AMINO ACIDS METABOLISM 2011 1 Text Harpers Biochemistry

AMINO ACIDS METABOLISM 2011년 1학기 Text : Harper’s Biochemistry Refs: Devlin’s, Lippincott’s Ch. 27. Biosynthesis of the Nutritionally Nonessential Amino Acids Ch. 28. Catabolism of Proteins & of Amino Acid Nitrogen Ch. 29. Catabolism of the Carbon Skeletons of Amino Acids Ch. 30. Conversion of Amino Acids to Specialized Products Byung Kwan Jin, Ph. D. Professor/Director Dept. of Biochemistry & Molecular Biology Neurodegeneration Control Research Center School of Medicine Kyung Hee University

Chapter 27. Biosynthesis of the Nutritionally Nonessential Amino Acids Biomedical Importance * Medical implications related to the Amino acid (AA) deficiency: If nutritionally essential AAs are absent from the diet, or are present in inadequate amounts * Amino acid deficiency: kwashiorkor, marasmus
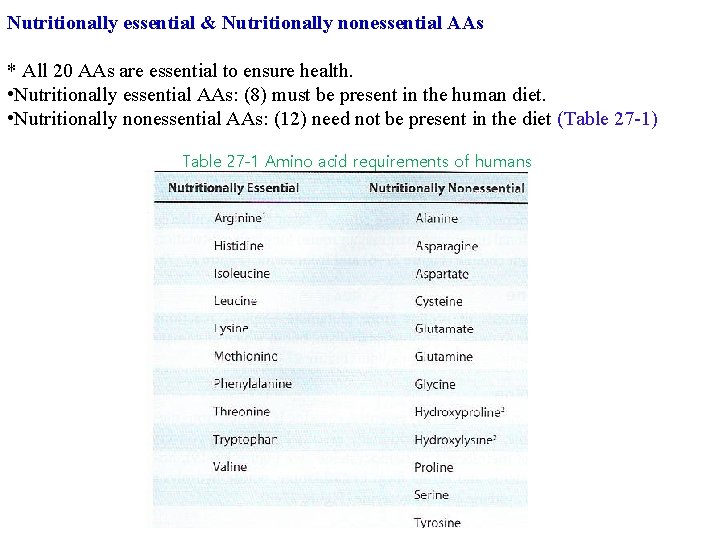
Nutritionally essential & Nutritionally nonessential AAs * All 20 AAs are essential to ensure health. • Nutritionally essential AAs: (8) must be present in the human diet. • Nutritionally nonessential AAs: (12) need not be present in the diet (Table 27 -1) Table 27 -1 Amino acid requirements of humans
Chapter 27. Biosynthesis of the Nutritionally Nonessential Amino Acids * Reactions and intermediates involved in the Biosynthesis (by human tissues) of the 12 nutritionally nonessential 12 AAs * Selected nutritional and metabolic disorders associated with metaboilsm Carbons NH 3
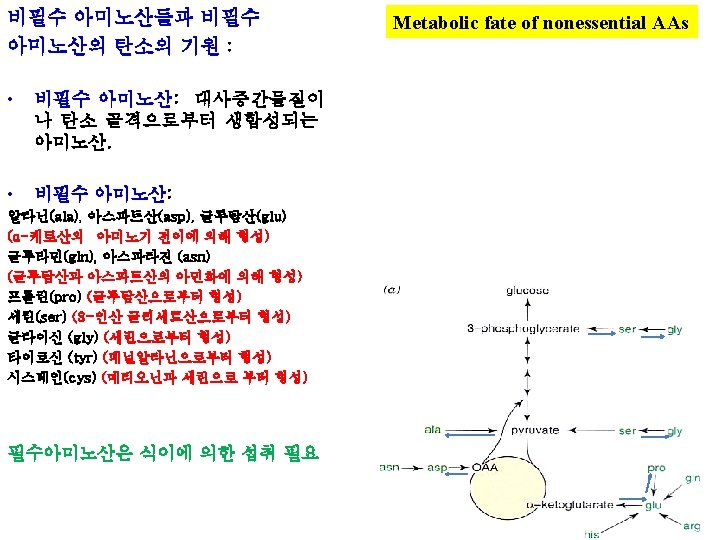
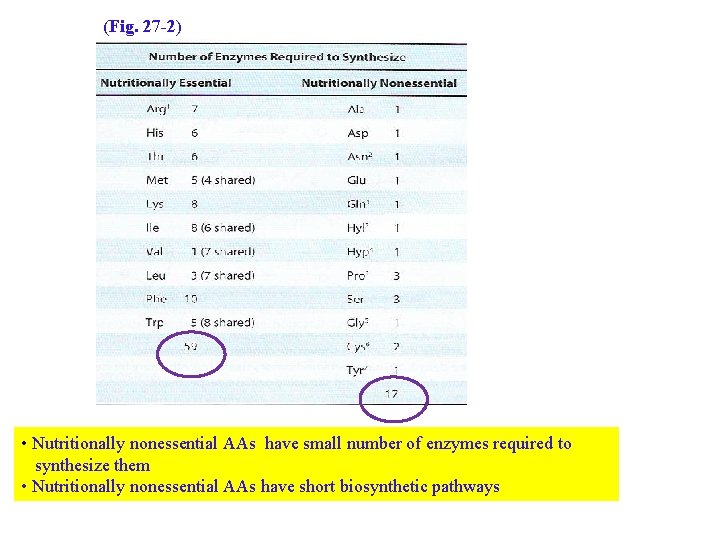
(Fig. 27 -2) • Nutritionally nonessential AAs have small number of enzymes required to synthesize them • Nutritionally nonessential AAs have short biosynthetic pathways

Glutamate dehydrogenase Glutamine synthetase Aminotransferase play central roles in AA biosynthesis
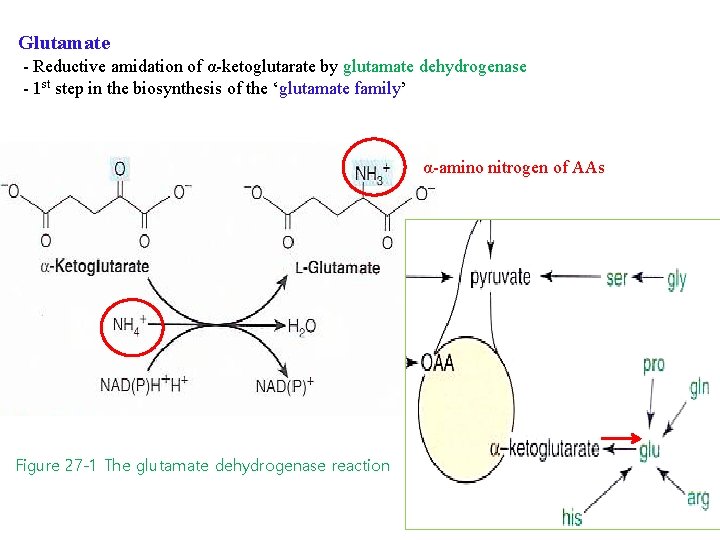
Glutamate - Reductive amidation of α-ketoglutarate by glutamate dehydrogenase - 1 st step in the biosynthesis of the ‘glutamate family’ α-amino nitrogen of AAs Figure 27 -1 The glutamate dehydrogenase reaction
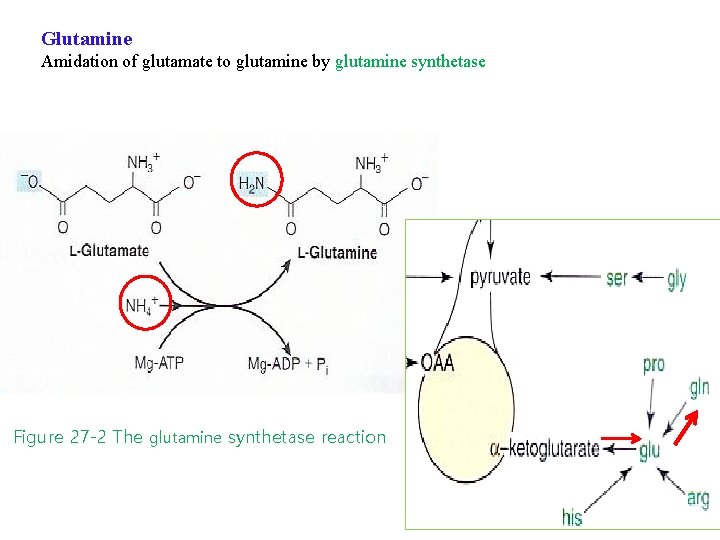
Glutamine Amidation of glutamate to glutamine by glutamine synthetase Figure 27 -2 The glutamine synthetase reaction
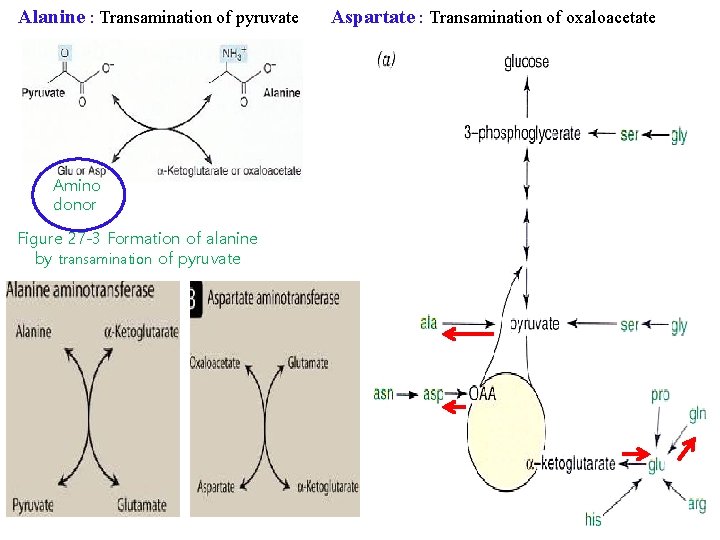
Alanine : Transamination of pyruvate Amino donor Figure 27 -3 Formation of alanine by transamination of pyruvate Aspartate : Transamination of oxaloacetate
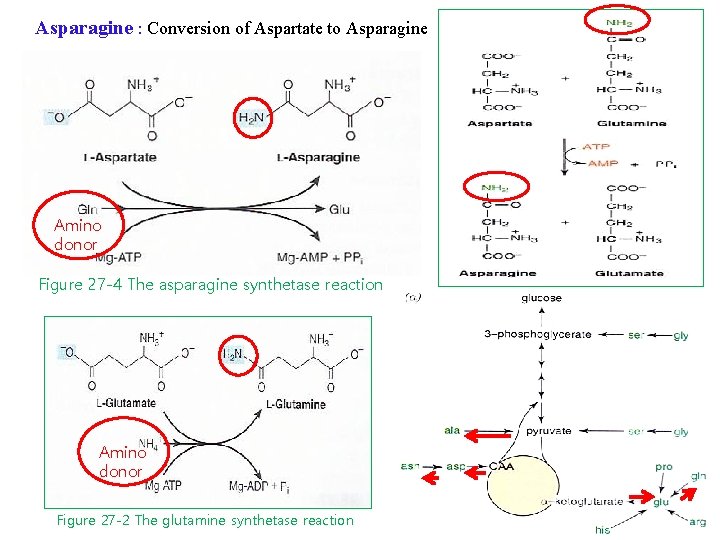
Asparagine : Conversion of Aspartate to Asparagine Amino donor Figure 27 -4 The asparagine synthetase reaction Amino donor Figure 27 -2 The glutamine synthetase reaction
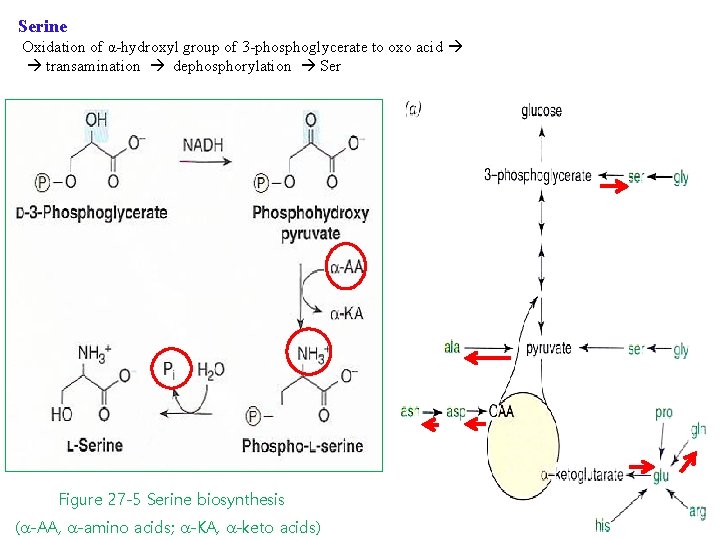
Serine Oxidation of α-hydroxyl group of 3 -phosphoglycerate to oxo acid transamination dephosphorylation Ser Figure 27 -5 Serine biosynthesis (a-AA, a-amino acids; a-KA, a-keto acids)
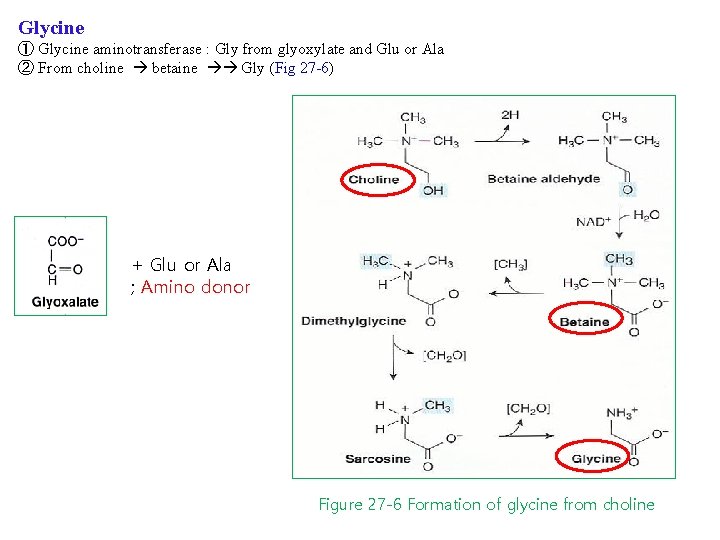
Glycine ① Glycine aminotransferase : Gly from glyoxylate and Glu or Ala ② From choline betaine Gly (Fig 27 -6) + Glu or Ala ; Amino donor Figure 27 -6 Formation of glycine from choline
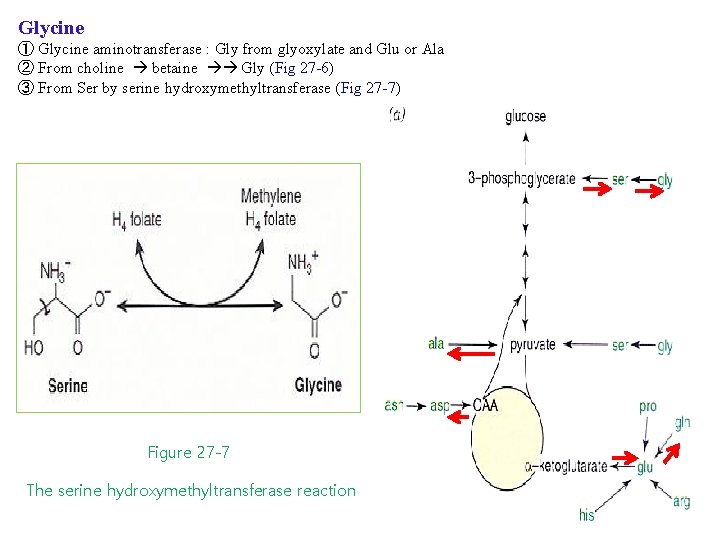
Glycine ① Glycine aminotransferase : Gly from glyoxylate and Glu or Ala ② From choline betaine Gly (Fig 27 -6) ③ From Ser by serine hydroxymethyltransferase (Fig 27 -7) Figure 27 -7 The serine hydroxymethyltransferase reaction
Glycine ① Glycine aminotransferase : Gly from glyoxylate and Glu or Ala ② From choline betaine Gly (Fig 27 -6) ③ From Ser by serine hydroxymethyltransferase (Fig 27 -7) ④ Mitochondrial Gly synthase complex (Fig. 29 -6)
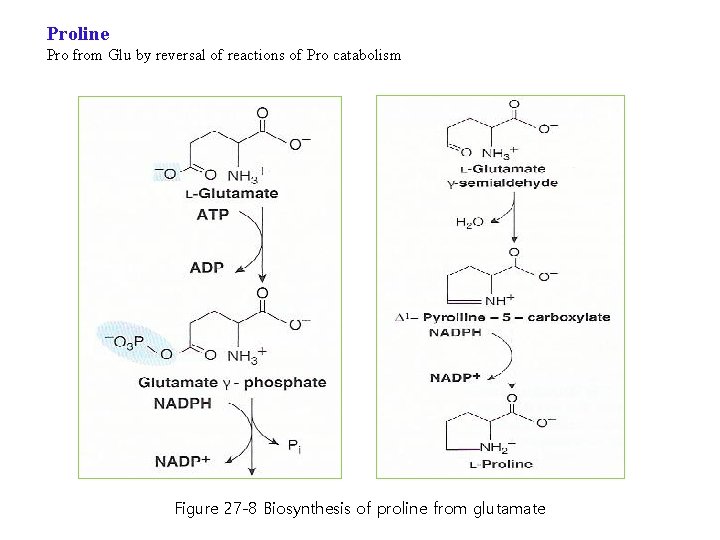
Proline Pro from Glu by reversal of reactions of Pro catabolism Figure 27 -8 Biosynthesis of proline from glutamate
Proline Pro from Glu by reversal of reactions of Pro catabolism
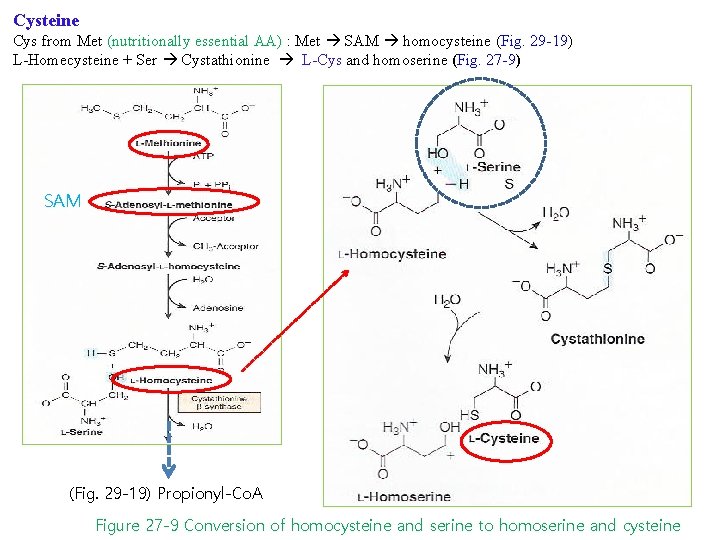
Cysteine Cys from Met (nutritionally essential AA) : Met SAM homocysteine (Fig. 29 -19) L-Homecysteine + Ser Cystathionine L-Cys and homoserine (Fig. 27 -9) SAM (Fig. 29 -19) Propionyl-Co. A Figure 27 -9 Conversion of homocysteine and serine to homoserine and cysteine
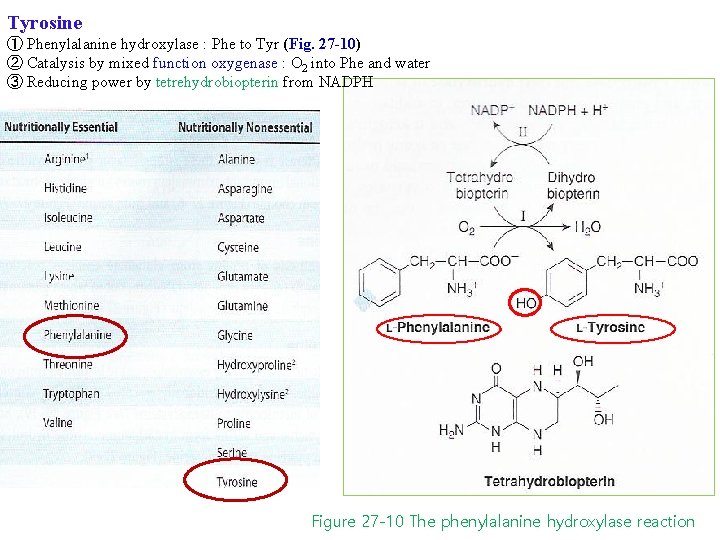
Tyrosine ① Phenylalanine hydroxylase : Phe to Tyr (Fig. 27 -10) ② Catalysis by mixed function oxygenase : O 2 into Phe and water ③ Reducing power by tetrehydrobiopterin from NADPH Figure 27 -10 The phenylalanine hydroxylase reaction
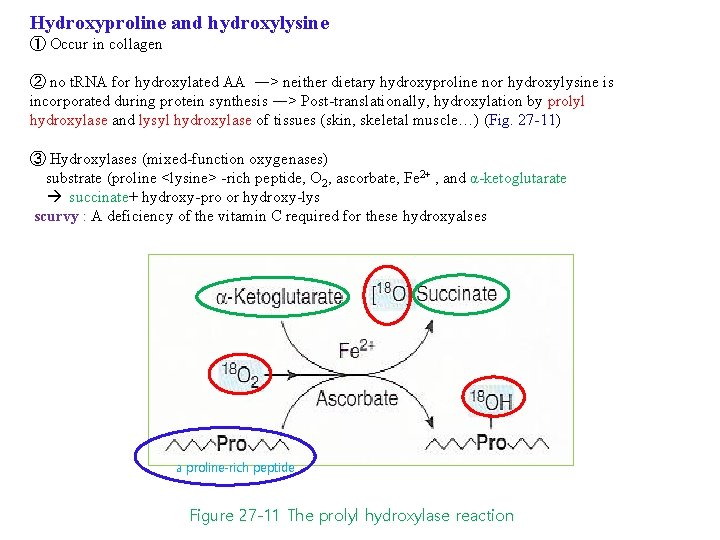
Hydroxyproline and hydroxylysine ① Occur in collagen ② no t. RNA for hydroxylated AA ㅡ> neither dietary hydroxyproline nor hydroxylysine is incorporated during protein synthesis ㅡ> Post-translationally, hydroxylation by prolyl hydroxylase and lysyl hydroxylase of tissues (skin, skeletal muscle…) (Fig. 27 -11) ③ Hydroxylases (mixed-function oxygenases) substrate (proline <lysine> -rich peptide, O 2, ascorbate, Fe 2+ , and α-ketoglutarate succinate+ hydroxy-pro or hydroxy-lys scurvy : A deficiency of the vitamin C required for these hydroxyalses a proline-rich peptide Figure 27 -11 The prolyl hydroxylase reaction
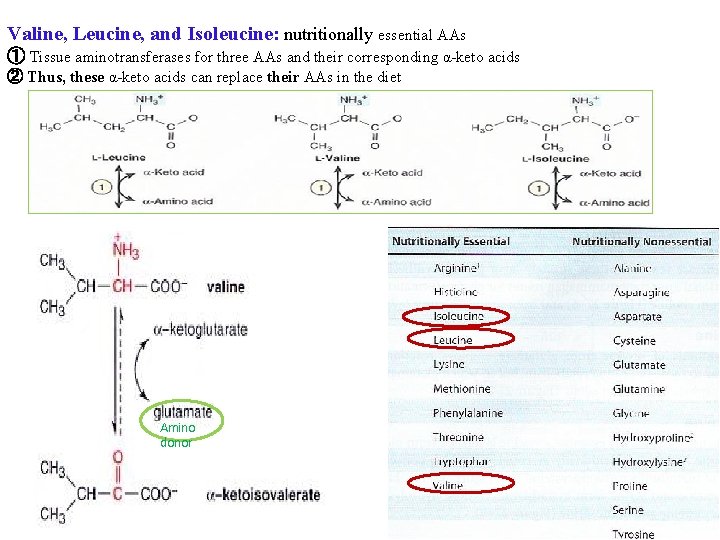
Valine, Leucine, and Isoleucine: nutritionally essential AAs ① Tissue aminotransferases for three AAs and their corresponding α-keto acids ② Thus, these α-keto acids can replace their AAs in the diet Amino donor
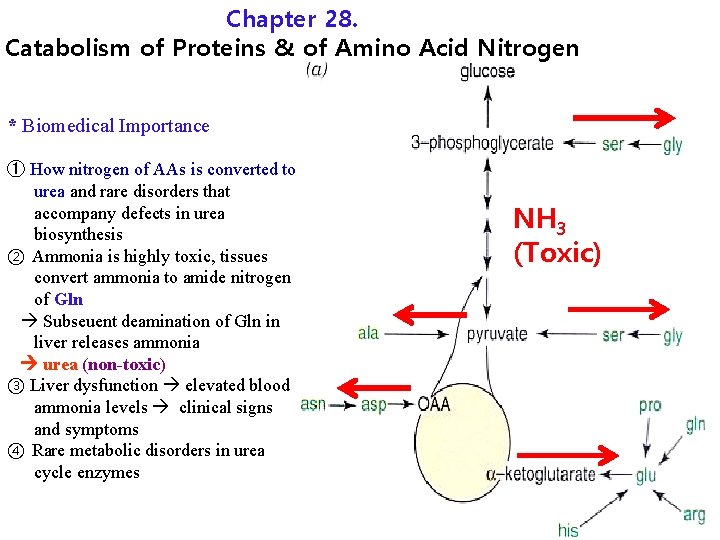
Chapter 28. Catabolism of Proteins & of Amino Acid Nitrogen * Biomedical Importance ① How nitrogen of AAs is converted to urea and rare disorders that accompany defects in urea biosynthesis ② Ammonia is highly toxic, tissues convert ammonia to amide nitrogen of Gln Subseuent deamination of Gln in liver releases ammonia urea (non-toxic) ③ Liver dysfunction elevated blood ammonia levels clinical signs and symptoms ④ Rare metabolic disorders in urea cycle enzymes NH 3 (Toxic)
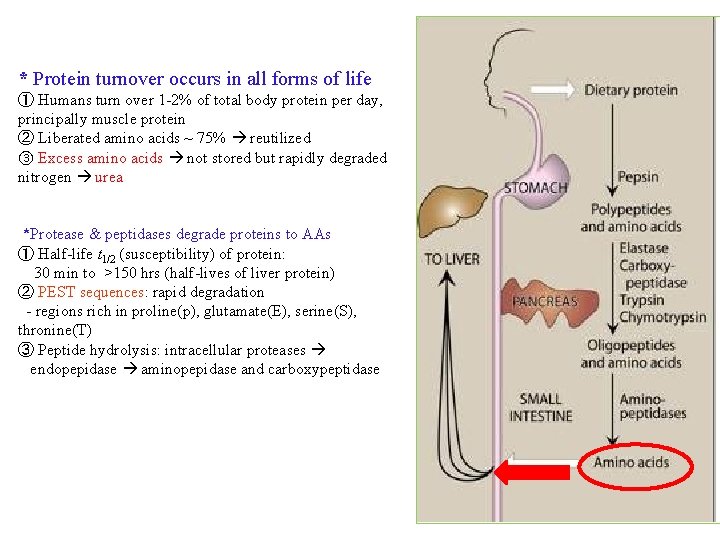
* Protein turnover occurs in all forms of life ① Humans turn over 1 -2% of total body protein per day, principally muscle protein ② Liberated amino acids ~ 75% reutilized ③ Excess amino acids not stored but rapidly degraded nitrogen urea *Protease & peptidases degrade proteins to AAs ① Half-life t 1/2 (susceptibility) of protein: 30 min to >150 hrs (half-lives of liver protein) ② PEST sequences: rapid degradation - regions rich in proline(p), glutamate(E), serine(S), thronine(T) ③ Peptide hydrolysis: intracellular proteases endopepidase aminopepidase and carboxypeptidase
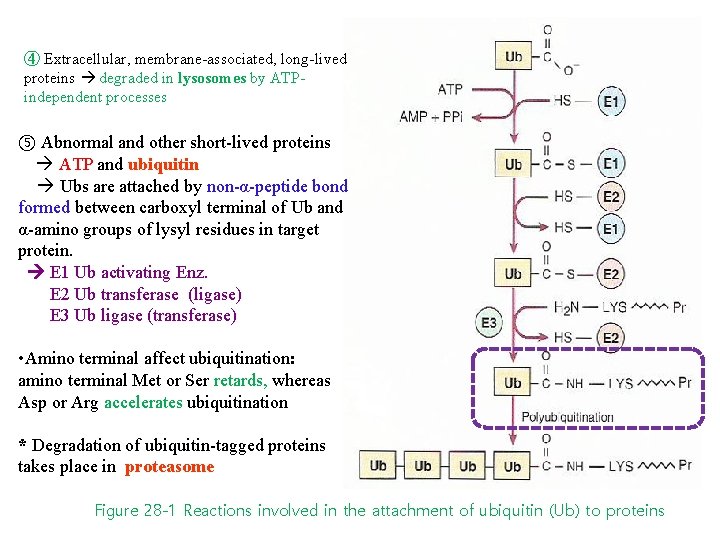
④ Extracellular, membrane-associated, long-lived proteins degraded in lysosomes by ATPindependent processes ⑤ Abnormal and other short-lived proteins ATP and ubiquitin Ubs are attached by non-α-peptide bond formed between carboxyl terminal of Ub and α-amino groups of lysyl residues in target protein. E 1 Ub activating Enz. E 2 Ub transferase (ligase) E 3 Ub ligase (transferase) • Amino terminal affect ubiquitination: amino terminal Met or Ser retards, whereas Asp or Arg accelerates ubiquitination * Degradation of ubiquitin-tagged proteins takes place in proteasome Figure 28 -1 Reactions involved in the attachment of ubiquitin (Ub) to proteins
The multiple ubiquitin-tagging hypothesis (1981) : Novel prize for the discovery of ubiquitin-mediated protein degradation (2004) Technion-Israel



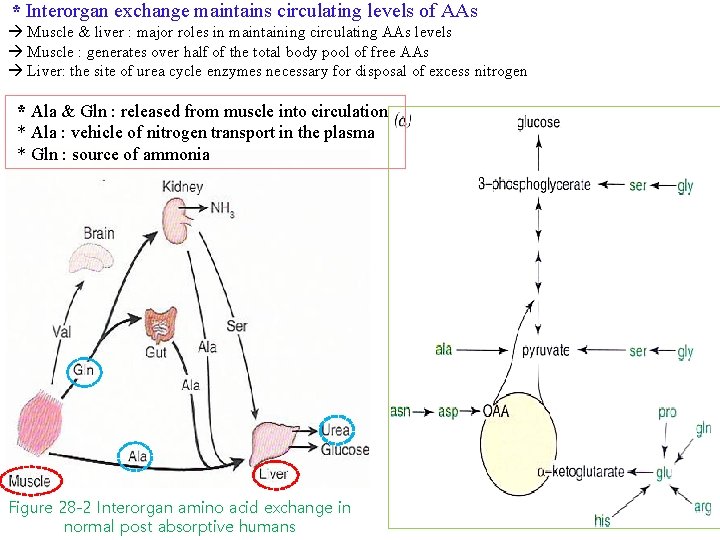
* Interorgan exchange maintains circulating levels of AAs Muscle & liver : major roles in maintaining circulating AAs levels Muscle : generates over half of the total body pool of free AAs Liver: the site of urea cycle enzymes necessary for disposal of excess nitrogen * Ala & Gln : released from muscle into circulation * Ala : vehicle of nitrogen transport in the plasma * Gln : source of ammonia Figure 28 -2 Interorgan amino acid exchange in normal post absorptive humans
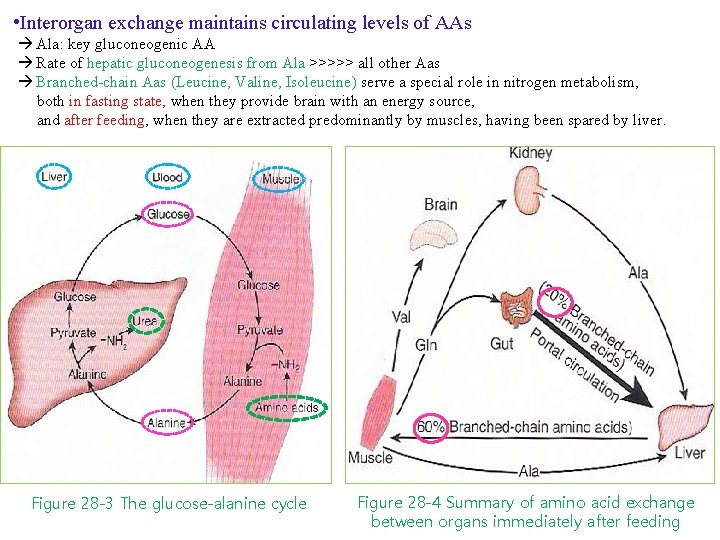
• Interorgan exchange maintains circulating levels of AAs Ala: key gluconeogenic AA Rate of hepatic gluconeogenesis from Ala >>>>> all other Aas Branched-chain Aas (Leucine, Valine, Isoleucine) serve a special role in nitrogen metabolism, both in fasting state, when they provide brain with an energy source, and after feeding, when they are extracted predominantly by muscles, having been spared by liver. Figure 28 -3 The glucose-alanine cycle Figure 28 -4 Summary of amino acid exchange between organs immediately after feeding

• Animals convert α–amino nitrogen to varied end products ① Different animals excrete excess nitrogen as ammonia, uric acid, or urea Fish : ammonotelic (excrete ammonia; highly toxic) Birds : uricotelic (excrete uric acid) Humans : ureotelic (excrete nontoxic, water-soluble urea) ② High blood urea levels in renal disease are consequence, but not a cause, of impaired renal function
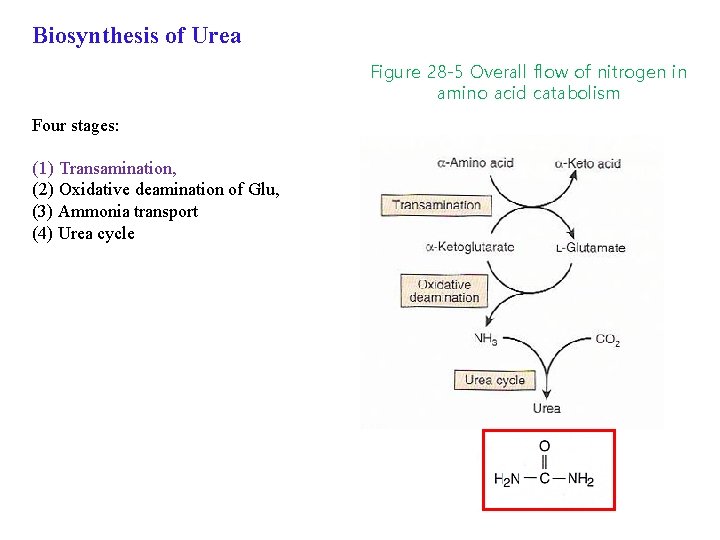
Biosynthesis of Urea Figure 28 -5 Overall flow of nitrogen in amino acid catabolism Four stages: (1) Transamination, (2) Oxidative deamination of Glu, (3) Ammonia transport (4) Urea cycle
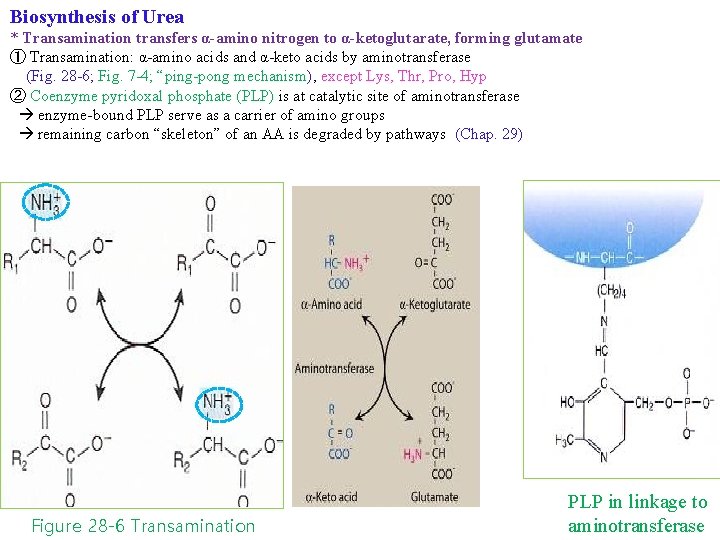
Biosynthesis of Urea * Transamination transfers α-amino nitrogen to α-ketoglutarate, forming glutamate ① Transamination: α-amino acids and α-keto acids by aminotransferase (Fig. 28 -6; Fig. 7 -4; “ping-pong mechanism), except Lys, Thr, Pro, Hyp ② Coenzyme pyridoxal phosphate (PLP) is at catalytic site of aminotransferase enzyme-bound PLP serve as a carrier of amino groups remaining carbon “skeleton” of an AA is degraded by pathways (Chap. 29) Figure 28 -6 Transamination PLP in linkage to aminotransferase
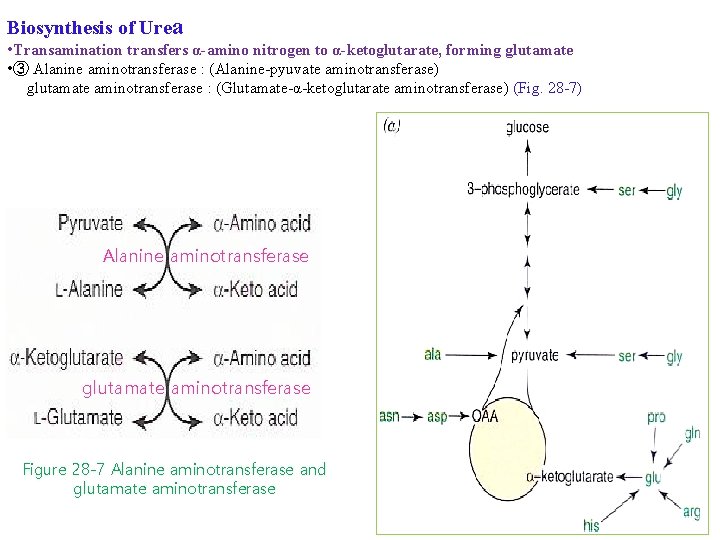
Biosynthesis of Urea • Transamination transfers α-amino nitrogen to α-ketoglutarate, forming glutamate • ③ Alanine aminotransferase : (Alanine-pyuvate aminotransferase) glutamate aminotransferase : (Glutamate-α-ketoglutarate aminotransferase) (Fig. 28 -7) Alanine aminotransferase glutamate aminotransferase Figure 28 -7 Alanine aminotransferase and glutamate aminotransferase
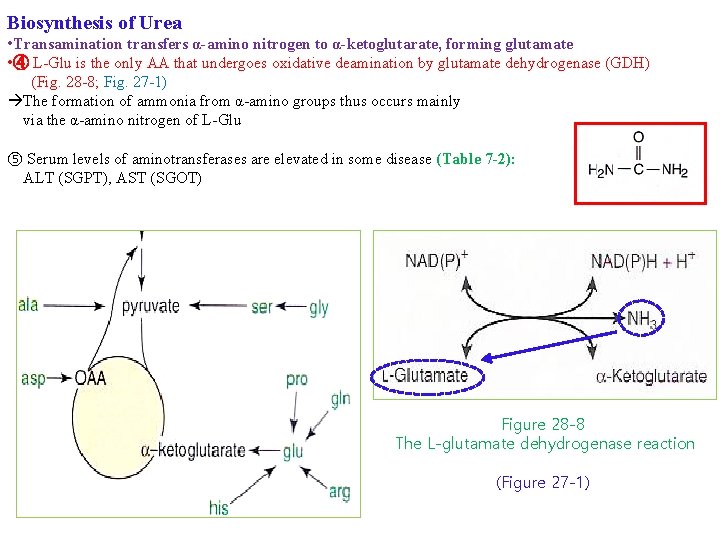
Biosynthesis of Urea • Transamination transfers α-amino nitrogen to α-ketoglutarate, forming glutamate • ④ L-Glu is the only AA that undergoes oxidative deamination by glutamate dehydrogenase (GDH) (Fig. 28 -8; Fig. 27 -1) The formation of ammonia from α-amino groups thus occurs mainly via the α-amino nitrogen of L-Glu ⑤ Serum levels of aminotransferases are elevated in some disease (Table 7 -2): ALT (SGPT), AST (SGOT) Figure 28 -8 The L-glutamate dehydrogenase reaction (Figure 27 -1)
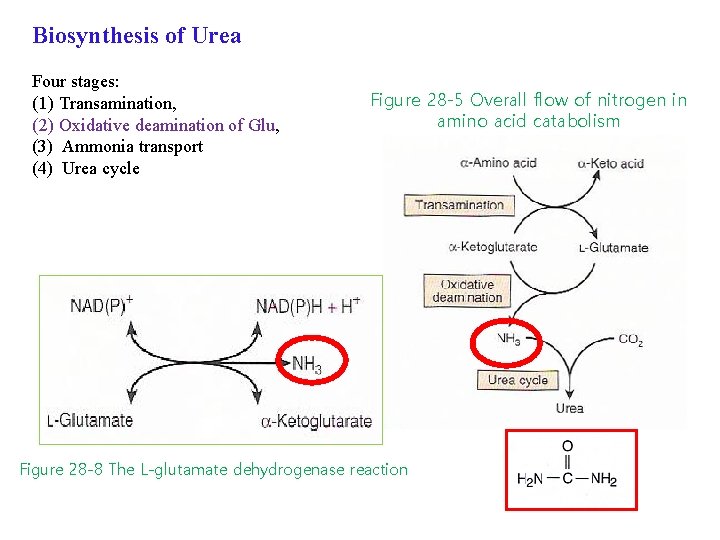
Biosynthesis of Urea Four stages: (1) Transamination, (2) Oxidative deamination of Glu, (3) Ammonia transport (4) Urea cycle Figure 28 -5 Overall flow of nitrogen in amino acid catabolism Figure 28 -8 The L-glutamate dehydrogenase reaction
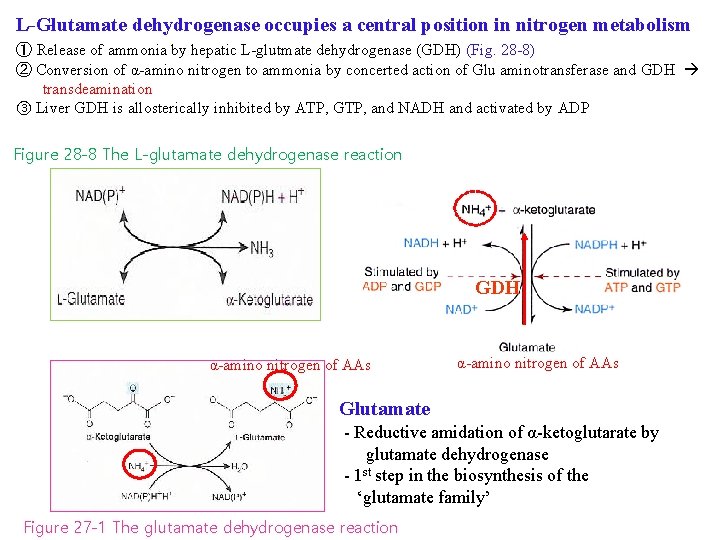
L-Glutamate dehydrogenase occupies a central position in nitrogen metabolism ① Release of ammonia by hepatic L-glutmate dehydrogenase (GDH) (Fig. 28 -8) ② Conversion of α-amino nitrogen to ammonia by concerted action of Glu aminotransferase and GDH transdeamination ③ Liver GDH is allosterically inhibited by ATP, GTP, and NADH and activated by ADP Figure 28 -8 The L-glutamate dehydrogenase reaction GDH α-amino nitrogen of AAs Glutamate - Reductive amidation of α-ketoglutarate by glutamate dehydrogenase - 1 st step in the biosynthesis of the ‘glutamate family’ Figure 27 -1 The glutamate dehydrogenase reaction
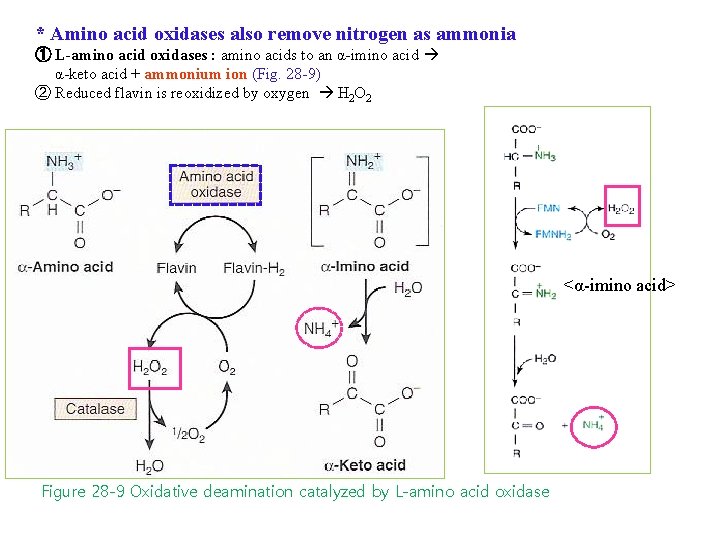
* Amino acid oxidases also remove nitrogen as ammonia ① L-amino acid oxidases : amino acids to an α-imino acid α-keto acid + ammonium ion (Fig. 28 -9) ② Reduced flavin is reoxidized by oxygen H 2 O 2 <α-imino acid> Figure 28 -9 Oxidative deamination catalyzed by L-amino acid oxidase

Ammonia intoxification is life-threatening ① Ammonia produced by enteric bacteria and absorbed into portal venous blood and produced by tissues removed from circulation by liver and converted to urea ② Ammonia is toxic to CNS; 10 -20 μg/d. L in peripheral blood ③ Portal blood bypass the liver Ammonia intoxification: tremor, slurred speech, coma, death ④ Ammonia in brain+ α-ketoglutarate Glu : depletion of α-ketoglutarate impairs TCA cycle in neurons GDH
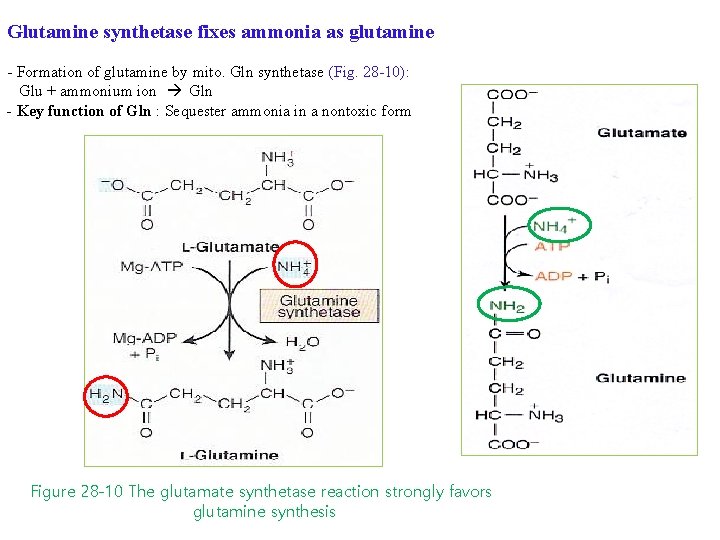
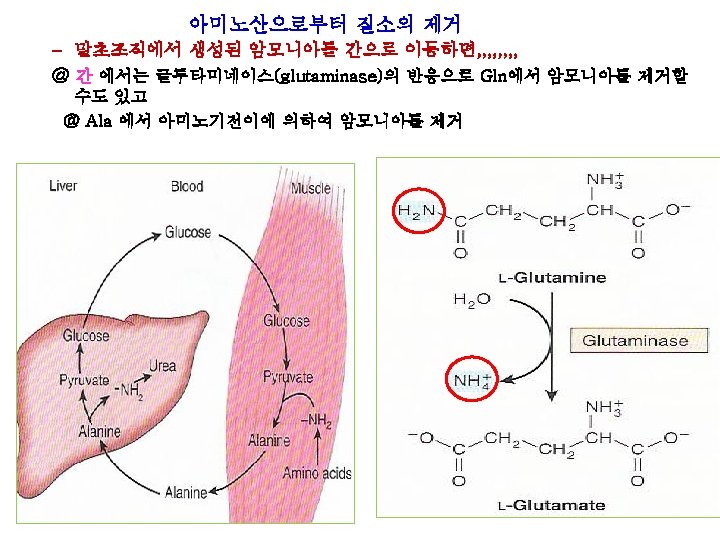
Glutamine synthetase fixes ammonia as glutamine - Formation of glutamine by mito. Gln synthetase (Fig. 28 -10): Glu + ammonium ion Gln - Key function of Gln : Sequester ammonia in a nontoxic form Figure 28 -10 The glutamate synthetase reaction strongly favors glutamine synthesis
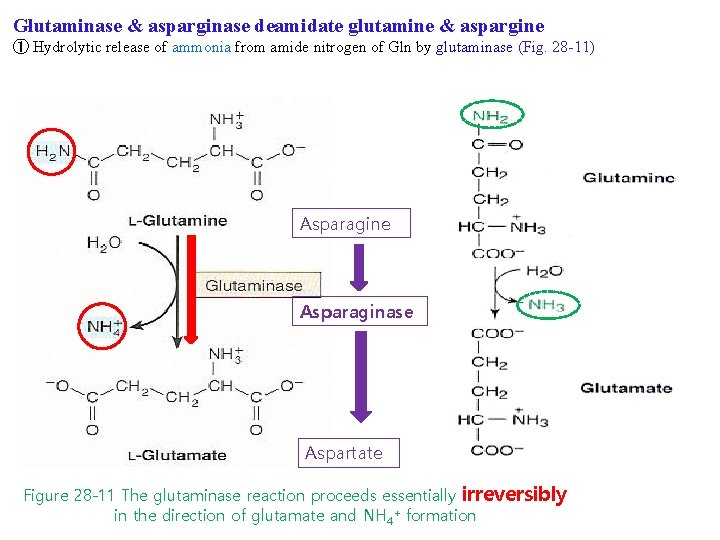
Glutaminase & asparginase deamidate glutamine & aspargine ① Hydrolytic release of ammonia from amide nitrogen of Gln by glutaminase (Fig. 28 -11) Asparagine Asparaginase Aspartate Figure 28 -11 The glutaminase reaction proceeds essentially irreversibly in the direction of glutamate and NH 4+ formation
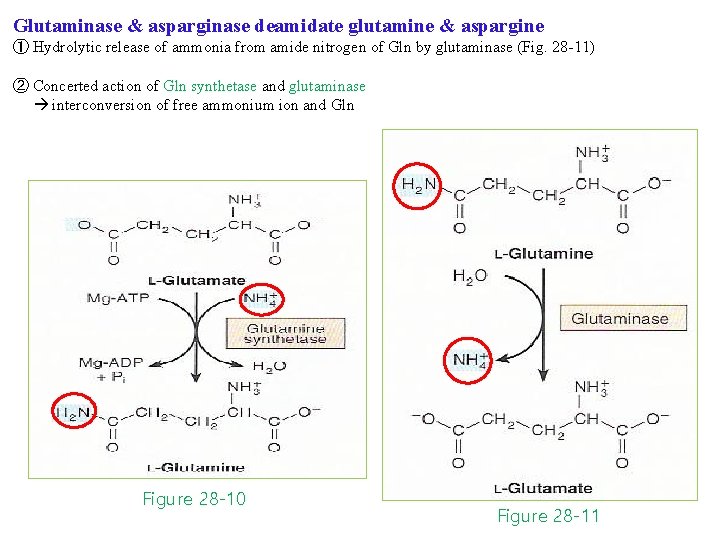
Glutaminase & asparginase deamidate glutamine & aspargine ① Hydrolytic release of ammonia from amide nitrogen of Gln by glutaminase (Fig. 28 -11) ② Concerted action of Gln synthetase and glutaminase interconversion of free ammonium ion and Gln Figure 28 -10 Figure 28 -11
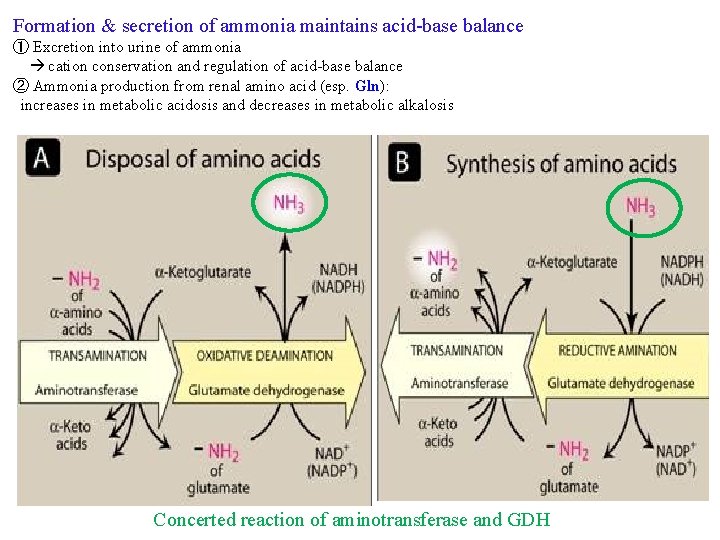
Formation & secretion of ammonia maintains acid-base balance ① Excretion into urine of ammonia cation conservation and regulation of acid-base balance ② Ammonia production from renal amino acid (esp. Gln): increases in metabolic acidosis and decreases in metabolic alkalosis Concerted reaction of aminotransferase and GDH
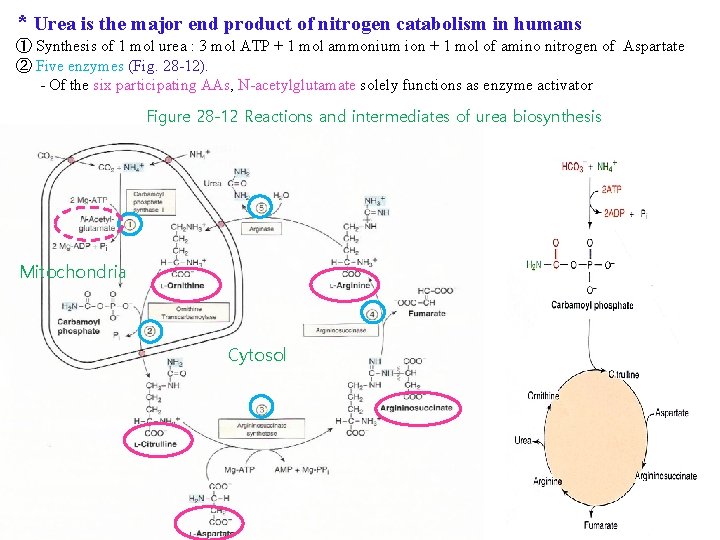
* Urea is the major end product of nitrogen catabolism in humans ① Synthesis of 1 mol urea : 3 mol ATP + 1 mol ammonium ion + 1 mol of amino nitrogen of Aspartate ② Five enzymes (Fig. 28 -12). - Of the six participating AAs, N-acetylglutamate solely functions as enzyme activator Figure 28 -12 Reactions and intermediates of urea biosynthesis Mitochondria Cytosol
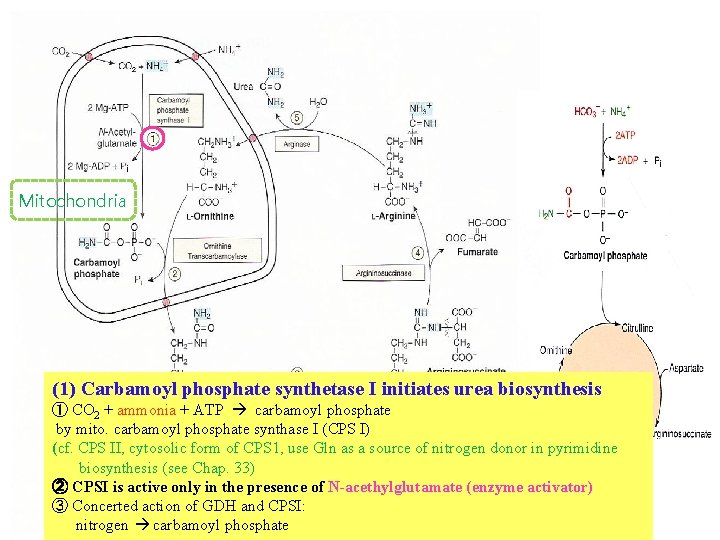
Mitochondria (1) Carbamoyl phosphate synthetase I initiates urea biosynthesis ① CO 2 + ammonia + ATP carbamoyl phosphate by mito. carbamoyl phosphate synthase I (CPS I) (cf. CPS II, cytosolic form of CPS 1, use Gln as a source of nitrogen donor in pyrimidine biosynthesis (see Chap. 33) ② CPSI is active only in the presence of N-acethylglutamate (enzyme activator) ③ Concerted action of GDH and CPSI: nitrogen carbamoyl phosphate
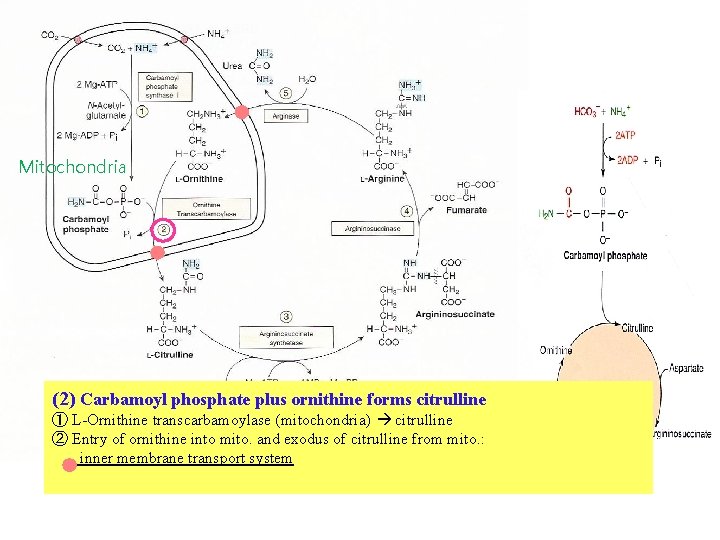
Mitochondria (2) Carbamoyl phosphate plus ornithine forms citrulline ① L-Ornithine transcarbamoylase (mitochondria) citrulline ② Entry of ornithine into mito. and exodus of citrulline from mito. : inner membrane transport system
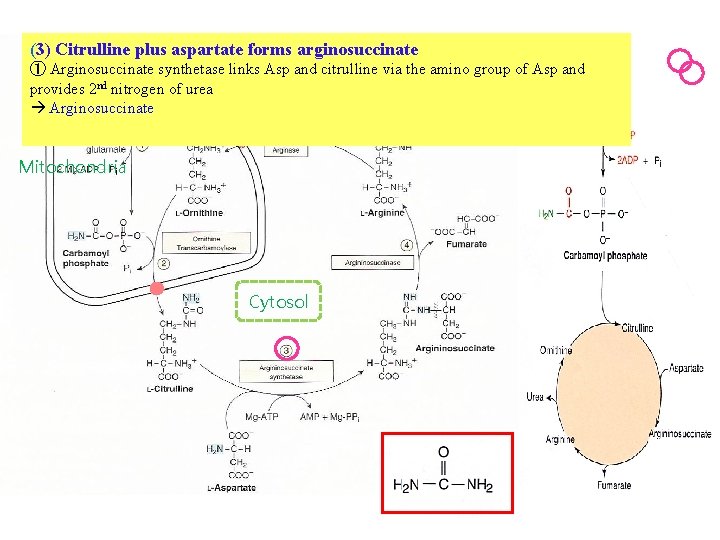
(3) Citrulline plus aspartate forms arginosuccinate ① Arginosuccinate synthetase links Asp and citrulline via the amino group of Asp and provides 2 nd nitrogen of urea Arginosuccinate Mitochondria Cytosol
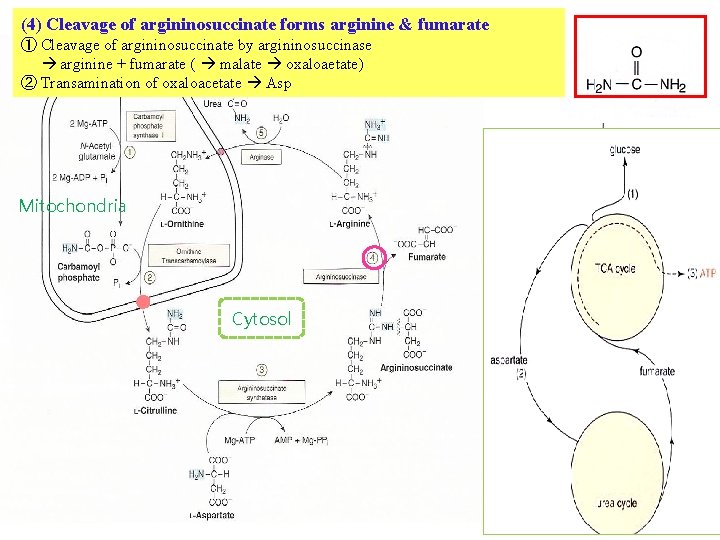
(4) Cleavage of argininosuccinate forms arginine & fumarate ① Cleavage of argininosuccinate by argininosuccinase arginine + fumarate ( malate oxaloaetate) ② Transamination of oxaloacetate Asp Mitochondria Cytosol
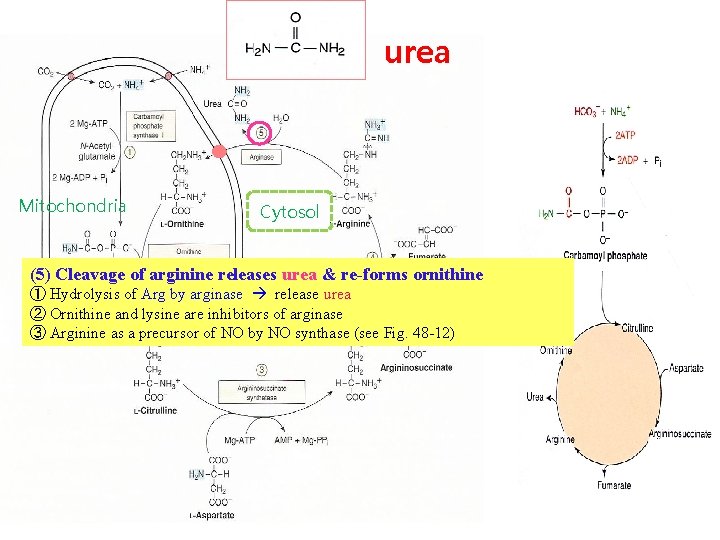
urea Mitochondria Cytosol (5) Cleavage of arginine releases urea & re-forms ornithine ① Hydrolysis of Arg by arginase release urea ② Ornithine and lysine are inhibitors of arginase ③ Arginine as a precursor of NO by NO synthase (see Fig. 48 -12)
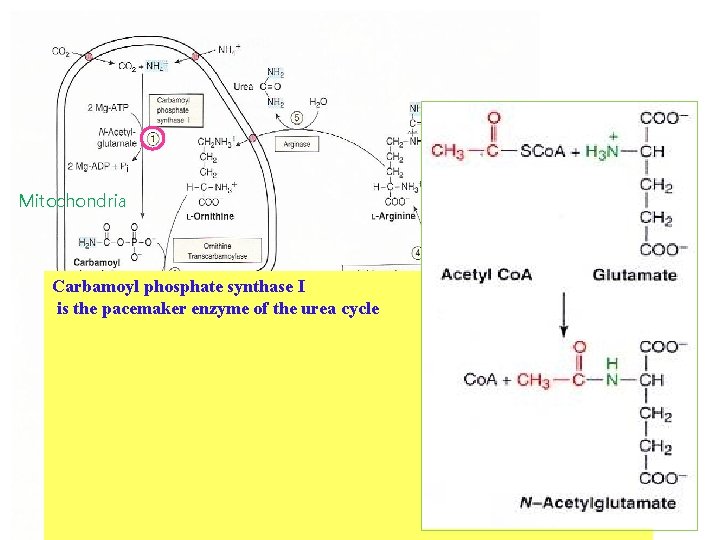
Mitochondria Carbamoyl phosphate synthase I is the pacemaker enzyme of the urea cycle
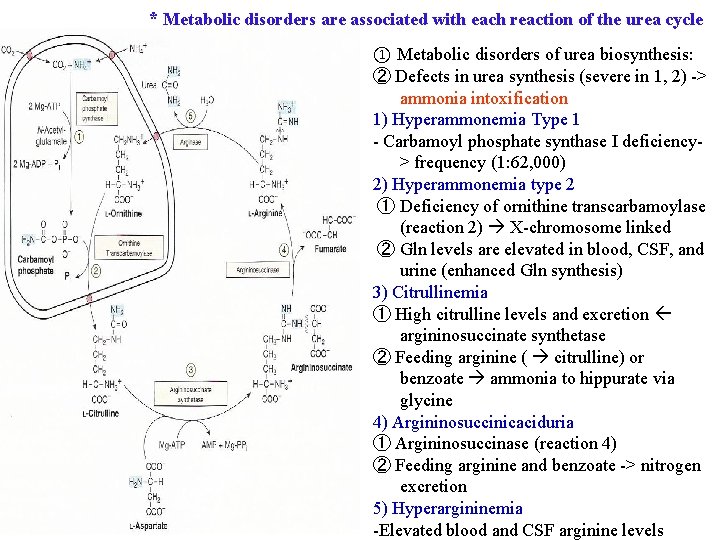
* Metabolic disorders are associated with each reaction of the urea cycle ① Metabolic disorders of urea biosynthesis: ② Defects in urea synthesis (severe in 1, 2) -> ammonia intoxification 1) Hyperammonemia Type 1 - Carbamoyl phosphate synthase I deficiency> frequency (1: 62, 000) 2) Hyperammonemia type 2 ① Deficiency of ornithine transcarbamoylase (reaction 2) X-chromosome linked ② Gln levels are elevated in blood, CSF, and urine (enhanced Gln synthesis) 3) Citrullinemia ① High citrulline levels and excretion argininosuccinate synthetase ② Feeding arginine ( citrulline) or benzoate ammonia to hippurate via glycine 4) Argininosuccinicaciduria ① Argininosuccinase (reaction 4) ② Feeding arginine and benzoate -> nitrogen excretion 5) Hyperargininemia -Elevated blood and CSF arginine levels
- Slides: 53