An Introduction to Radiation Protection Nuclear Reactor Health




































- Slides: 42

An Introduction to Radiation Protection Nuclear Reactor Health Physics An Introduction to Radiation Protection 6 e 6 e © 2012 Martin, Cole/Hodder. Education An Introduction to Radiation Protection © 2012 Martin, Harbison, Beach, Cole/Hodder

Introduction • Nuclear fission, chain reactions, criticality • Fission products, transuranic elements, neutron activation products • Power reactor systems, components of reactor • Sources of radiation on a reactor • Sources of radioactive contamination on reactor • Research reactor radiological hazards • Fuel storage ponds • Fuel cycle facilities and storage of spent fuel An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

History of Nuclear Fission • Nuclear fission – discovered in 1938 - first fission reactor operated in Chicago in 1942 • Rapid development of nuclear power plants in 1960 s and 1970 s: slowed down from 1980 s onwards • At end of 2011, 435 nuclear plants in operation, 61 under construction, 156 planned and 343 proposed • Special issues in relation to nuclear power: – protection of operators and maintainers – treatment and disposal or storage of radioactive waste – risks to people and environment from potential accidents An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

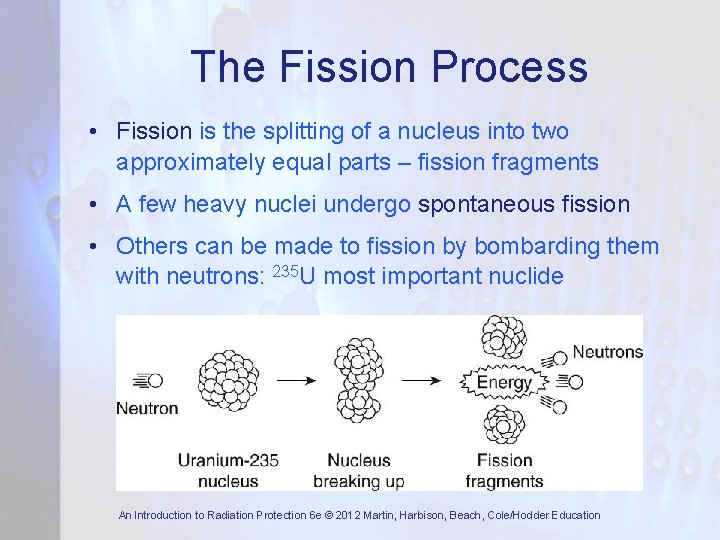
The Fission Process • Fission is the splitting of a nucleus into two approximately equal parts – fission fragments • A few heavy nuclei undergo spontaneous fission • Others can be made to fission by bombarding them with neutrons: 235 U most important nuclide An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Fission Neutrons • Fission fragments are so unstable that they give off neutrons, between 2 and 4 per fission • Most fission neutrons are emitted almost instantaneously - prompt neutrons • Some are released seconds or even minutes after fission - delayed neutrons • The release of neutrons in fission results in – the possibility of a chain reaction – the production of transuranic elements, e. g. plutonium – the activation of structural and other materials An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Chain Reactions • Of the 2 – 4 neutrons produced per fission: – some are captured in non-fission reactions – minimized by excluding materials with high ncapture cross-section – some escape from core (leakage) – minimized by increasing size of core and putting neutron reflector around it – remainder are available to cause further fissions An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Criticality • An assembly of fissile material is said to be critical if: nf = nabs + nesc + 1 nf is number of neutrons from each fission nabs is number of neutrons absorbed nesc is number of neutrons escaping • Critical mass: minimum mass of fuel needed to sustain a chain reaction An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

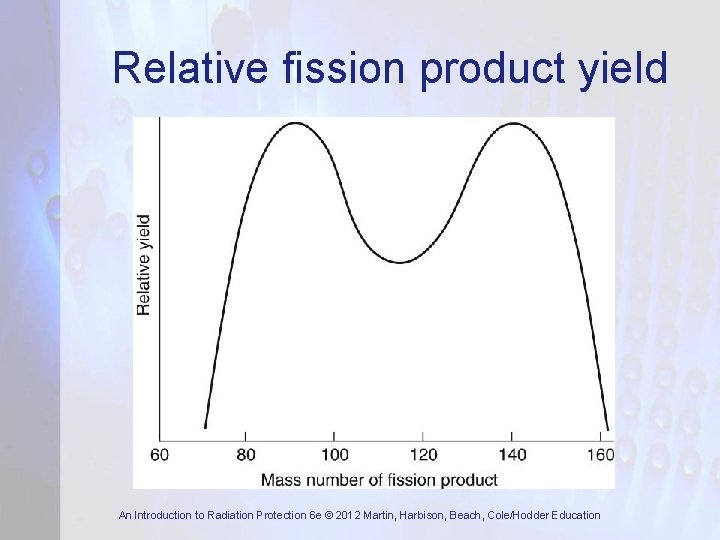
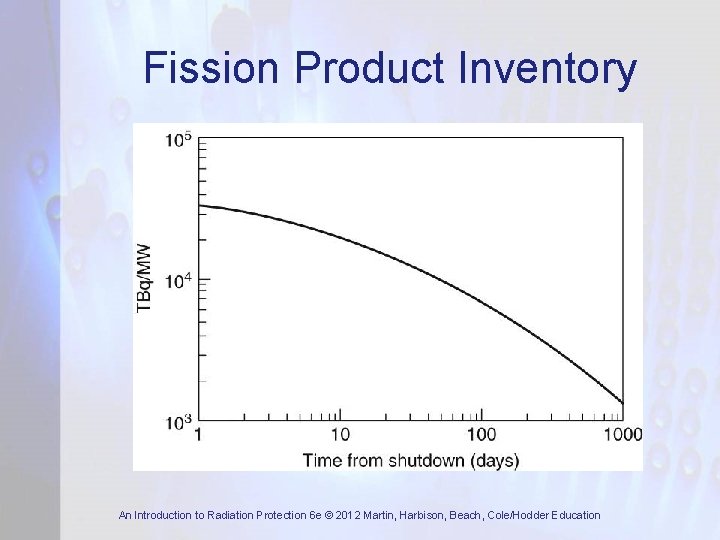
Fission Products • In fission, atoms split in many different ways • Fission produces about 300 different nuclides – most are neutron-rich and decay by β-emission • Double-peak distribution in mass number of fission products with masses around 97 and 135 most likely • Fission product half-lives vary from a fraction of sec up to very many years • Fission product inventory builds up in fuel over its period of irradiation in reactor An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Relative fission product yield An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Fission Product Inventory An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Transuranic Elements • Successive capture reactions and β-decays result in the generation of transuranic elements (elements beyond U, such as Pu, Am and Cm) • Neutron capture in 238 U is very important 238 U(n, γ) 239 U β 239 Np β 239 Pu • This process converts 238 U (which is not significantly fissile) into 239 Pu which is fissile • However, 239 Pu is very long-lived (T½ = 24, 300 years) and, unlike U, is also highly radiotoxic An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Activation Products • Activation products result from neutron capture in reactor structural materials and in coolant • Many different activation products, e. g: 60 Co T½ = 5. 22 y, emits 2 energetic γ-rays 59 Fe T½ = 45 days, decays by β emission 63 Ni T½ = 96 y, decays by β emission 16 N T½ = 7. 2 s, emits 6. 1 Me. V γ-rays An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

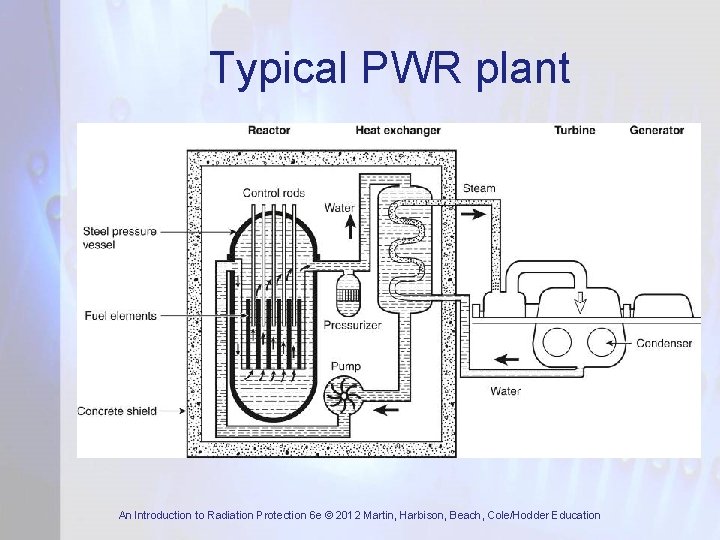
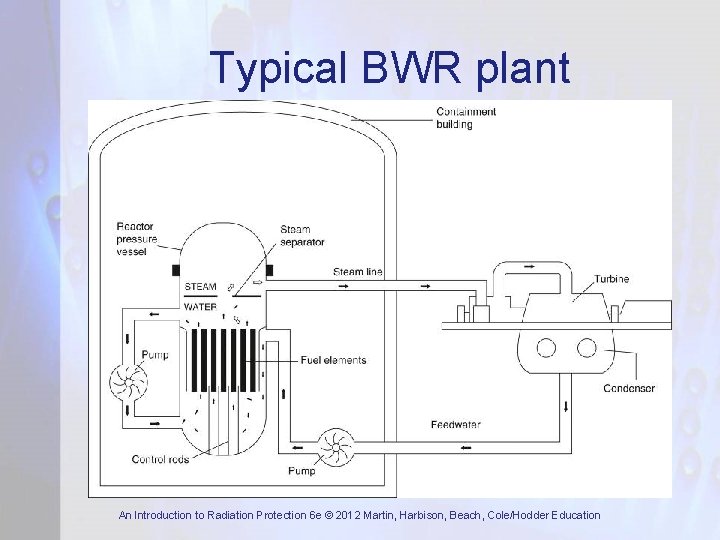
Power Reactor Systems • Majority of commercial power reactors are light water reactors (LWRs). They are of two types: – Pressurised water reactors (PWR): very high cooling water pressure prevents bulk boiling – Boiling water reactors (BWR): cooling water boils and steam is passed directly to turbines • CANDU reactor, developed in Canada, uses heavy water (D 2 O) as coolant and moderator • In UK, two generations of gas (CO 2) - cooled reactors were developed, Magnox and AGR An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Possible Future Reactor Systems • Liquid-metal fast breeder reactors (LMFBRs) – Research and development over more than 50 y – Original concept - breed enough Pu, by neutron capture in 238 U, to meet ongoing fuel needs – Development has slowed because of high costs, safety concerns and current plentiful supply of U • High-temperature gas-cooled reactors (HTGRs) use helium gas to cool ceramic U fuel. Present many novel engineering/ radiological challenges An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

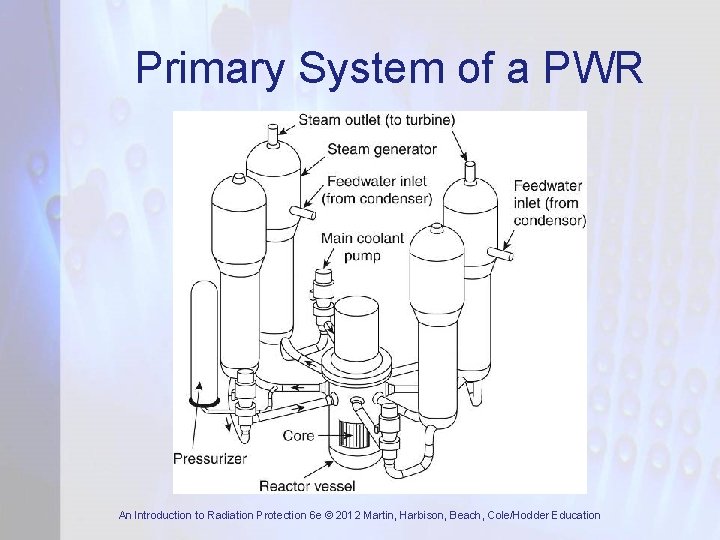
Primary System of a PWR An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Typical PWR plant An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Typical BWR plant An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Basic Reactor Components • Core which contains the fissile material • Control system to control the fission rate • Moderator to slow down the fast neutrons • Cooling system to remove heat generated by fission • Radiation shield (biological shield) to ensure radiation doses to plant workers and members of public are acceptable An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Reactor Core • Contains fuel assemblies or fuel elements • Fuel element consists of fissile material in a metal can or covered by metal cladding • Fissile material is normally U • Natural U is 0. 7% 235 U and 99. 3% 238 U • Most power reactors use enriched U • Moderator – reduces energy of fission neutrons to thermal – where fission occurs An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Control System • Criticality and power level are controlled by control rods made from materials of high neutron absorption cross-section (B or Cd) • Control system is based on detectors around core which measure neutron flux • If neutron flux exceeds a preset value or is increasing too rapidly detectors sense it and control rods are inserted automatically An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Cooling System • Energy released during fission causes temperature rise in fuel and cladding • This heat is removed by the coolant and used to raise steam to drive turbines • In LWRs the coolant is water while in gascooled reactors it is CO 2 or He • Most coolants become radioactive to some extent so integrity of cooling system needs to be very high An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Biological Shield • To attenuate n and γ radiation from core and cooling system to acceptable levels • Lead, concrete, iron, water and polythene • For power reactors the required factor of reduction may be ~ 108 to 109 • Weaknesses can lead to streaming paths • Primary shield around reactor core • Secondary shield around cooling system An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Reactor Refuelling • Only a small percentage (~ 0. 3%) of the U atoms in a fuel element are burned up • When a fuel element reaches its required burn-up it must be removed and replaced • PWR and BWR are refuelled during shutdowns, at intervals of 1 – 2 years • Other reactors such as CANDU, RBMK and AGR can be refuelled on-load An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Radiation Hazards from Reactors • Reactors present a lesser radiation hazard when operating than when shutdown • During operation, shielding and operational controls ensure acceptable radiation levels at working positions • During shut-down, exposure control is more difficult because of need to carry out nonroutine jobs on radioactive systems • Reactor accidents will be dealt with later An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Sources of Radiation An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Radiation from Core • Fission neutrons – initially fast but neutrons of all energies emerge from surface of biological shield • Fission γ-rays, energies from 0. 25 - 7 Me. V • Fission product decay γ-rays – emitted for many years after reactor shutdown • γ-rays from neutron capture reactions in reactor structural materials and shielding • Decay γ-rays, from activated materials An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Radiation from Coolant • Decay γ-rays from coolant activation products: 16 N(T½ = 7. 2 s, 6. 1 Me. V γ-rays) 41 Ar (T½ = 1. 8 h, 1. 29 Me. V γ-ray) • Decay γ-rays from activated corrosion products: in LWRs there is build-up of CRUD in coolant system • FP contamination, U contamination of fuel element surfaces, U impurity in fuel cladding or FP release from damaged fuel An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Contamination Hazards from Reactors • Most γ-emitters are also β-emitters • β radiation is important if a radioactive leak from reactor system causes contamination • Tritium (3 H) - important β-emitter in all reactor systems. T½ = 12. 3 y • Coolant leaks - release of gaseous fission products such as 88 Kr, 138 Xe, plus 41 Ar • In BWR, steam carries 16 N into turbines An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Containment of Reactor Contamination • A reactor operating at 1000 MW contains about 2 x 108 TBq of fission products • Requires three levels of containment – First: fuel can or fuel cladding – Second: boundary of primary systems, i. e. RPV and coolant system – Third: structure within which whole system is housed - “reactor containment” An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Hazards during Reactor Shutdown • External radiation hazard – to personnel working on primary system due to radioactivity within the system. Dose rate falls rapidly over first 24 h after shut-down. On reactors with corrosion problems, dose levels of 10 -100 m. Sv/h can arise • Contamination hazard - from leakage of reactor coolant - normally contains fission products and activated corrosion products An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Research Reactors • About 240 research reactors worldwide, fuel enrichment generally higher (~ 20%) than power reactors (3 – 5%) • Many applications, including materials testing, basic research, nuclear medicine and production of radioactive sources • Main radiological problems usually arise from the experimental or test equipment An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Fuel Storage Ponds • Spent fuel rods generate considerable heat and intense radiation. Generally stored in fuel storage pond at the reactor for some months or years to allow radioactive decay • Storage ponds pose two special hazards: – Criticality - fissile material in pond must not be allowed to “go critical” so fuel elements are stored in a safe configuration and their movements in the pond are strictly controlled – Loss of shielding - pond water loss must be prevented and fuel handling accidents safeguarded against An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Pond Instrumentation • Three types of instrument are used to give warning of hazardous conditions: – Installed γ monitors: at least three instruments are needed to measure the γ-ray dose rate from the fuel: must not be shielded from pond surface – Pond water counter: continuously monitors pond water for β activity and gives early warning of damaged fuel – Air monitor: continuous airborne particle monitor to warn of high airborne contamination An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Nuclear Fuel Cycle • Front-end operations – mining, extraction of uranium, uranium enrichment and fuel fabrication to produce fuel for reactors • Back-end operations – all the activities necessary to ensure safe management of irradiated fuel and its associated radioactive wastes An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Uranium Mining • Uranium ore is obtained from both underground mining and open-pit working • Main radiological issue – airborne radon and its decay products in the mine atmosphere: controlled by ventilation • Radiation doses to underground mine workers are typically ~ 10 m. Sv per year • Open pit operations, worker doses small, ~ 2 - 3 m. Sv per year, mostly external An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Uranium Enrichment • Enrichment - concentration of 235 U is increased from its natural level of 0. 7% to about 3. 5% (for most power reactors) • Enrichment process - uses gaseous UF 6 • Two main enrichment processes - gaseous diffusion and gas centrifuge • Main safety issue - high chemical toxicity of UF 6. Radiation not a significant issue An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Fuel Fabrication • In first step, UF 6 is converted into uranium dioxide (UO 2) powder • UO 2 powder then fabricated into pellets and sintered to create ceramic pellets • Pellets are ground, stacked in a metal alloy can which is filled with He and sealed • Contamination control and shielding are not significant issues in fuel fabrication • Doses to workers generally low, ~ 1 m. Sv/y An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Fuel Reprocessing (1) • Spent fuel is either stored or transported to a fuel reprocessing plant where it is usually held in a cooling pond for ~ 5 y • Steps in reprocessing – dismantling and shearing of fuel pins; – dissolution in nitric acid; – chemical separation of resulting raffinate into unused U, Pu and highly-active waste An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Fuel Reprocessing (2) • Reprocessing plant - much greater radiological challenge than a reactor • Intensely radioactive solutions require multilevel containment and thick shielding • Remote operations and maintenance • Containment of Pu stream vital • Highly-active waste stream, special issues • Operator exposures, now only few m. Sv/y An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Long-Term Storage of Irradiated Fuel • Wet storage - With good water chemistry, fuel cladding should retain its integrity for decades. Ponds require active monitoring, cooling, water treatment and ventilation. • Dry storage - in casks which provide shielding and high-integrity containment. Heat is dissipated by conduction and natural convection - passive safety. • Operator doses should be low for either. An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Summary (1) • Fission - splitting of nucleus into two fission fragments • Neutron chain reaction - neutrons from one fission cause another fission to occur • Critical mass - smallest amount of fissile material needed for a sustained chain reaction • Reactor system - core, control rods, moderator, coolant and biological shield • Sources of radiation when operating – fission neutrons and γ-rays, fission product decay γ-rays, neutron capture γ-rays and activation product γ-rays • Shut-down sources - fission and activation product decay γ-rays An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education

Summary (2) • Contamination - may occur due to coolant leaks or maintenance operations • Containment - vital to contain fission products to prevent overexposures • Fuel storage ponds - two special hazards: loss of shielding and criticality • Fuel reprocessing plant - chemical separation of fuel into uranium, plutonium and waste streams. Severe radiological problems • Long-term storage - wet storage in ponds, good water chemistry vital. Dry storage in casks similar to transport containers. No significant radiological problems An Introduction to Radiation Protection 6 e © 2012 Martin, Harbison, Beach, Cole/Hodder Education