AN INTRODUCTION TO ELECTRODE POTENTIALS KNOCKHARDY PUBLISHING KNOCKHARDY

AN INTRODUCTION TO ELECTRODE POTENTIALS KNOCKHARDY PUBLISHING

KNOCKHARDY PUBLISHING ELECTRODE POTENTIALS INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at AS and A 2 level Chemistry. It is based on the requirements of the AQA and OCR specifications but is suitable for other examination boards. Individual students may use the material at home for revision purposes or it may be used for classroom teaching if an interactive white board is available. Accompanying notes on this, and the full range of AS and A 2 topics, are available from the KNOCKHARDY SCIENCE WEBSITE at. . . www. argonet. co. uk/users/hoptonj/sci. htm Navigation is achieved by. . . either clicking on the grey arrows at the foot of each page or using the left and right arrow keys on the keyboard

ELECTRODE POTENTIALS CONTENTS • Types of half cells • Cell potential • The standard hydrogen electrode • Measuring electrode potentials • The electrochemical series • Combining half cells • Cell diagrams • Uses of E° values

ELECTRODE POTENTIALS Before you start it would be helpful to… • Recall the definitions of oxidation and reduction • Be able to balance simple ionic equations • Have a knowledge of simple circuitry

TYPES OF HALF CELL These are systems involving oxidation or reduction METALS IN CONTACT WITH SOLUTIONS OF THEIR IONS Reaction Cu 2+(aq) + 2 e¯ Electrode copper Solution Cu 2+(aq) (1 M) Potential + 0. 34 V Reaction Zn 2+(aq) + 2 e¯ Electrode zinc Solution Zn 2+(aq) (1 M) Potential - 0. 76 V Cu(s) - 1 M copper sulphate solution Zn(s) - 1 M zinc sulphate solution

TYPES OF HALF CELL These are systems involving oxidation or reduction GASES IN CONTACT WITH SOLUTIONS OF THEIR IONS Reaction Electrode 2 H+(aq) + 2 e¯ platinum H 2(g) Solution H+(aq) (1 M) Gas hydrogen ( 1 atm pressure ) Potential 0. 00 V Reaction Cl 2(aq) + 2 e¯ Electrode platinum Solution Cl¯(aq) (1 M) Gas chorine ( 1 atm pressure ) Potential + 1. 36 V - 1 M HCl or 0. 5 M H 2 SO 4 2 Cl¯(g) - 1 M sodium chloride

TYPES OF HALF CELL These are systems involving oxidation or reduction SOLUTIONS OF IONS IN TWO DIFFERENT OXIDATION STATES Reaction Electrode Fe 3+(aq) + e¯ platinum Fe 2+(aq) Solution Fe 3+(aq) (1 M) and Fe 2+(aq) (1 M) Potential + 0. 77 V SOLUTIONS OF OXIDISING AGENTS IN ACID SOLUTION Reaction Mn. O 4¯ + 8 H+(aq) + 5 e¯ Electrode platinum Solution Mn. O 4¯(aq) (1 M) and Potential + 1. 52 V Mn 2+(aq) + 4 H 2 O(l) Mn 2+(aq) (1 M) and H+(aq)

CELL POTENTIAL Each electrode / electrolyte combination has its own half-reaction which sets up a potential difference The value is affected by. . . Measurement BUT. . . TEMPERATURE PRESSURE OF ANY GASES SOLUTION CONCENTRATION it is impossible to measure the potential of a single electrode… you can measure the potential difference between two electrodes it is measured relative to a reference cell under standard conditions The ultimate reference is the STANDARD HYDROGEN ELECTRODE. However, as it is difficult to set up, secondary standards are used.

THE STANDARD HYDROGEN ELECTRODE The ultimate reference is the STANDARD HYDROGEN ELECTRODE. However as it is difficult to set up secondary standards are used. 298 K (25°C) SOLUTION OF 1 M H+(aq) e. g. 1 M HCl or 0. 5 M H 2 SO 4 HYDROGEN GAS AT 1 ATMOSPHERE PRESSURE PLATINUM ELECTRODE The standard hydrogen electrode is assigned an E° value of 0. 00 V.
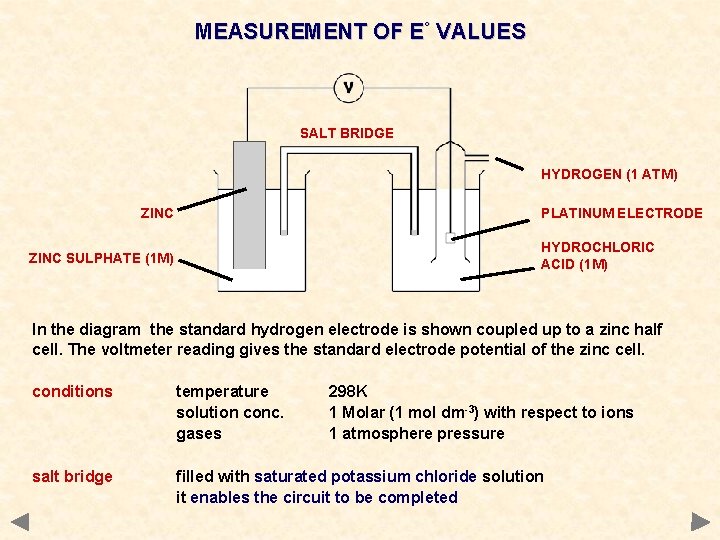
MEASUREMENT OF E° VALUES SALT BRIDGE HYDROGEN (1 ATM) ZINC PLATINUM ELECTRODE HYDROCHLORIC ACID (1 M) ZINC SULPHATE (1 M) In the diagram the standard hydrogen electrode is shown coupled up to a zinc half cell. The voltmeter reading gives the standard electrode potential of the zinc cell. conditions temperature solution conc. gases 298 K 1 Molar (1 mol dm-3) with respect to ions 1 atmosphere pressure salt bridge filled with saturated potassium chloride solution it enables the circuit to be completed
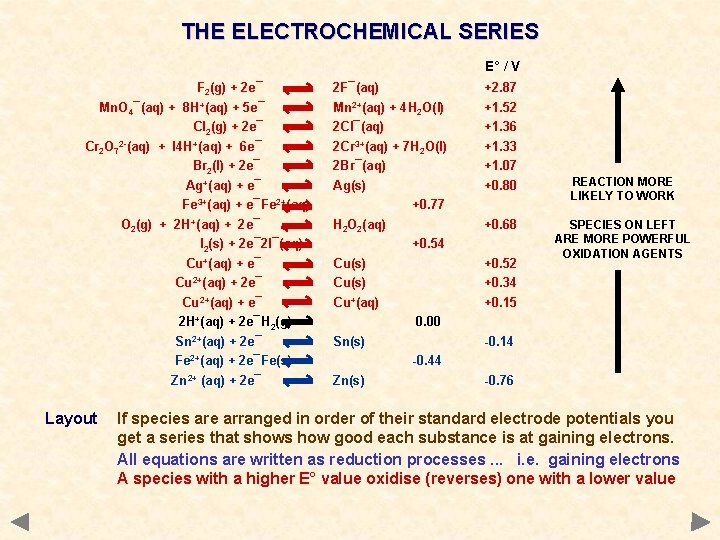
THE ELECTROCHEMICAL SERIES E° / V F 2(g) + 2 e¯ 2 F¯(aq) +2. 87 Mn. O 4¯(aq) + 8 H+(aq) + 5 e¯ Mn 2+(aq) + 4 H 2 O(l) +1. 52 Cl 2(g) + 2 e¯ Cr 2 O 72 -(aq) + I 4 H+(aq) + 6 e¯ Br 2(l) + 2 e¯ 2 Cl¯(aq) 2 Cr 3+(aq) + 7 H 2 O(l) 2 Br¯(aq) +1. 36 +1. 33 +1. 07 Ag(s) +0. 80 REACTION MORE LIKELY TO WORK +0. 68 SPECIES ON LEFT ARE MORE POWERFUL OXIDATION AGENTS Ag+(aq) + e¯ Fe 3+(aq) + e¯Fe 2+(aq) O 2(g) + 2 H+(aq) + 2 e¯ I 2(s) + 2 e¯ 2 I¯(aq) Cu+(aq) + e¯ Cu 2+(aq) + 2 e¯ Cu 2+(aq) + e¯ 2 H+(aq) + 2 e¯H 2(g) Sn 2+(aq) + 2 e¯ Fe 2+(aq) + 2 e¯Fe(s) Zn 2+ (aq) + 2 e¯ Layout +0. 77 H 2 O 2(aq) +0. 54 Cu(s) Cu+(aq) +0. 52 +0. 34 +0. 15 0. 00 Sn(s) -0. 14 -0. 44 Zn(s) -0. 76 If species are arranged in order of their standard electrode potentials you get a series that shows how good each substance is at gaining electrons. All equations are written as reduction processes. . . i. e. gaining electrons A species with a higher E° value oxidise (reverses) one with a lower value

THE ELECTROCHEMICAL SERIES E° / V F 2(g) + 2 e¯ 2 F¯(aq) +2. 87 Mn. O 4¯(aq) + 8 H+(aq) + 5 e¯ Mn 2+(aq) + 4 H 2 O(l) +1. 52 Cl 2(g) + 2 e¯ Cr 2 O 72 -(aq) + I 4 H+(aq) + 6 e¯ Br 2(l) + 2 e¯ 2 Cl¯(aq) 2 Cr 3+(aq) + 7 H 2 O(l) 2 Br¯(aq) +1. 36 +1. 33 +1. 07 Ag(s) +0. 80 Ag+(aq) + e¯ Fe 3+(aq) + e¯Fe 2+(aq) O 2(g) + 2 H+(aq) + 2 e¯ I 2(s) + 2 e¯ 2 I¯(aq) Cu+(aq) + e¯ Cu 2+(aq) + 2 e¯ Cu 2+(aq) + e¯ 2 H+(aq) + 2 e¯H 2(g) Sn 2+(aq) + 2 e¯ Fe 2+(aq) + 2 e¯Fe(s) Zn 2+ (aq) + 2 e¯ +0. 77 H 2 O 2(aq) +0. 68 +0. 54 Cu(s) Cu+(aq) +0. 52 +0. 34 +0. 15 AN EQUATION WITH A HIGHER E° VALUE WILL REVERSE AN EQUATION WITH A LOWER VALUE 0. 00 Sn(s) -0. 14 -0. 44 Zn(s) -0. 76 Application Chlorine is a more powerful oxidising agent - it has a higher E° Chlorine will get its electrons by reversing the iodine equation Cl 2(g) + 2 e¯ ——> 2 Cl¯(aq) and 2 I¯(aq) ——> I 2(s) + 2 e¯ Overall equation is Cl 2(g) + 2 I¯(aq) ——> I 2(s) + 2 Cl¯(aq)

SECONDARY STANDARDS Why? The standard hydrogen electrode (SHE) is difficult to set up it is easier to choose a more convenient secondary standard the secondary standard has been calibrated against the SHE Calomel • the calomel electrode contains Hg 2 Cl 2 • it has a standard electrode potential of +0. 27 V • is used as the LH electrode to determine the potential of an unknown • to get the value of the other cell ADD 0. 27 V to the measured cell potential

ELECTROCHEMICAL CELLS • • • electrochemical cells contain two electrodes each electrode / electrolyte combination has its own half-reaction the electrons produced by one half reaction are available for the other oxidation occurs at the anode reduction occurs at the cathode. ANODE Zn(s) ——> Zn 2+(aq) + 2 e¯ OXIDATION CATHODE Cu 2+(aq) + 2 e¯ ——> Cu(s) REDUCTION The resulting cell has a potential difference (voltage) called the cell potential which depends on the difference between the two potentials It is affected by. . . • • current temperature pressure of any gases solution concentrations
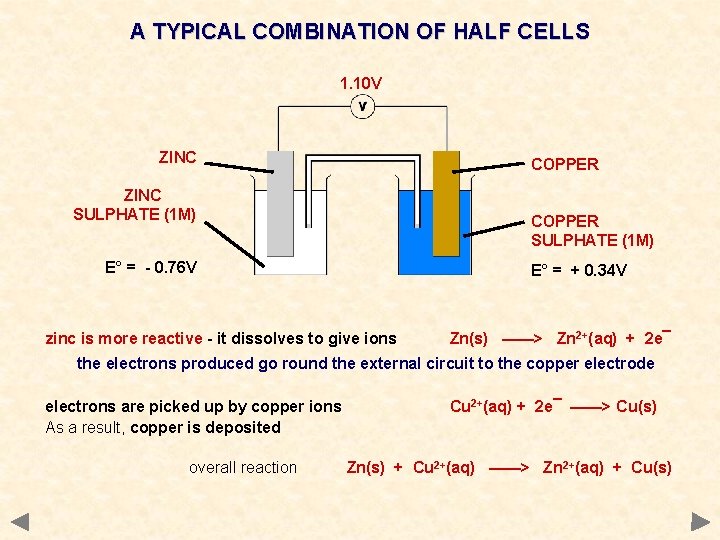
A TYPICAL COMBINATION OF HALF CELLS 1. 10 V ZINC COPPER ZINC SULPHATE (1 M) COPPER SULPHATE (1 M) E° = - 0. 76 V E° = + 0. 34 V zinc is more reactive - it dissolves to give ions Zn(s) ——> Zn 2+(aq) + 2 e¯ the electrons produced go round the external circuit to the copper electrode electrons are picked up by copper ions As a result, copper is deposited overall reaction Cu 2+(aq) + 2 e¯ ——> Cu(s) Zn(s) + Cu 2+(aq) ——> Zn 2+(aq) + Cu(s)

CELL DIAGRAMS These give a diagrammatic representation of what is happening in a cell. • Place the cell with the more positive E° value on the RHS of the diagram. Cu 2+(aq) + 2 e¯ Cu(s) E° = + 0. 34 V put on the RHS Zn 2+(aq) + 2 e¯ Zn(s) E° = - 0. 76 V put on the LHS Zn Zn 2+ Cu
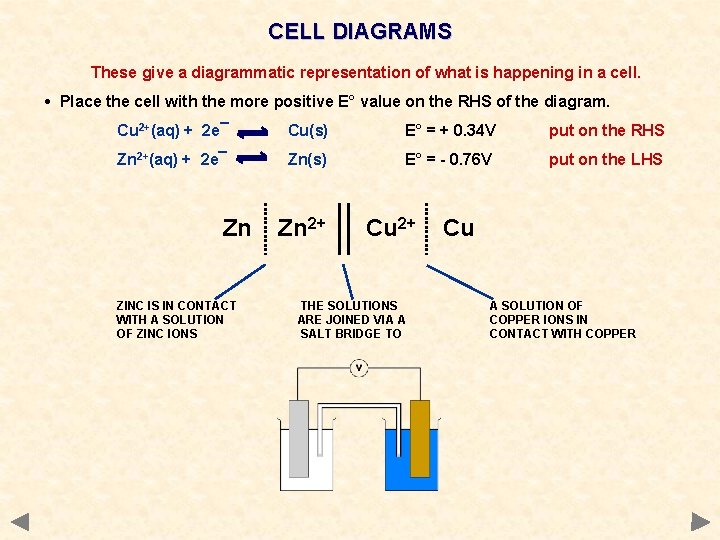
CELL DIAGRAMS These give a diagrammatic representation of what is happening in a cell. • Place the cell with the more positive E° value on the RHS of the diagram. Cu 2+(aq) + 2 e¯ Cu(s) E° = + 0. 34 V put on the RHS Zn 2+(aq) + 2 e¯ Zn(s) E° = - 0. 76 V put on the LHS Zn ZINC IS IN CONTACT WITH A SOLUTION OF ZINC IONS Zn 2+ Cu 2+ THE SOLUTIONS ARE JOINED VIA A SALT BRIDGE TO Cu A SOLUTION OF COPPER IONS IN CONTACT WITH COPPER
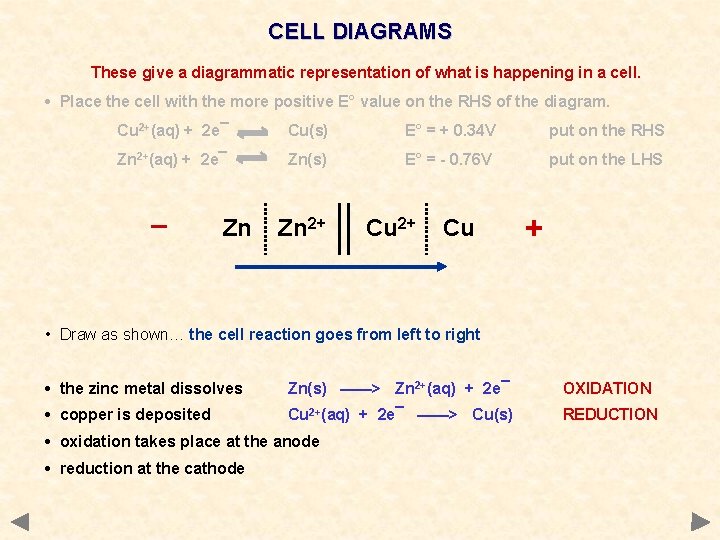
CELL DIAGRAMS These give a diagrammatic representation of what is happening in a cell. • Place the cell with the more positive E° value on the RHS of the diagram. Cu 2+(aq) + 2 e¯ Cu(s) E° = + 0. 34 V put on the RHS Zn 2+(aq) + 2 e¯ Zn(s) E° = - 0. 76 V put on the LHS _ Zn Zn 2+ Cu + • Draw as shown… the cell reaction goes from left to right • the zinc metal dissolves Zn(s) ——> Zn 2+(aq) + 2 e¯ OXIDATION • copper is deposited Cu 2+(aq) + 2 e¯ ——> Cu(s) REDUCTION • oxidation takes place at the anode • reduction at the cathode
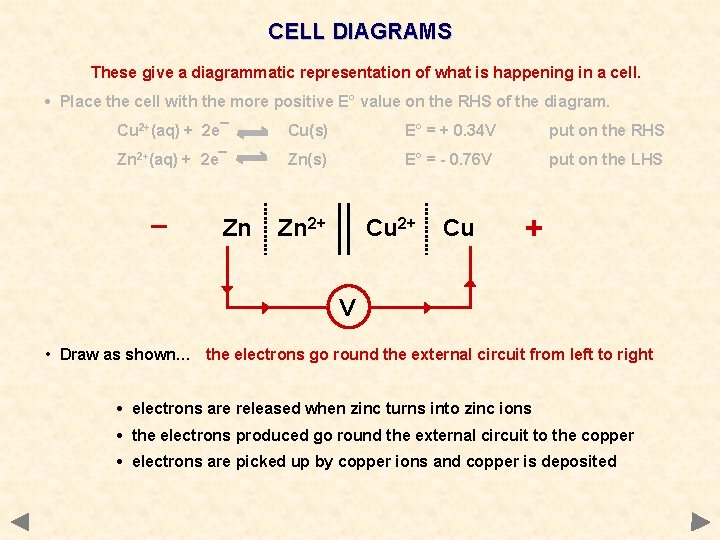
CELL DIAGRAMS These give a diagrammatic representation of what is happening in a cell. • Place the cell with the more positive E° value on the RHS of the diagram. Cu 2+(aq) + 2 e¯ Cu(s) E° = + 0. 34 V put on the RHS Zn 2+(aq) + 2 e¯ Zn(s) E° = - 0. 76 V put on the LHS _ Zn Zn 2+ Cu + V • Draw as shown… the electrons go round the external circuit from left to right • electrons are released when zinc turns into zinc ions • the electrons produced go round the external circuit to the copper • electrons are picked up by copper ions and copper is deposited
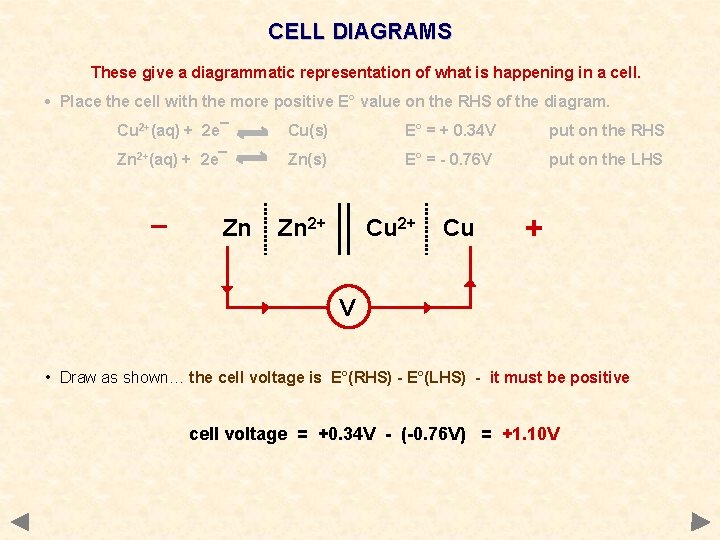
CELL DIAGRAMS These give a diagrammatic representation of what is happening in a cell. • Place the cell with the more positive E° value on the RHS of the diagram. Cu 2+(aq) + 2 e¯ Cu(s) E° = + 0. 34 V put on the RHS Zn 2+(aq) + 2 e¯ Zn(s) E° = - 0. 76 V put on the LHS _ Zn Zn 2+ Cu + V • Draw as shown… the cell voltage is E°(RHS) - E°(LHS) - it must be positive cell voltage = +0. 34 V - (-0. 76 V) = +1. 10 V

USE OF Eo VALUES - WILL IT WORK? E° values Can be used to predict the feasibility of redox and cell reactions In theory ANY REDOX REACTION WITH A POSITIVE E° VALUE WILL WORK In practice, it proceeds if the E° value of the reaction is greater than + 0. 40 V An equation with a more positive E° value reverse a less positive one

USE OF Eo VALUES - WILL IT WORK? An equation with a more positive E° value reverse a less positive one What happens if an Sn(s) / Sn 2+(aq) and a Cu(s) / Cu 2+(aq) cell are connected? Write out the equations Cu 2+(aq) + 2 e¯ Cu(s) ; E° = +0. 34 V Sn 2+(aq) + 2 e¯ Sn(s) ; E° = -0. 14 V the half reaction with the more positive E° value is more likely to work it gets the electrons by reversing the half reaction with the lower E° value therefore Cu 2+(aq) ——> Cu(s) Sn(s) the overall reaction is Cu 2+(aq) + Sn(s) ——> and Sn 2+(aq) ——> Sn 2+(aq) + Cu(s) the cell voltage is the difference in E° values. . . (+0. 34) - (-0. 14) = + 0. 48 V

USE OF Eo VALUES - WILL IT WORK? An equation with a more positive E° value reverse a less positive one Will this reaction be spontaneous? Sn(s) + Cu 2+(aq) ——> Sn 2+(aq) + Cu(s) Write out the appropriate equations Cu 2+(aq) + 2 e¯ Cu(s) ; E° = +0. 34 V as reductions with their E° values Sn 2+(aq) + 2 e¯ Sn(s) ; E° = - 0. 14 V The reaction which takes place will involve the more positive one reversing the other i. e. Cu 2+(aq) ——> Cu(s) and The cell voltage will be the difference in E° values and will be positive. . . Sn(s) ——> Sn 2+(aq) (+0. 34) - (- 0. 14) = + 0. 48 V If this is the equation you want then it will be spontaneous If it is the opposite equation (going the other way) it will not be spontaneous

USE OF Eo VALUES - WILL IT WORK? An equation with a more positive E° value reverse a less positive one Will this reaction be spontaneous? Sn(s) + Cu 2+(aq) ——> Sn 2+(aq) + Cu(s) Split equation into two half equations Cu 2+(aq) Sn(s) + 2 e¯ ——> Sn 2+(aq) Cu(s) + 2 e¯ Find the electrode potentials Cu 2+(aq) + 2 e¯ Cu(s) ; E° = +0. 34 V and the usual equations Sn 2+(aq) + 2 e¯ Sn(s) ; E° = - 0. 14 V Reverse one equation and its sign Sn(s) ——> Sn 2+(aq) Combine the two half equations Sn(s) + Cu 2+(aq) Add the two numerical values + 2 e¯ ; E° = +0. 14 V ——> Sn 2+(aq) + Cu(s) (+0. 34 V) + (+ 0. 14 V) = +0. 48 V If the value is positive the reaction will be spontaneous

REVISION CHECK What should you be able to do? Recall the different types of half cells Recall the structure of the standard hydrogen electrode Recall the methods used to calculate standard electrode potentials Write balanced full and half equations representing electrochemical processes Know that a reaction can be spontaneous if it has a positive E value Calculate if a reaction is feasible by finding its E value CAN YOU DO ALL OF THESE? YES NO

You need to go over the relevant topic(s) again Click on the button to return to the menu

WELL DONE! Try some past paper questions

AN INTRODUCTION TO ELECTRODE POTENTIALS THE END © 2004 JONATHAN HOPTON & KNOCKHARDY PUBLISHING
- Slides: 28