AMS Analysis of Uranium Isotopes and Trace Elements















- Slides: 27

AMS Analysis of Uranium Isotopes and Trace Elements in UOC Samples Prof. William E. (Liam) Kieser André E. Lalonde AMS Laboratory and Dept of Physics and Earth Sciences University of Ottawa International Conference on Nuclear Security February 2020

Overview Uranium Ore Concentrate (UOC) Project Description AMS Analysis of Trace Actinide Isotopes in UOCs Advantages of Accelerator Mass Spectrometry (AMS) AMS Analysis of Trace Medium Mass Isotopes in UOCs How AMS Works Summary and Outlook

The Task: - 307 Uranium Oxide Concentrate Samples from mines and mills around the world. - Find trace elements or even isotopes that could help distinguish one sample from another. If something looks interesting, develop new techniques to explore it - The Tools: At the André E. Lalonde Accelerator Mass Spectrometry Lab: For less abundant elements and isotopes, an Accelerator Mass Spectrometer X -

The A. E. Lalonde Accelerator Mass Spectrometer

Introduction to Accelerator Mass Spectrometry Why add an accelerator to a mass spectrometer? The additional ion energy enables: a) Destruction of molecular isobars and reduction of some atomic isobars b) Noise-free single atom counting and some elemental identification The Results: a) Isotope ratios down to 10 -15 b) Mass sensitivities in the femtogram range Applications: Earth and Planetary Sciences, Environmental History and current monitoring, Bio-medical research, Archeological Science, Forensics

Isobaric Interferences: Isobars are atoms of a different element or molecules that have nearly the same mass as the atoms to be analyzed – these must be eliminated for accurate measurement Atomic Isobars: Several isobars can be eliminated if we use negative rather than positive ions: 14 N for 14 C analysis 26 Mg for 26 Al analysis 129 Xe for 129 I analysis Others can be attenuated using higher energy and interacting with gases or foils: e. g. 10 B for 10 Be analysis Molecular Isobars: e. g. 12 CH 2, 13 CH, 7 Li 2 for 14 C analysis The accelerator enables both these techniques as well as providing noise-free, single atom counting in the detector. X Most molecules are broken apart removing their outer electrons – needs higher energies and interaction with a gas or foil

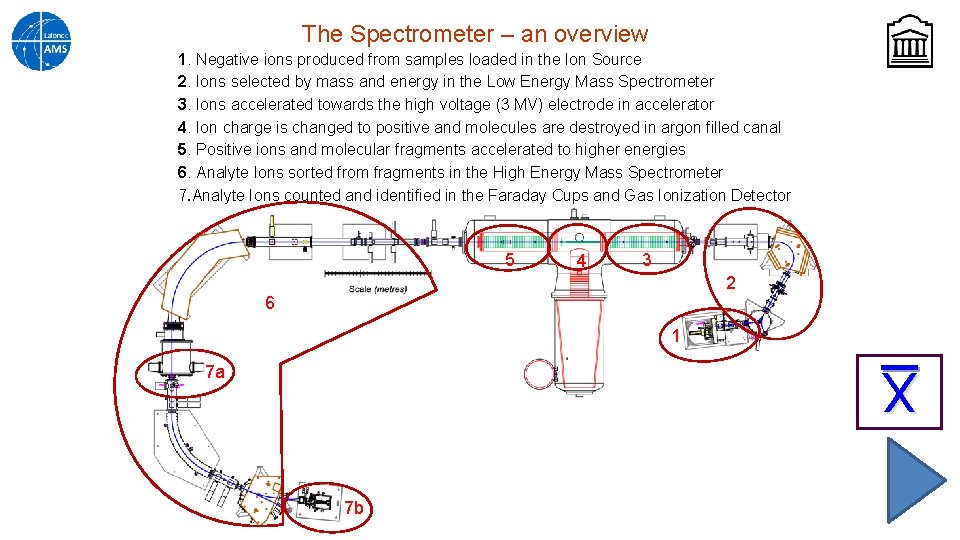
The Spectrometer – an overview 1. Negative ions produced from samples loaded in the Ion Source 2. Ions selected by mass and energy in the Low Energy Mass Spectrometer 3. Ions accelerated towards the high voltage (3 MV) electrode in accelerator 4. Ion charge is changed to positive and molecules are destroyed in argon filled canal 5. Positive ions and molecular fragments accelerated to higher energies 6. Analyte Ions sorted from fragments in the High Energy Mass Spectrometer 7. Analyte Ions counted and identified in the Faraday Cups and Gas Ionization Detector 5 4 3 2 6 1 X 7 a 7 b

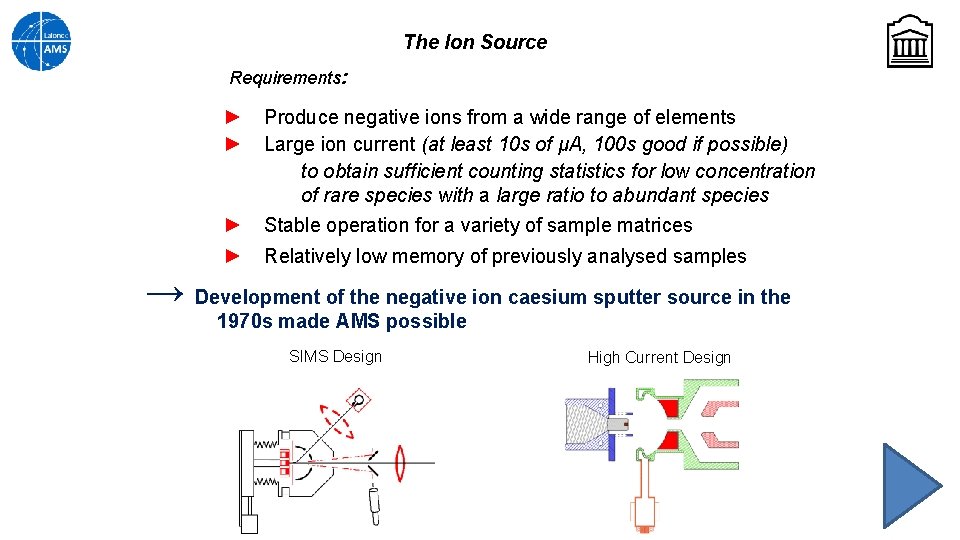
The Ion Source Requirements: ► ► ► Produce negative ions from a wide range of elements Large ion current (at least 10 s of μA, 100 s good if possible) to obtain sufficient counting statistics for low concentration of rare species with a large ratio to abundant species Stable operation for a variety of sample matrices ► Relatively low memory of previously analysed samples → Development of the negative ion caesium sputter source in the 1970 s made AMS possible SIMS Design High Current Design

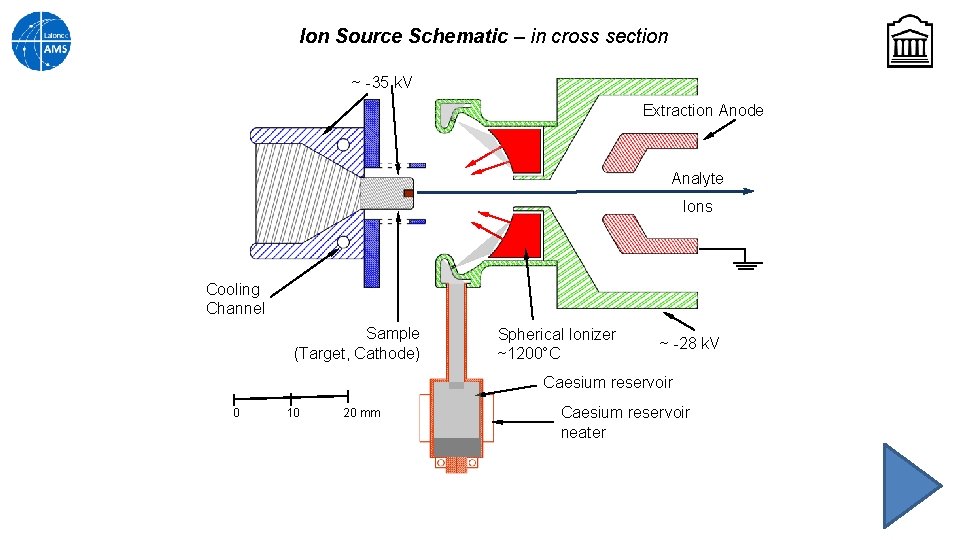
Ion Source Schematic – in cross section ~ -35 k. V Extraction Anode Analyte Ions Cooling Channel Sample (Target, Cathode) Spherical Ionizer ~1200°C ~ -28 k. V Caesium reservoir 0 10 20 mm Caesium reservoir neater

The SO-110 high current ion source 200 Sample Carousel Focusing, Steering Electrodes Sample Insertion Actuator Ion Beam Source Chamber Faraday Cup

The SO-110 high current ion source head Target (~ -35 k. V) Caesium delivery tube Ionizer Location

The SO-110 – target assembly For solid materials – compress into a 1. 3 mm Φ pellet in a replaceable Al, Cu or SS cylinder – provides the micro- environment for the conversion of CO 2 into negative carbon ions – one assembly must be prepared for each 14 C measurement X For gases

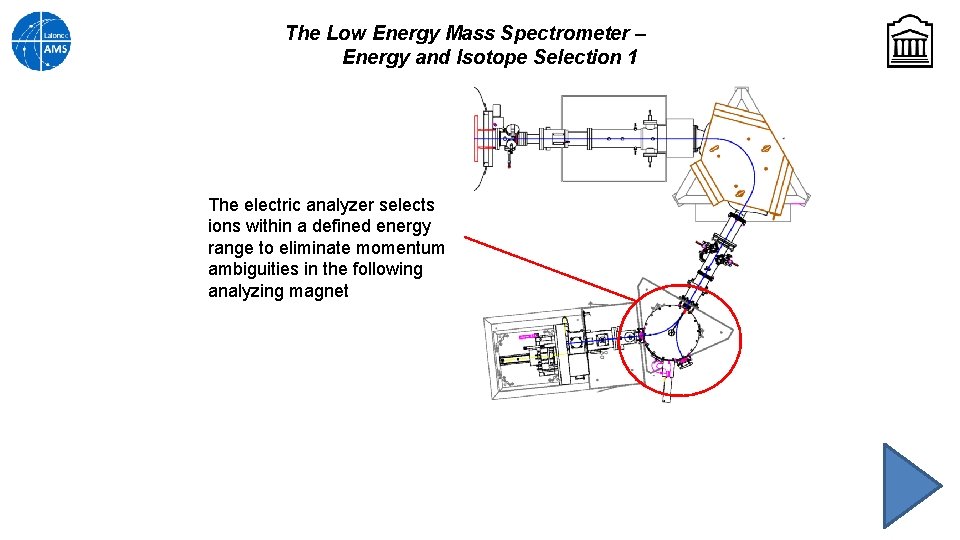
The Low Energy Mass Spectrometer – Energy and Isotope Selection 1 The electric analyzer selects ions within a defined energy range to eliminate momentum ambiguities in the following analyzing magnet

The Low Energy Mass Spectrometer – Energy and Isotope Selection 2 Although the magnet selects ions by momentum – with the electric analyzer, this defines the mass to be injected. Changing the magnetic field is too slow for isotope selection, so the momentum of the ions is locally changed by acceleration or deceleration. To do this, the vacuum box inside the magnet is insulated from the rest of the beam line and a voltage is applied to this box. Isotope Voltage Temporary Energy Momentum Time Carbon-12 +2, 927. 5 V 37. 9275 ke. V 30. 1705 (u-ke. V)1/2 100 µs Carbon-13 +10. 0 V 35, 010. 0 ke. V 30. 1705 (u-ke. V)1/2 100 µs Carbon-14 -2, 490. 7 V 32. 5093 ke. V 30. 175 (u-ke. V)1/2 2 -3 ms

The Low Energy Magnetic Analyzer Upper yoke and coil removed Insulators Vacuum Box Upper Pole

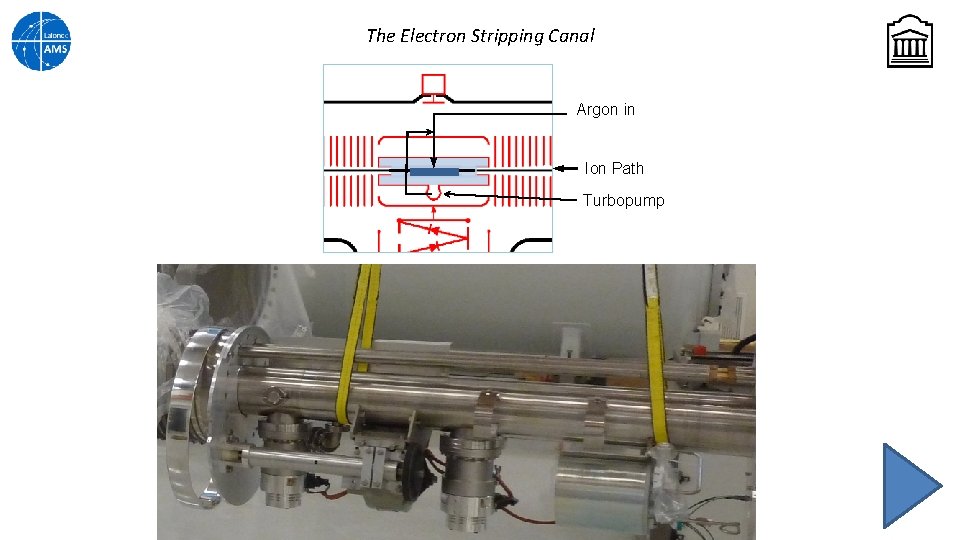
The Electron Stripping Canal Argon in Ion Path Turbopump

The Accelerator Column Assembled Column inside pressure vessel

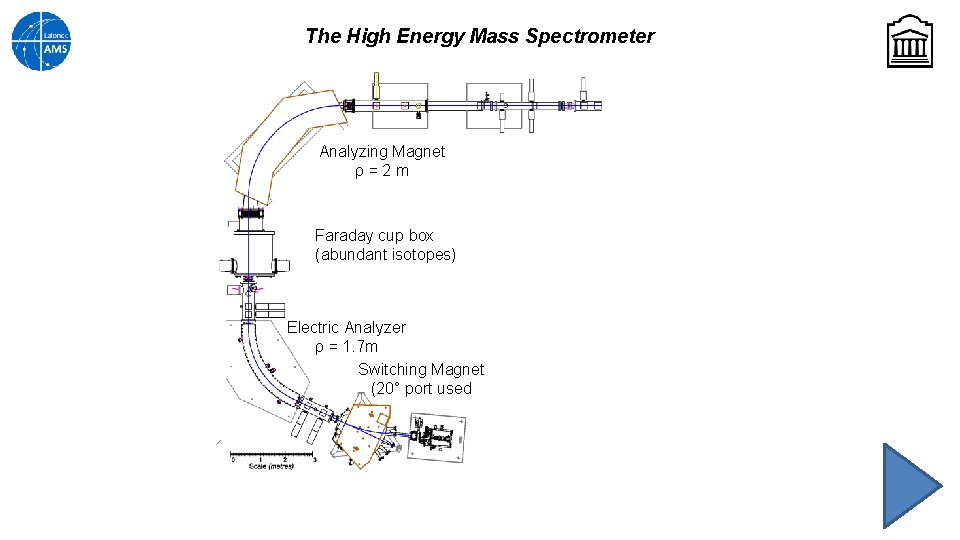
The High Energy Mass Spectrometer Analyzing Magnet ρ=2 m Faraday cup box (abundant isotopes) Electric Analyzer ρ = 1. 7 m Switching Magnet (20° port used

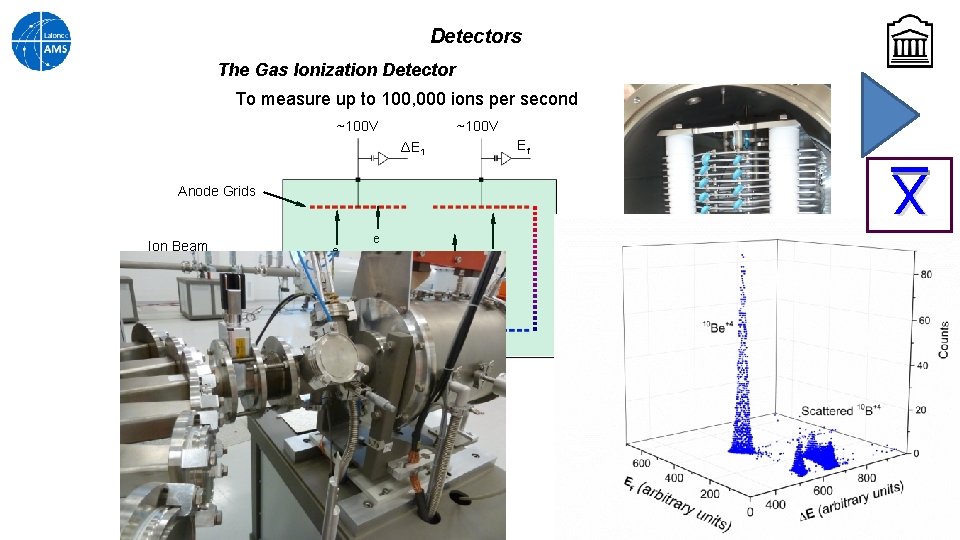
Detectors The Gas Ionization Detector To measure up to 100, 000 ions per second ~100 V Ef ΔE 1 X Anode Grids Ion Beam e e Isobutane gas – 5 -25 mbar 75 nm thick Si. N window Cathode Grid

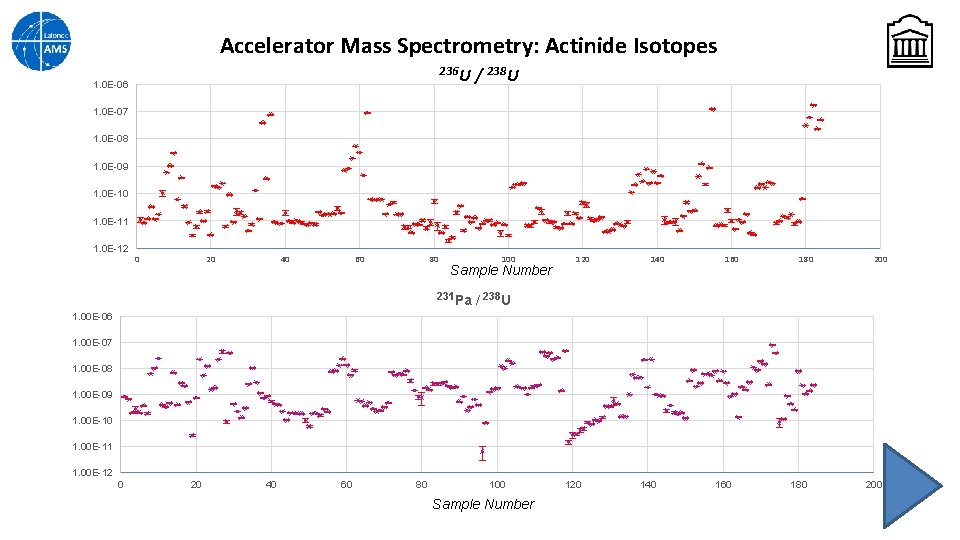
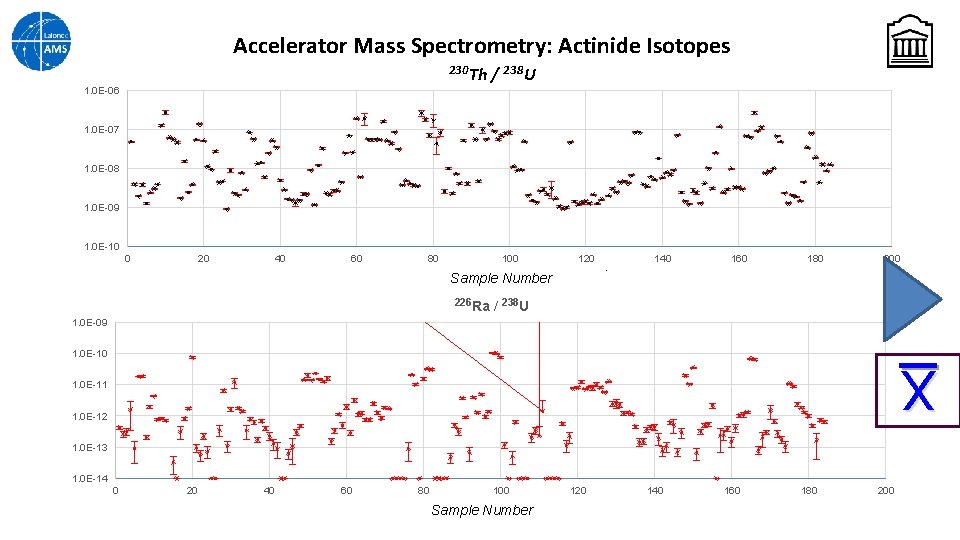
Accelerator Mass Spectrometry: Actinide Isotopes Normally Uranium isotopes are measured with high precision by TIMS, but sample preparation is time-consuming and throughput can be slow UOC samples can be prepared for an AMS ion source by mechanically mixing the powder with a fluorinating agent (e. g. Pb. F 2) and pressing this mixture into the target holder. The AMS system can be programmed to analyze several lowabundance isotopes in the gas ionization detector during one sequence (Slow Sequential Injection). An actinides measurement was set up to measure 238 U current and counts of 236 U, 231 Pa, 230 Th and 226 Ra Problem: Just as in IRMS, AMS needs to measure to a standard. So far, no actinide standards exist (but NRC is working on it)

Accelerator Mass Spectrometry: Actinide Isotopes 236 U 1. 0 E-06 / 238 U 1. 0 E-07 1. 0 E-08 1. 0 E-09 1. 0 E-10 1. 0 E-11 1. 0 E-12 0 20 40 60 80 100 Sample Number 231 Pa 120 140 160 180 200 / 238 U 1. 00 E-06 1. 00 E-07 1. 00 E-08 1. 00 E-09 1. 00 E-10 1. 00 E-11 1. 00 E-12 0 20 40 60 80 100 Sample Number 120 140 160 180 200


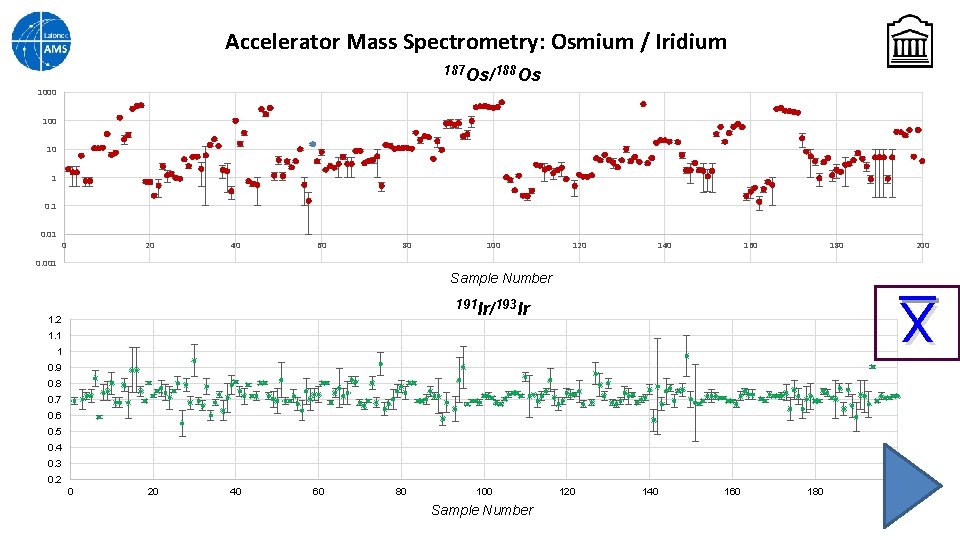
Accelerator Mass Spectrometry: Osmium / Iridium UOC samples can also be prepared for an AMS ion source without mechanically mixing the powder with a fluorinating agent. In this case an oxide beam is produced which, for actinides, would not be as efficient. Unmixed UOC samples can be used to analyze trace isotopes of interest. Of a number of isotopes tried, osmium and iridium looked promising and gave appropriate particle currents. A measurement was set up to look at the ratios 187 Os/188 Os and 191 Ir/193 Ir Again, no standards are available for this material, so the data reported are simply based on the transmission of the instrument.

Accelerator Mass Spectrometry: Osmium / Iridium 187 Os/188 Os 1000 10 1 0. 01 0 20 40 60 80 100 120 140 160 180 200 0. 001 Sample Number X 191 Ir/193 Ir 1. 2 1. 1 1 0. 9 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0 20 40 60 80 100 Sample Number 120 140 160 180 200

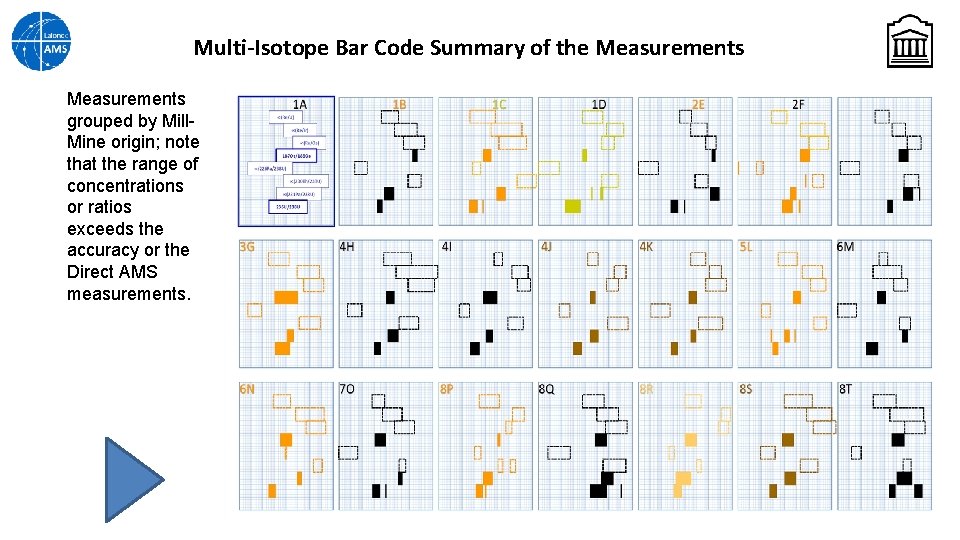
Multi-Isotope Bar Code Summary of the Measurements grouped by Mill. Mine origin; note that the range of concentrations or ratios exceeds the accuracy or the Direct AMS measurements.

Summary 1. AMS analysis, using simple samle preparation can provide actinide measurements which show noticeable differences, but require standard reference materials for reliable accuracy 2. Even simpler sample preparation can provide other trace element measurements which also show noticeable differences, but again, there require standard reference materials for reliable accuracy 3. Ranges of isotope ratios and trace elemental concentrations can provide provenance information from the analysis of UOC samples. 4. We are awaiting the results of the NRC global UOC calibration exercise, so that these measurements can be reported with greater accuracy

u. Ottawa Advanced Research Complex Home of the Jan Veizer Stable Isotope Laboratory the André E. Lalonde AMS Laboratory Investigators, Affiliations and Acknowledgements A. E. Litherland Ian D. Clark, Liam Kieser, Jack Cornett Xiao-Lei Zhao, , Gilles St-Jean, Norm St-Jean, , Iso. Trace Laboratory, University of Toronto A. E. Lalonde AMS Laboratory, University of Ottawa Funding from: Centre for Security Science Canada Foundation for Innovation Ontario Research Fund B