Amino acids Rgroups nonpolar acidic basic proteins condensation
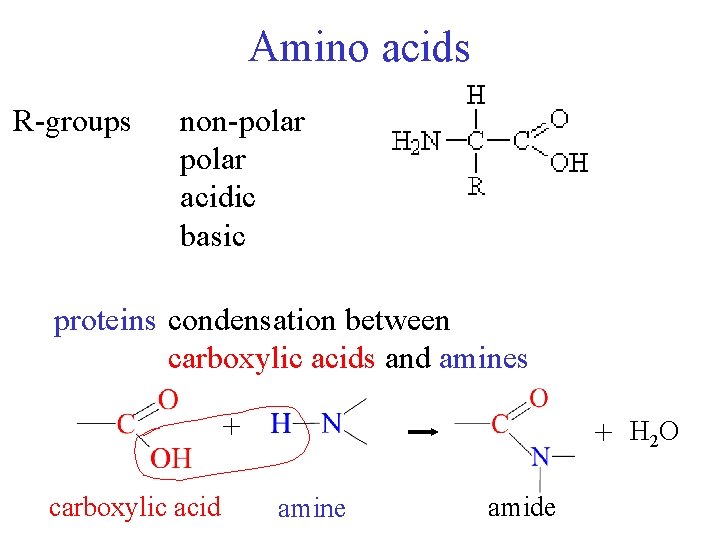
Amino acids R-groups non-polar acidic basic proteins condensation between carboxylic acids and amines + carboxylic acid + H 2 O amine amide
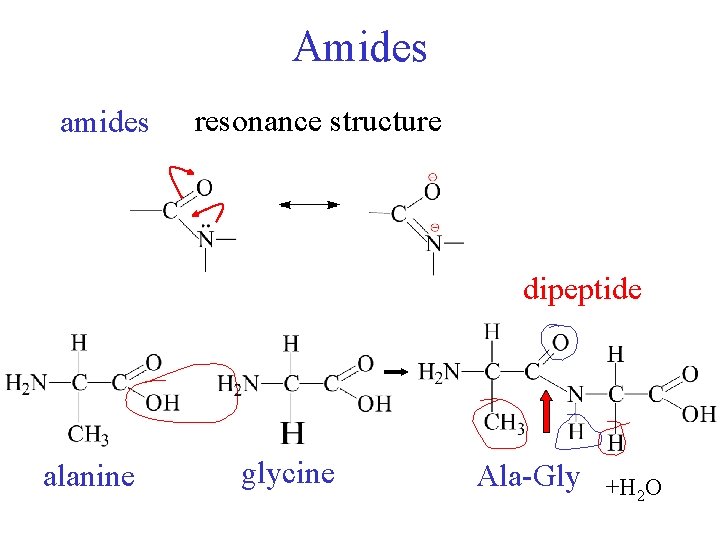
Amides amides resonance structure dipeptide alanine glycine Ala-Gly +H 2 O
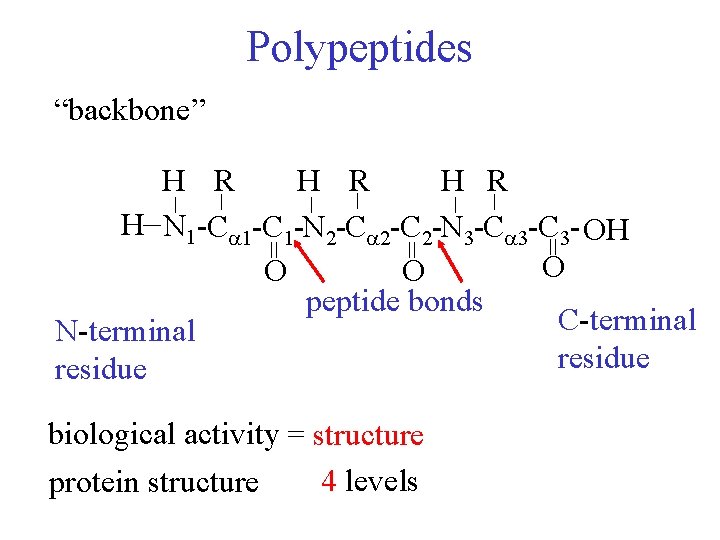
Polypeptides “backbone” _ _ _ H R H R _ H N 1 -C 1 -N 2 -C 2 -N 3 -C 3 - OH O O O peptide bonds C-terminal N-terminal residue = = = biological activity = structure 4 levels protein structure
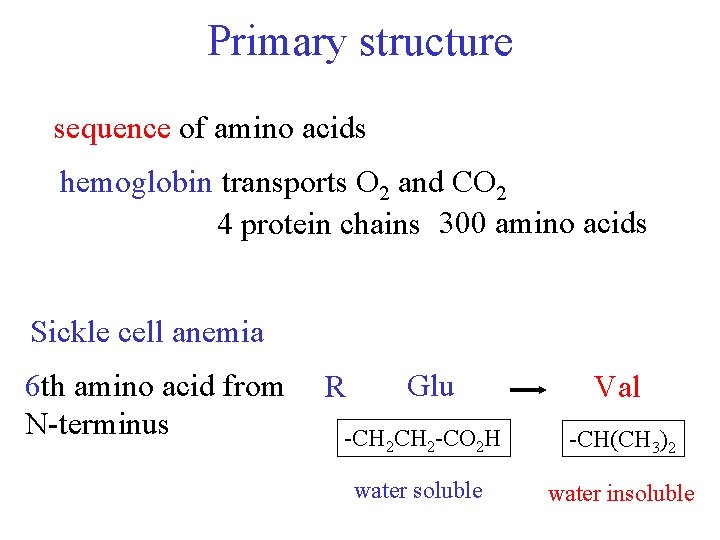
Primary structure sequence of amino acids hemoglobin transports O 2 and CO 2 4 protein chains 300 amino acids Sickle cell anemia 6 th amino acid from N-terminus R Glu Val -CH 2 -CO 2 H -CH(CH 3)2 water soluble water insoluble
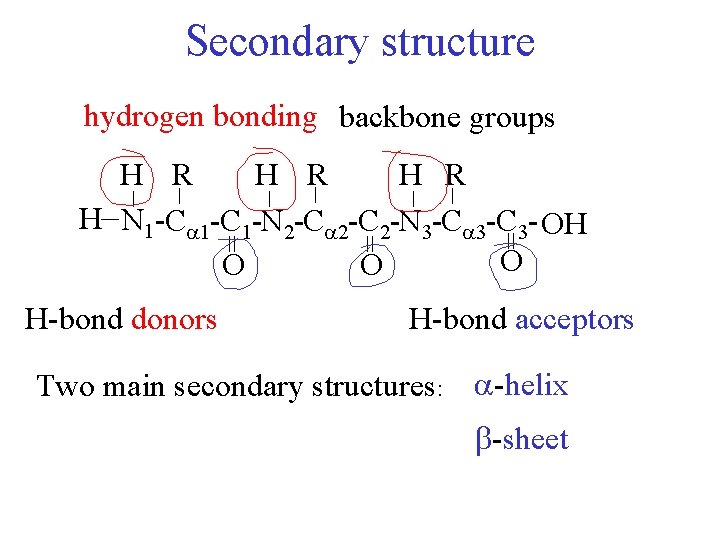
Secondary structure hydrogen bonding backbone groups = = = H-bond donors _ _ _ H R H R _ H N 1 -C 1 -N 2 -C 2 -N 3 -C 3 - OH O O O H-bond acceptors Two main secondary structures: -helix -sheet
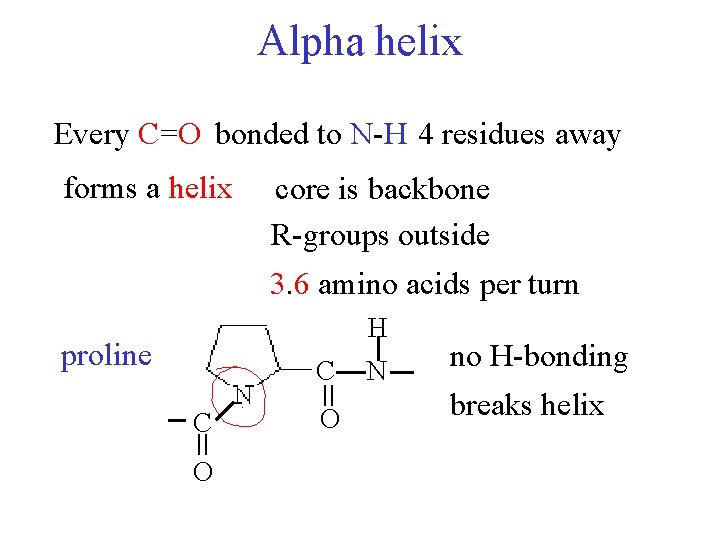
Alpha helix Every C=O bonded to N-H 4 residues away forms a helix core is backbone R-groups outside 3. 6 amino acids per turn H proline = C O N no H-bonding breaks helix

Beta sheet Every C=O bonded to N-H far apart in 1 o structure on different chains peptide chains extended side-by-side maximal H-bonding for anti-parallel chains small R-groups above and below the sheet if not -helix or -sheet random coil

Fibrous Proteins 1 o structure amino acid sequence 2 o structure - helix -sheet H-bonding between C=O- and N-H+ of backbone some proteins only have 1 o and 2 o structure: fibroin (silk) keratin hair collagen skin -sheet - helix insoluble in H 2 O non-polar residues
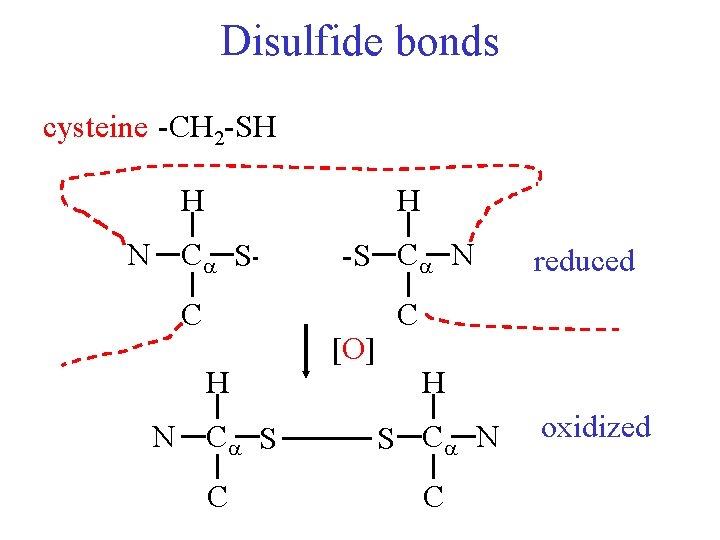
Disulfide bonds cysteine -CH 2 -SH H H N C S-H C H H-S C N [O] reduced C H N C S S C N C C oxidized

Tertiary structure Primary structure sequence of amino acids Ala-Phe-Ser-Val-Glu-His-Ile-Met-Arg-Asp-Val-His-Asn-Gly Ala-Phe-Ser-Val- Glu-His-Ile-Met-Arg-Asp-Val-His-Asn-Gly- Non-polar Polar Acidic or Basic Alanine Phenylalanine Valine Isoleusine Methionine Glycine Serine Histidine Glutamic acid Arginine Aspartic acid Asperagine
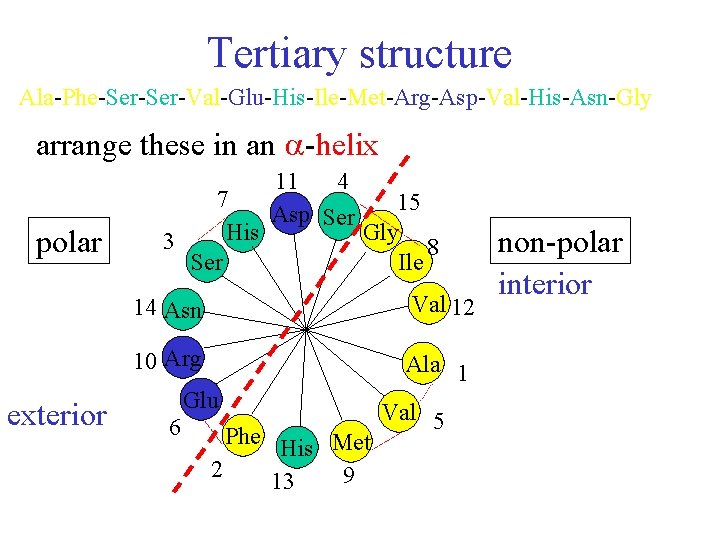
Tertiary structure Ala-Phe-Ser-Val-Glu-His-Ile-Met-Arg-Asp-Val-His-Asn-Gly arrange these in an -helix polar exterior 7 His 3 Ser 11 4 Asp Ser 15 Gly 8 Ile 14 Asn Val 12 10 Arg Ala 1 Glu 6 Phe 2 His Met 9 13 Val 5 non-polar interior
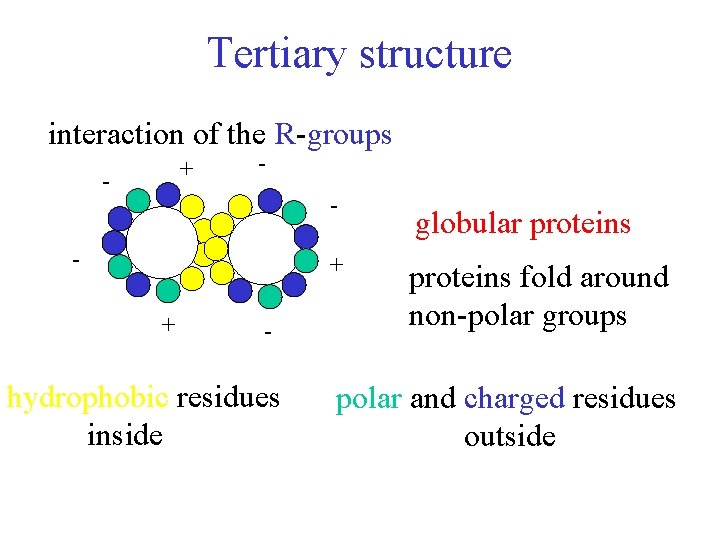
Tertiary structure interaction of the R-groups + - - + + - hydrophobic residues inside globular proteins fold around non-polar groups polar and charged residues outside

Tertiary structure interactions of R-groups 1. Hydrophobic interactions non-polar R-groups LDF 2. Hydrogen bonding polar R-groups between H-bond donors and acceptors 3. Ionic bonds (salt bridges) acidic and basic R-groups ion-ion 4. Covalent bonds (disulfide) cysteins
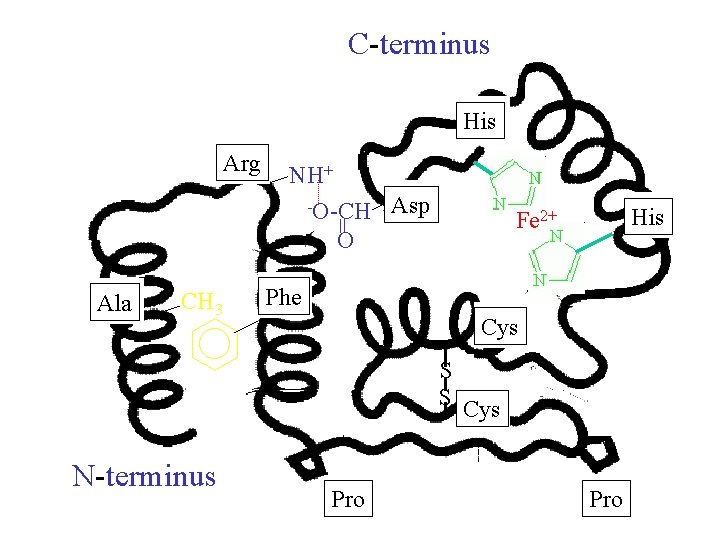
C-terminus His Arg NH+ Asp O Ala CH 3 Phe Cys S S N-terminus His Fe 2+ = -O-CH Pro Cys Pro
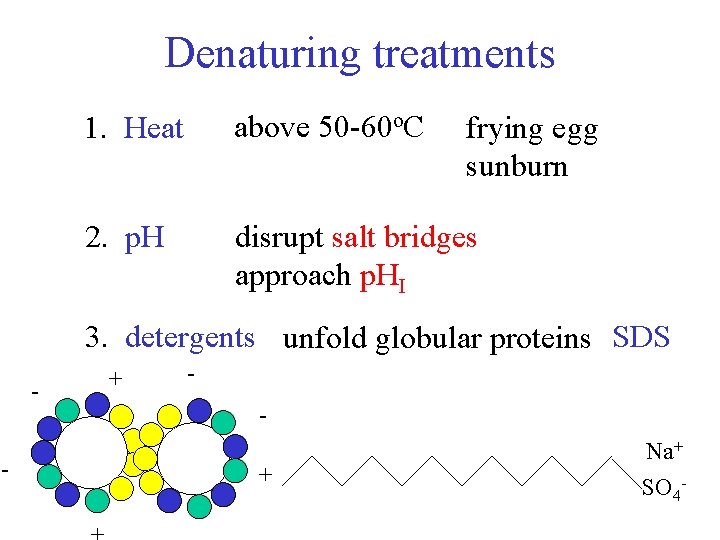
Denaturing treatments 1. Heat above 50 -60 o. C 2. p. H disrupt salt bridges approach p. HI frying egg sunburn 3. detergents unfold globular proteins SDS - + + Na+ SO 4 -
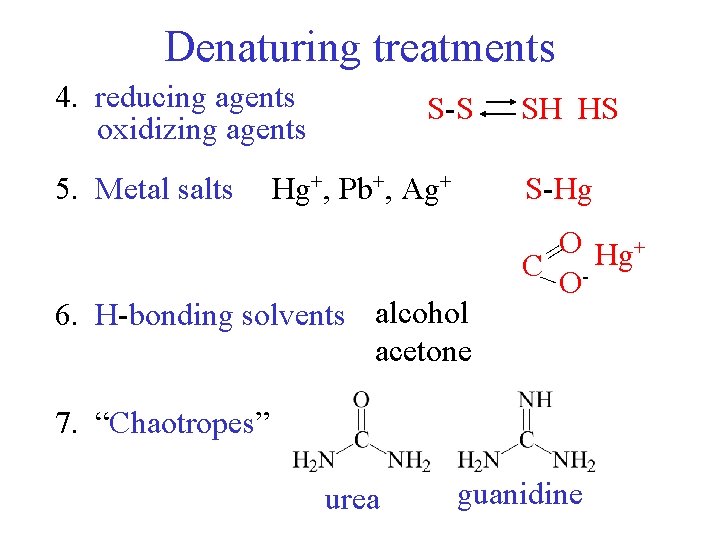
Denaturing treatments 4. reducing agents oxidizing agents 5. Metal salts S-S Hg+, Pb+, Ag+ SH HS S-Hg 6. H-bonding solvents alcohol acetone O Hg+ = C_ O 7. “Chaotropes” urea guanidine
- Slides: 16