An Introduction to Metabolism and Enzymes Metabolism is

An Introduction to Metabolism and Enzymes
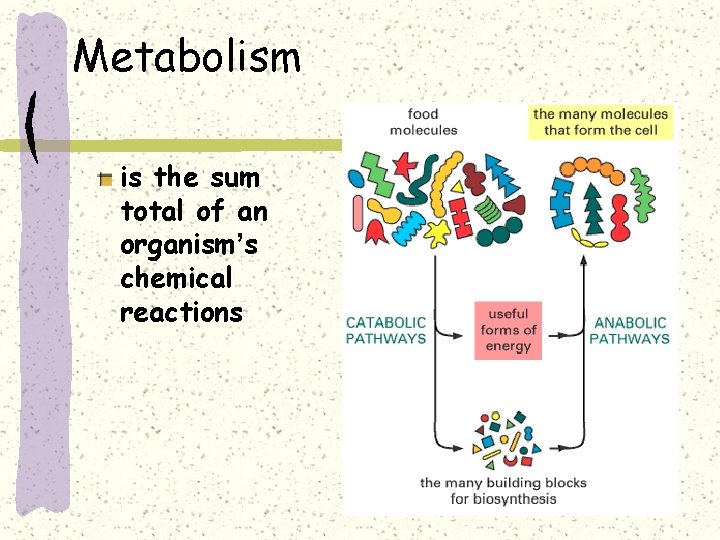
Metabolism is the sum total of an organism’s chemical reactions
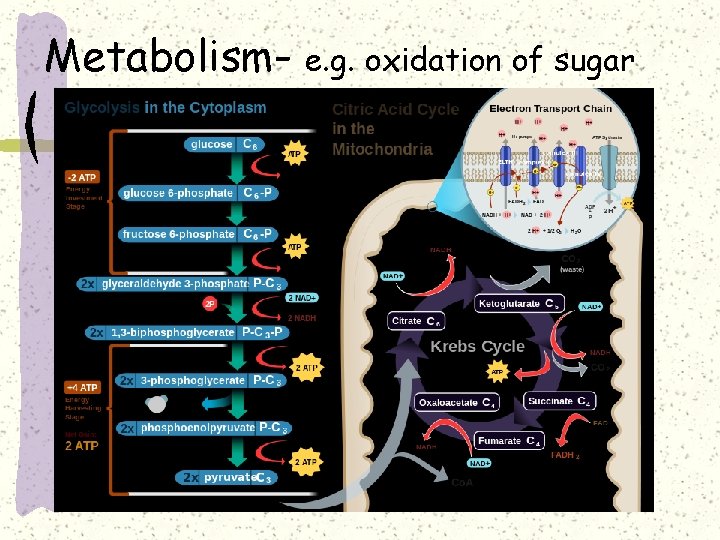
Metabolism- e. g. oxidation of sugar
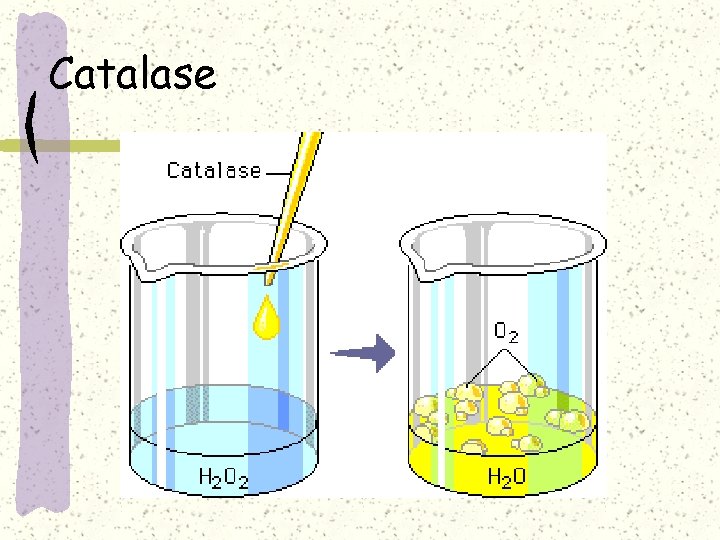
Catalase

Facts to be explained n Enzymes are proteins n Enzymes are required in minute quantities n Enzymes show striking specificity. They catalyze specific reactions involving specific substrates n Enzymes increase the speed of a chemical reaction - Both forward and backward reaction n The action of enzyme is greatly affected by temperatures. n Enzymatic activity is affected by H+ ions concentration (p. H) n The rate of reaction is affected by substrate and enzyme concentration n Enzyme itself is not used up in the reaction it catalyzes so it can be used over again and again
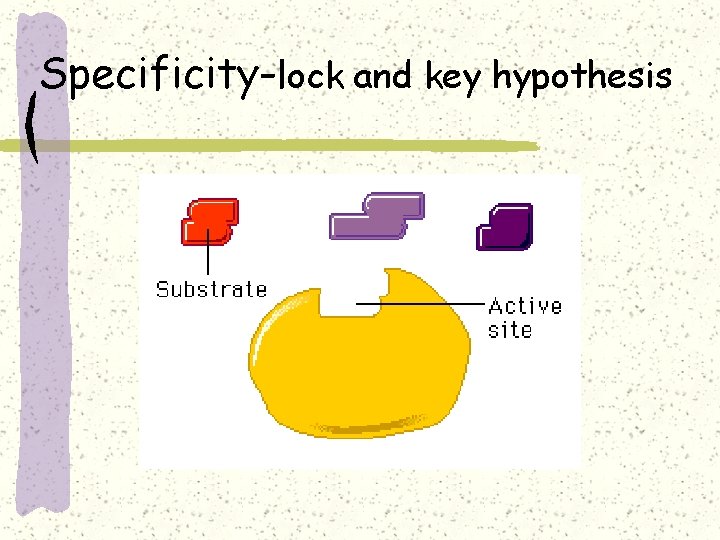
Specificity-lock and key hypothesis
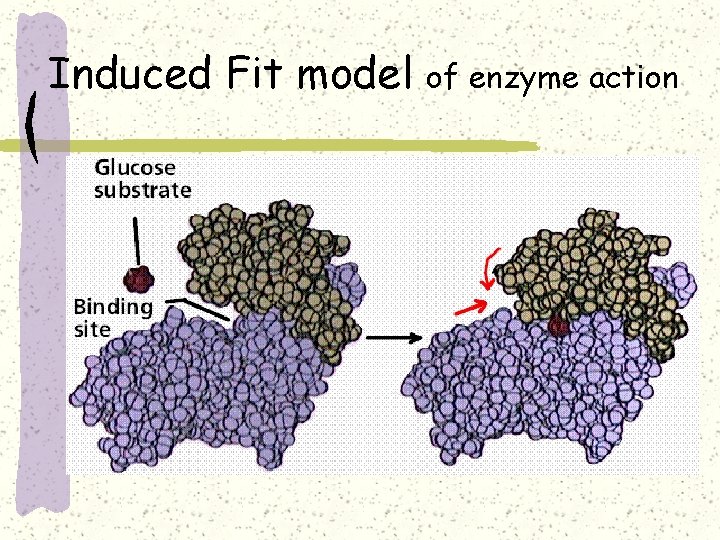
Induced Fit model of enzyme action
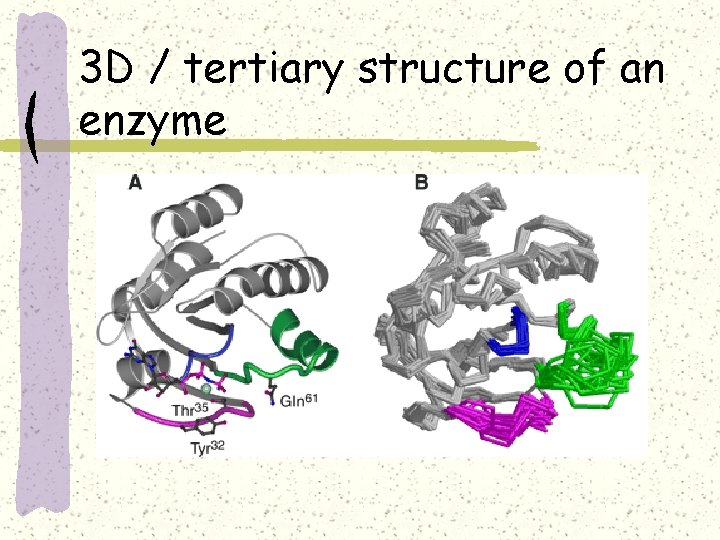
3 D / tertiary structure of an enzyme
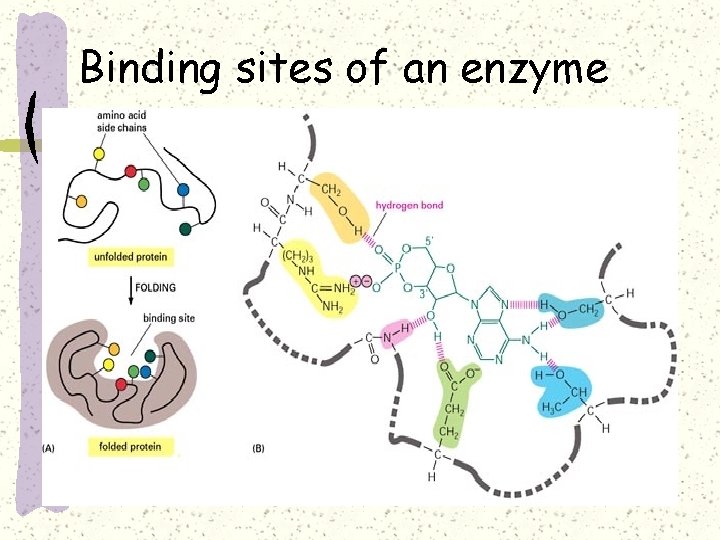
Binding sites of an enzyme
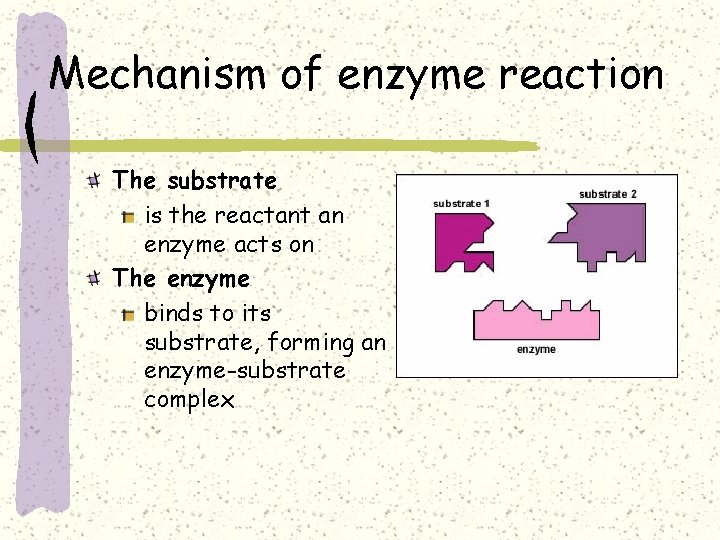
Mechanism of enzyme reaction The substrate is the reactant an enzyme acts on The enzyme binds to its substrate, forming an enzyme-substrate complex
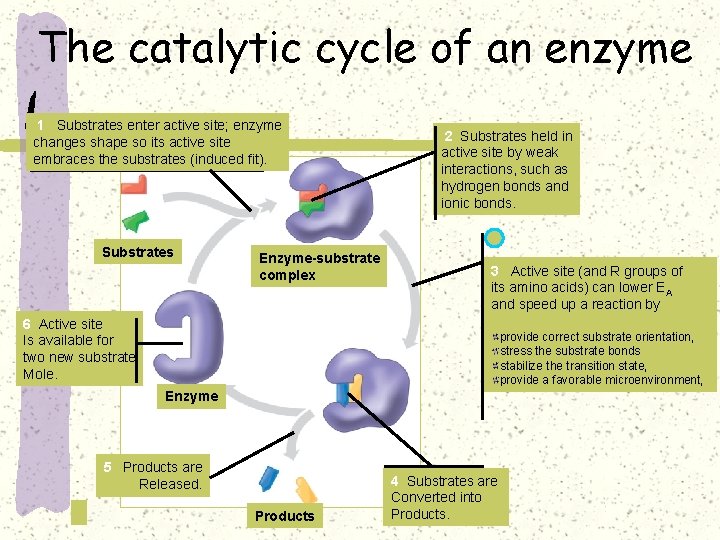
The catalytic cycle of an enzyme 1 Substrates enter active site; enzyme changes shape so its active site embraces the substrates (induced fit). Substrates Enzyme-substrate complex 2 Substrates held in active site by weak interactions, such as hydrogen bonds and ionic bonds. 3 Active site (and R groups of its amino acids) can lower EA and speed up a reaction by 6 Active site Is available for two new substrate Mole. provide correct substrate orientation, stress the substrate bonds stabilize the transition state, provide a favorable microenvironment, Enzyme 5 Products are Released. Products 4 Substrates are Converted into Products.

Interaction between Active site and substrate fitting of a substrate brings chemical groups of the active site into positions that enhance their ability to catalyze the chemical reaction
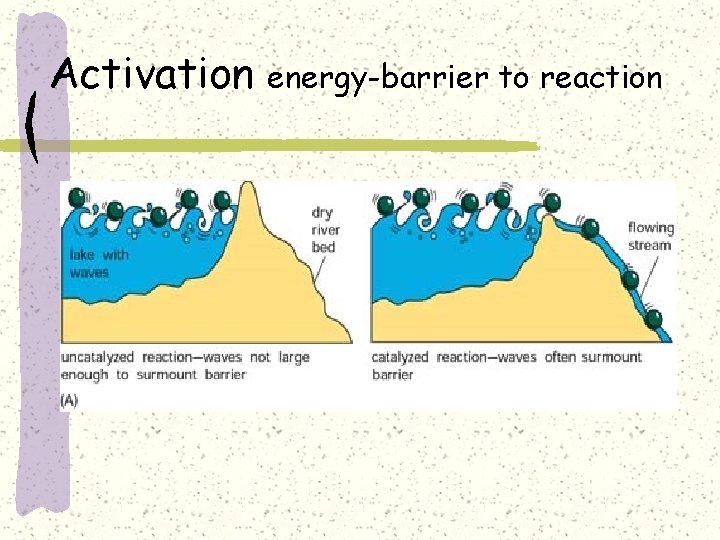
Activation energy-barrier to reaction
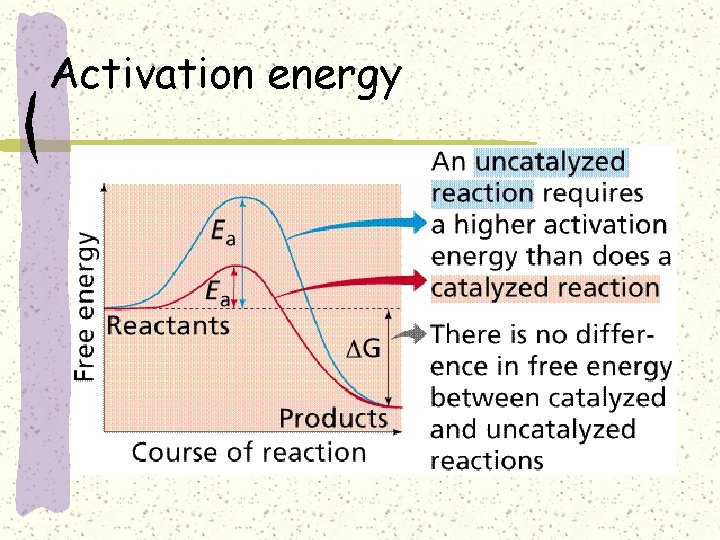
Activation energy

The active site can lower an EA barrier by Orienting substrates correctly Straining substrate bonds Stabilizing transition state Providing a favorable microenvironment
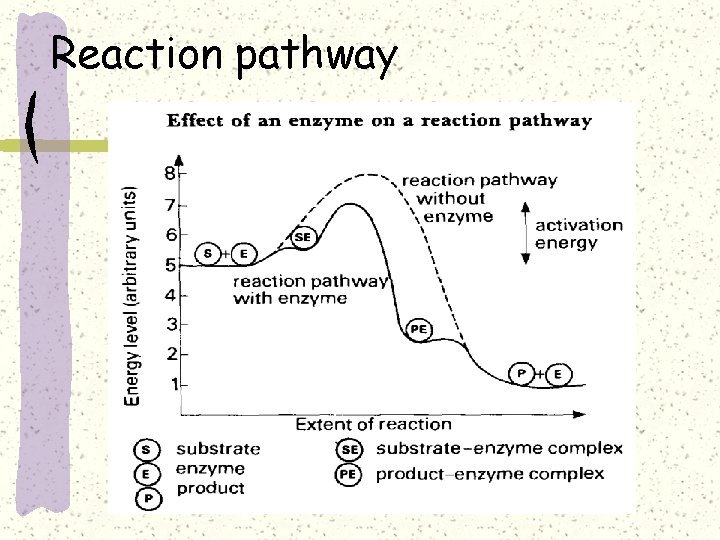
Reaction pathway
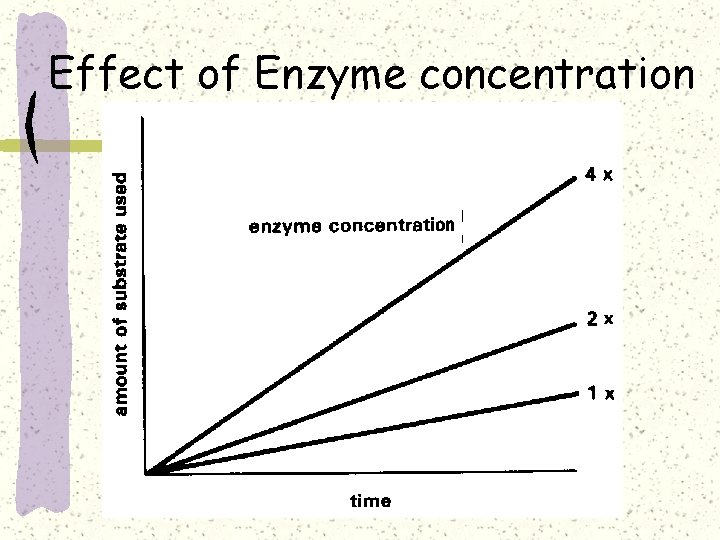
Effect of Enzyme concentration
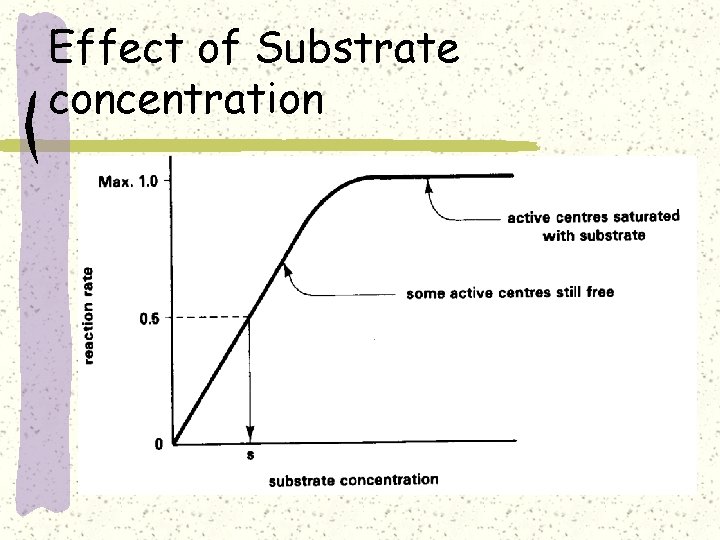
Effect of Substrate concentration
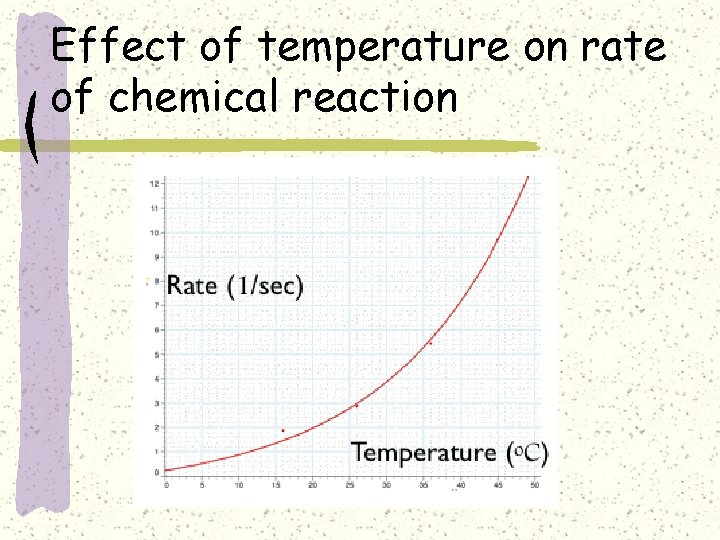
Effect of temperature on rate of chemical reaction
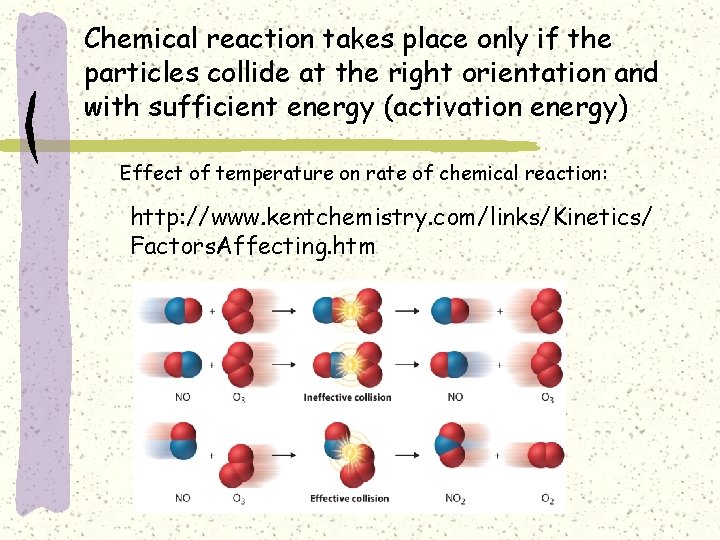
Chemical reaction takes place only if the particles collide at the right orientation and with sufficient energy (activation energy) Effect of temperature on rate of chemical reaction: http: //www. kentchemistry. com/links/Kinetics/ Factors. Affecting. htm
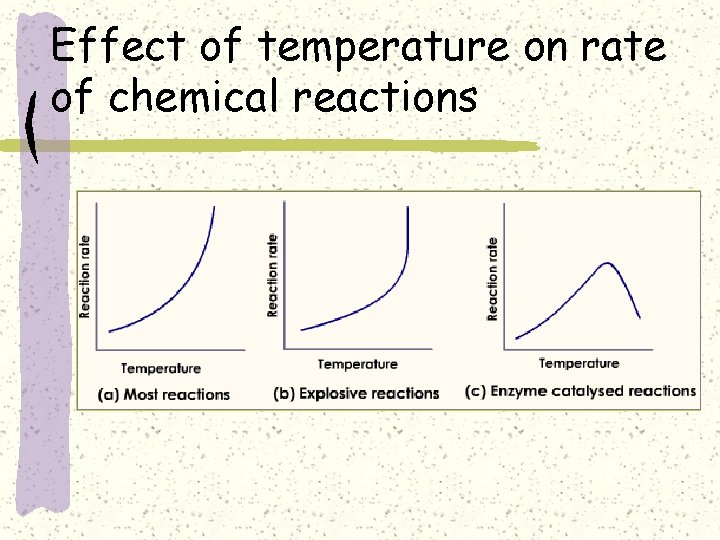
Effect of temperature on rate of chemical reactions
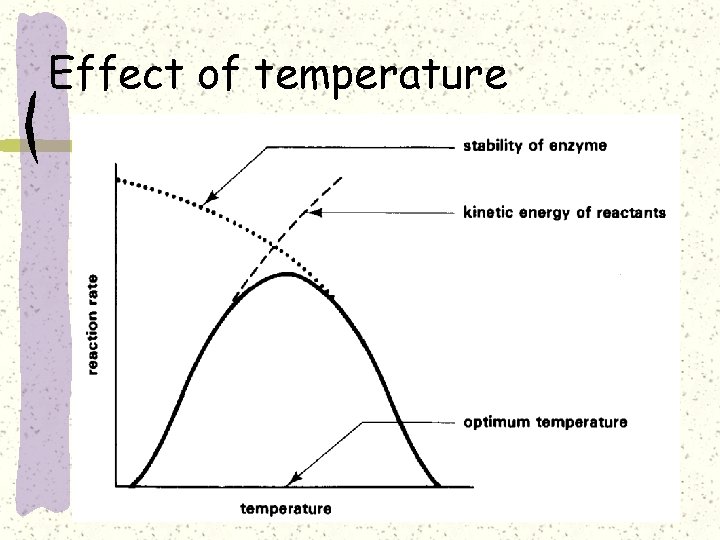
Effect of temperature
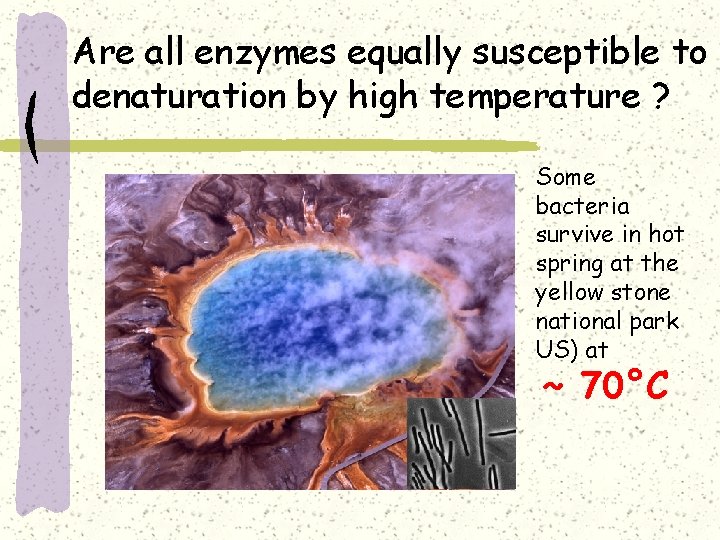
Are all enzymes equally susceptible to denaturation by high temperature ? Some bacteria survive in hot spring at the yellow stone national park US) at ~ 70°C

Some bacteria survive in deep sea hydrothermal vent at > 110°C

Enzyme application_thermophilic
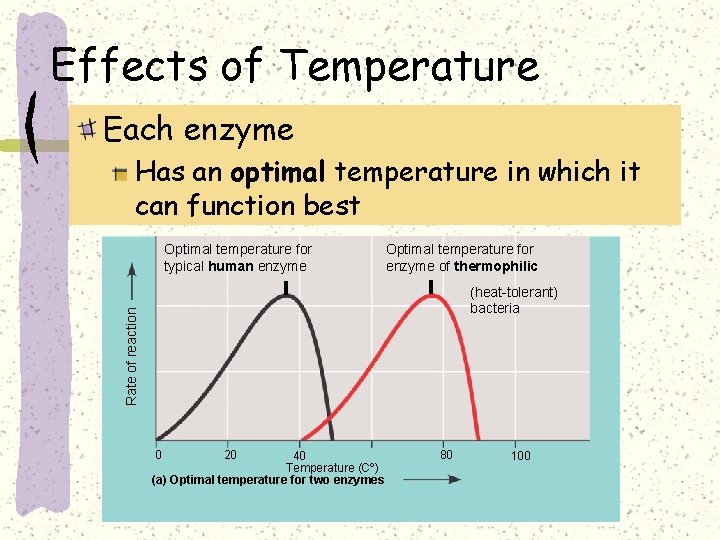
Effects of Temperature Each enzyme Has an optimal temperature in which it can function best Optimal temperature for typical human enzyme Optimal temperature for enzyme of thermophilic Rate of reaction (heat-tolerant) bacteria 0 20 40 Temperature (Cº) (a) Optimal temperature for two enzymes 80 100
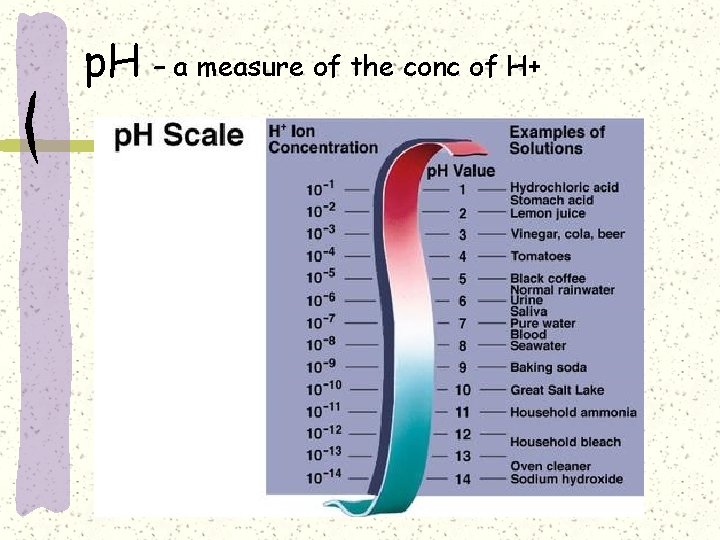
p. H – a measure of the conc of H+
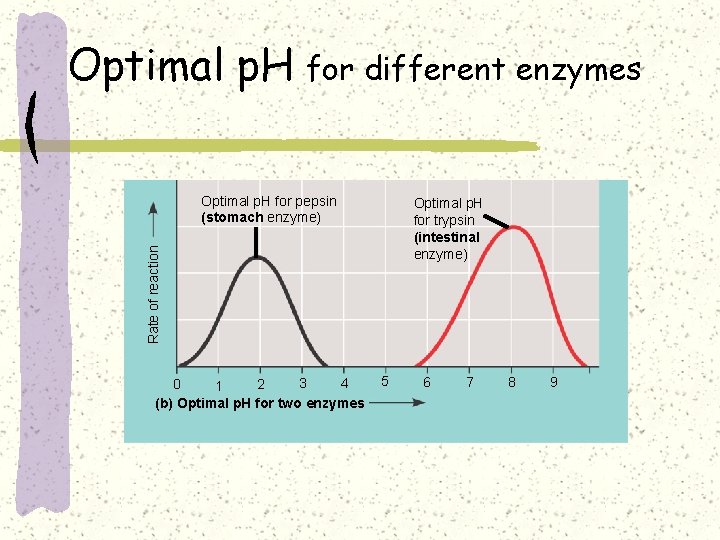
Optimal p. H for different enzymes Optimal p. H for pepsin (stomach enzyme) Rate of reaction Optimal p. H for trypsin (intestinal enzyme) 3 4 0 2 1 (b) Optimal p. H for two enzymes 5 6 7 8 9
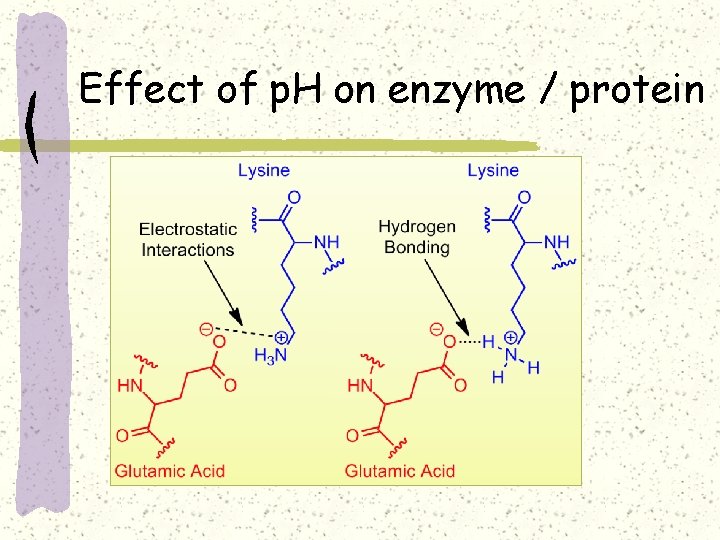
Effect of p. H on enzyme / protein
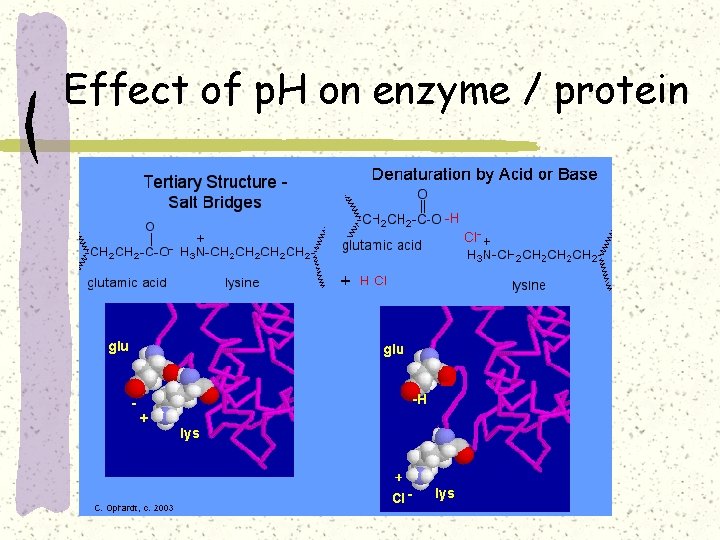
Effect of p. H on enzyme / protein
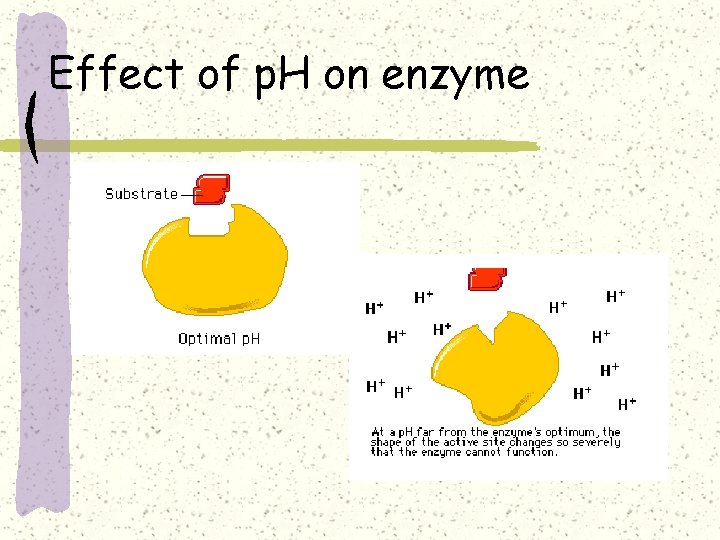
Effect of p. H on enzyme

Coenzymes -- Non-protein molecules that are enzyme helpers Some enzymes work only in the presence of other molecules E. g. NAD+ in dehydrogenation Some vitamins are important because they are co-enzymes e. g. Vitamin B groups
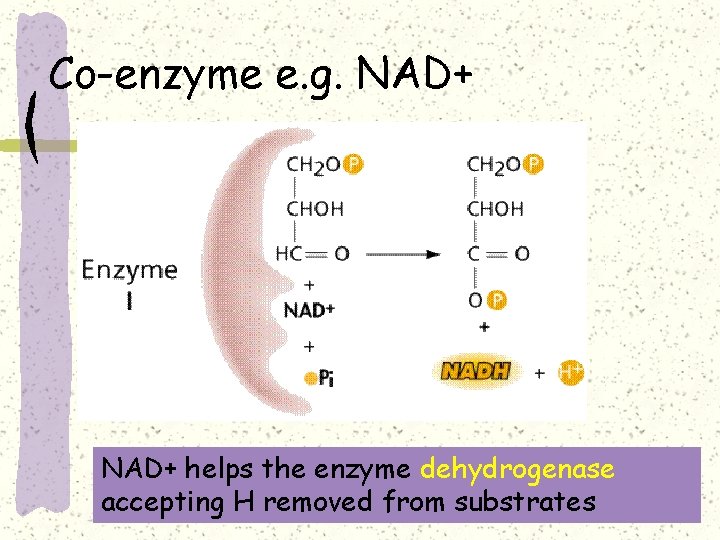
Co-enzyme e. g. NAD+ helps the enzyme dehydrogenase accepting H removed from substrates
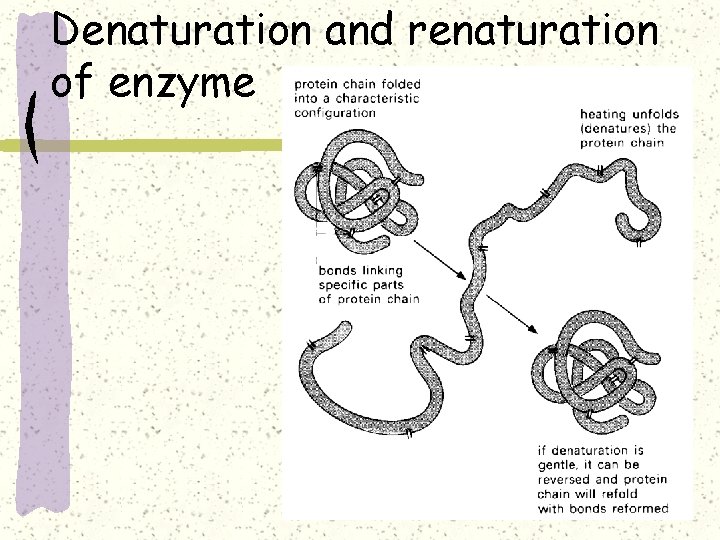
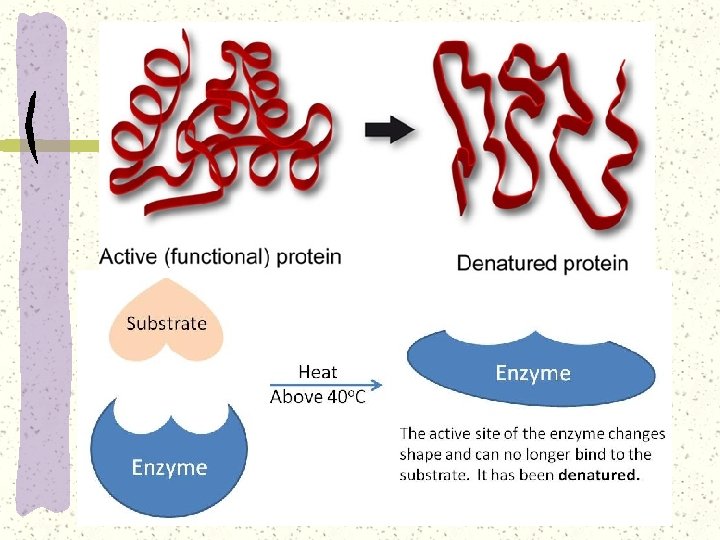
Denaturation and renaturation of enzyme

Denaturation of enzyme

Denaturation of enzyme It is the loss of the specific tertiary structure of a protein molecule. Lose its normal biological function. The change may be temporary or permanent, but the amino acid sequence (primary structure) of the protein could remain un____ --renaturation, though rare, might occurs and restore the enzyme function.
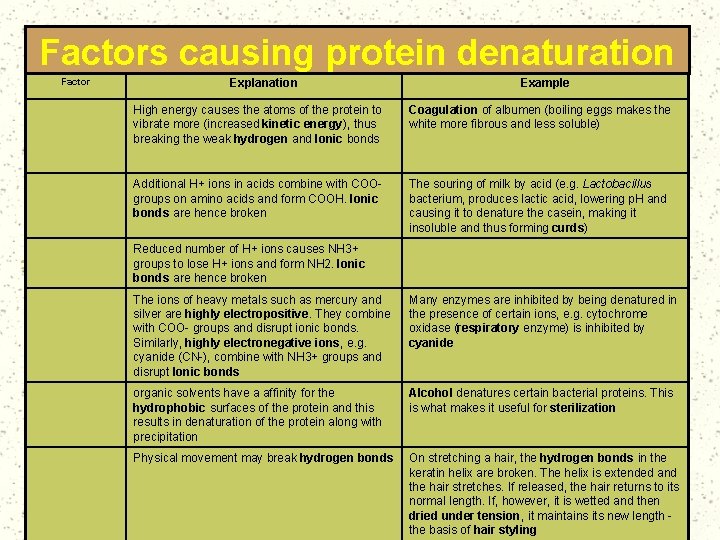
Factors causing protein denaturation Factor Explanation Example Heat or radiation (UV, infra. Red) High energy causes the atoms of the protein to vibrate more (increased kinetic energy), thus breaking the weak hydrogen and Ionic bonds Coagulation of albumen (boiling eggs makes the white more fibrous and less soluble) Acids Additional H+ ions in acids combine with COOgroups on amino acids and form COOH. Ionic bonds are hence broken The souring of milk by acid (e. g. Lactobacillus bacterium, produces lactic acid, lowering p. H and causing it to denature the casein, making it insoluble and thus forming curds) Alkalis Reduced number of H+ ions causes NH 3+ groups to lose H+ ions and form NH 2. Ionic bonds are hence broken Inorganic chemicals The ions of heavy metals such as mercury and silver are highly electropositive. They combine with COO- groups and disrupt ionic bonds. Similarly, highly electronegative ions, e. g. cyanide (CN-), combine with NH 3+ groups and disrupt Ionic bonds Many enzymes are inhibited by being denatured in the presence of certain ions, e. g. cytochrome oxidase (respiratory enzyme) is inhibited by cyanide Organic chemicals organic solvents have a affinity for the hydrophobic surfaces of the protein and this results in denaturation of the protein along with precipitation Alcohol denatures certain bacterial proteins. This is what makes it useful for sterilization Mechanical force Physical movement may break hydrogen bonds On stretching a hair, the hydrogen bonds in the keratin helix are broken. The helix is extended and the hair stretches. If released, the hair returns to its normal length. If, however, it is wetted and then dried under tension, it maintains its new length the basis of hair styling
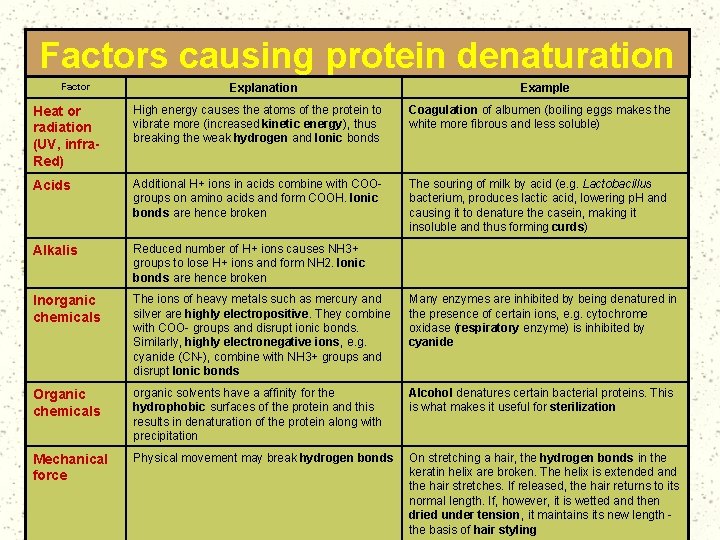
Factors causing protein denaturation Factor Explanation Example Heat or radiation (UV, infra. Red) High energy causes the atoms of the protein to vibrate more (increased kinetic energy), thus breaking the weak hydrogen and Ionic bonds Coagulation of albumen (boiling eggs makes the white more fibrous and less soluble) Acids Additional H+ ions in acids combine with COOgroups on amino acids and form COOH. Ionic bonds are hence broken The souring of milk by acid (e. g. Lactobacillus bacterium, produces lactic acid, lowering p. H and causing it to denature the casein, making it insoluble and thus forming curds) Alkalis Reduced number of H+ ions causes NH 3+ groups to lose H+ ions and form NH 2. Ionic bonds are hence broken Inorganic chemicals The ions of heavy metals such as mercury and silver are highly electropositive. They combine with COO- groups and disrupt ionic bonds. Similarly, highly electronegative ions, e. g. cyanide (CN-), combine with NH 3+ groups and disrupt Ionic bonds Many enzymes are inhibited by being denatured in the presence of certain ions, e. g. cytochrome oxidase (respiratory enzyme) is inhibited by cyanide Organic chemicals organic solvents have a affinity for the hydrophobic surfaces of the protein and this results in denaturation of the protein along with precipitation Alcohol denatures certain bacterial proteins. This is what makes it useful for sterilization Mechanical force Physical movement may break hydrogen bonds On stretching a hair, the hydrogen bonds in the keratin helix are broken. The helix is extended and the hair stretches. If released, the hair returns to its normal length. If, however, it is wetted and then dried under tension, it maintains its new length the basis of hair styling
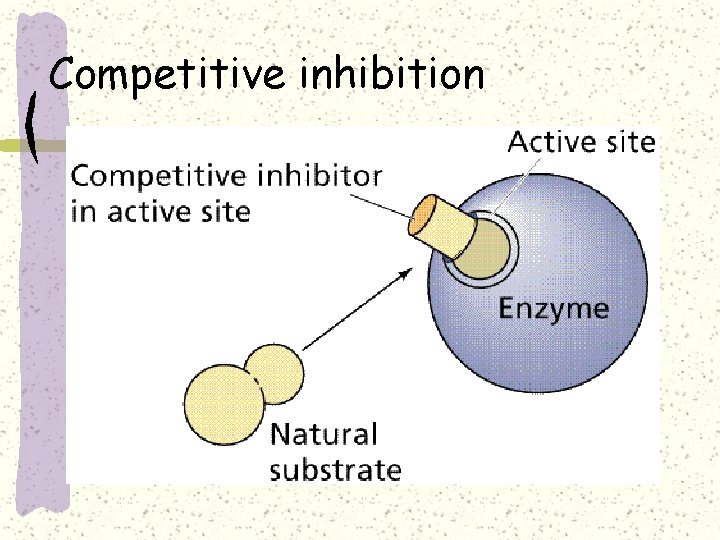
Competitive inhibition
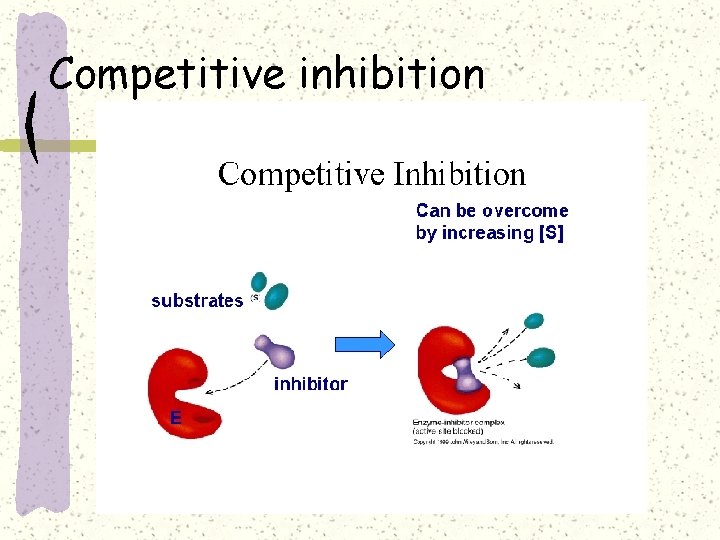
Competitive inhibition
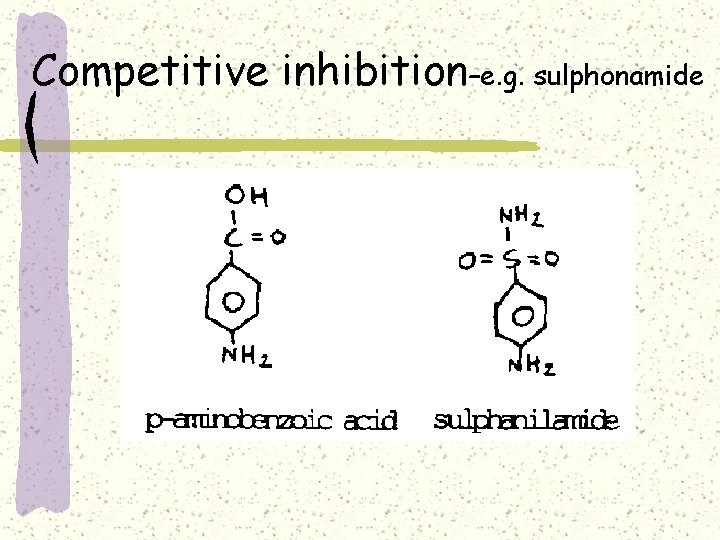
Competitive inhibition–e. g. sulphonamide
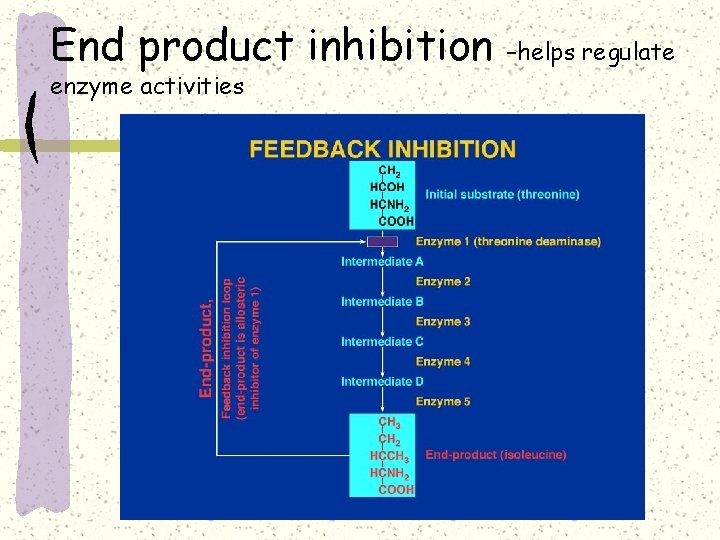
End product inhibition enzyme activities –helps regulate
End product inhibition
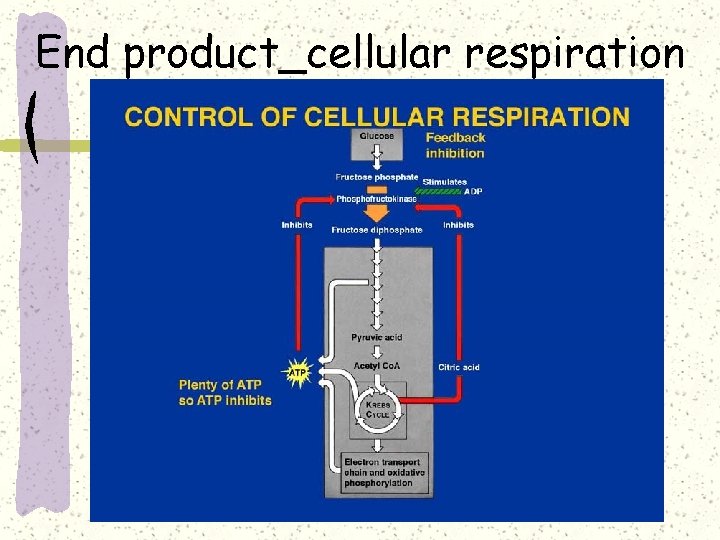
End product_cellular respiration
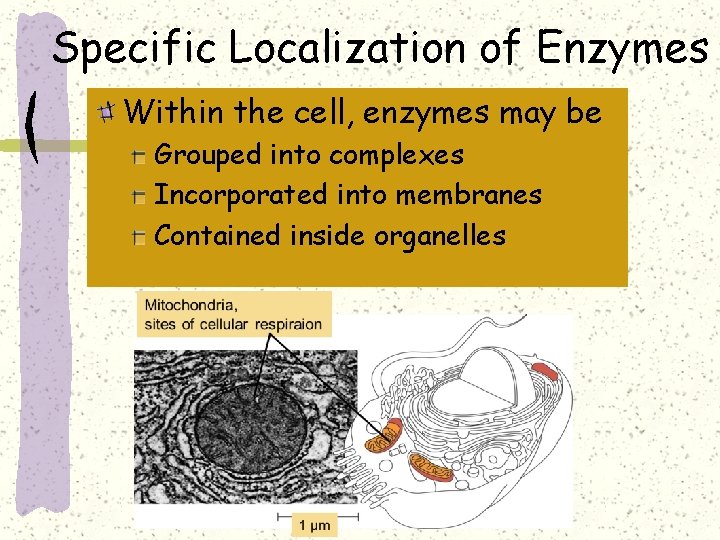
Specific Localization of Enzymes Within the cell, enzymes may be Grouped into complexes Incorporated into membranes Contained inside organelles

• Is 37 degree Celcius the optimum temperature for human enzyme? • Why then our body temperature is 37 degree Celcius?

• Are all enzymes denatured above 80 C?
- Slides: 48