An introduction to Microbial Oceanography Microbial photoautotrophy Ricardo

An introduction to Microbial Oceanography Microbial photoautotrophy Ricardo M Letelier Outline: - Evolution and Diversity - Photosynthesis light and dark reactions - Measuring techniques - Photo-aclimation and adaptation - Expanding the concept of marine phototrophy - Some present and future challenges 2008 Center for Microbial Oceanography: Research and Education (CMORE) Summer Course

Global Heat Engine v Solar energy impinges on Earth unequally v Most insolation at the equator v Large temperature gradients between equator and Poles v Air & ocean movements controlled by temperature gradient v Net transference of heat from equator to poles.
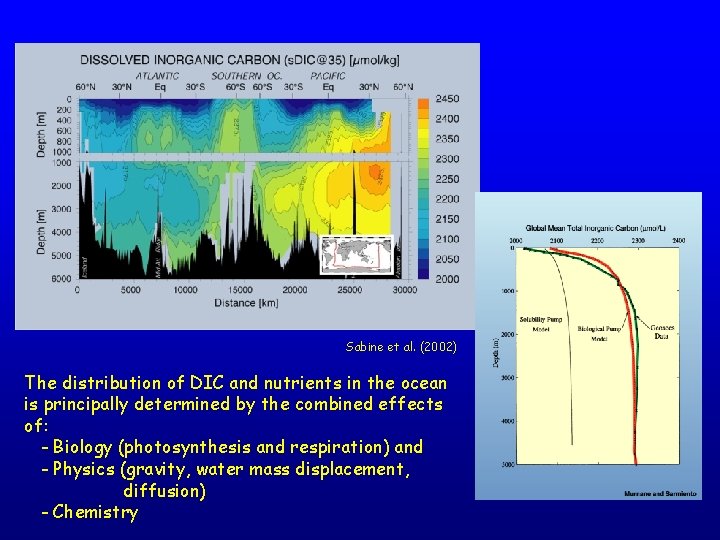
Sabine et al. (2002) The distribution of DIC and nutrients in the ocean is principally determined by the combined effects of: - Biology (photosynthesis and respiration) and - Physics (gravity, water mass displacement, diffusion) - Chemistry
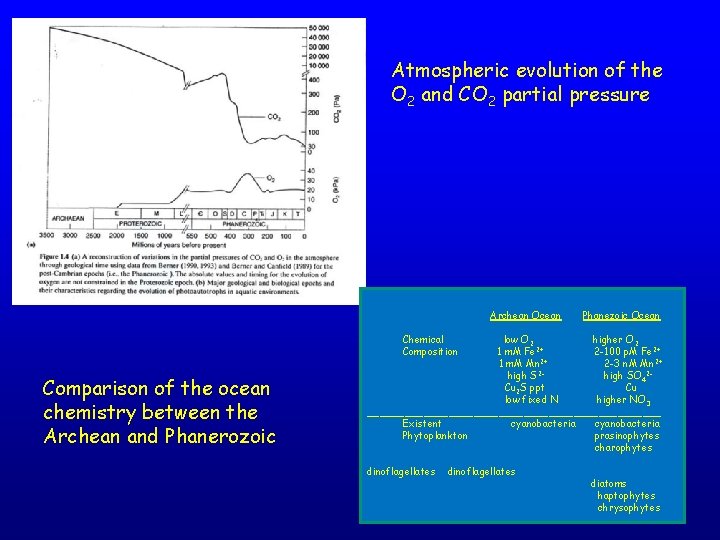
Atmospheric evolution of the O 2 and CO 2 partial pressure Archean Ocean Phanezoic Ocean Chemical Composition Comparison of the ocean chemistry between the Archean and Phanerozoic low O 2 higher O 2 1 m. M Fe 2+ 2 -100 p. M Fe 2+ 1 m. M Mn 2+ 2 -3 n. M Mn 2+ high S 2 high SO 42 Cu 2 S ppt Cu low fixed N higher NO 3 ________________________ Existent cyanobacteria Phytoplankton prasinophytes charophytes dinoflagellates diatoms haptophytes chrysophytes
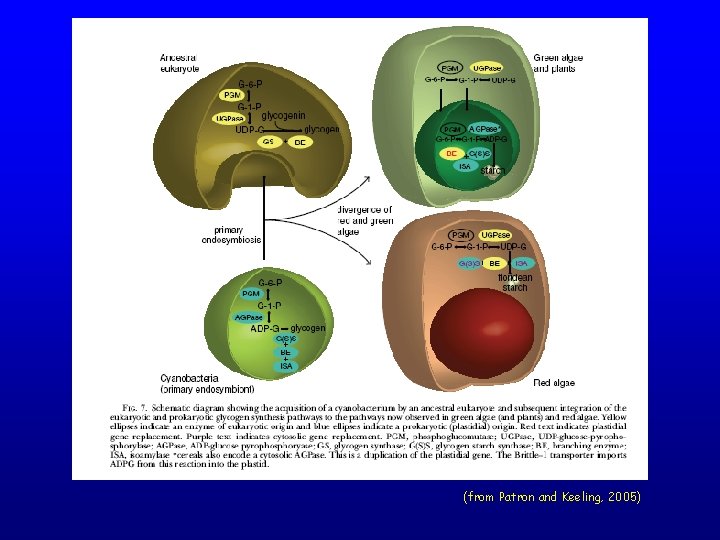
(from Patron and Keeling, 2005)

Physiological changes originated through the evolution of an aerobic environment O 2 derivatives: Protective mechanisms: singlet oxygen peroxide superoxide anion radials hydroxide radicals carotenoids (zeaxanthin) OH scavengers superoxide dismutase (Fe and/or Mn) peroxidase (Fe) catalase (Fe) Metal e- carriers Fe _______________________ Oxidation of Fe 2+, Mn 2+ and S 2 -: virtual depletion of the reduced forms by phanezoic

Subsequent evolution of photoautotrophs: Alternate transition metals used superoxide dismutase with Cu and Zn (charophyceae) Aerobic respiration (instead of anaerobic with S 2 - as terminal e- acceptor) Cyanobacteria use cytochrome c (Fe) for e- transport (in the absence of Cu) If Cu is present then synthesis of plastocyanin is induced. Aerobic respiration in eukaryotes uses cytochrome oxidase (Cu).

12 -16 major groups, 2 dominant, 4 frequent Classes Common Names Bacillariophyceae diatoms Dinophyceae dinoflagellates Prymnesiophytes coccolithophorids (Haptophyceae) Cyanophyceae cyanobacteria Prochlorophyceae prochlorophytes various nanoflagellates

Distinguishing features: sizes and shapes linear dimensions (1 m - 1 mm) cell volume (4 -5 orders of magnitude) flagella (presence, number, evenness, mastigonemes) cell wall cell organelles (shape and type) pigments storage products molecular markers (from Larkum and Vesk, 2003)

Molecular Phylogeny based on r. RNA Sequences: - Prokaryote 16 s r. RNA - organelle 16 s r. RNA - eukaryotes 18 s r. RNA & 80 s ribosomes Other Molecular techniques: - HPLC pigments - Inmunofluorescence antibodies - RNA & DNA probes - Metabolites (from Raven, J. Phycol. 2005)
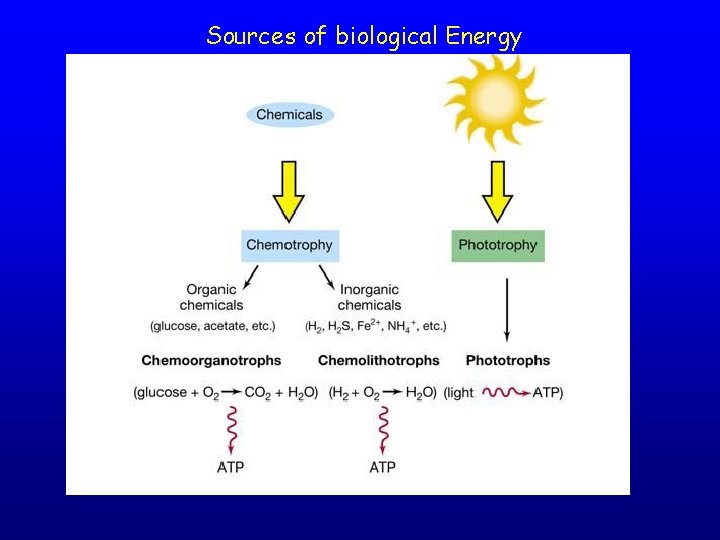
Sources of biological Energy
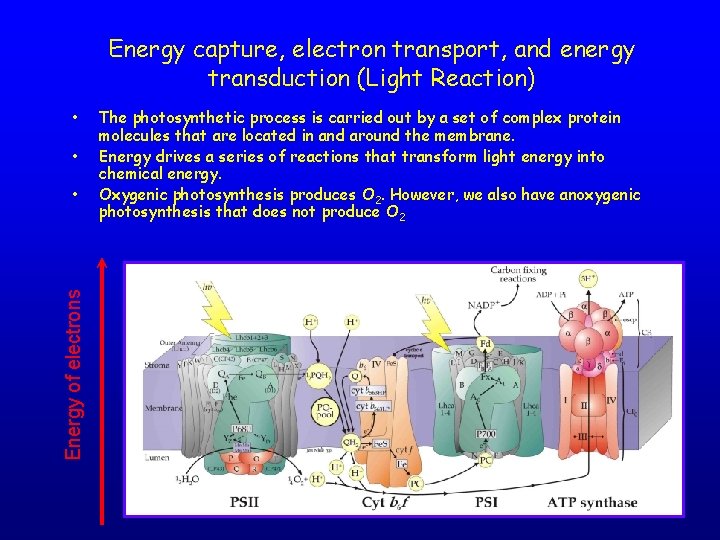
Energy capture, electron transport, and energy transduction (Light Reaction) • • Energy of electrons • The photosynthetic process is carried out by a set of complex protein molecules that are located in and around the membrane. Energy drives a series of reactions that transform light energy into chemical energy. Oxygenic photosynthesis produces O 2. However, we also have anoxygenic photosynthesis that does not produce O 2
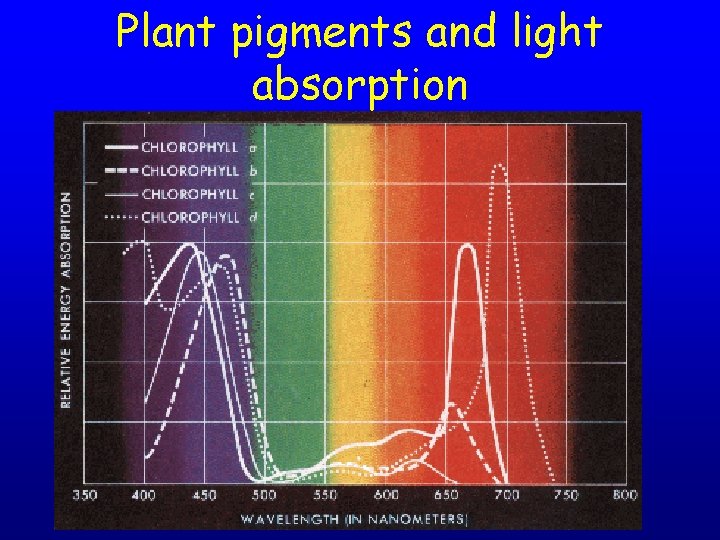
Plant pigments and light absorption
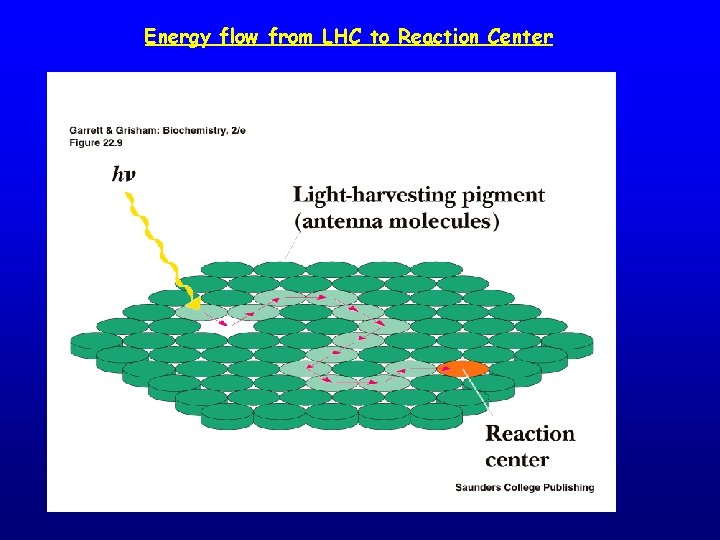
Energy flow from LHC to Reaction Center
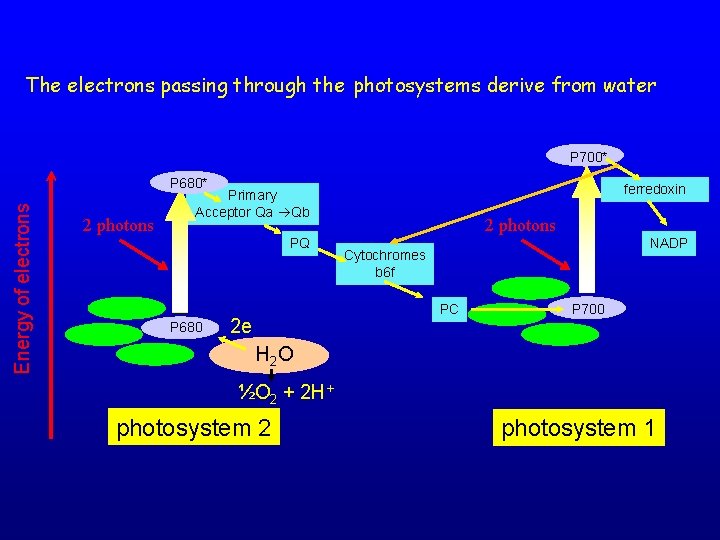
The electrons passing through the photosystems derive from water P 700* Energy of electrons P 680* 2 photons ferredoxin Primary Acceptor Qa Qb PQ P 680 2 photons NADP Cytochromes b 6 f PC 2 e P 700 H 2 O ½O 2 + 2 H+ photosystem 2 photosystem 1

Final result H 2 O + NAD(P)+ 1/2 O 2 + NAD(P)H + H+ The reaction requires: 26 Kcal per e 220 k. J per mol However, once the electron reaches the ferredoxin, the electron pathway becomes branched, enabling redox free energy to enter other metabolic pathways in the chloroplast. - transfer of electron to nitrite reductase - glutamate synthase - thioredoxin reductase - PSI cyclic electron flow - Mehler reaction (pseudo cyclic electron flow) that leads to reduction of molecular oxygen
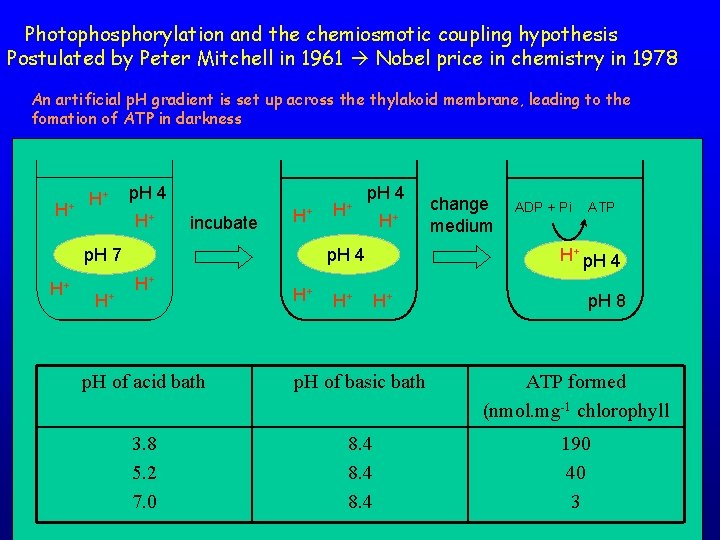
Photophosphorylation and the chemiosmotic coupling hypothesis Postulated by Peter Mitchell in 1961 Nobel price in chemistry in 1978 An artificial p. H gradient is set up across the thylakoid membrane, leading to the fomation of ATP in darkness H+ H+ p. H 4 H+ incubate H+ p. H 7 H+ H+ H+ p. H 4 H+ H+ H+ change medium ADP + Pi ATP H+ p. H 4 H+ p. H 8 p. H of acid bath p. H of basic bath ATP formed (nmol. mg-1 chlorophyll 3. 8 5. 2 7. 0 8. 4 190 40 3

The Light Reactions: • Light absorption takes place in an “Antenna” made of photosynthetic pigments • Pigments funnel energy to a central pigment (a Chlorophyll a, embedded in a photosynthetic membrane) known as the Reaction Centre (RC), • Energy is then used to liberate an electron from the RC • This electron is then transferred from protein to protein along the “Electron Transport Chain”, losing energy and moving protons across the membrane • Finally the electron is used to reduce NADP+ to NADPH • The protons are used to produce ATP via the enzyme ATPase

In the electron transfer process the redox free energy is converted in two forms of energy: i) A chemical potential (p. H gradient across the membrane) created by: - release of protons during the oxidation of water by PSII - the translocation of protons from the stroma and the lumen by the coupled reactions of PSII and the bf complex. Result Dp. H = p. Hin – p. Hout ii) An electric potential across the membrane that is created by - primary charge separation at the reaction center - charge transfer by the cytochrome b 6 f complex (these reactions transfer uncompensated charge across the photosynthetic membrane) Result Transmembrane electric potential (ΔΨ=Ψin-Ψout) Together, these two forms of energy make up the proton electrochemical potential (ΔμH+)
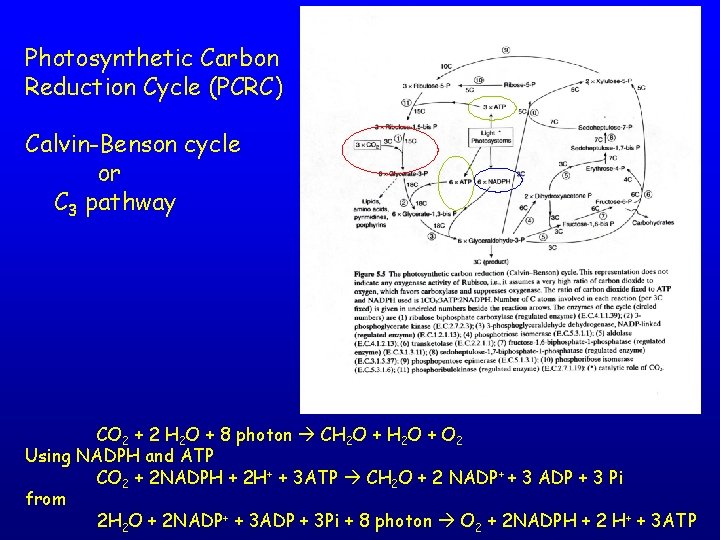
Photosynthetic Carbon Reduction Cycle (PCRC) Calvin-Benson cycle or C 3 pathway CO 2 + 2 H 2 O + 8 photon CH 2 O + O 2 Using NADPH and ATP CO 2 + 2 NADPH + 2 H+ + 3 ATP CH 2 O + 2 NADP+ + 3 ADP + 3 Pi from 2 H 2 O + 2 NADP+ + 3 ADP + 3 Pi + 8 photon O 2 + 2 NADPH + 2 H+ + 3 ATP

Ru. BPc (Ribulose biphosphate carboxylase) MW = 500, 000 Not overly adaptative for fixing CO 2 At CO 2 ~ 10 M and O 2 ~ 250 M In air equilibrated solution Ru. BPc/ Ru. BPo = 5 Ru. BPo is part of photorespiration, runs counter to reductive carboxylation, consumes energy and requires additional enzymes. Problems are intensified by depletion of CO 2 in cell and Production of O 2
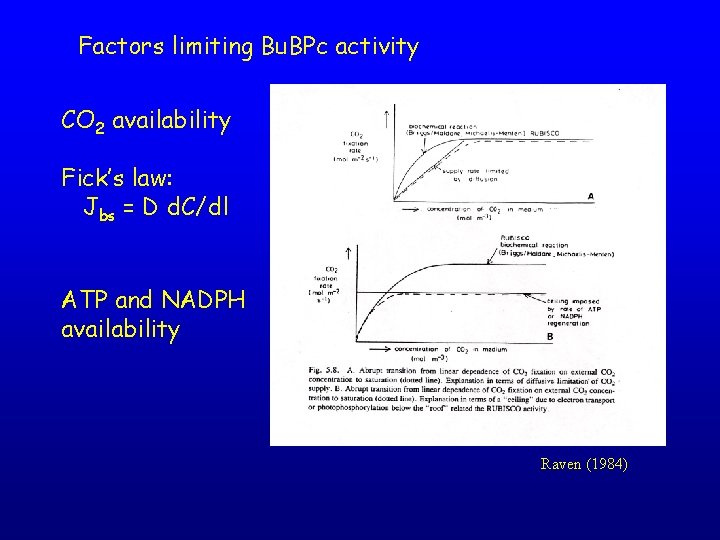
Factors limiting Bu. BPc activity CO 2 availability Fick’s law: Jbs = D d. C/dl ATP and NADPH availability Raven (1984)
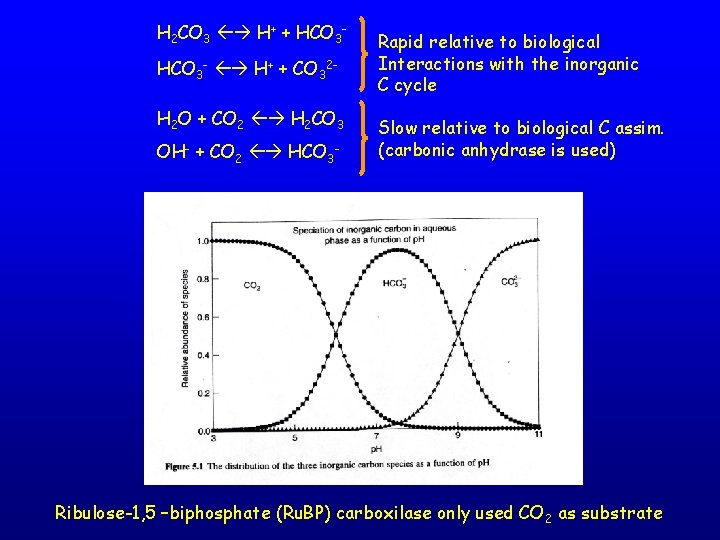
H 2 CO 3 H+ + HCO 3 - H+ + CO 32 H 2 O + CO 2 H 2 CO 3 OH- + CO 2 HCO 3 - Rapid relative to biological Interactions with the inorganic C cycle Slow relative to biological C assim. (carbonic anhydrase is used) Ribulose-1, 5 –biphosphate (Ru. BP) carboxilase only used CO 2 as substrate
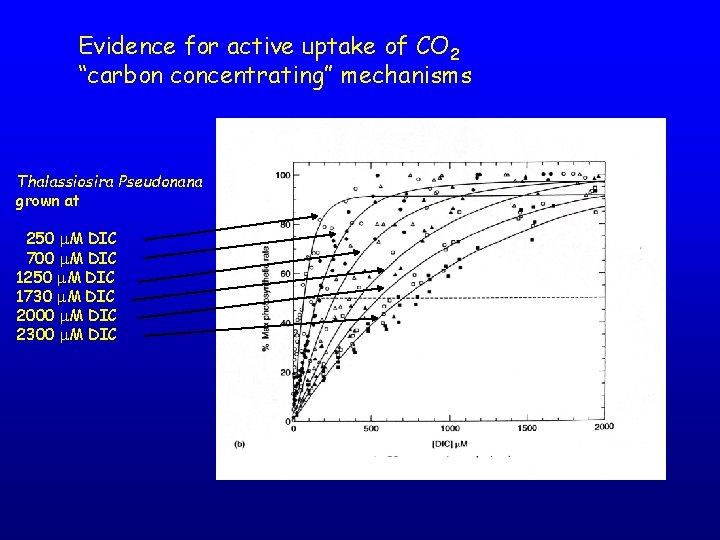
Evidence for active uptake of CO 2 “carbon concentrating” mechanisms Thalassiosira Pseudonana grown at 250 M DIC 700 M DIC 1250 M DIC 1730 M DIC 2000 M DIC 2300 M DIC

Measuring photosynthetic rates in marine microbial assemblages -Oxygen evolution in light and dark incubation bottles -14 C or 13 C incorporation rates -Variable Fluorescence approach
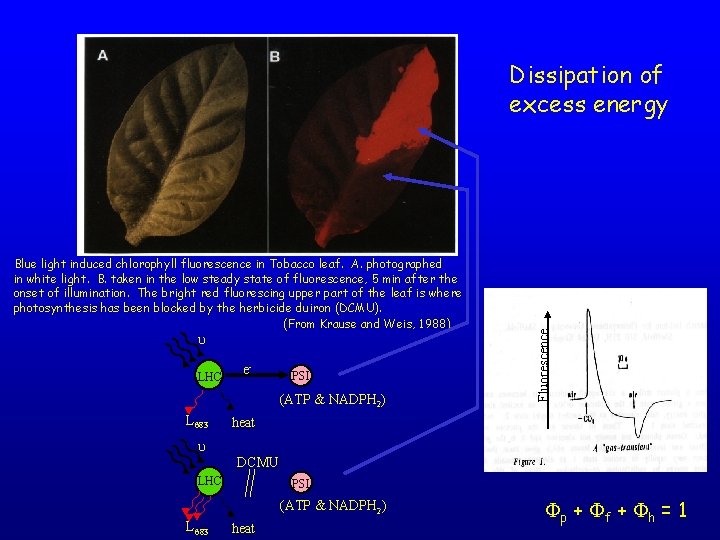
Blue light induced chlorophyll fluorescence in Tobacco leaf. A. photographed in white light. B. taken in the low steady state of fluorescence, 5 min after the onset of illumination. The bright red fluorescing upper part of the leaf is where photosynthesis has been blocked by the herbicide duiron (DCMU). (From Krause and Weis, 1988) u LHC e- PSI (ATP & NADPH 2) L 683 Fluorescence Dissipation of excess energy heat u DCMU LHC PSI (ATP & NADPH 2) L 683 heat Fp + Ff + Fh = 1
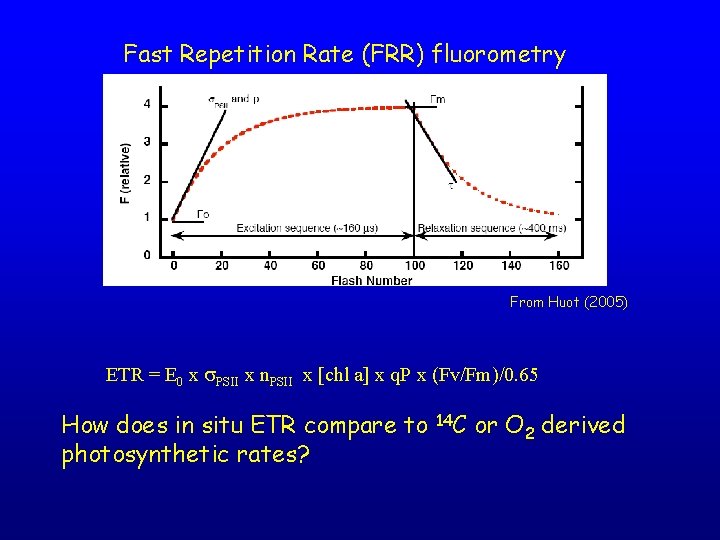
Fast Repetition Rate (FRR) fluorometry From Huot (2005) ETR = E 0 x s. PSII x n. PSII x [chl a] x q. P x (Fv/Fm)/0. 65 How does in situ ETR compare to photosynthetic rates? 14 C or O 2 derived
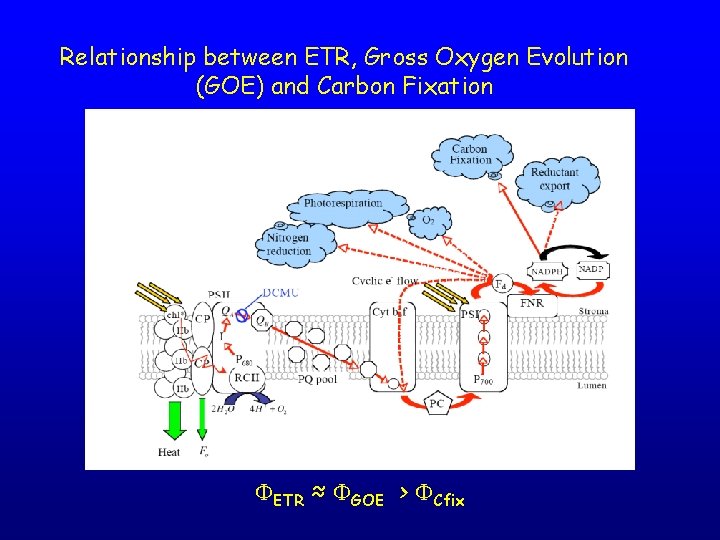
Relationship between ETR, Gross Oxygen Evolution (GOE) and Carbon Fixation FETR ≈ FGOE > FCfix

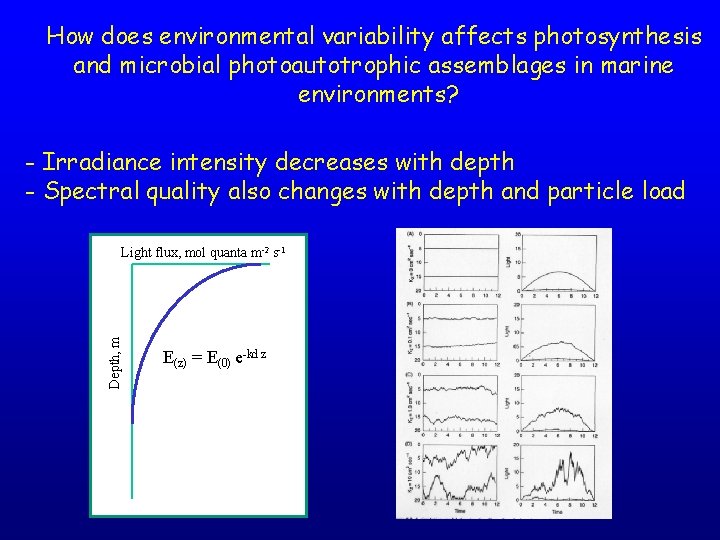
How does environmental variability affects photosynthesis and microbial photoautotrophic assemblages in marine environments? - Irradiance intensity decreases with depth - Spectral quality also changes with depth and particle load Depth, m Light flux, mol quanta m-2 s-1 E(z) = E(0) e-kd z
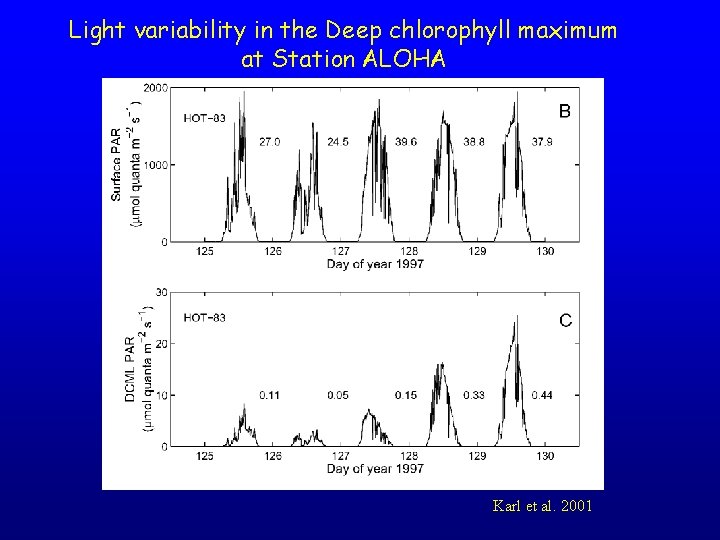
Light variability in the Deep chlorophyll maximum at Station ALOHA Karl et al. 2001

Temporal scales of aclimation and adaptation From J. Allen, 2002
![Changes in absorption cross sections as a result of increase [chl a] under low Changes in absorption cross sections as a result of increase [chl a] under low](http://slidetodoc.com/presentation_image/58ecd733e54209271eba60eebfee56a9/image-33.jpg)
Changes in absorption cross sections as a result of increase [chl a] under low light condition
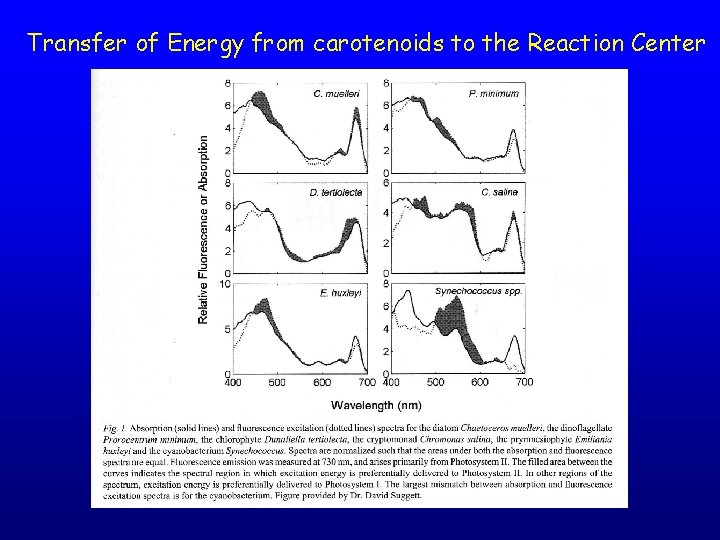
Transfer of Energy from carotenoids to the Reaction Center

Photoadaptation mechanisms: • Xanthophyll cycle: Involves the de-epoxidation of specific carotenoids, lowering the excitation state from a transferring level to a receiving level allows thermal dissipation of excess energy. • State transition: Allows the redistribution of energy from the LHC to PSII or PSI • Ru. Bisco or nitrate reductase activation/deactivation • Changes in antennae size and/or density of RC. • Changes in growth rates (controls of balanced growth rates)

Xanthophill cycle photoprotective mechanism
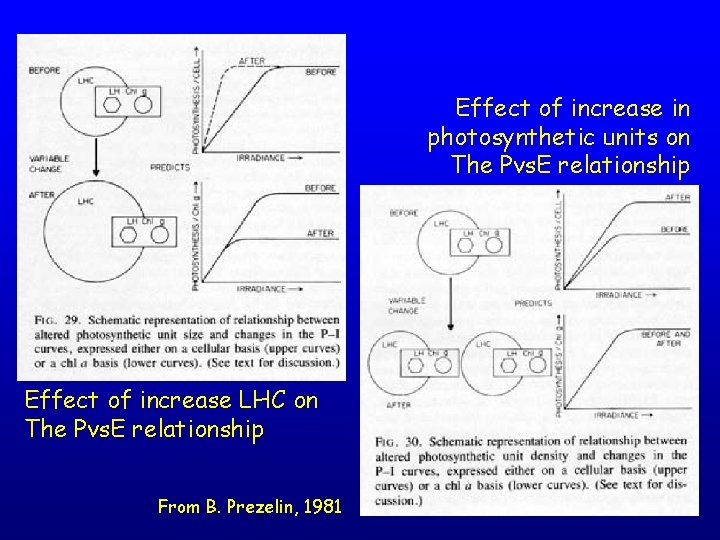
Effect of increase in photosynthetic units on The Pvs. E relationship Effect of increase LHC on The Pvs. E relationship From B. Prezelin, 1981
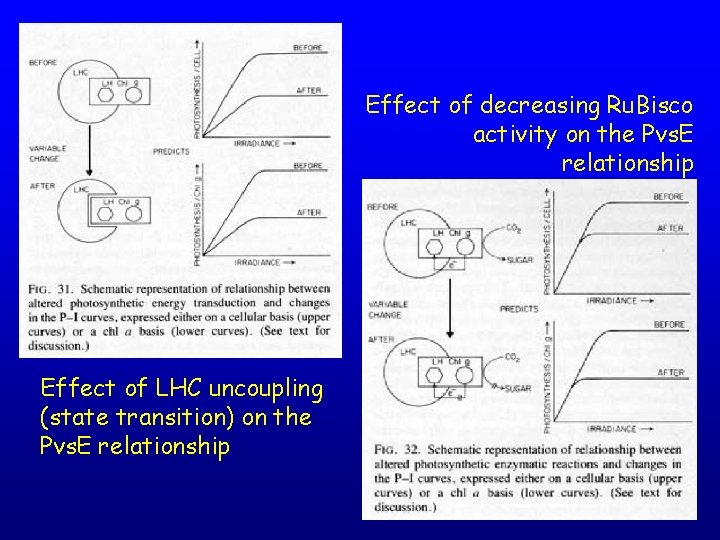
Effect of decreasing Ru. Bisco activity on the Pvs. E relationship Effect of LHC uncoupling (state transition) on the Pvs. E relationship
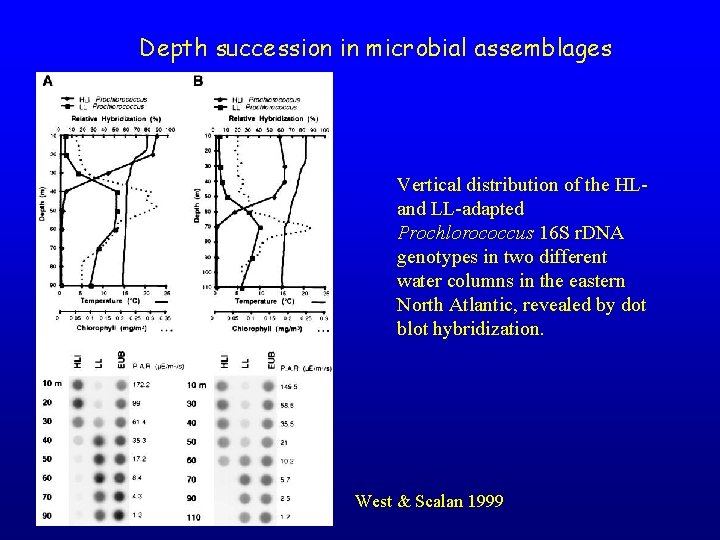
Depth succession in microbial assemblages Vertical distribution of the HLand LL-adapted Prochlorococcus 16 S r. DNA genotypes in two different water columns in the eastern North Atlantic, revealed by dot blot hybridization. West & Scalan 1999
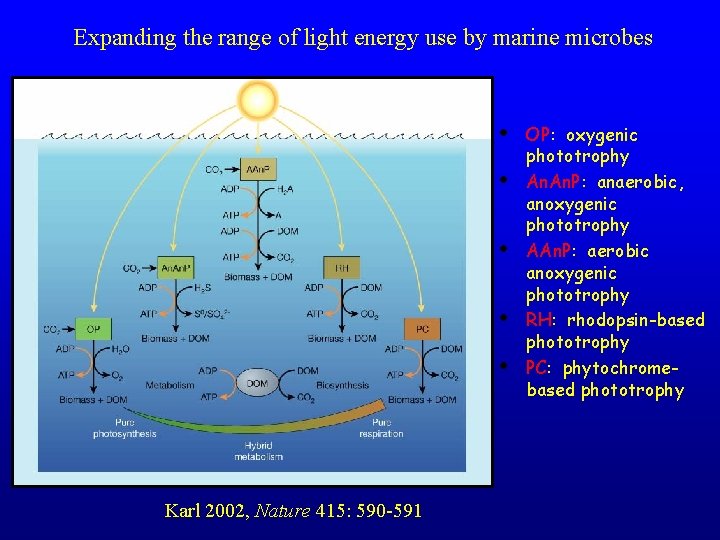
Expanding the range of light energy use by marine microbes • • • Karl 2002, Nature 415: 590 -591 OP: oxygenic phototrophy An. P: anaerobic, anoxygenic phototrophy AAn. P: aerobic anoxygenic phototrophy RH: rhodopsin-based phototrophy PC: phytochromebased phototrophy
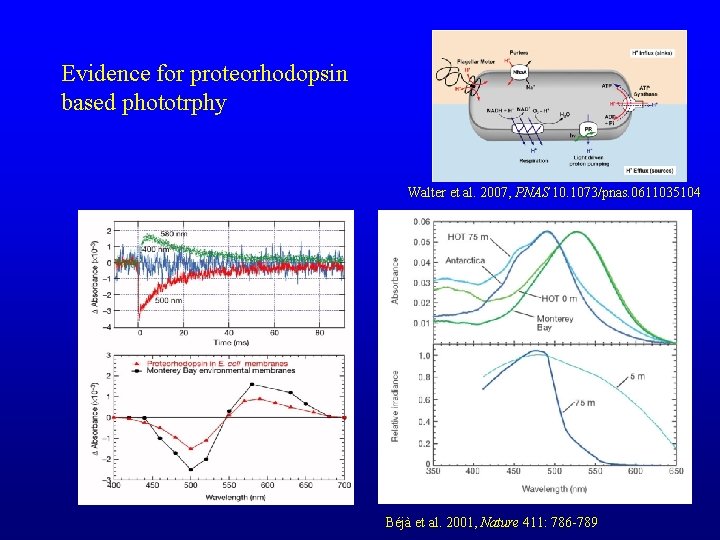
Evidence for proteorhodopsin based phototrphy Walter et al. 2007, PNAS 10. 1073/pnas. 0611035104 Béjà et al. 2001, Nature 411: 786 -789
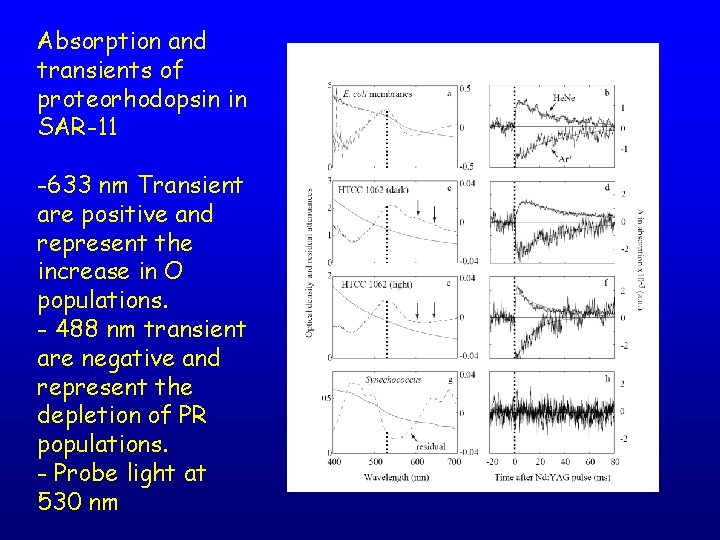
Absorption and transients of proteorhodopsin in SAR-11 -633 nm Transient are positive and represent the increase in O populations. - 488 nm transient are negative and represent the depletion of PR populations. - Probe light at 530 nm
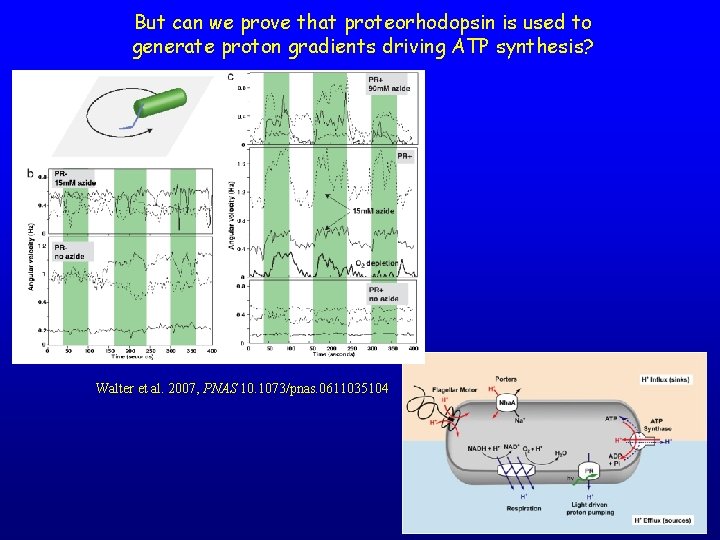
But can we prove that proteorhodopsin is used to generate proton gradients driving ATP synthesis? Walter et al. 2007, PNAS 10. 1073/pnas. 0611035104
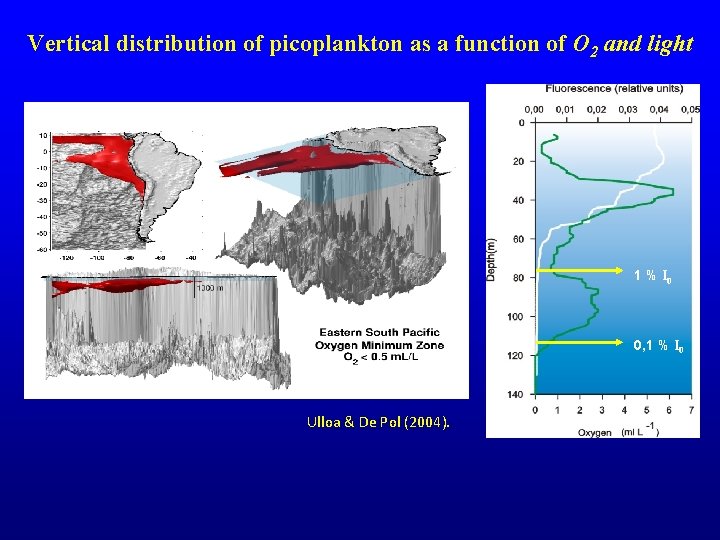
Vertical distribution of picoplankton as a function of O 2 and light 1 % I 0 0, 1 % I 0 Ulloa & De Pol (2004).
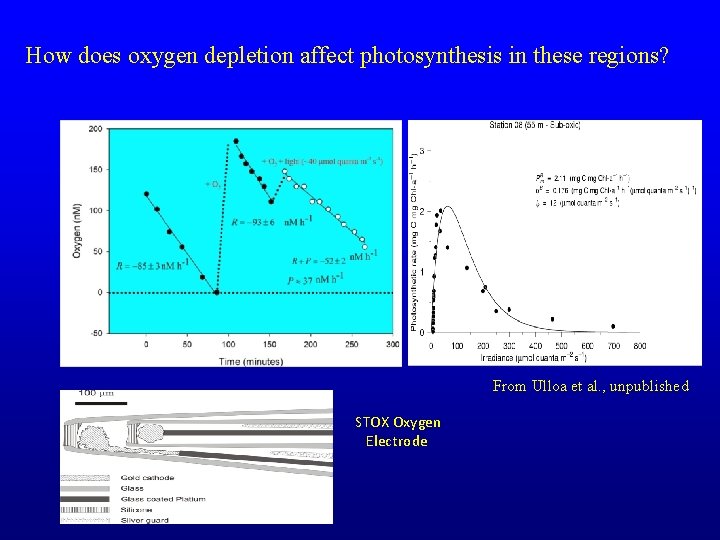
How does oxygen depletion affect photosynthesis in these regions? From Ulloa et al. , unpublished STOX Oxygen Electrode

Some present and future challenges: - Are we done discovering new taxa and pathways? - Do we understand the fluctuating environment experienced by microbes and their scale of response? - How do we characterize the breath of microbial phototrophy and its ecological role? - How do we develop and integrate new technologies in marine phototrophy research?
- Slides: 46