Amphoteric Al By Dr Prem D Sattsangi 2010

Amphoteric “Al” By: Dr. Prem D. Sattsangi © 2010 - and Christopher L. Byers (programmer)

Meet Amphoteric Actor, “Al. ” METAL NON-METAL . . . . This is Alex, called “Al” for short (an amphoteric actor).
Profiles for “Al” METAL NON-METAL . . . .
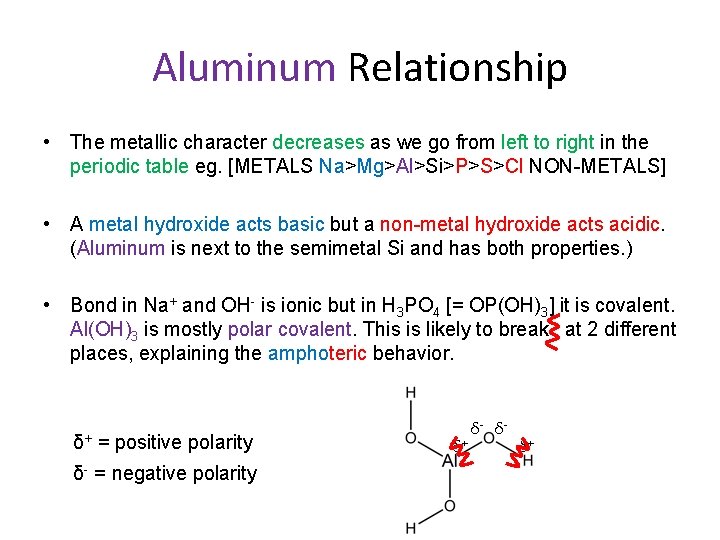
Aluminum Relationship • The metallic character decreases as we go from left to right in the periodic table eg. [METALS Na>Mg>Al>Si>P>S>Cl NON-METALS] • A metal hydroxide acts basic but a non-metal hydroxide acts acidic. (Aluminum is next to the semimetal Si and has both properties. ) • Bond in Na+ and OH- is ionic but in H 3 PO 4 [= OP(OH)3] it is covalent. Al(OH)3 is mostly polar covalent. This is likely to break at 2 different places, explaining the amphoteric behavior. δ+ = positive polarity δ- = negative polarity δ+ δ- δ- δ+
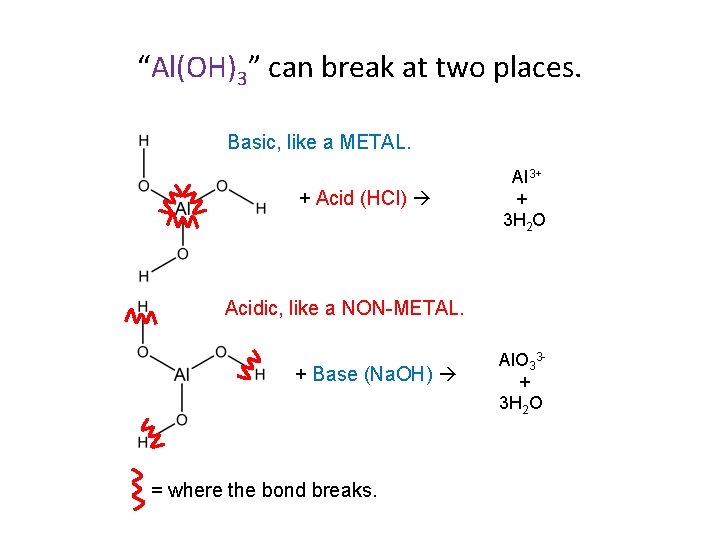
“Al(OH)3” can break at two places. Basic, like a METAL. + Acid (HCl) Al 3+ + 3 H 2 O Acidic, like a NON-METAL. + Base (Na. OH) = where the bond breaks. Al. O 33+ 3 H 2 O
![METAL and NON-METAL Hydroxides [METALS Na>Mg>Al>Si>P>S>Cl NON-METALS] Al(OH)3 METAL Hydroxide NON-METAL Hydroxide A. Ionic METAL and NON-METAL Hydroxides [METALS Na>Mg>Al>Si>P>S>Cl NON-METALS] Al(OH)3 METAL Hydroxide NON-METAL Hydroxide A. Ionic](http://slidetodoc.com/presentation_image_h2/990ea3f5257288a0fb734e5a97d37e66/image-6.jpg)
METAL and NON-METAL Hydroxides [METALS Na>Mg>Al>Si>P>S>Cl NON-METALS] Al(OH)3 METAL Hydroxide NON-METAL Hydroxide A. Ionic eg. Na+ OHB. Covalent eg. Al(OH)3 Basic: Reacts with Acidic: Reacts with Base
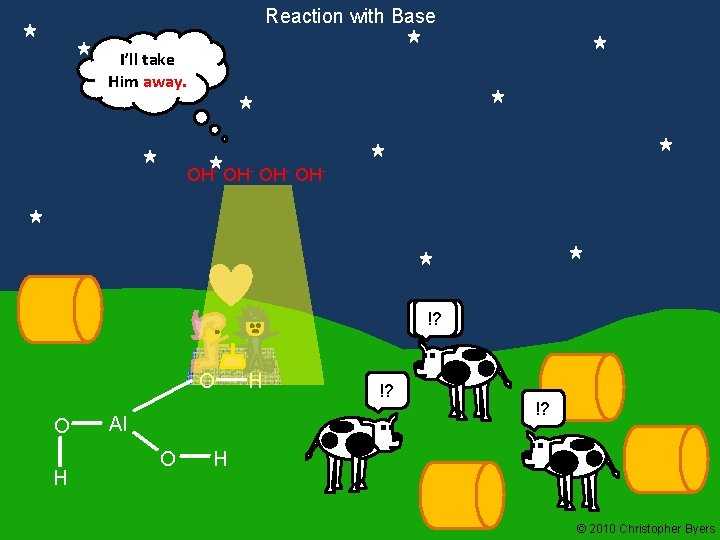
Reaction with Base I’ll take Him away. OH- OH- Moo !? O O H Al O H !? H © 2010 Christopher Byers
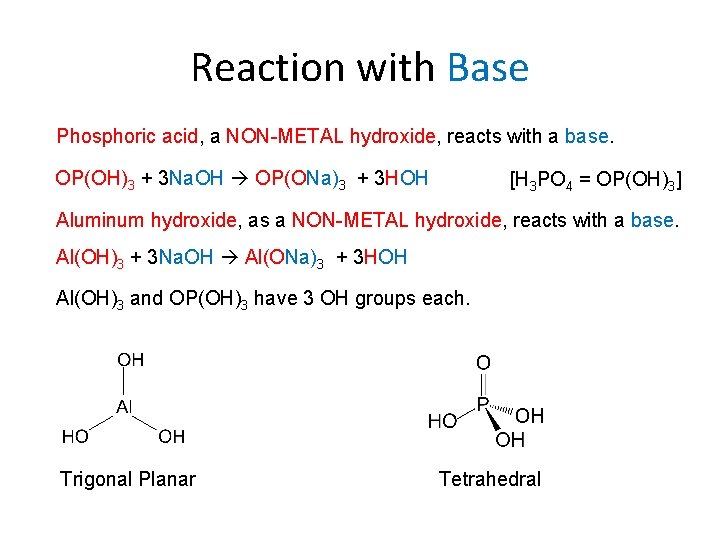
Reaction with Base Phosphoric acid, a NON-METAL hydroxide, reacts with a base. OP(OH)3 + 3 Na. OH OP(ONa)3 + 3 HOH [H 3 PO 4 = OP(OH)3] Aluminum hydroxide, as a NON-METAL hydroxide, reacts with a base. Al(OH)3 + 3 Na. OH Al(ONa)3 + 3 HOH Al(OH)3 and OP(OH)3 have 3 OH groups each. Trigonal Planar Tetrahedral
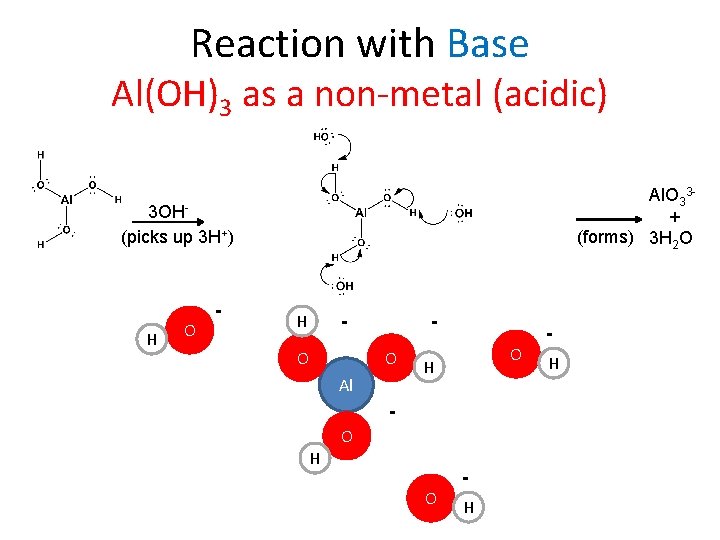
Reaction with Base Al(OH)3 as a non-metal (acidic) Al. O 33+ (forms) 3 H 2 O 3 OH(picks up 3 H+) H O - H - O O Al O H O H H
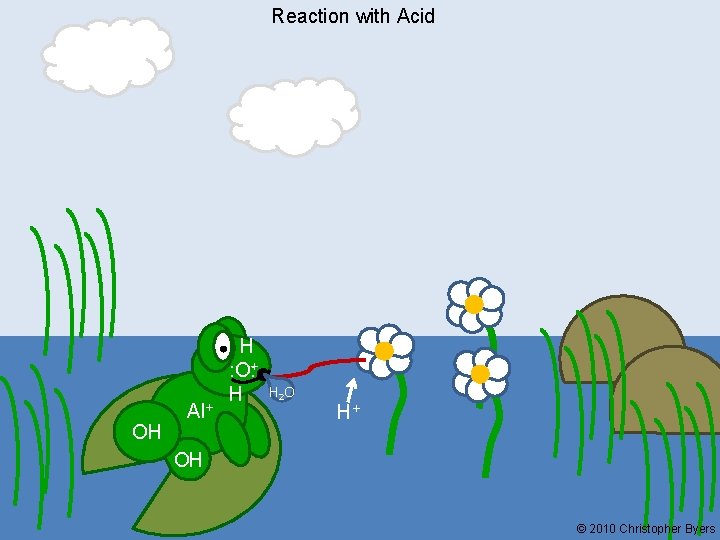
Reaction with Acid OH Al+ H : O: + : O H H 2 O H+ OH © 2010 Christopher Byers

Reaction with Acid Sodium hydroxide, an ionic METAL hydroxide, reacts with an acid. Na. OH + HCl Na. Cl + HOH Aluminum hydroxide, as a covalent METAL hydroxide, reacts with an acid by catalysis. Al(OH)3 + 3 HCl Al. Cl 3 + 3 HOH Both have similar results.
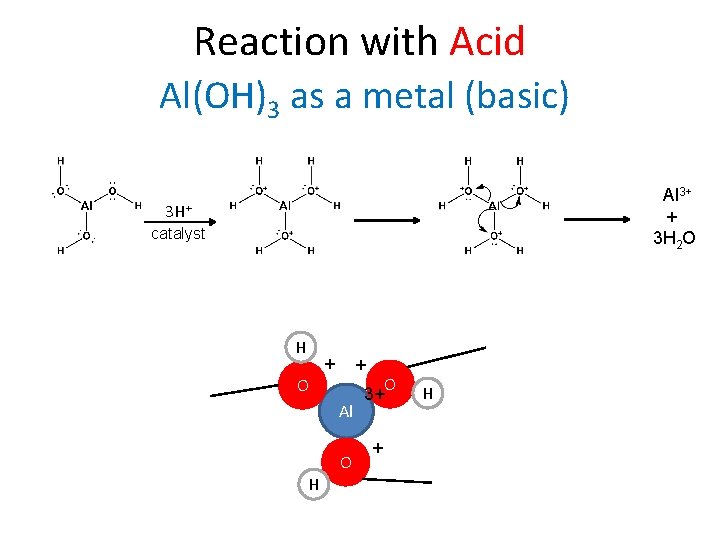
Reaction with Acid Al(OH)3 as a metal (basic) Al 3+ + 3 H 2 O 3 H+ catalyst H H + + + O Al O H H O 3+ H + +

Amphoteric Concept • What does it mean to be amphoteric? • Amphoteric means capable of functioning as either an acid or a base. • How is Aluminum amphoteric? • In presence of a base Al(OH)3 releases 3 H+. • eg. Al(OH)3 + 3 Na. OH Al(ONa)3 + 3 H 2 O. • In presence of an acid Al(OH)3 releases 3 OH-. • eg. Al(OH)3 + 3 HCl Al. Cl 3 + 3 H 2 O.

PARTY TIME! Hmmm… Actually, that’s all folks. Enjoy the rest of your day!. . . Are they gone? Then, it’s time to… What! you’re still here? GO! !
- Slides: 14