An Introduction to Statistical Thermodynamics Kinetic Model of





















- Slides: 28

An Introduction to Statistical Thermodynamics

Kinetic Model of Gas molecules typically collide with a wall or other molecules about once every ns. Each molecule has a different speed, si. For N molecules, the root-meansquare (rms) speed, c, is: ( ) If n moles of gas, with a molar mass of M, are in a volume of V, then the pressure is: Total mass

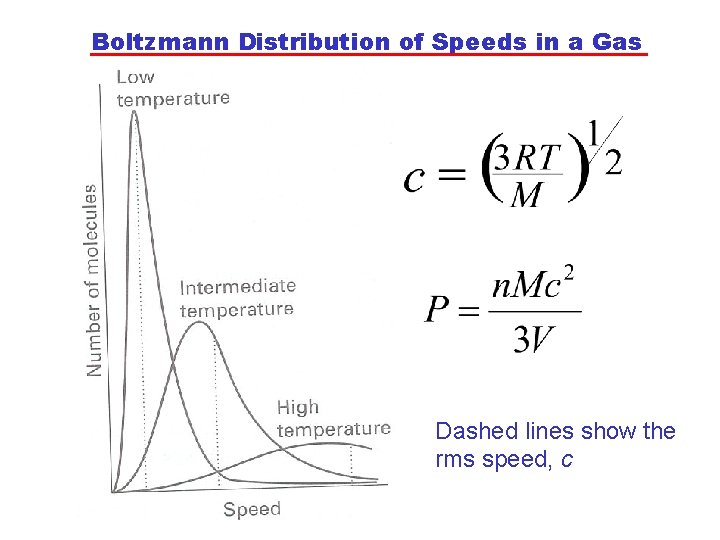
Kinetic Model of Gas As PV=n. RT for an ideal gas, we can solve for c as a function of T: There is a proportionality between the rms speed of molecules and the square root of temperature. Note: There is a distribution of speeds, which is described by the Boltzmann distribution.

Boltzmann Distribution of Speeds in a Gas Dashed lines show the rms speed, c

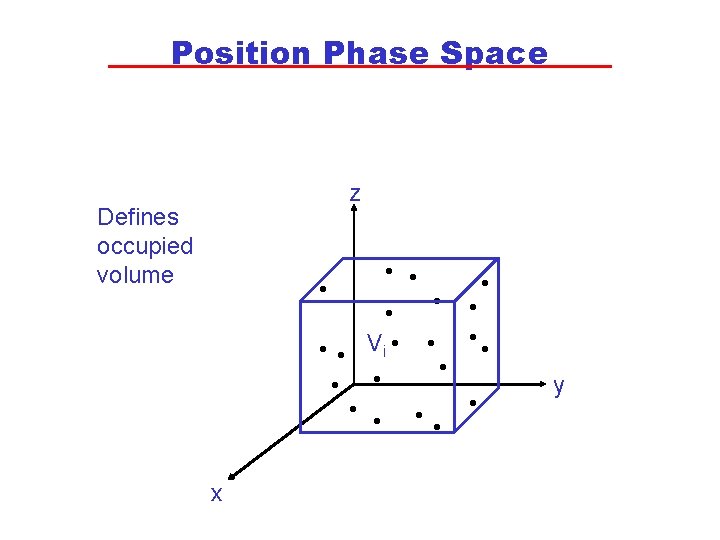
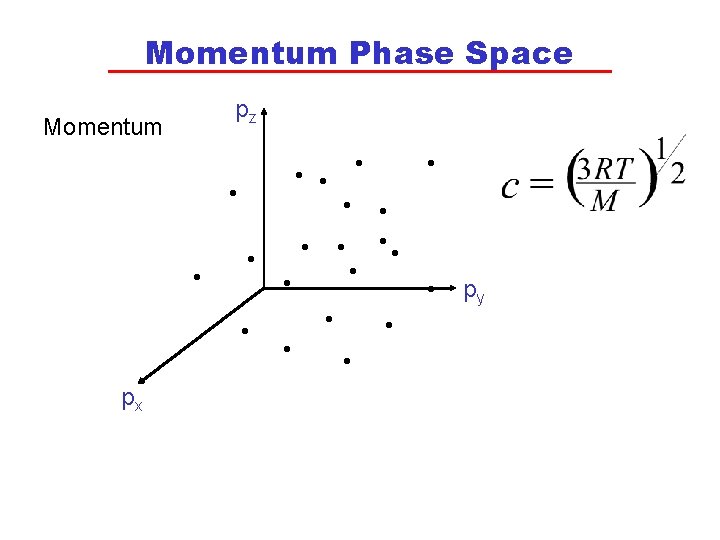
Statistical Approach • Statistical thermodynamics uses a mathematical description of the distribution of particle positions and speeds to calculate the state variables for a system and to obtain values of the state functions, such as U and S. • The position of each molecule is given in “phase space” by the co-ordinates of x, y and z. • The momentum (a vector that is the product of mass and velocity) of each molecule is represented in “phase space” by three vectors: px, py and pz. • Neither position nor momentum can both be known with certainty but are only known within a range in phase space.

Position Phase Space z Defines occupied volume • • • Vi • • • x y

Momentum Phase Space pz Momentum • • • px • • • • • py

Statistical Approach • The positions of molecules in phase space can be used to determine the bulk state variables for the system. • Obvious correlation between positions and volume, V. • Pressure, P can be related to the rms speed, c, which is related to momentum of all the molecules in the system. • Temperature, T is related to the square of the rms speed, which is likewise related to the momentum. • Several different arrangements of molecules in phase space can correspond to the same bulk state (P, V, T). • The number of arrangements of molecules for a particular state of the system is called thermodynamic probability, .

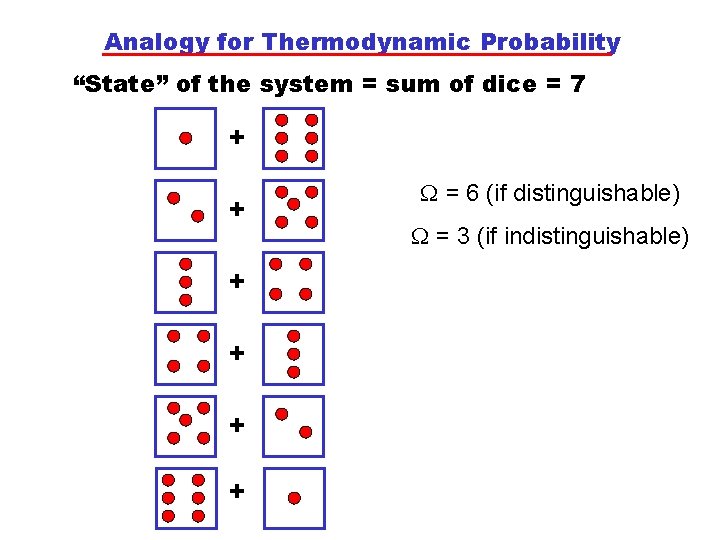
Analogy for Thermodynamic Probability “State” of the system = sum of dice = 7 + + + = 6 (if distinguishable) = 3 (if indistinguishable)

Analogy for Thermodynamic Probability “State” of the system = sum of dice = 2 + =1 (indistinguishable) • Conclude that a “state” of 7 is more likely to be found as it has a higher probability, . • In a “real” system, each state (Pi, Vi, Ti) will have a certain i. • As one mole contains ~1023 molecules, tends to be a very large number! • The equilibrium state of a system is most likely and hence must have the largest .

Relationship between and S • In an isolated system, or thermodynamic Universe, molecules will spontaneously move from a low state to a high state. • Thus the for the system approaches a maximum. • Previously we saw that in a thermally-isolated system (or t. d. Universe), S approaches a maximum. • CONCLUDE that there must be some proportionality between and S.

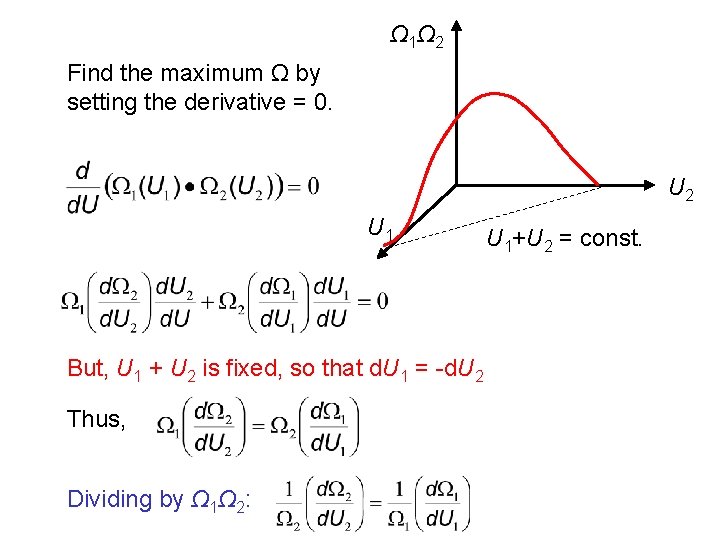
Two systems with differing internal energies (U 1 and U 2) and differing Ω are placed into contact. Together they define a thermodynamic universe. U 1 Ω 1(U 1) U 2 Ω 2(U 2) We know that the two systems will reach thermodynamic equilibrium over time. Total energy for the t. d. universe: U = U 1 + U 2 Ω for the universe is Ω = Ω 1Ω 2 The equilibrium macroscopic state will correspond to the maximum Ω (i. e. most likely state). What will be the values for Ω 1 and Ω 2 at equilibrium?

Ω 1Ω 2 Find the maximum Ω by setting the derivative = 0. U 2 U 1 But, U 1 + U 2 is fixed, so that d. U 1 = -d. U 2 Thus, Dividing by Ω 1Ω 2: U 1+U 2 = const.

We note that If then d (ln Ω 1) = d (ln Ω 2 ) Substituting, we find the condition for thermal equilibrium that is satisfied when Ω is a maximum: We say that System 1 and 2 are at the “same temperature” (T 1 = T 2) and have the same thermal energy, k. T. Comparing to the condition above, we write that: k is the Boltzmann constant.

Classical Definition of Temperature and the Boltzmann Equation Recall the differential form of the First Law of Thermodynamics: d. U = d. Q + d. W = Td. S - Pd. V Now find the partial derivative w. r. t. S while holding V constant: Giving us a new definition of T We recall that such that Comparing the two definitions for temperature, we see that: S = k lnΩ

Ludwig Boltzmann (1844 -1906)

W = thermodynamic probability,

Boltzmann Equation Relates the microscopic state of a system (molecular level) to its macroscopic state. Voted one of the ten most beautiful equations in a survey by Physics World! Significance: An increase in the entropy of a system corresponds to it going from a less probable state to a more probable state.

Calculating the Thermodynamic Probability Imagine there are N=10 positions in phase space, and you are putting j molecules into these positions. If the molecules are indistinguishable, there is only one way to arrange 10 molecules, so = 1! Here we are assuming single occupancy of sites. In position/momentum space, multiple occupancy is allowed!

How many ways are there to arrange four indistinguishable molecules on ten sites? If the particles were distinguishable, there would be 10 x 9 x 8 x 7 ways. That is, the first particle would have 10 sites to choose from; the second particle would have 9 sites, etc. Generalising for j particles on N sites: If the particles are indistinguishable, the number or arrangements are reduced by j!

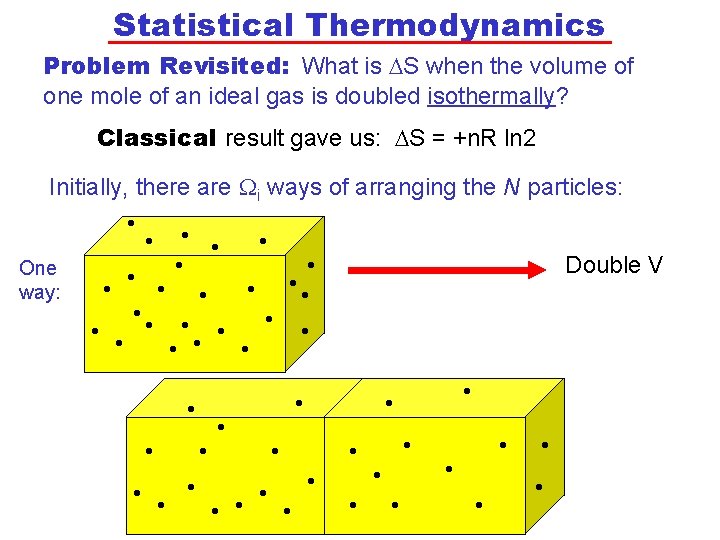
Statistical Thermodynamics Problem Revisited: What is DS when the volume of one mole of an ideal gas is doubled isothermally? Classical result gave us: DS = +n. R ln 2 Initially, there are i ways of arranging the N particles: • • Double V One • • • • way: • • • • • • • • • • •



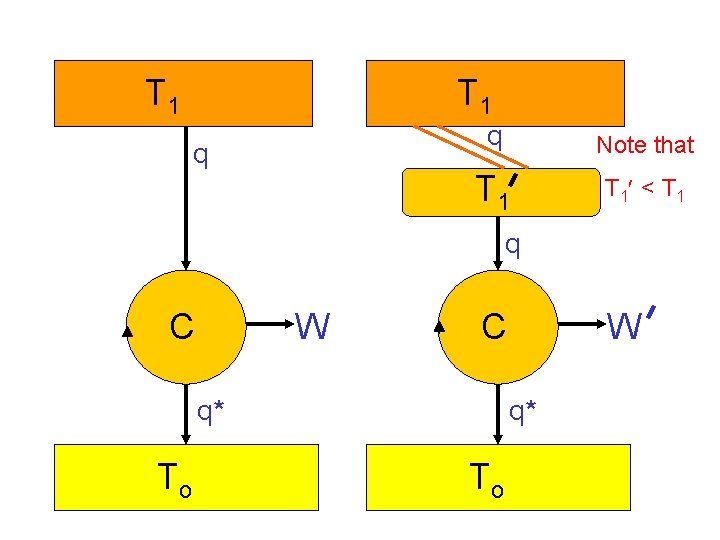
T 1 q q Note that T 1 < T 1 q C W C q* To W q* To

Question: How much less work are we doing as a result of the “extra” heat flow?


Boltzmann Equation For a thermodynamic Universe, we saw that: SUniverse The Universe can consist of several parts (systems and reservoirs) each with their own entropy, so: SUniverse = S 1 + S 2 + S 3 +. . . Probabilities are multiplicative, so: SUniverse = S 1 + S 2 + S 3 +. . . 1 2 3…. But, since S 1 1 and S 2 2 , etc. , then: S 1 + S 2 + S 3 +. . . 1+ 2+ 3+. . . How can this be? Boltzmann thought about this problem….

Boltzmann Equation We note a property of logarithms: ln 1+ ln 2+ ln 3+. . . ln ( 1 2 3…. ) The conditions are therefore satisfied if S ln . Then, S 1 + S 2 + S 3 +. . . ln 1+ ln 2+ ln 3+. . . but also S 1 + S 2 + S 3 +. . . ln( 1 2 3…. ), as required. Boltzmann derived a relationship in which k is the constant of proportionality: S = k ln with k = 1. 38 x 10 -23 JK-1. Voted one of the ten most beautiful equations in a survey by Physics World!