. 092 Tec. Eco, N-Mg route Cambridge & University of Novacem Rome Novacem After Klaus Lackner? 1

Magnesium Phosphate Cements • Chemical cements that rely on the precipitation of insoluble magnesium phosphate from a mix of magnesium oxide and a soluble phosphate. • Some of the oldest binders known (dung +Mg. O) • Potentially very green – if the magnesium oxide used is made with no releases or via the nesquehonite (N-Mg route) and – a way can be found to utilise waste phosphate from intensive agriculture and fisheries e. g. feedlots. (Thereby solving another environmental problem) Criteria Good Energy Requirements and Chemical Releases, Reabsorption (Sequestration? ) The Mg. O used could be made without releases Speed and Ease of Implementation Rapid adoption possible Barriers to Deployment Cost/Benefit Use of Wastes? or Allow Use of Wastes? Performance Engineering Thermal Architectural Safety Audience 1 Audience 2 Economies of scale issue for Mg. O to overcome With technology could use waste phosphate reducing water pollution Excellent all round High thermal capacity No issues Bad There is not much phosphate on the planet If barrier overcome (see below) Permissions and rewards systems see http: //www. tececo. com/sustainability. permissions_rewa rds. php. Must find a way to extract phosphate from organic pollution.

Sorel Type Cements and Derivatives are all nano or mechano composites relying on a mix of ionic, co-valent and polar bonding. There a very large number of permutations and combinations and thus a large number of patents Criteria Good Energy Requirements and Chemical Releases, Reabsorption (Sequestration? ) The Mg. O used could be made without releases Speed and Ease of Implementation More could be used Barriers to Deployment Cost/Benefit Economies of scale issue for Mg. O to overcome Use of Wastes? or Allow Use of Wastes? Performance Engineering Excellent except Thermal High thermal capacity Architectural Safety No issues Audience 1 Audience 2 Bad If barrier overcome (see below) Not waterproof even with modification. Not waterproof Not waterpoof, salt affect metals

Magnesium Carbonate Cements • Magnesite (Mg. CO 3) and the di, tri, and pentahydrates known as barringtonite (Mg. CO 3· 2 H 2 O), nesquehonite (Mg. CO 3· 3 H 2 O), and lansfordite (Mg. CO 3· 5 H 2 O), respectively. • Some basic forms such as artinite (Mg. CO 3·Mg(OH)2· 3 H 2 O), hydromagnestite (4 Mg. CO 3·Mg(OH)2· 4 H 2 O) and dypingite (4 Mg. CO 3· Mg(OH)2· 5 H 2 O) also occur as minerals. • We pointed out as early as 2001 that magnesium carbonates are ideal for sequestration as building materials mainly because a higher proportion of CO 2 than with calcium can be bound and significant strength can be achieved. • The significant strength is a result of increased density through carbonation (high molar volume increases) and the microstructure developed by some forms.

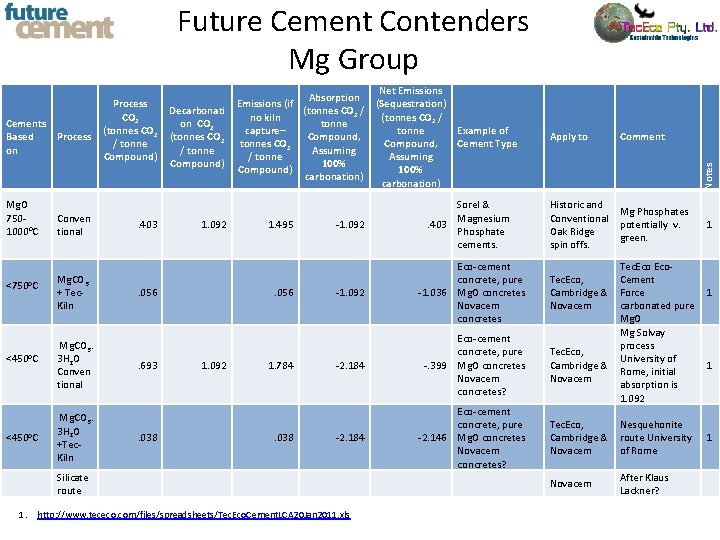
Future Cement Contenders Mg Group Mg. O 7501000 o. C <750 o. C <450 o. C Process Conven tional Mg. CO 3 + Tec. Kiln Mg. CO 3. 3 H 2 O Conven tional Mg. CO 3. 3 H 2 O +Tec. Kiln . 403 Decarbonati on CO 2 (tonnes CO 2 / tonne Compound) 1. 092 . 056 . 693 . 038 Net Emissions Absorption Emissions (if (Sequestration) (tonnes CO 2 / no kiln (tonnes CO 2 / tonne capture– tonne Compound, tonnes CO 2 Compound, Assuming / tonne Assuming 100% Compound) 100% carbonation) 1. 495 . 056 1. 092 1. 784 . 038 Example of Cement Type -1. 092 Sorel & Magnesium . 403 Phosphate cements. -1. 092 Eco-cement concrete, pure -1. 036 Mg. O concretes Novacem concretes -2. 184 Eco-cement concrete, pure -. 399 Mg. O concretes Novacem concretes? -2. 184 Eco-cement concrete, pure -2. 146 Mg. O concretes Novacem concretes? Silicate route 1. http: //www. tececo. com/files/spreadsheets/Tec. Eco. Cement. LCA 20 Jan 2011. xls Apply to Comment Historic and Conventional Oak Ridge spin offs. Mg Phosphates potentially v. green. Notes Cements Based on Process CO 2 (tonnes CO 2 / tonne Compound) 1 Tec. Eco, Cement Cambridge & Force 1 Novacem carbonated pure Mg. O Mg Solvay process Tec. Eco, University of Cambridge & 1 Rome, initial Novacem absorption is 1. 092 Tec. Eco, Nesquehonite Cambridge & route University Novacem of Rome Novacem After Klaus Lackner? 1

Tec. Eco-Cements (Tec-Kiln) Eco-Cements are blends of one or more hydraulic cements and relatively high proportions of reactive magnesia with or without pozzolans and supplementary cementitious additions. They will only carbonate in gas permeable substrates forming strong fibrous minerals. Water vapour and CO 2 must be available for carbonation to ensue. Eco-Cements can be used in a wide range of products from foamed concretes to bricks, blocks and pavers, mortars renders, grouts and pervious concretes such as our own permeacocrete. Somewhere in the vicinity of the Pareto proportion (80%) of conventional concretes could be replaced by Eco-Cement. Left: Recent Eco-Cement blocks made, transported and erected in a week. Laying and Eco-Cement floor. Eco-Cement mortar & Eco-cement mud bricks. Right: Eco-Cement permeacocretes and foamed concretes Criteria Good Bad Energy Requirements and Chemical Releases, The Mg. O used could be made without releases and Reabsorption (Sequestration? ) using the N-Mg route Speed and Ease of Implementation Barriers to Deployment Cost/Benefit Use of Wastes? or Allow Use of Wastes? Performance Engineering Thermal Architectural Safety Audience 1 Audience 2 Easily implemented as no carbonation rooms etc reqd. Permissions and rewards systems see http: //www. tececo. com/sustainability. permissions_rewa rds. php. We need carbon trading! Economies of scale issue for Mg. O to overcome A vast array of wastes can be incorporated Excellent Engineered thermal capacity and conductivity. Need to be handled gently in the first few days

Tec. Eco-Cements (Tec-Kiln, N-Mg route) Scope for Reducing Energy Using Waste Heat? Initial weight loss below 100" consists almost entirely of water (1. 3 molecules per molecule of nesquehonite). Between 100 and 1500 C volatilization of further water is associated with a small loss of carbon dioxide (~3 -5 %). From 1500 C to 2500 C, the residual water content varies between 0 -6 and 0 -2 molecules per molecule of Mg. C 03. Above 3000 C, loss of carbon dioxide becomes appreciable and is virtually complete by 4200 C, leaving Mg. O with a small residual water content. Energy could be saved using a two stage calcination process using waste energy for the first stage. Nesquehonite courtesy of Vincenzo Ferrini, university of Rome. Dell, R. M. and S. W. Weller (1959). "The Thermal Decomposition of Nesquehonite Mg. CO 3 3 H 20 And Magnesium Ammonium Carbonate Mg. CO 3 (NH 4)2 CO 3 4 H 2 O. " Trans Faraday Soc 55(10): 2203 - 2220. Criteria Good Energy Requirements and Chemical Releases, Reabsorption (Sequestration? ) The total process uses two Speed and Ease of Implementation Barriers to Deployment Cost/Benefit Use of Wastes? or Allow Use of Wastes? Performance Engineering Thermal Architectural Safety Audience 1 Audience 2 Easily implemented as no carbonation rooms etc reqd. Bad Permissions and rewards systems see http: //www. tececo. com/sustainability. permissions_rewa rds. php. We need carbon trading! Economies of scale issue for Mg. O to overcome A vast array of wastes can be incorporated Excellent Engineered thermal capacity and conductivity. Need to be handled gently in the first few days

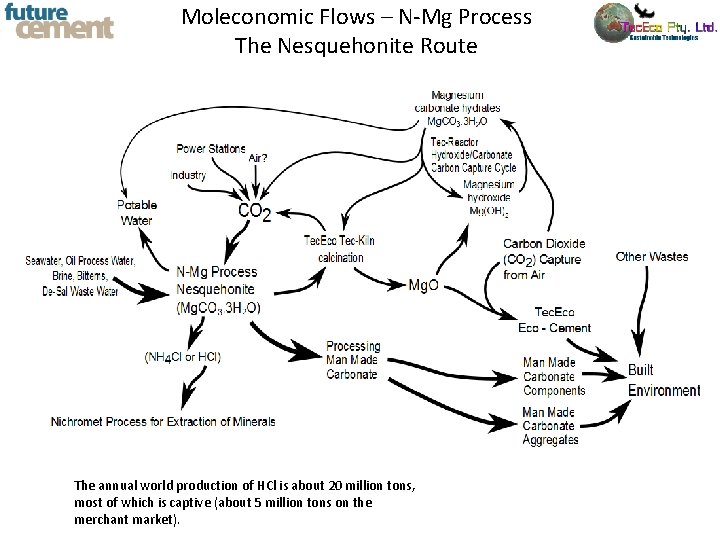
Moleconomic Flows – N-Mg Process The Nesquehonite Route The annual world production of HCl is about 20 million tons, most of which is captive (about 5 million tons on the merchant market).

The Tec-Reactor Hydroxide Carbonate Capture Cycle • The solubility of carbon dioxide gas in seawater – Increases as the temperature approached zero and – Is at a maxima around 4 o. C • This phenomenon is related to the chemical nature of CO 2 and water and • Can be utilised in a carbonate – hydroxide slurry process to capture CO 2 out of the air and release it for storage or use in a controlled manner

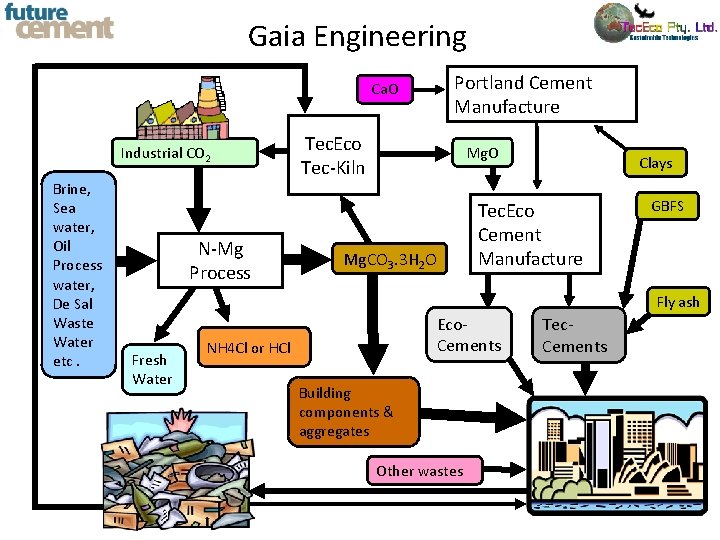
Gaia Engineering Portland Cement Manufacture Ca. O Industrial CO 2 Brine, Sea water, Oil Process water, De Sal Waste Water etc. N-Mg Process Tec. Eco Tec-Kiln Mg. O Mg. CO 3. 3 H 2 O Clays Tec. Eco Cement Manufacture GBFS Fly ash Fresh Water Eco. Cements NH 4 Cl or HCl Building components & aggregates Other wastes Tec. Cements

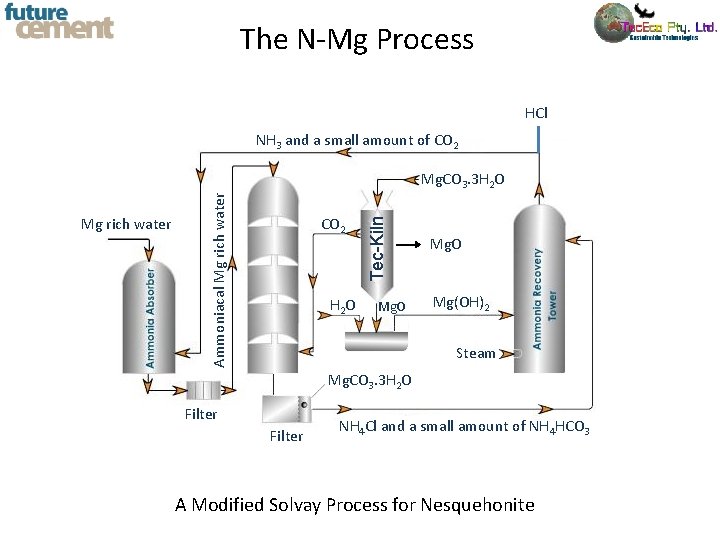
The N-Mg Process HCl NH 3 and a small amount of CO 2 H 2 O Tec-Kiln Mg rich water Ammoniacal Mg rich water Mg. CO 3. 3 H 2 O Mg(OH)2 Steam Mg. CO 3. 3 H 2 O Filter NH 4 Cl and a small amount of NH 4 HCO 3 A Modified Solvay Process for Nesquehonite

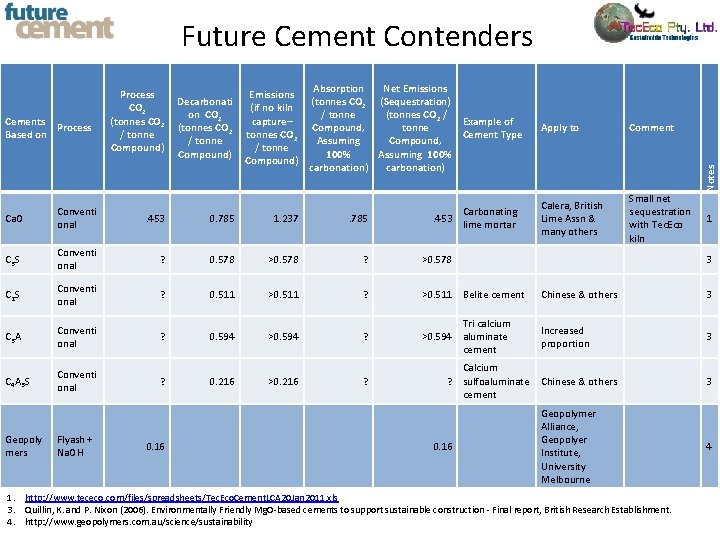
Cements Process Based on Process CO 2 (tonnes CO 2 / tonne Compound) Decarbonati on CO 2 (tonnes CO 2 / tonne Compound) Emissions (if no kiln capture– tonnes CO 2 / tonne Compound) Absorption Net Emissions (tonnes CO 2 (Sequestration) / tonne (tonnes CO 2 / Example of Compound, tonne Cement Type Assuming Compound, 100% Assuming 100% carbonation) Carbonating . 453 lime mortar Apply to Comment Calera, British Lime Assn & many others Small net sequestration with Tec. Eco kiln Notes Future Cement Contenders Ca. O Conventi onal . 453 0. 785 1. 237 . 785 C 3 S Conventi onal ? 0. 578 >0. 578 ? >0. 578 C 2 S Conventi onal ? 0. 511 >0. 511 ? >0. 511 Belite cement Chinese & others 3 C 3 A Conventi onal ? Tri calcium >0. 594 aluminate cement Increased proportion 3 C 4 A 3 S Conventi onal Geopoly mers Flyash + Na. OH ? ? 0. 16 0. 594 0. 216 >0. 594 >0. 216 ? 3 Calcium ? sulfoaluminate Chinese & others cement 0. 16 1 Geopolymer Alliance, Geopolyer Institute, University Melbourne 1. http: //www. tececo. com/files/spreadsheets/Tec. Eco. Cement. LCA 20 Jan 2011. xls 3. Quillin, K. and P. Nixon (2006). Environmentally Friendly Mg. O-based cements to support sustainable construction - Final report, British Research Establishment. 4. http: //www. geopolymers. com. au/science/sustainability 3 4

Ca. O-Lime Criteria Good Energy Requirements and Chemical Releases, Reabsorption (Sequestration? ) The Ca. O used could be made without Speed and Ease of Implementation Barriers to Deployment Cost/Benefit Use of Wastes? or Allow Use of Wastes? Performance Engineering Thermal Architectural Safety Audience 1 Audience 2 Easily implemented as no carbonation rooms etc reqd. Good Engineered thermal capacity and conductivity. An irritating dust Bad Permissions and rewards systems see http: //www. tececo. com/sustainability. permissions_rewa rds. php. We need carbon trading!

Geopolymers Criteria Good Energy Requirements and Chemical Releases, Reabsorption (Sequestration? ) Low provided we do not run out of fly ash Speed and Ease of Implementation Barriers to Deployment Cost/Benefit Use of Wastes? or Allow Use of Wastes? Performance Engineering Thermal Architectural Safety Audience 1 Audience 2 Process issues to be overcome Good but inconsistent Engineered thermal capacity and conductivity. Caustic liquors Bad Permissions and rewards systems see http: //www. tececo. com/sustainability. permissions_rewa rds. php. We need carbon trading!

Other Contenders • Belite cements suffer from a slower setting rate which could be accelerated with more aluminates rather than alite. Mg. O works to some extent as well althougth as yet we have not done enough work • Sulfoaluminate type cements have a low energy requirement.

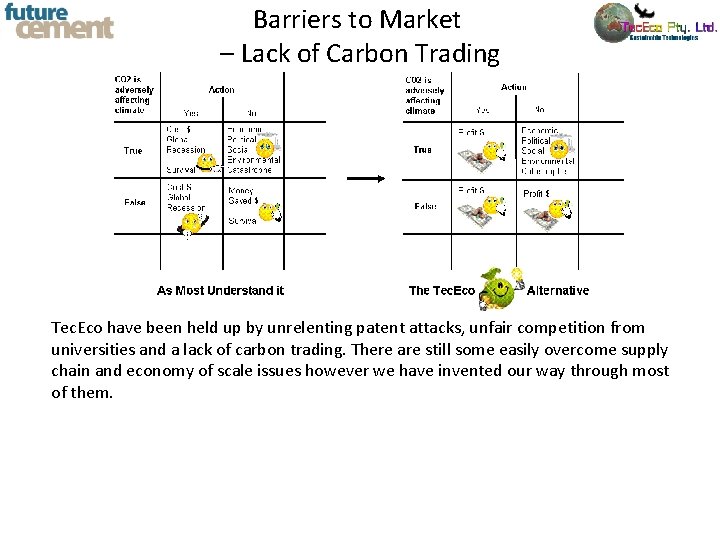
Barriers to Market - Patents Fierce competition whilst the world heats up reminds me of Nero. Perhaps a more co-operative approach is more appropriate. We face after all common supply chain, economic and technical issues. We should jointly be marketing to governments as new technologies are essential as the potential for emissions reduction and sequestration is enormous http: //www. google. com/patents? id=hh. YJ AAAAEBAJ&printsec=abstract&zoom=4#v =onepage&q&f=false

Barriers to Market – Lack of Carbon Trading Tec. Eco have been held up by unrelenting patent attacks, unfair competition from universities and a lack of carbon trading. There are still some easily overcome supply chain and economy of scale issues however we have invented our way through most of them.

The Concept of a Carbonate Built Environment 13 th July 2002 – Fred Pearce in New Scientist about Tec. Eco magnesium cement technology: “THERE is a way to make our city streets as green as the Amazon rainforest. Almost every aspect of the built environment, from bridges to factories to tower blocks, and from roads to sea walls, could be turned into structures that soak up carbon dioxide- the main greenhouse gas behind global warming. All we need to do is change the way we make cement. All we have to do is change the way we do things and do what a big old tree does – make our homes out of CO 2