AN INTRODUCTION TO TRANSITION METAL CHEMISTRY KNOCKHARDY PUBLISHING

AN INTRODUCTION TO TRANSITION METAL CHEMISTRY KNOCKHARDY PUBLISHING

DEFINITION OF TRANSITION ELEMENTS Definition D-block elements forming one or more stable ions with partially filled (incomplete) d-sub shells. Properties arise from an incomplete d sub-shell in atoms or ions

THE FIRST ROW TRANSITION ELEMENTS Metallicproperties All the transition elements are metals Strong metallic bonds due to small ionic size and close packing Higher melting, boiling points and densities than s-block metals K Ca Sc Ti V Cr Mn Fe Co m. p. (°C) 63 850 1400 1677 1917 1903 1244 1539 1495 density (g cm-3 ) 0. 86 1. 55 4. 5 6. 1 7. 2 7. 4 7. 9 3 8. 9
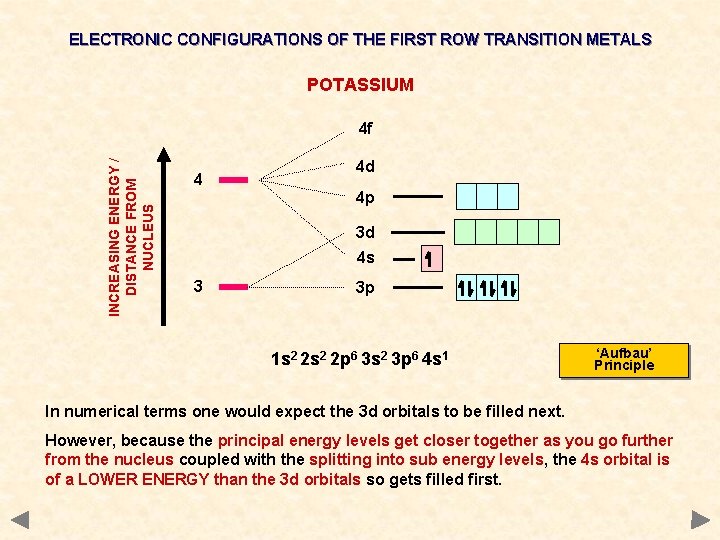
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS POTASSIUM INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 ‘Aufbau’ Principle In numerical terms one would expect the 3 d orbitals to be filled next. However, because the principal energy levels get closer together as you go further from the nucleus coupled with the splitting into sub energy levels, the 4 s orbital is of a LOWER ENERGY than the 3 d orbitals so gets filled first.
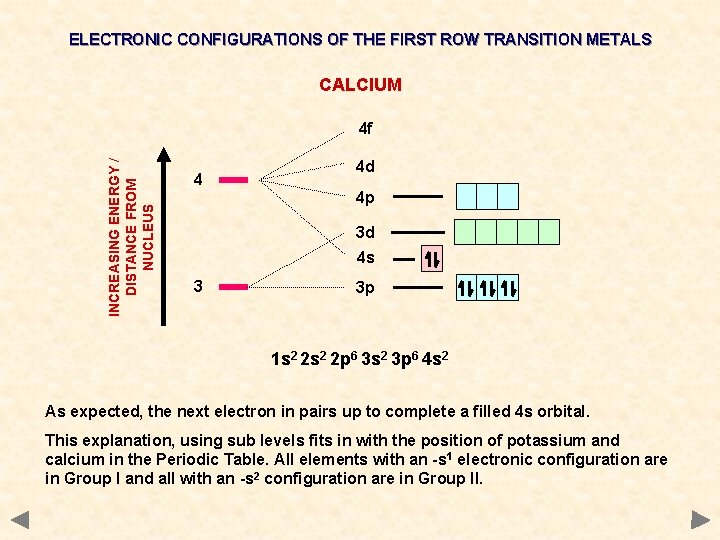
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS CALCIUM INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 As expected, the next electron in pairs up to complete a filled 4 s orbital. This explanation, using sub levels fits in with the position of potassium and calcium in the Periodic Table. All elements with an -s 1 electronic configuration are in Group I and all with an -s 2 configuration are in Group II.
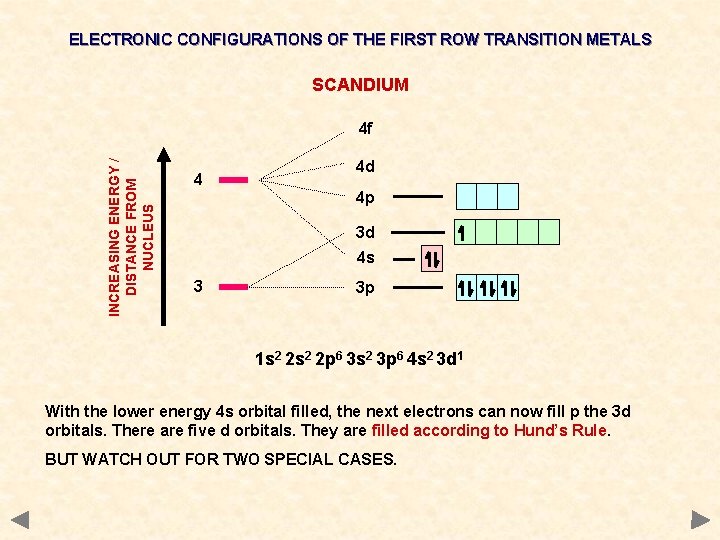
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS SCANDIUM INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 1 With the lower energy 4 s orbital filled, the next electrons can now fill p the 3 d orbitals. There are five d orbitals. They are filled according to Hund’s Rule. BUT WATCH OUT FOR TWO SPECIAL CASES.
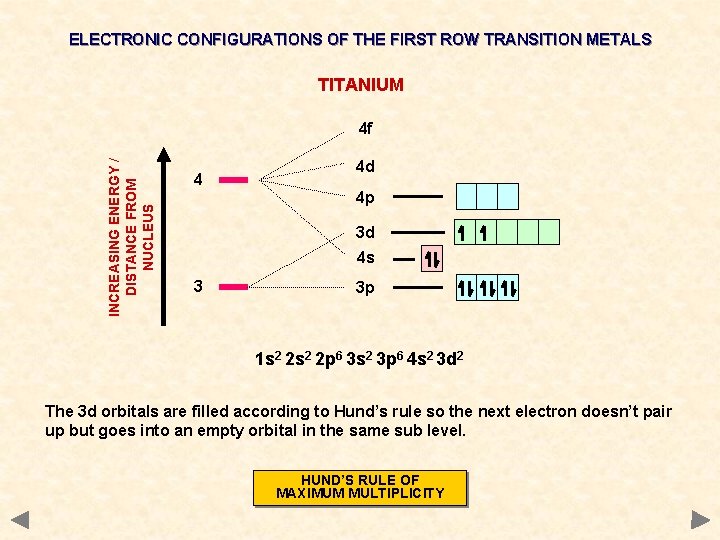
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS TITANIUM INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 2 The 3 d orbitals are filled according to Hund’s rule so the next electron doesn’t pair up but goes into an empty orbital in the same sub level. HUND’S RULE OF MAXIMUM MULTIPLICITY
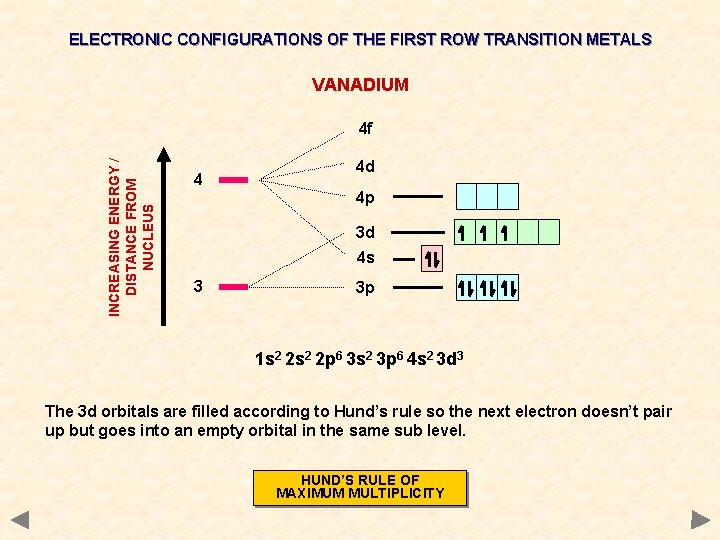
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS VANADIUM INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 3 The 3 d orbitals are filled according to Hund’s rule so the next electron doesn’t pair up but goes into an empty orbital in the same sub level. HUND’S RULE OF MAXIMUM MULTIPLICITY

ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS CHROMIUM INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 5 One would expect the configuration of chromium atoms to end in 4 s 2 3 d 4. To achieve a more stable arrangement of lower energy, one of the 4 s electrons is promoted into the 3 d to give six unpaired electrons with lower repulsion.
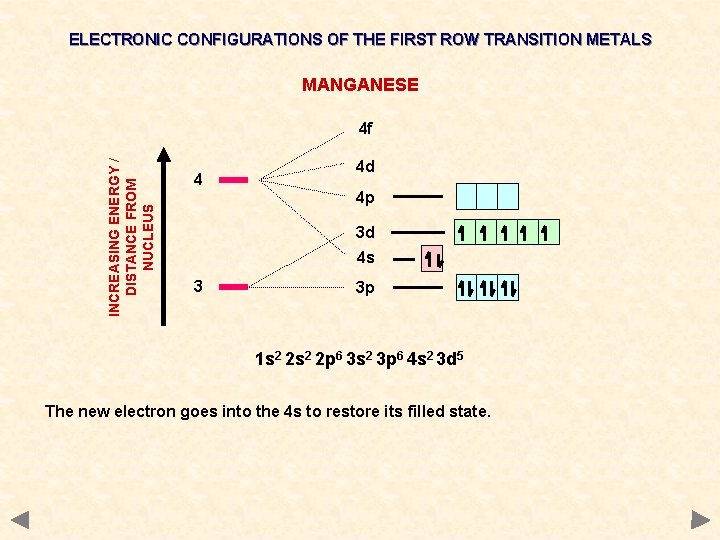
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS MANGANESE INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 5 The new electron goes into the 4 s to restore its filled state.
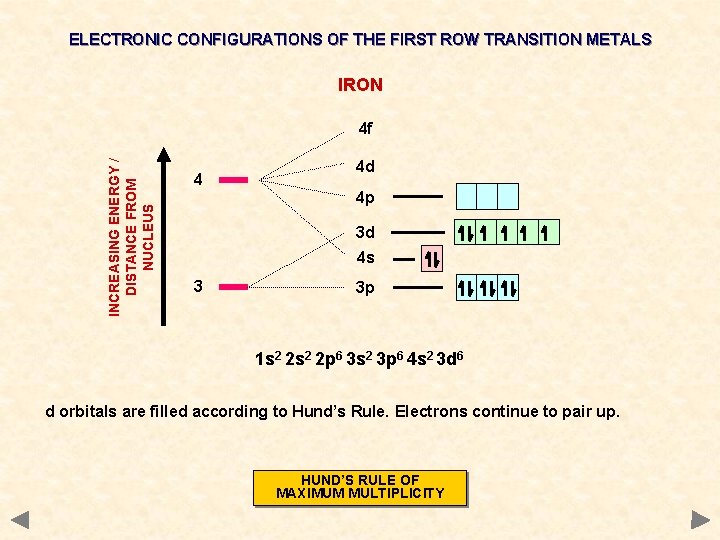
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS IRON INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 d orbitals are filled according to Hund’s Rule. Electrons continue to pair up. HUND’S RULE OF MAXIMUM MULTIPLICITY
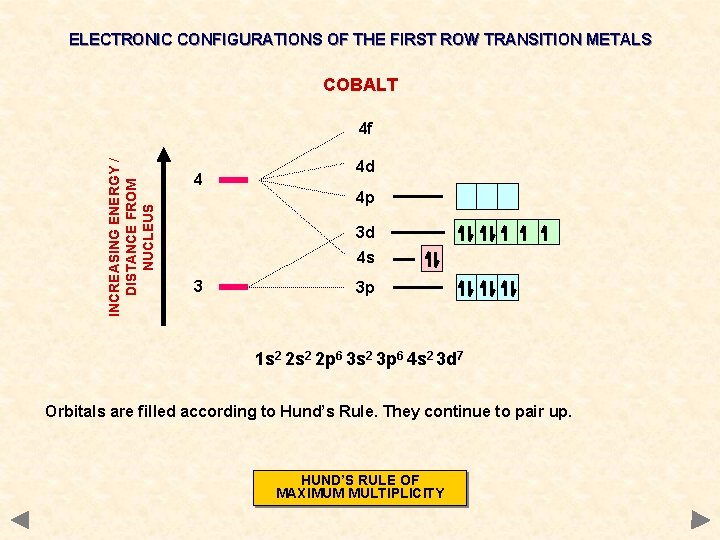
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS COBALT INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 7 Orbitals are filled according to Hund’s Rule. They continue to pair up. HUND’S RULE OF MAXIMUM MULTIPLICITY
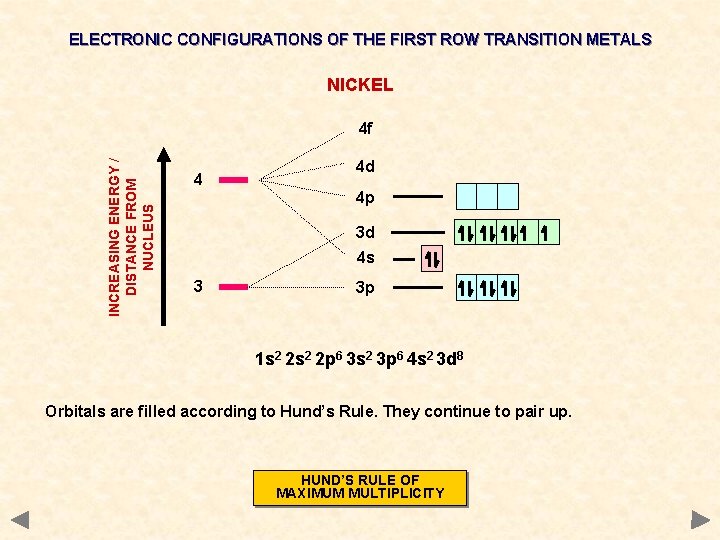
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS NICKEL INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 8 Orbitals are filled according to Hund’s Rule. They continue to pair up. HUND’S RULE OF MAXIMUM MULTIPLICITY
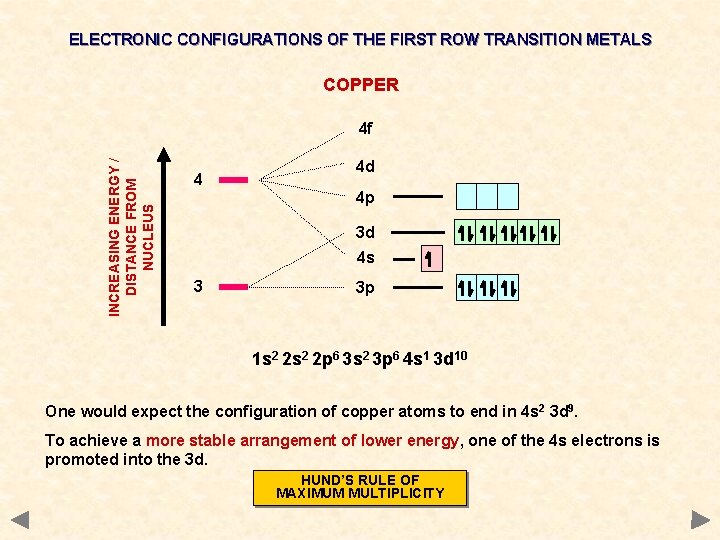
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS COPPER INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 10 One would expect the configuration of copper atoms to end in 4 s 2 3 d 9. To achieve a more stable arrangement of lower energy, one of the 4 s electrons is promoted into the 3 d. HUND’S RULE OF MAXIMUM MULTIPLICITY
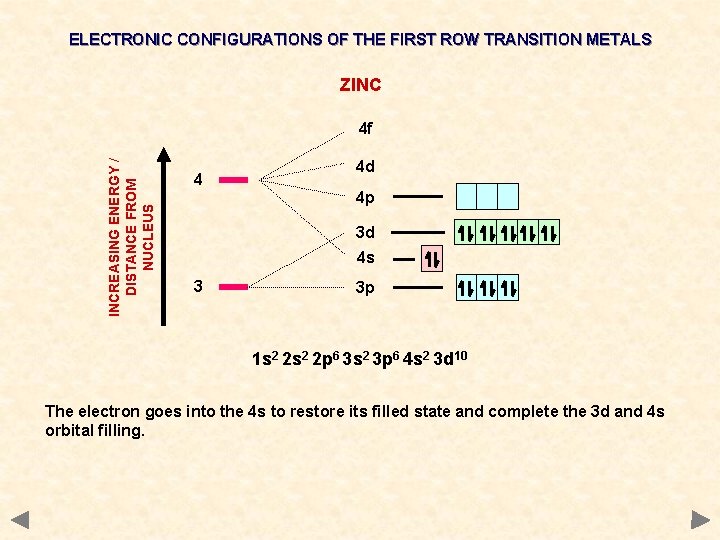
ELECTRONIC CONFIGURATIONS OF THE FIRST ROW TRANSITION METALS ZINC INCREASING ENERGY / DISTANCE FROM NUCLEUS 4 f 4 4 d 4 p 3 d 4 s 3 3 p 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 The electron goes into the 4 s to restore its filled state and complete the 3 d and 4 s orbital filling.
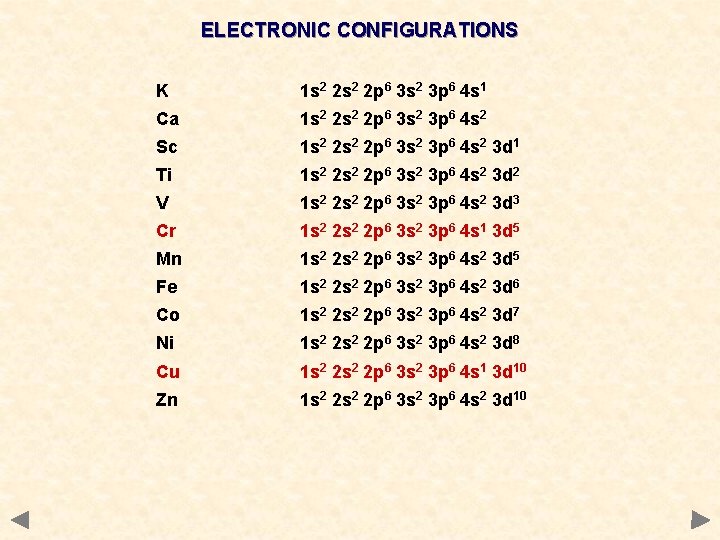
ELECTRONIC CONFIGURATIONS K 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 Ca 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 Sc 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 1 Ti 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 2 V 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 3 Cr 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 5 Mn 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 5 Fe 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 Co 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 7 Ni 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 8 Cu 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 10 Zn 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10

VARIABLE OXIDATION STATES Arises from the similar energies required for removal of 4 s and 3 d electrons When electrons are removed they come from the 4 s orbitals first Cu 1 s 2 2 p 6 3 s 2 3 p 6 3 d 10 4 s 1 Cu+ 1 s 2 2 p 6 3 s 2 3 p 6 3 d 10 Cu 2+ 1 s 2 2 p 6 3 s 2 3 p 6 3 d 9 Ti 1 s 2 2 p 6 3 s 2 3 p 6 3 d 2 4 s 2 Ti 2+1 s 2 2 p 6 3 s 2 3 p 6 3 d 2 Ti 3+1 s 2 2 s 2 p 6 3 s 2 3 p 6 3 d 1 Ti 4+1 s 2 2 p 6 3 s 2 3 p 6

COLOURED IONS A characteristic of transition metals is their ability to form coloured compounds Theory 1) Ions with a d 10 (full) or d 0 (empty) configuration are colourless 2) Ions with partially filled d-orbitals tend to be coloured 3) This is caused by the ease of transition of electrons between energy levels 4) Energy is absorbed when an electron is promoted to a higher level The frequency of light is proportional to the energy difference Ions with But Colorless: Not transition metal d 10 (full) Cu+, Ag+ Zn 2+ d 0 (empty) Sc 3+ Ti+4 compounds are white, nevertheless is a transition metal because of Ti+2 and Ti+3 which are colored
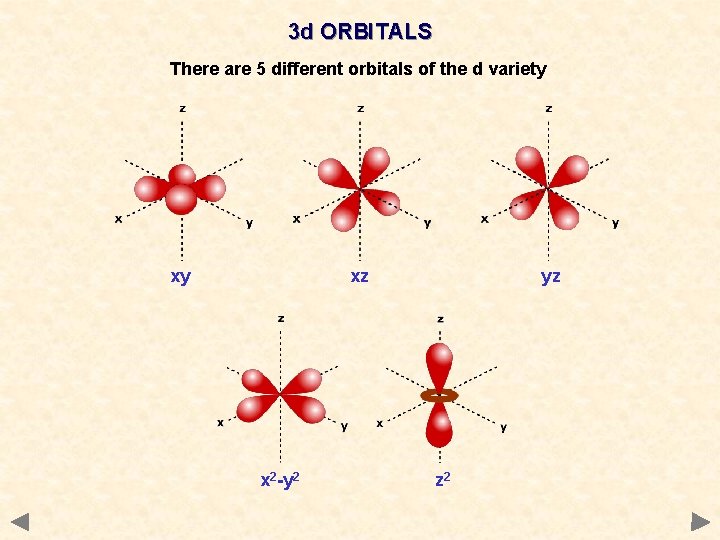
3 d ORBITALS There are 5 different orbitals of the d variety xy xz x 2 -y 2 yz z 2
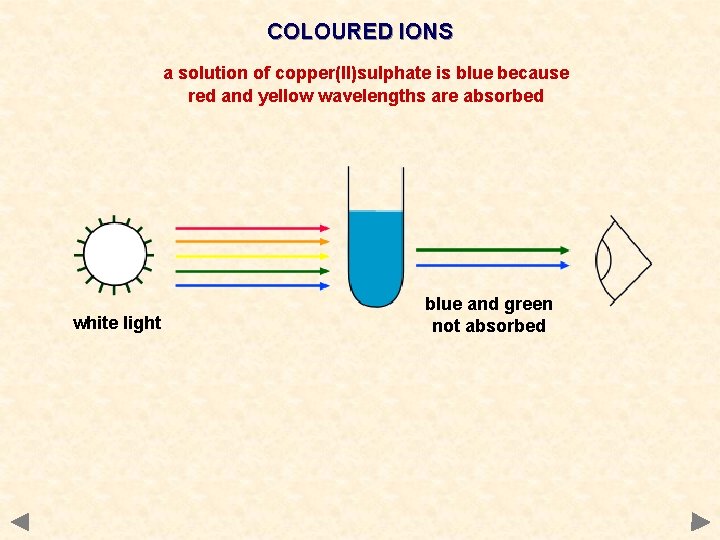
COLOURED IONS a solution of copper(II)sulphate is blue because red and yellow wavelengths are absorbed white light blue and green not absorbed
- Slides: 20