Amino Acids Biochemistry Free For All Part 1

Amino Acids Biochemistry Free For All

Part 1 - Amino Acids Structure and Chemistry • Building Blocks of Proteins • Essential Amino Acids • Basic Structure • Stereochemistry • Side Chain Chemistry • Properties • Ionization • Bonding

Amino Acids & Proteins • Workhorses of cell – Catalysis – Signaling – Structure – Energy/Gradient Generation • Proteins comprised of 20 -21 amino acids

Amino Acids Essential Amino Acids
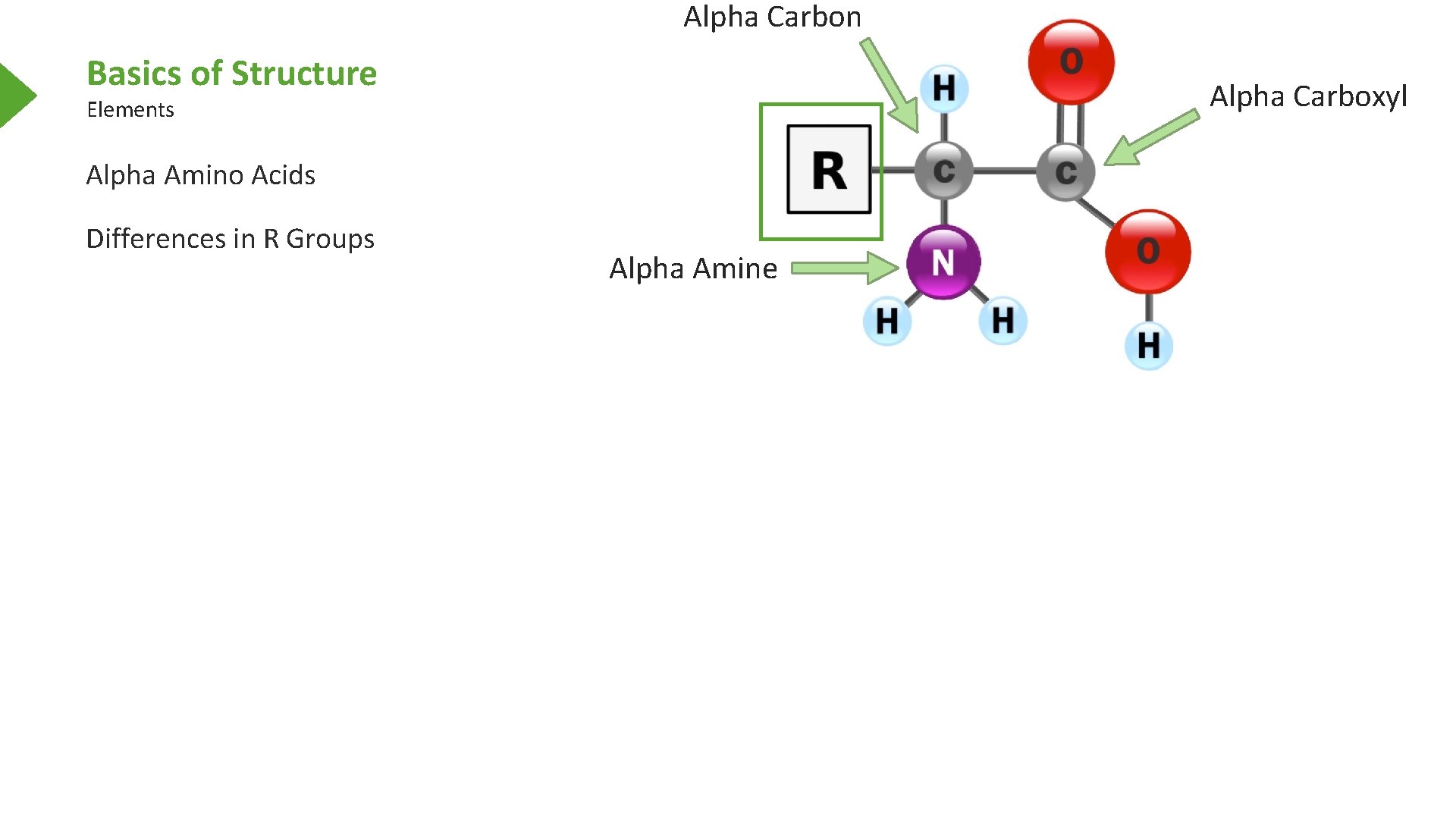
Alpha Carbon Basics of Structure Alpha Carboxyl Elements Alpha Amino Acids Differences in R Groups Alpha Amine
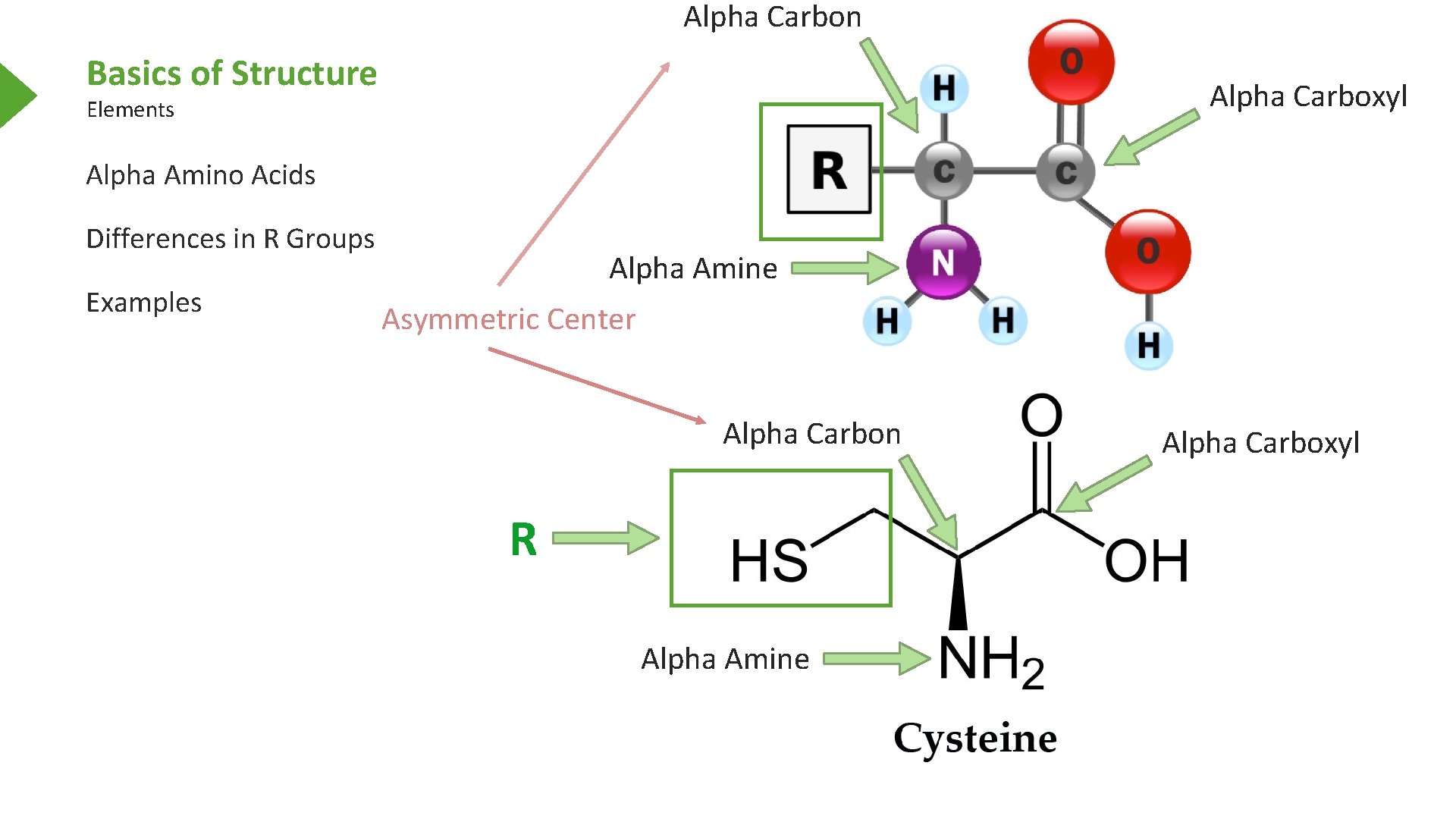
Alpha Carbon Basics of Structure Alpha Carboxyl Elements Alpha Amino Acids Differences in R Groups Examples Alpha Amine Asymmetric Center Alpha Carbon R Alpha Amine Alpha Carboxyl
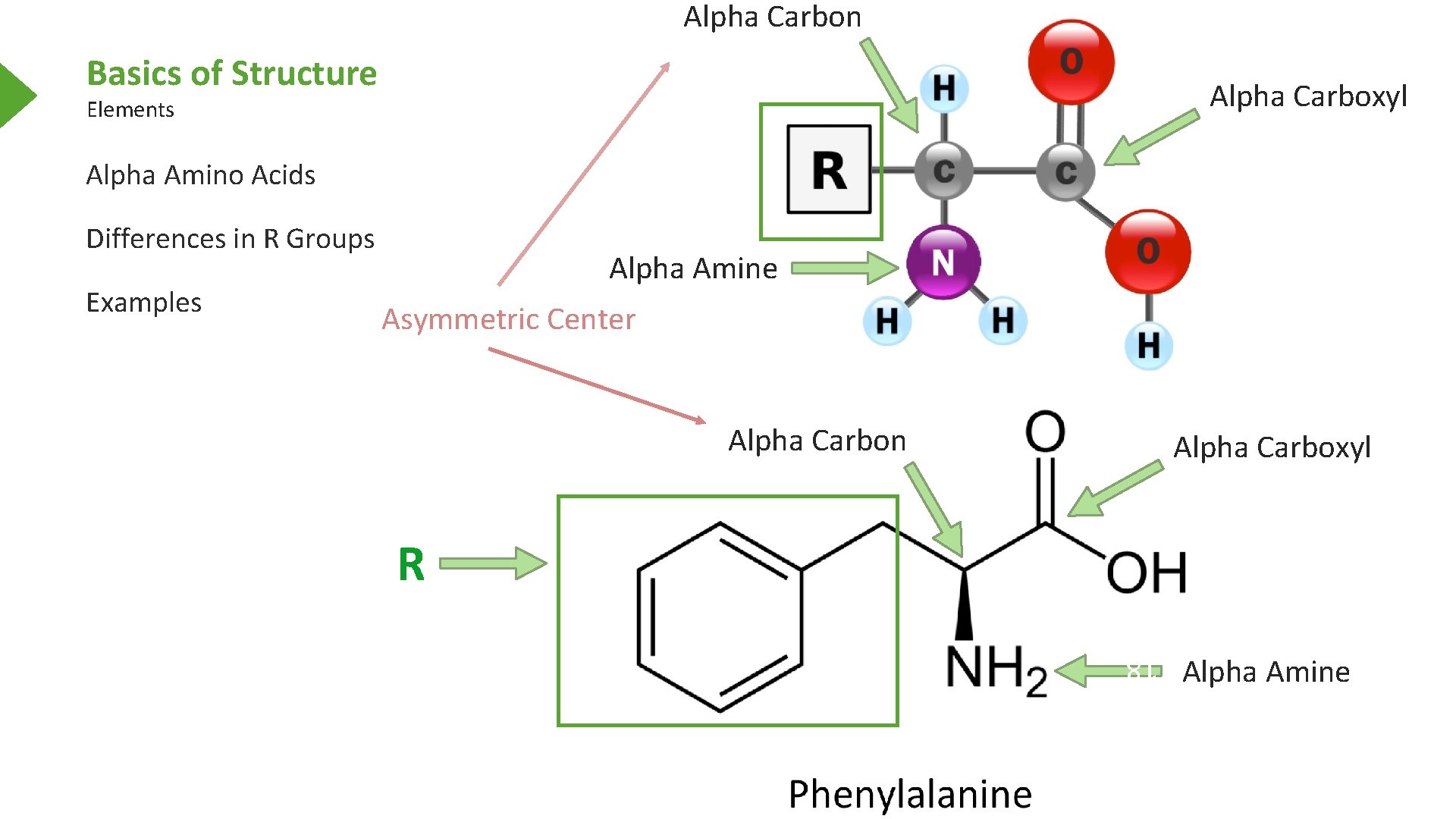
Alpha Carbon Basics of Structure Alpha Carboxyl Elements Alpha Amino Acids Differences in R Groups Asymmetric Center Alpha Carbon Alpha Carboxyl R 18 Examples Alpha Amine
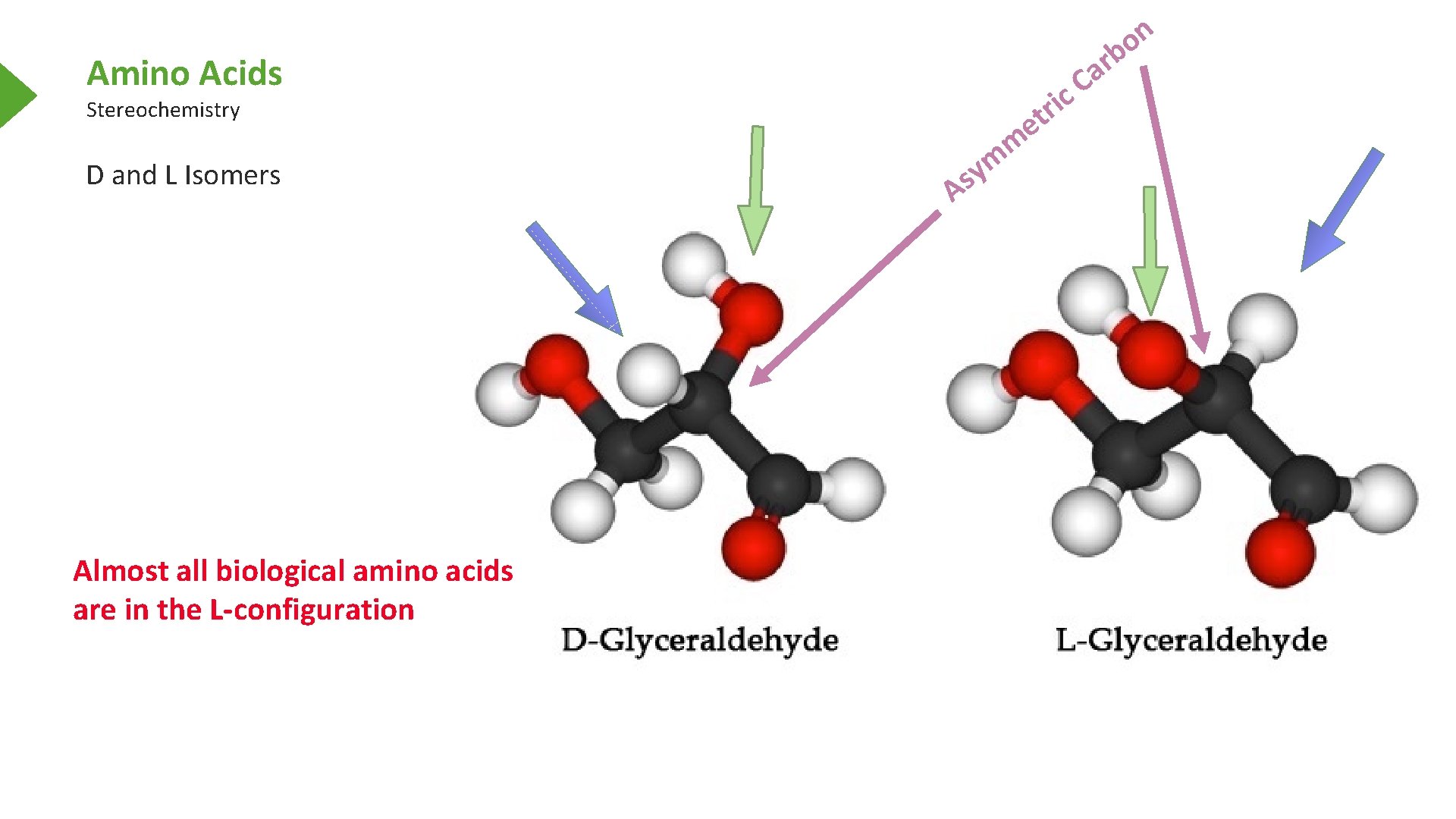
n o Amino Acids Stereochemistry D and L Isomers Almost all biological amino acids are in the L-configuration e i r t s A m m y C c b r a
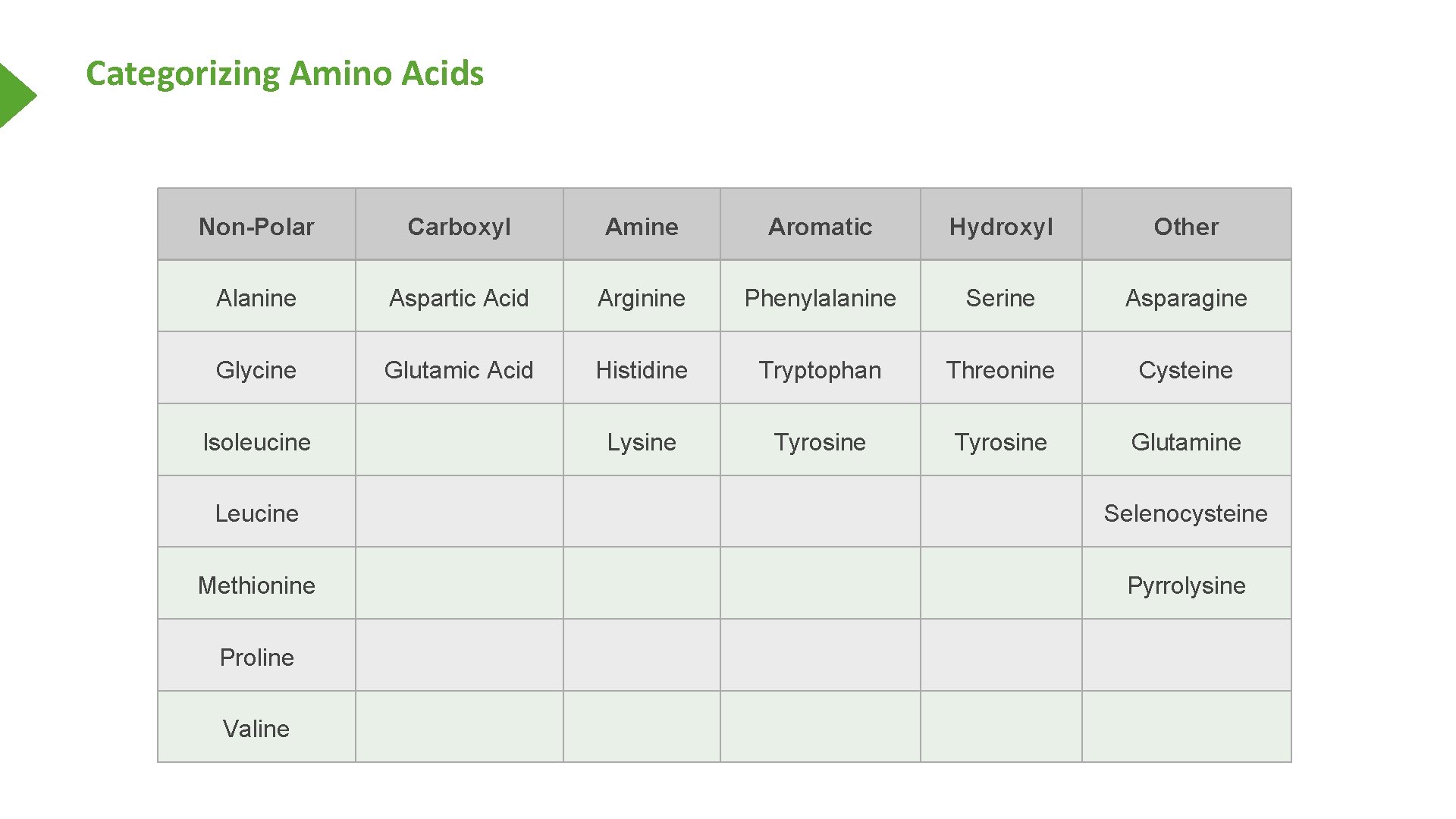
Categorizing Amino Acids Non-Polar Carboxyl Amine Aromatic Hydroxyl Other Alanine Aspartic Acid Arginine Phenylalanine Serine Asparagine Glycine Glutamic Acid Histidine Tryptophan Threonine Cysteine Lysine Tyrosine Glutamine Isoleucine Leucine Selenocysteine Methionine Pyrrolysine Proline Valine
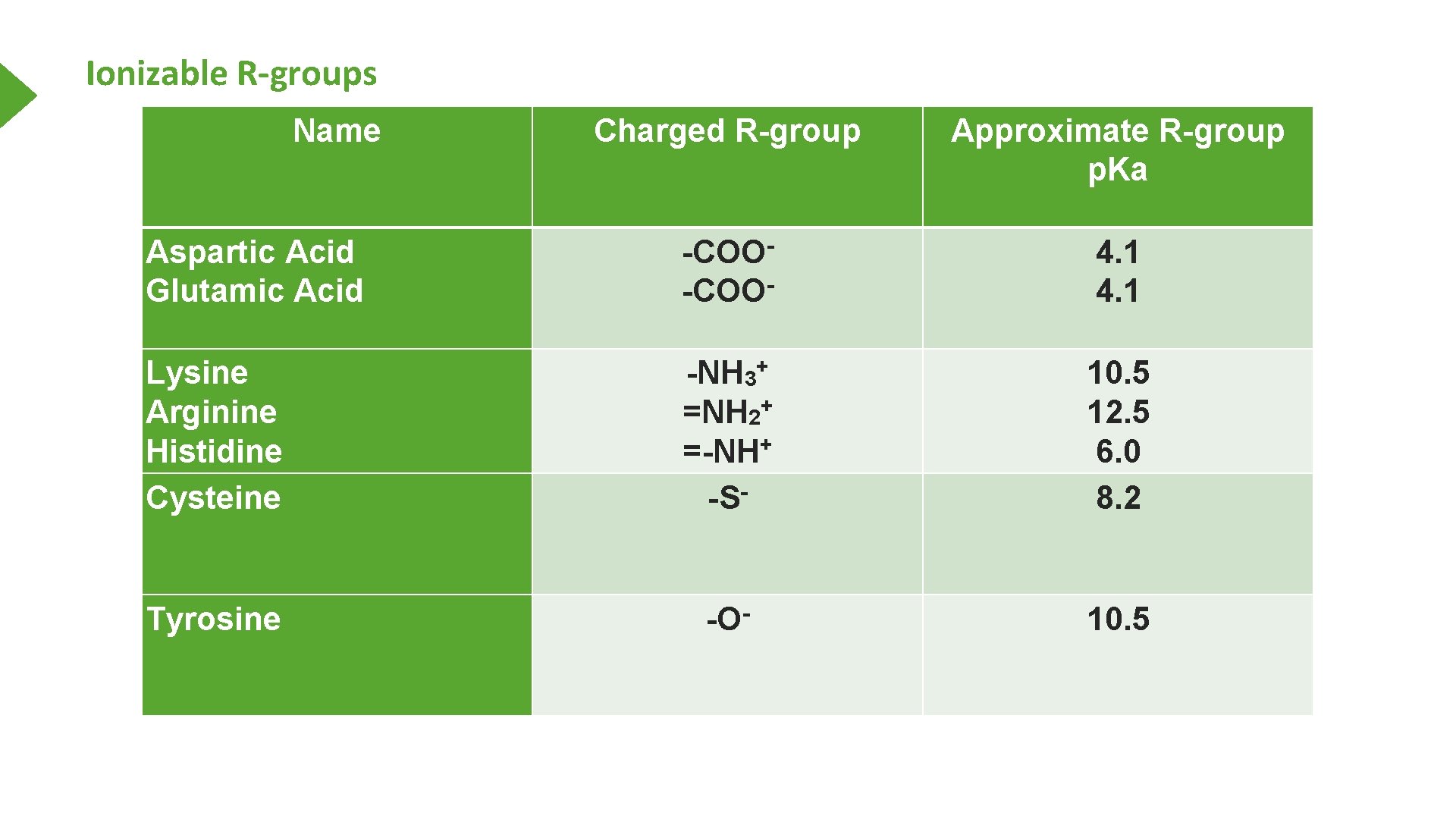
Ionizable R-groups Name Charged R-group Approximate R-group p. Ka Aspartic Acid Glutamic Acid -COO- 4. 1 Lysine Arginine Histidine Cysteine -NH 3+ =NH 2+ =-NH+ -S- 10. 5 12. 5 6. 0 8. 2 Tyrosine -O- 10. 5

Amino Acids Aromatic R-Groups Alpha Carbon Alpha Amine Tryptophan Alpha Carboxyl Tyrosine Phenylalanine
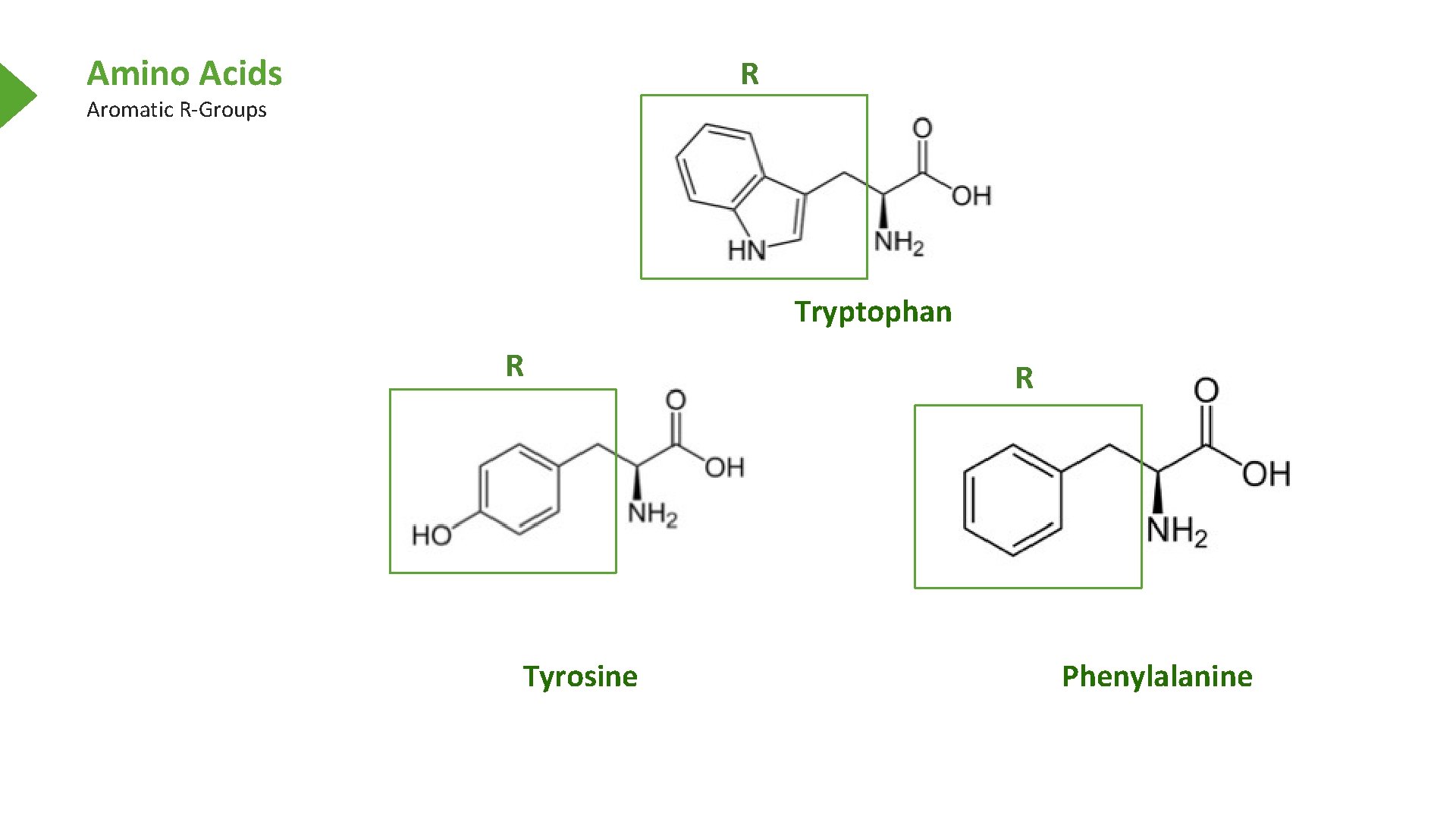
Amino Acids R Aromatic R-Groups Tryptophan R Tyrosine R Phenylalanine
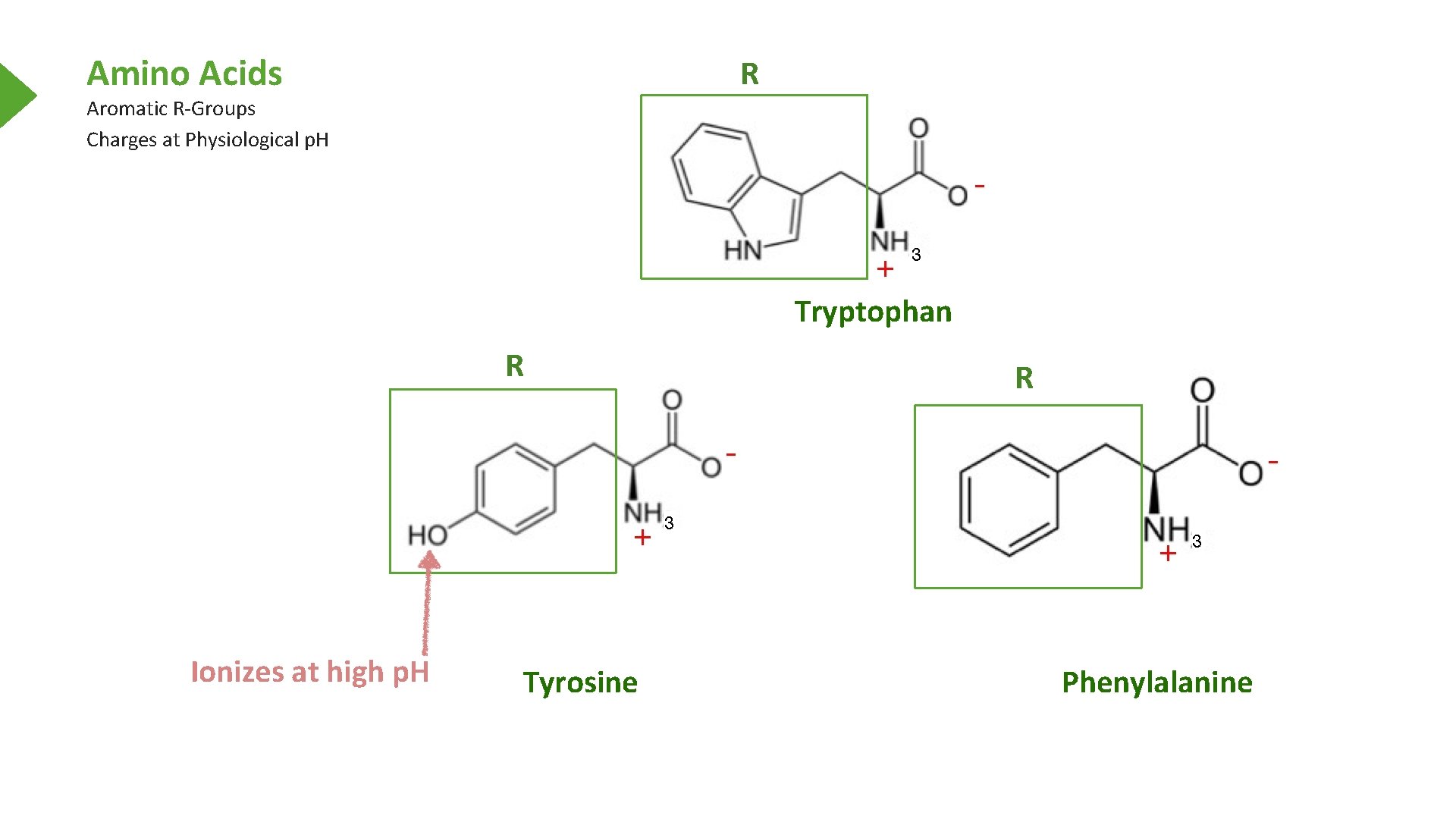
Amino Acids R Aromatic R-Groups Charges at Physiological p. H Tryptophan R Ionizes at high p. H Tyrosine R Phenylalanine
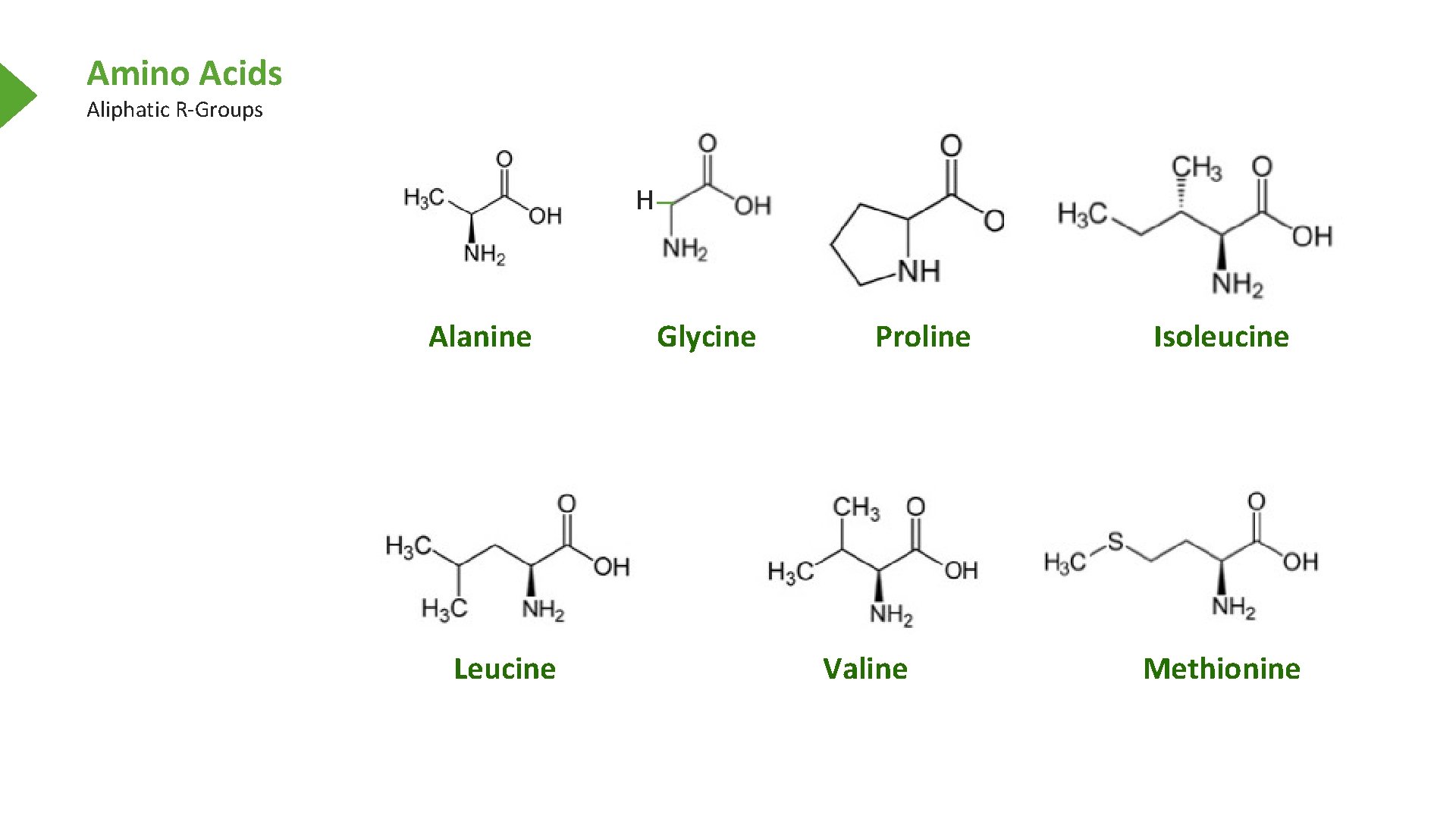
Amino Acids Aliphatic R-Groups H Alanine Leucine Glycine Proline Valine Isoleucine Methionine
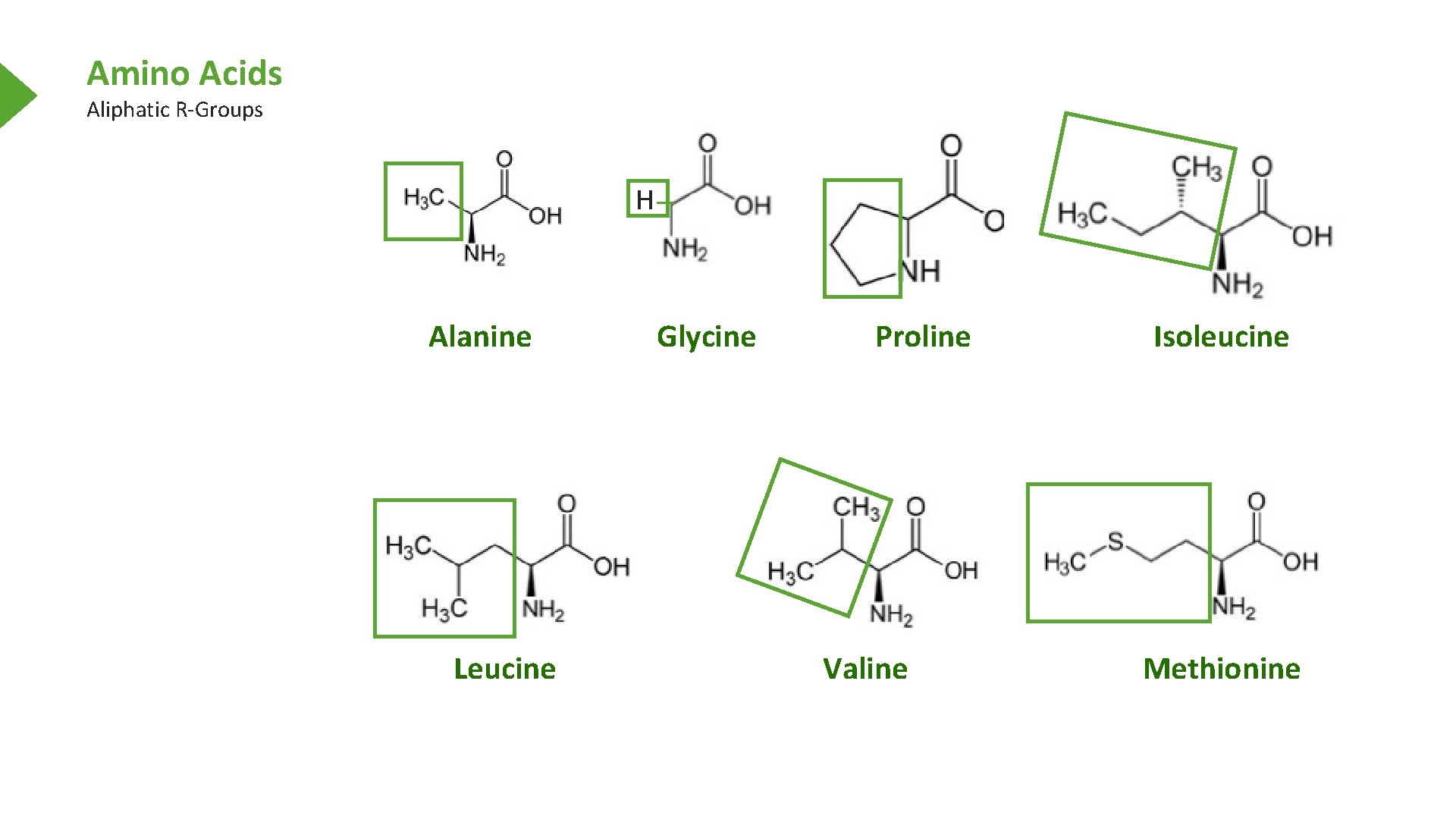
Amino Acids Aliphatic R-Groups H Alanine Leucine Glycine Proline Valine Isoleucine Methionine
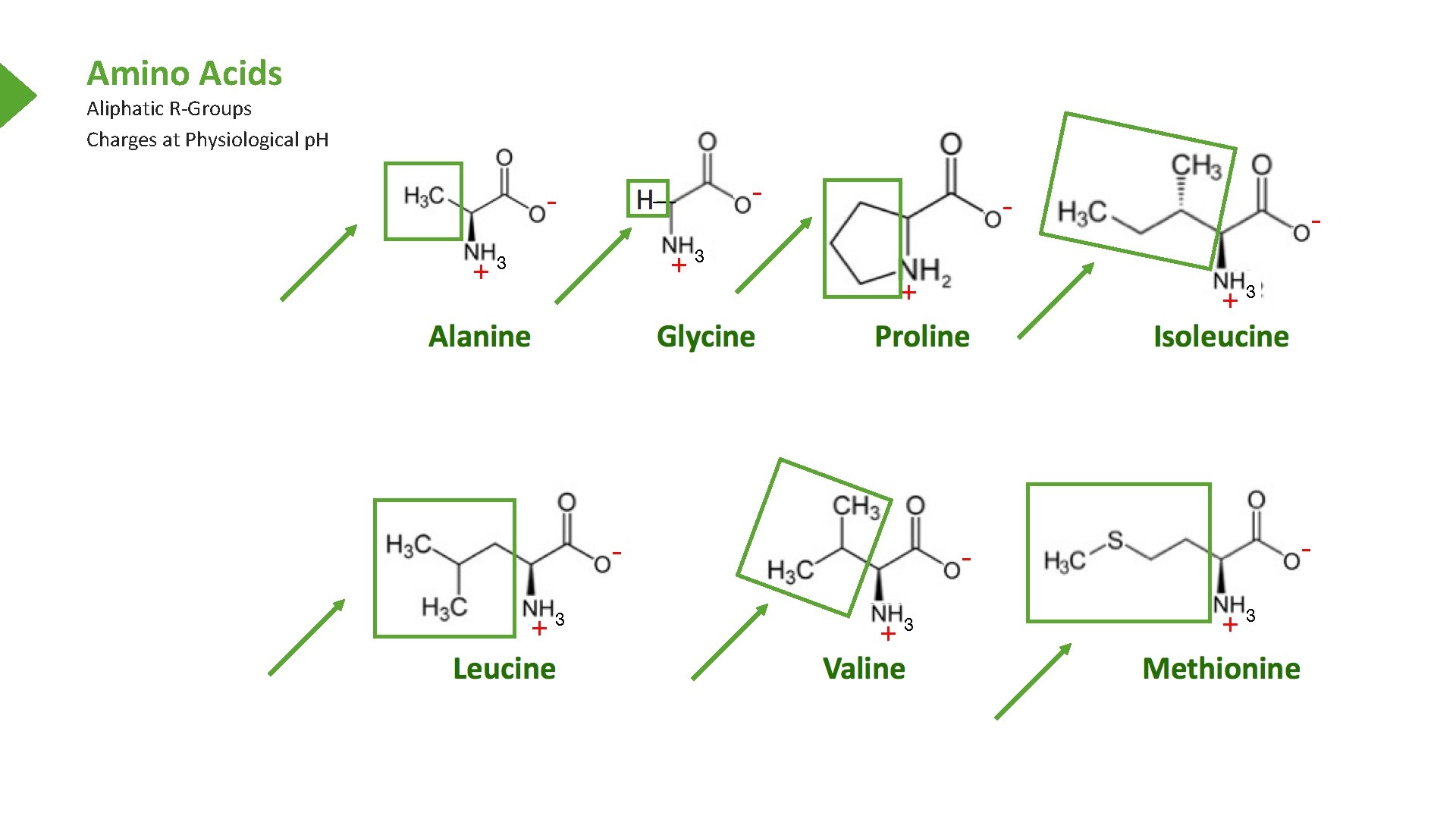
Amino Acids Aliphatic R-Groups Charges at Physiological p. H
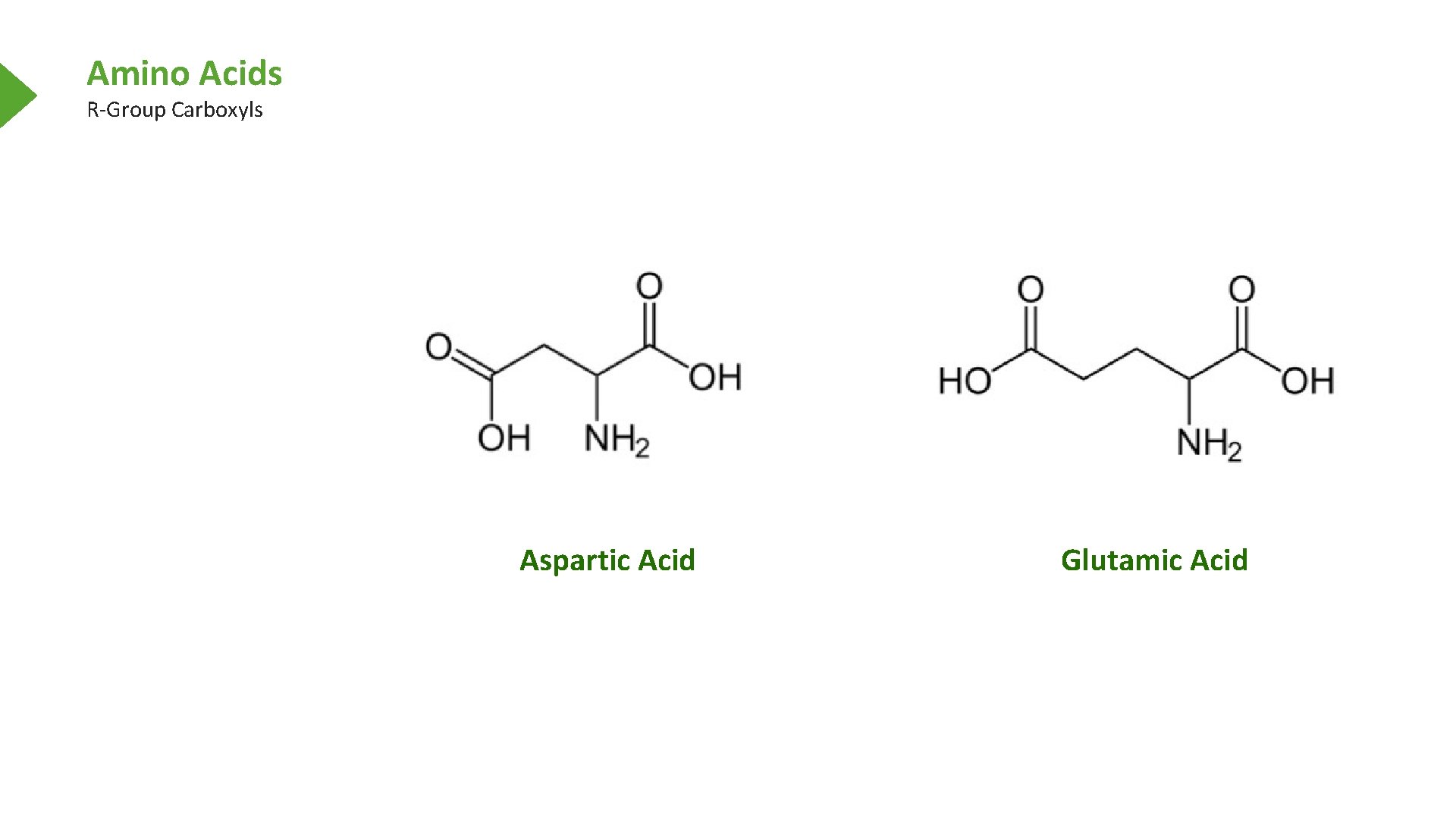
Amino Acids R-Group Carboxyls Aspartic Acid Glutamic Acid
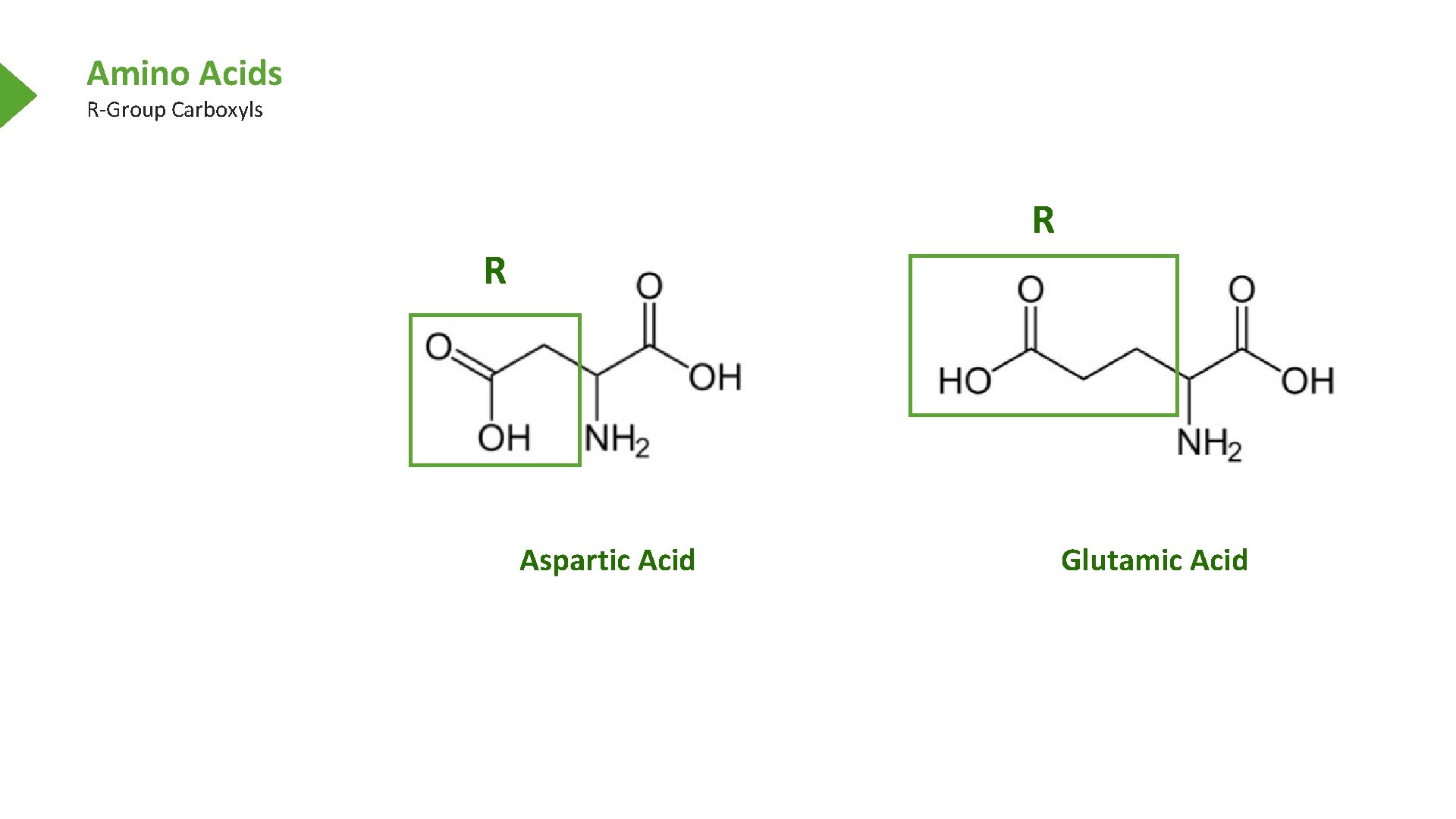
Amino Acids R-Group Carboxyls R R Aspartic Acid Glutamic Acid
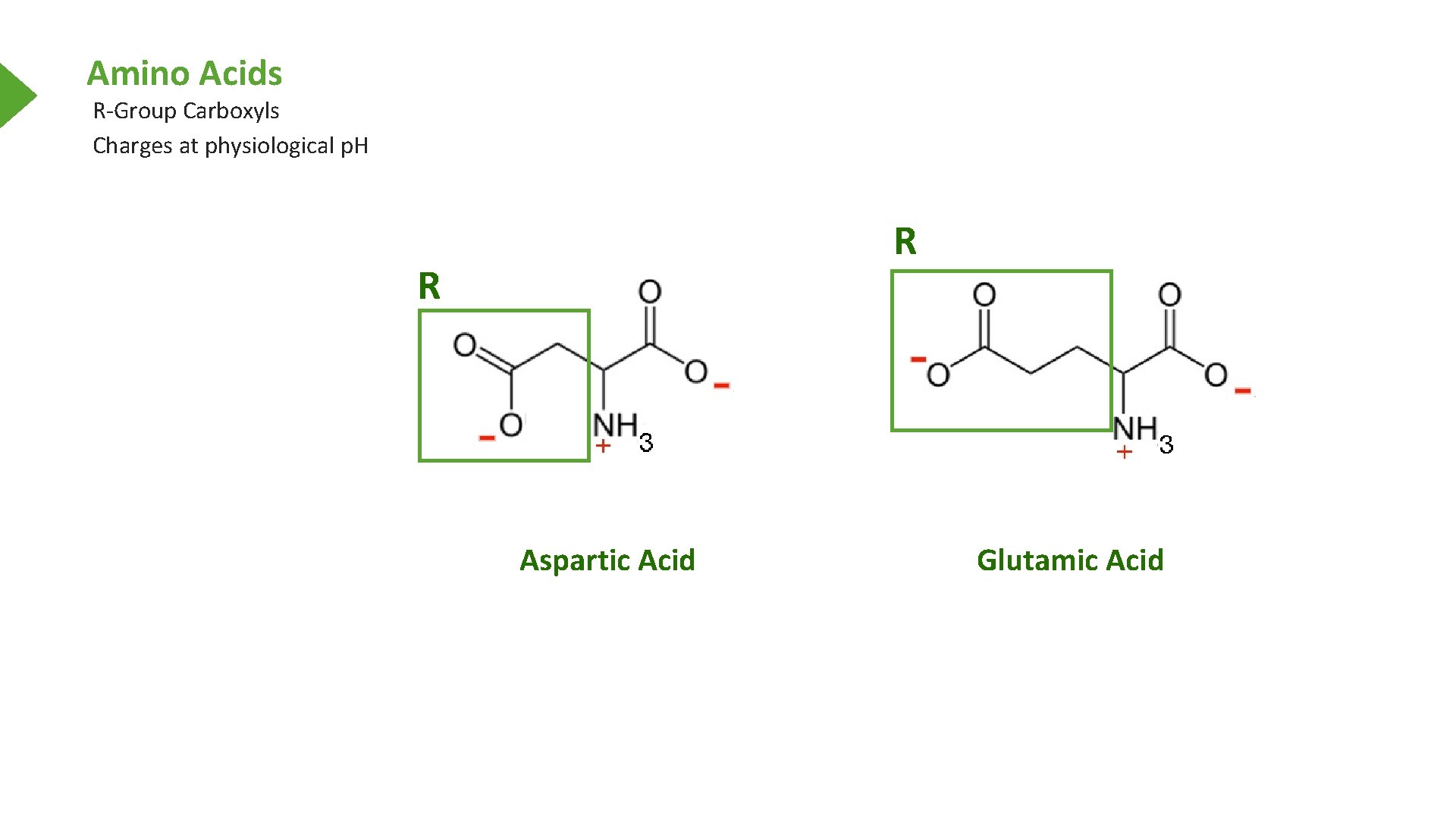
Amino Acids R-Group Carboxyls Charges at physiological p. H R R Aspartic Acid Glutamic Acid
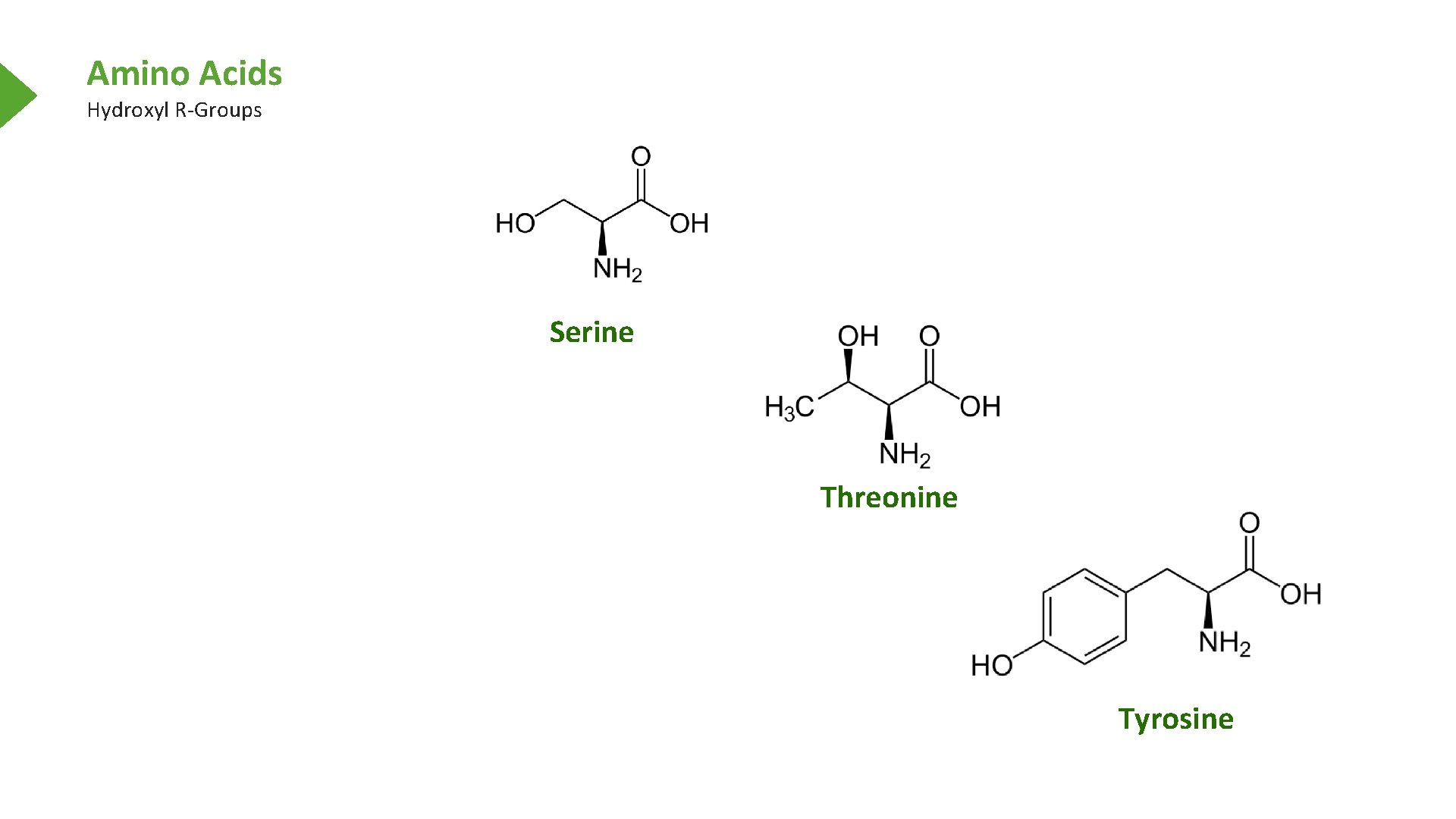
Amino Acids Hydroxyl R-Groups Serine Threonine Tyrosine

Amino Acids Hydroxyl R-Groups Serine Threonine Tyrosine
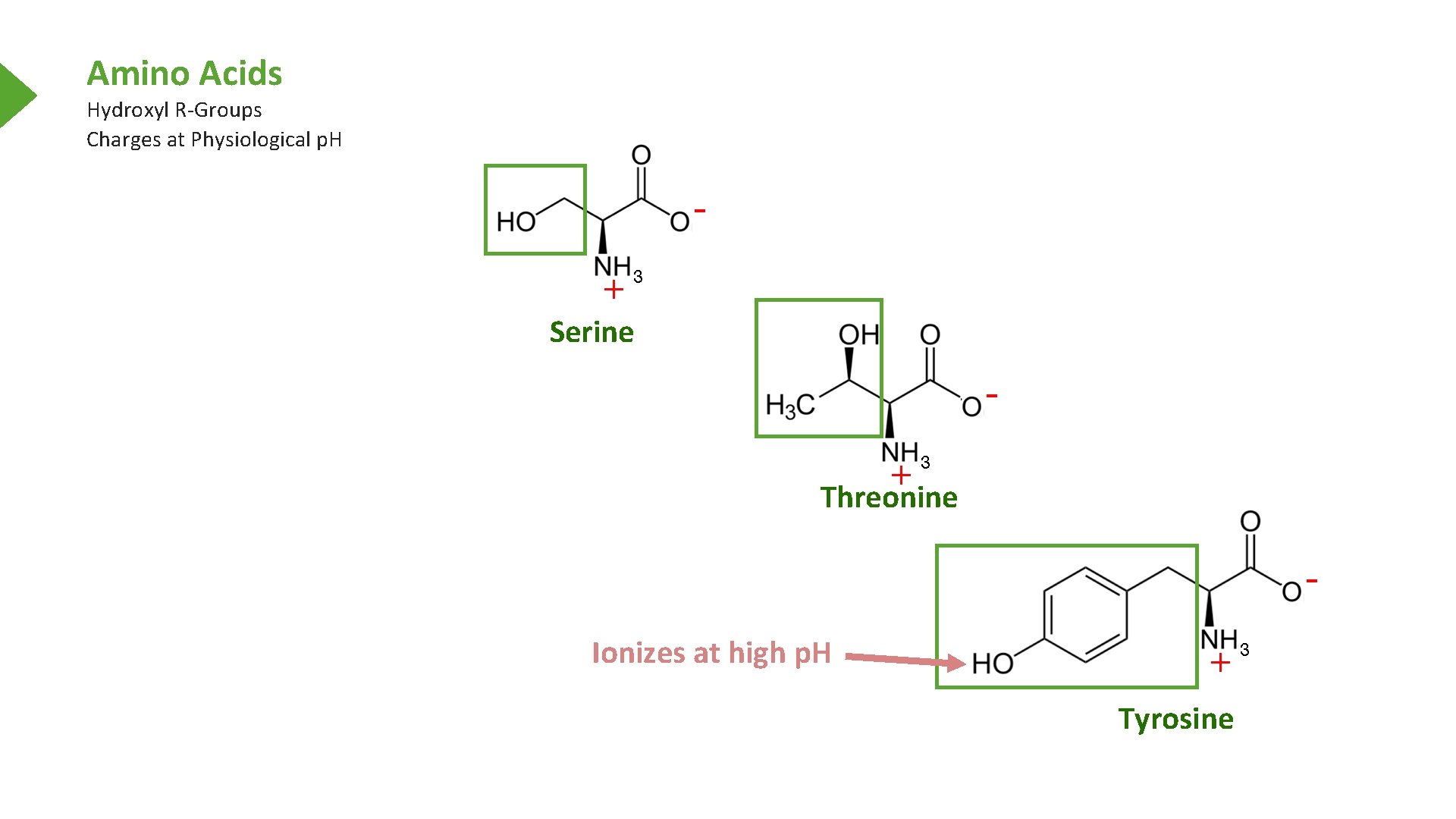
Amino Acids Hydroxyl R-Groups Charges at Physiological p. H Serine Threonine Ionizes at high p. H Tyrosine

Amino Acids Sulfhydryl R-Group Cysteine
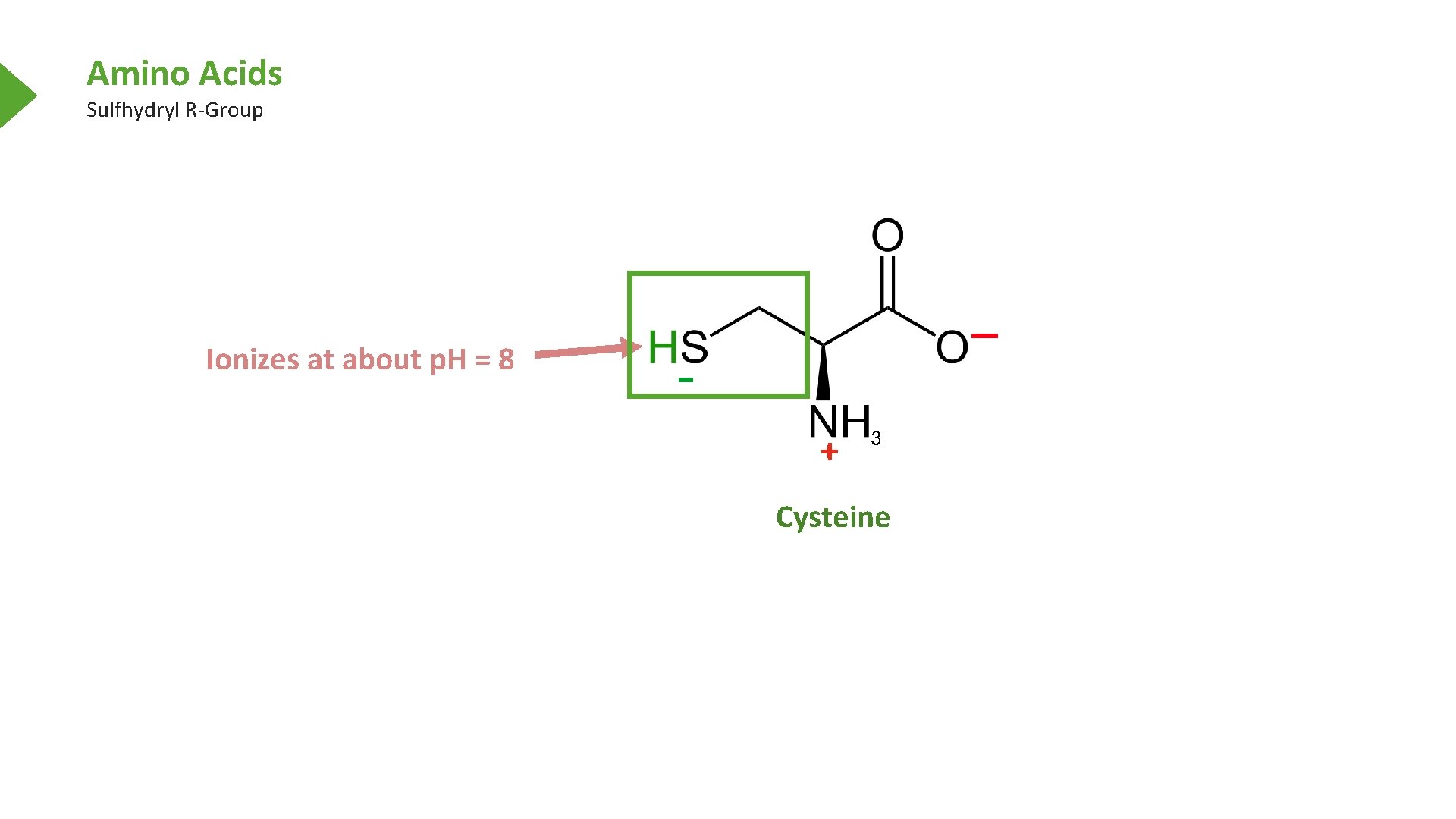
Amino Acids Sulfhydryl R-Group Ionizes at about p. H = 8 + Cysteine
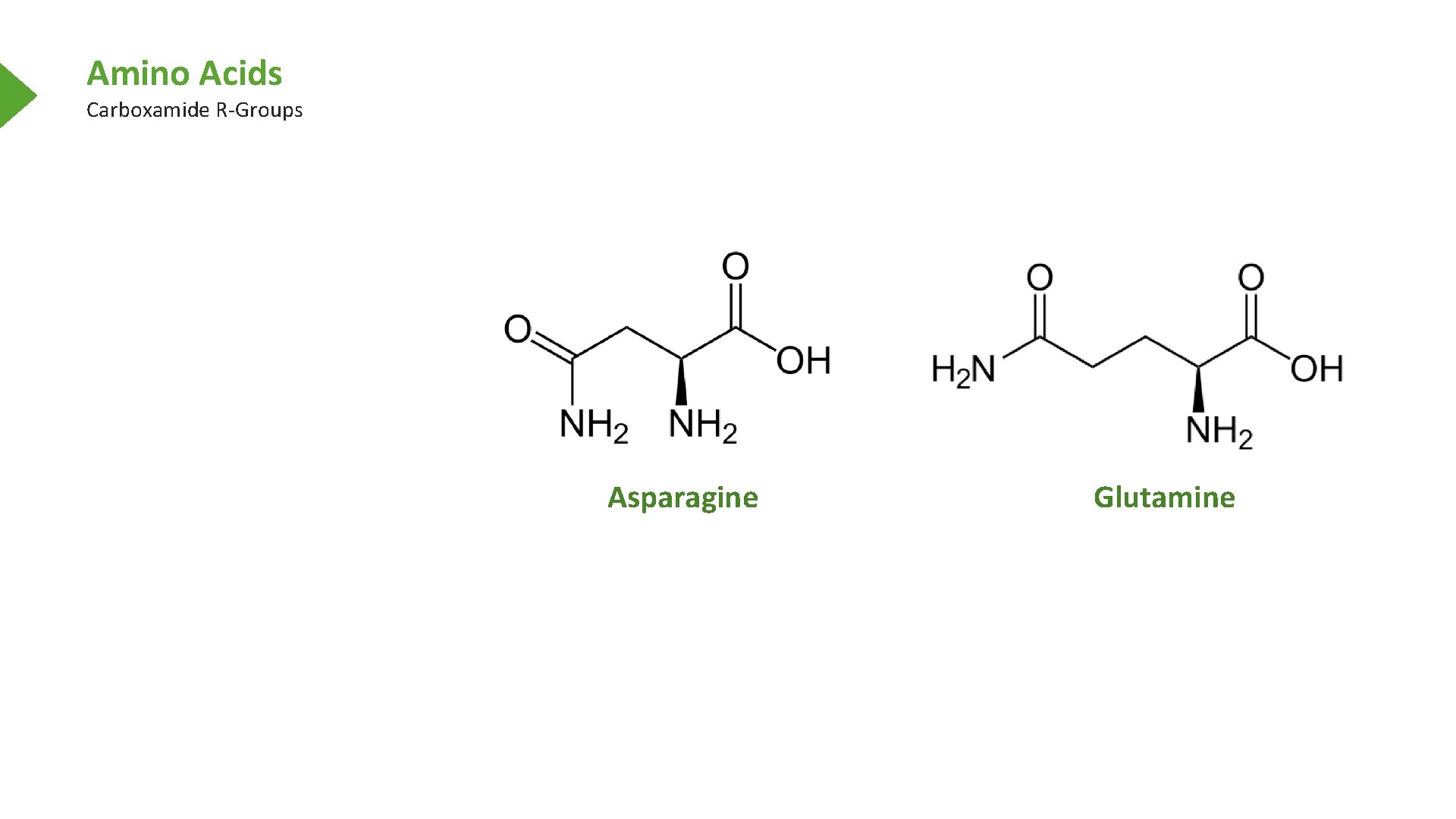
Amino Acids Carboxamide R-Groups Asparagine Glutamine
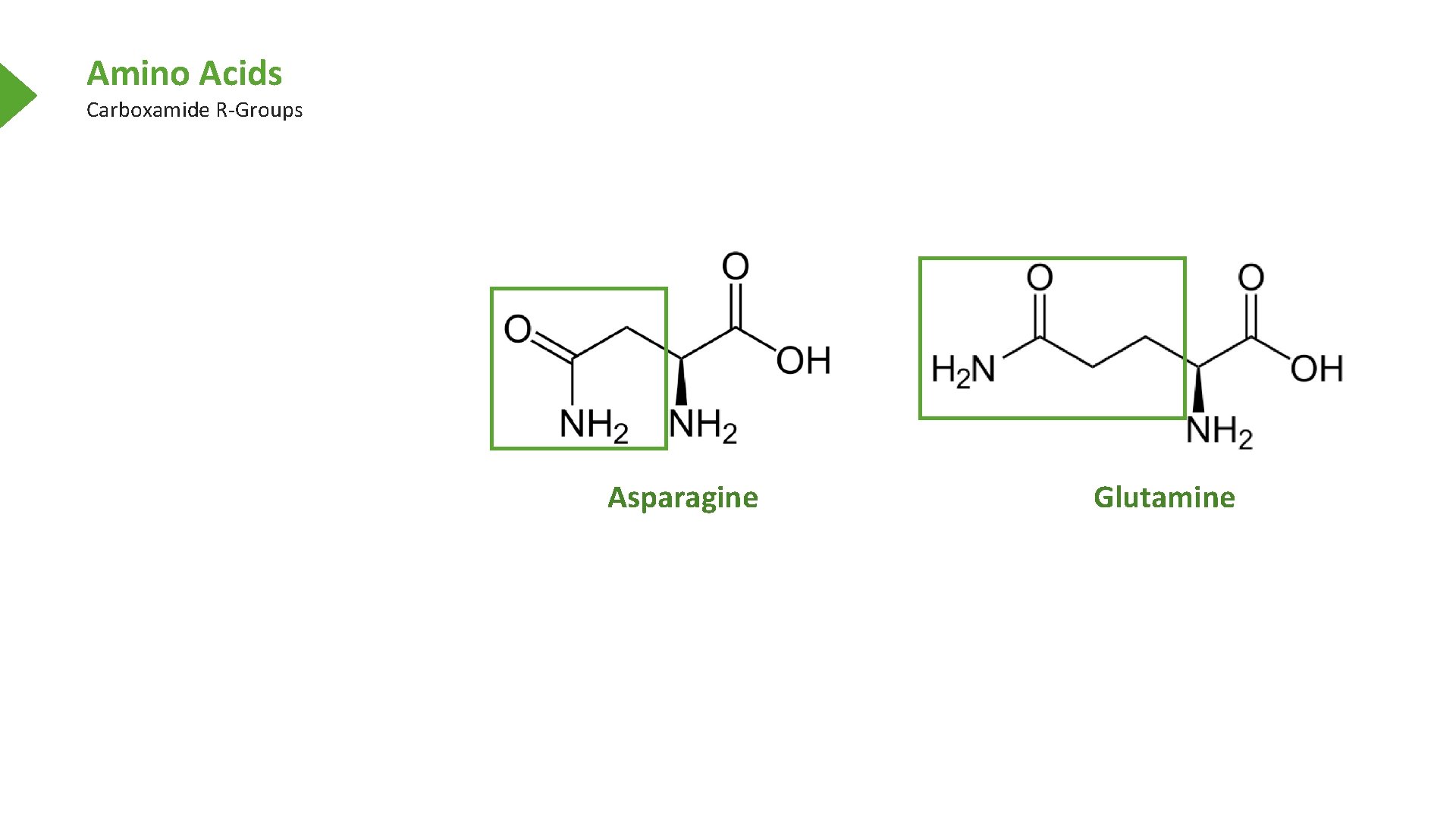
Amino Acids Carboxamide R-Groups Asparagine Glutamine

Amino Acids Carboxamide R-Groups Asparagine Glutamine
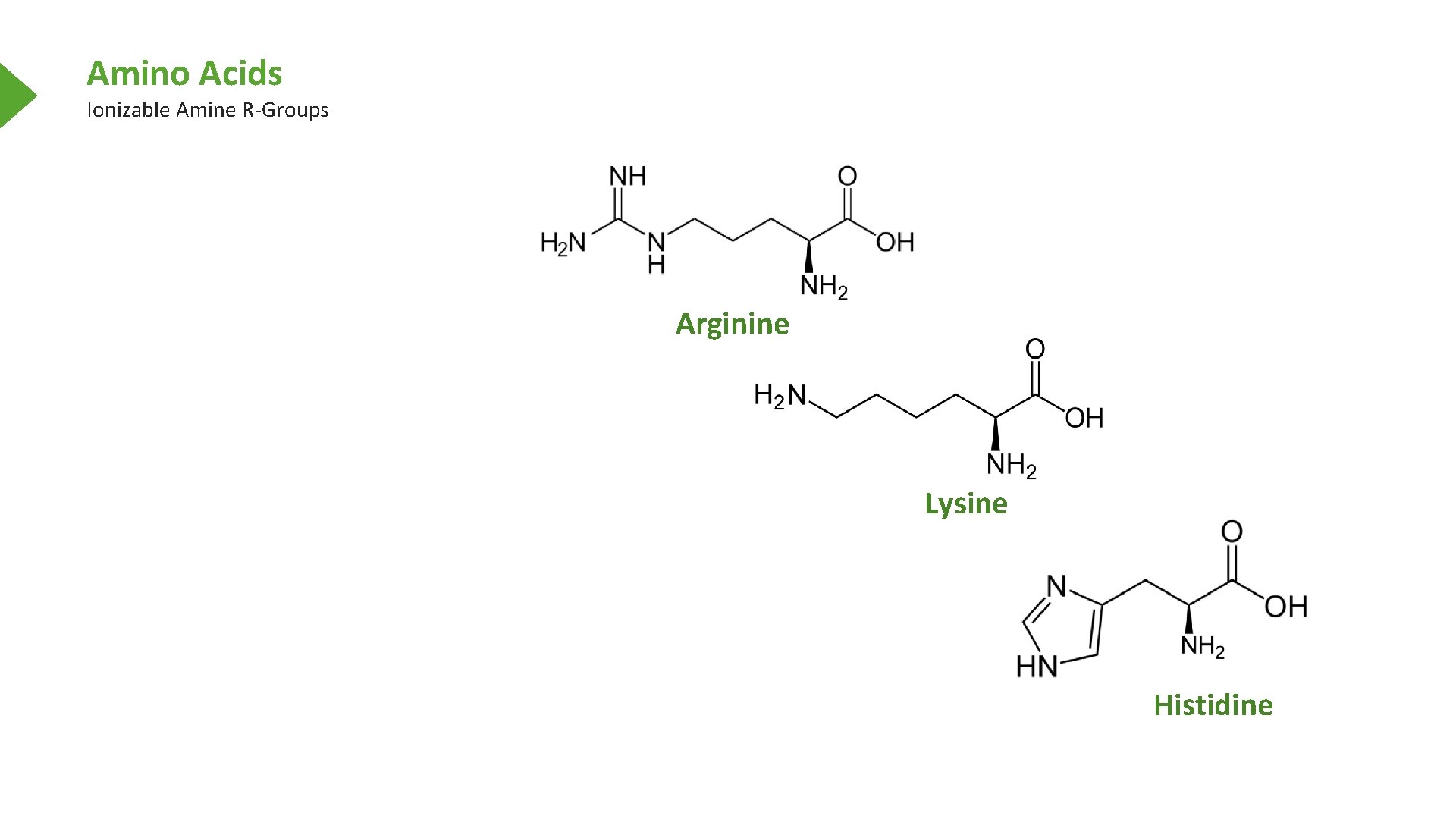
Amino Acids Ionizable Amine R-Groups Arginine Lysine Histidine
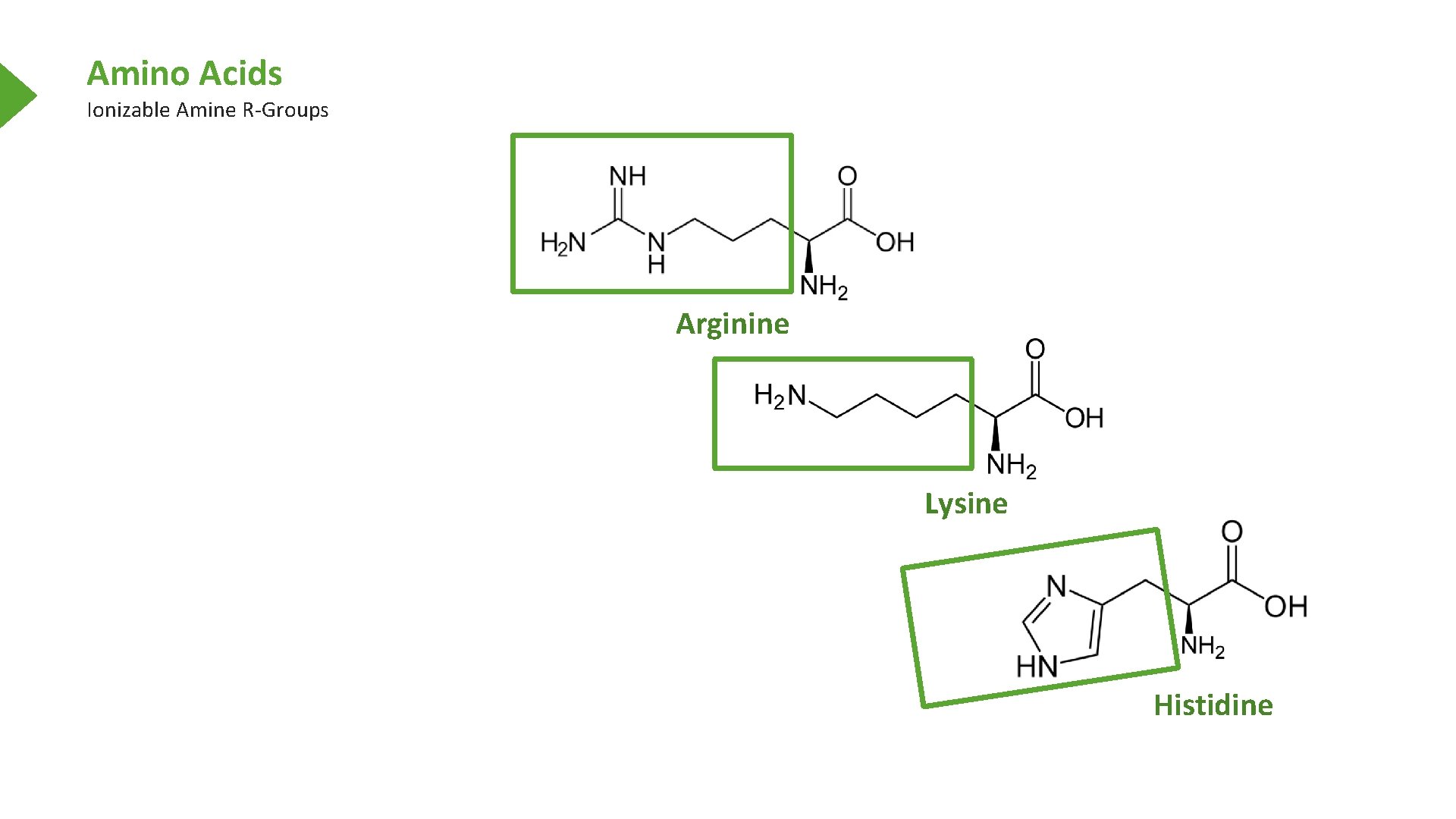
Amino Acids Ionizable Amine R-Groups Arginine Lysine Histidine
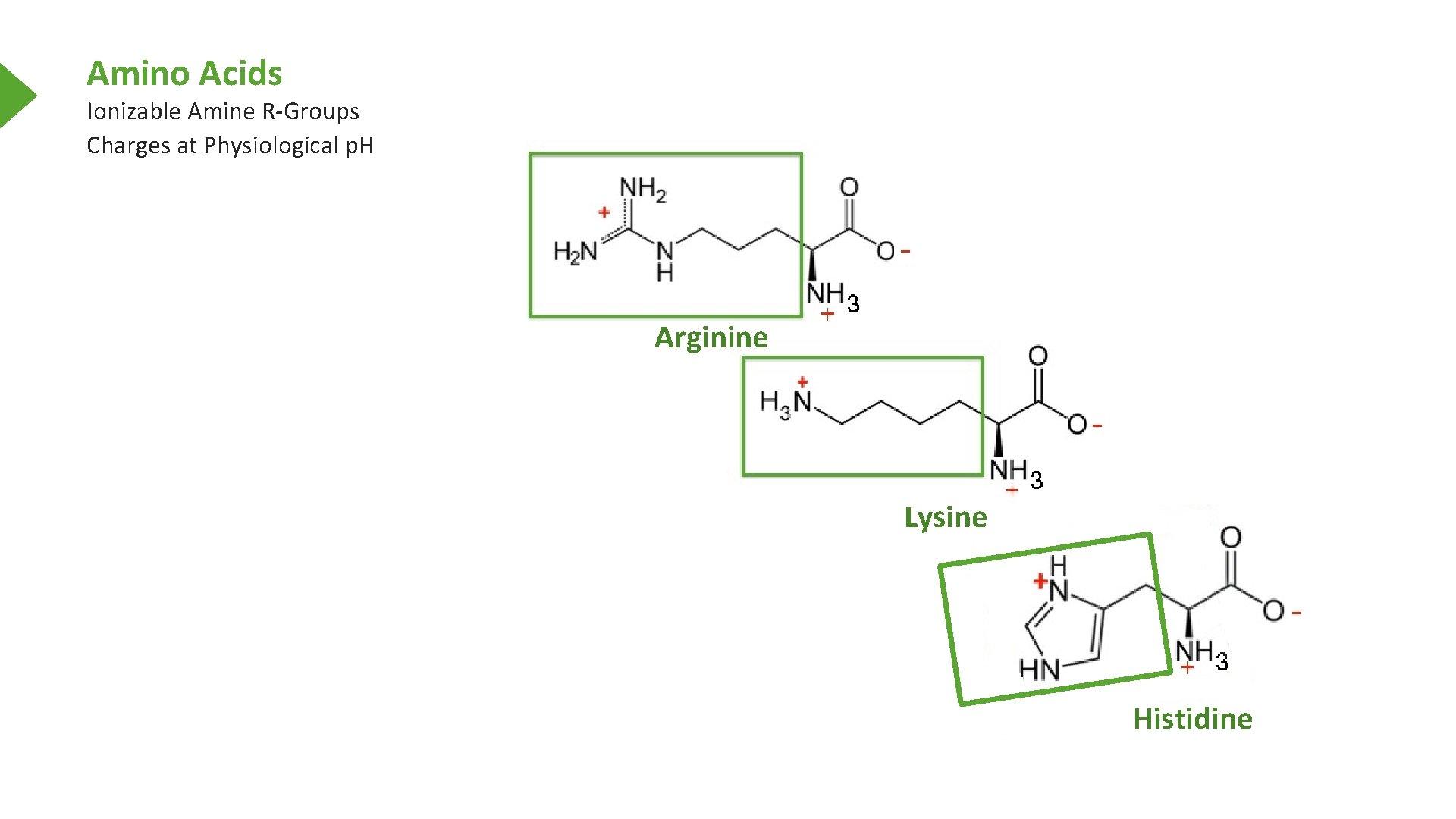
Amino Acids Ionizable Amine R-Groups Charges at Physiological p. H Arginine Lysine Histidine
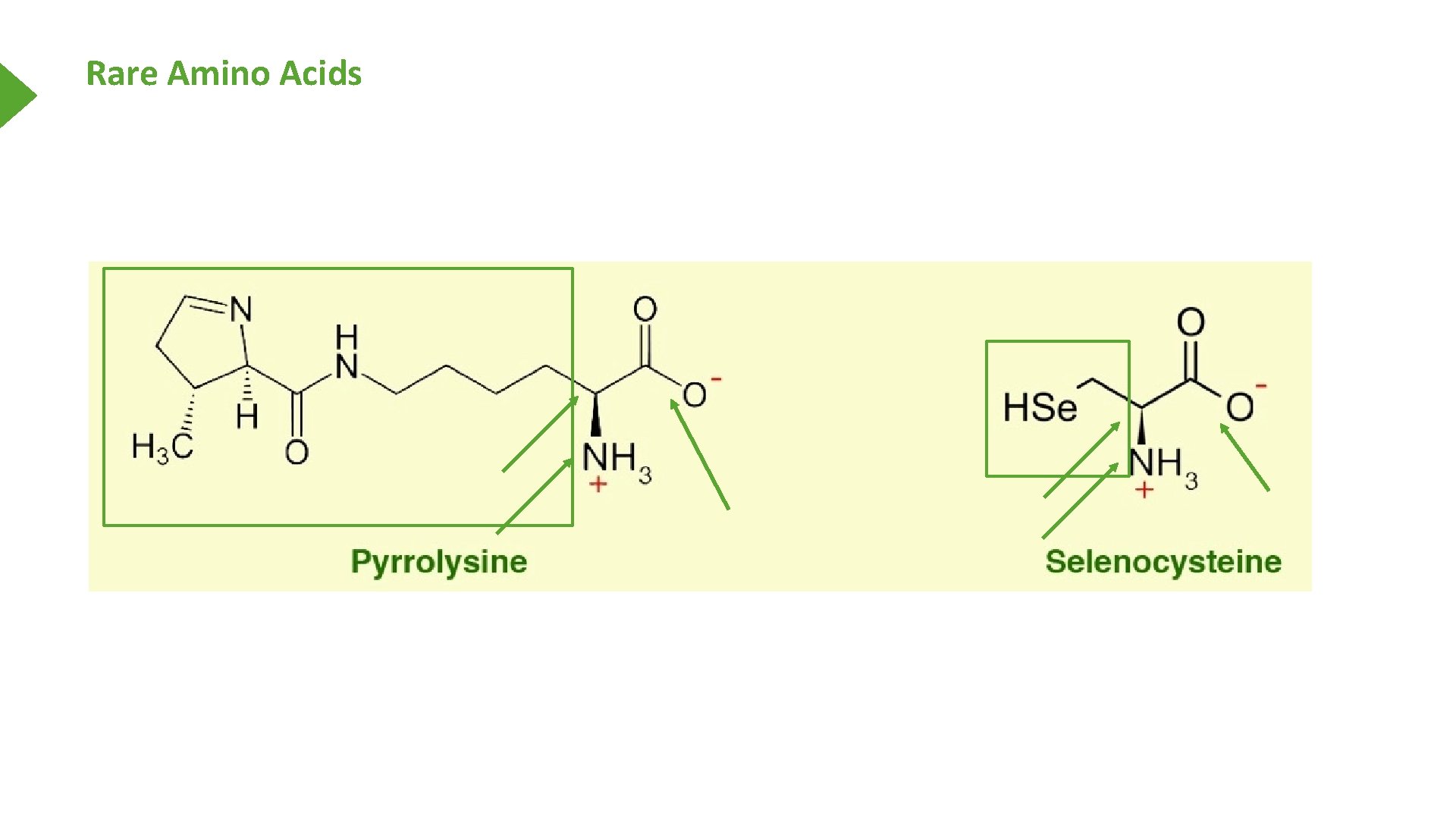
Rare Amino Acids
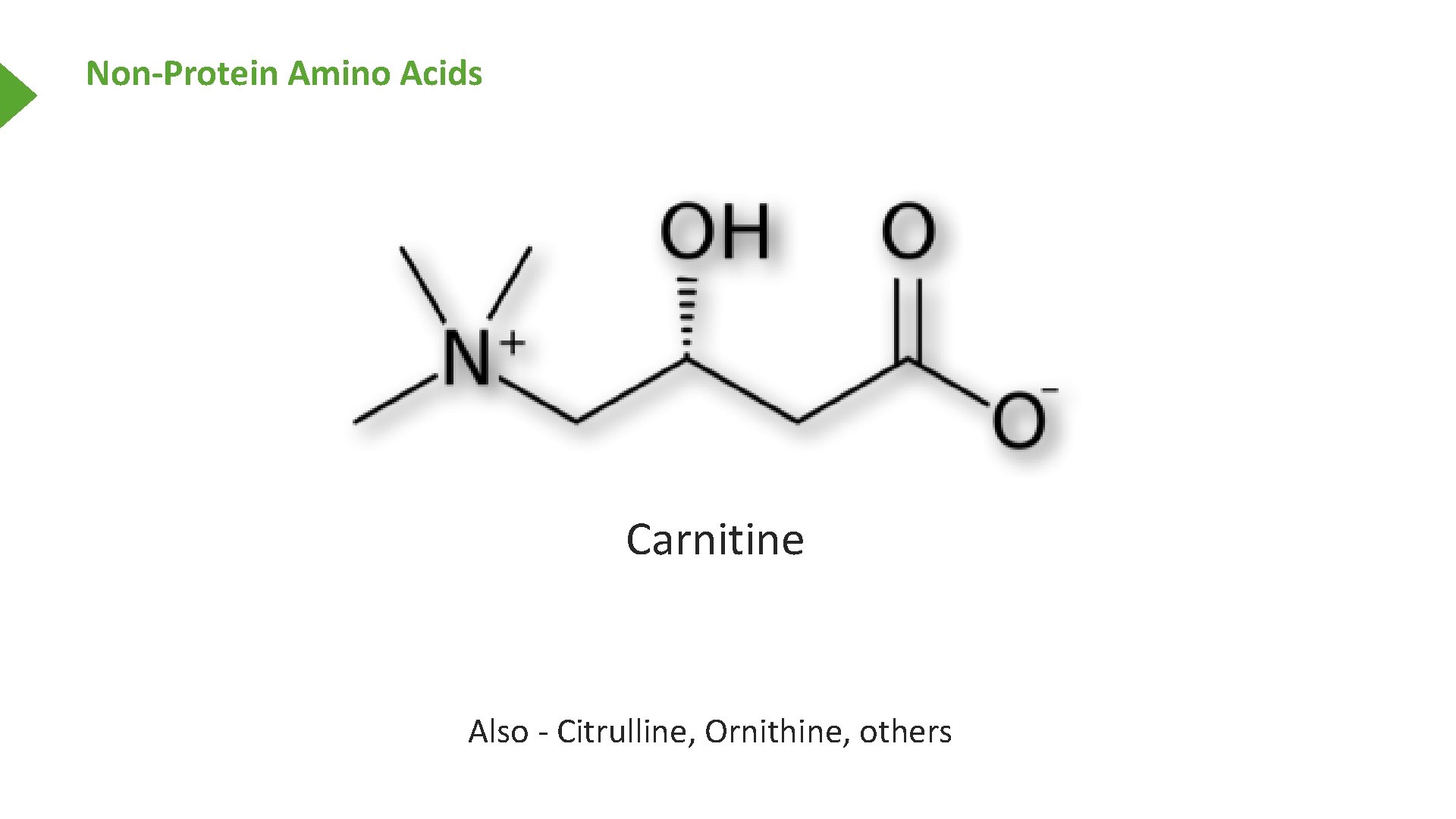
Non-Protein Amino Acids Carnitine Also - Citrulline, Ornithine, others
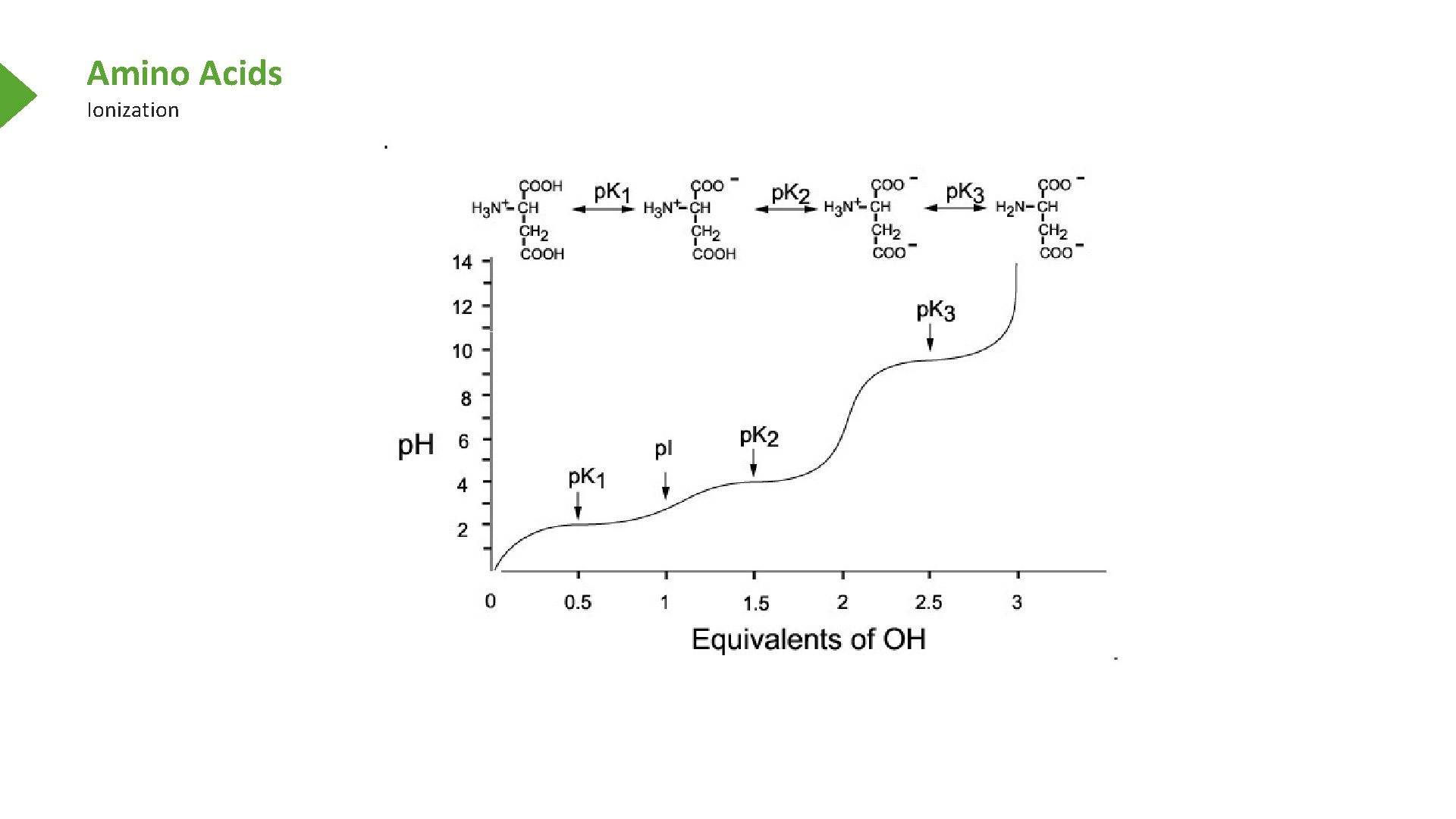
Amino Acids Ionization
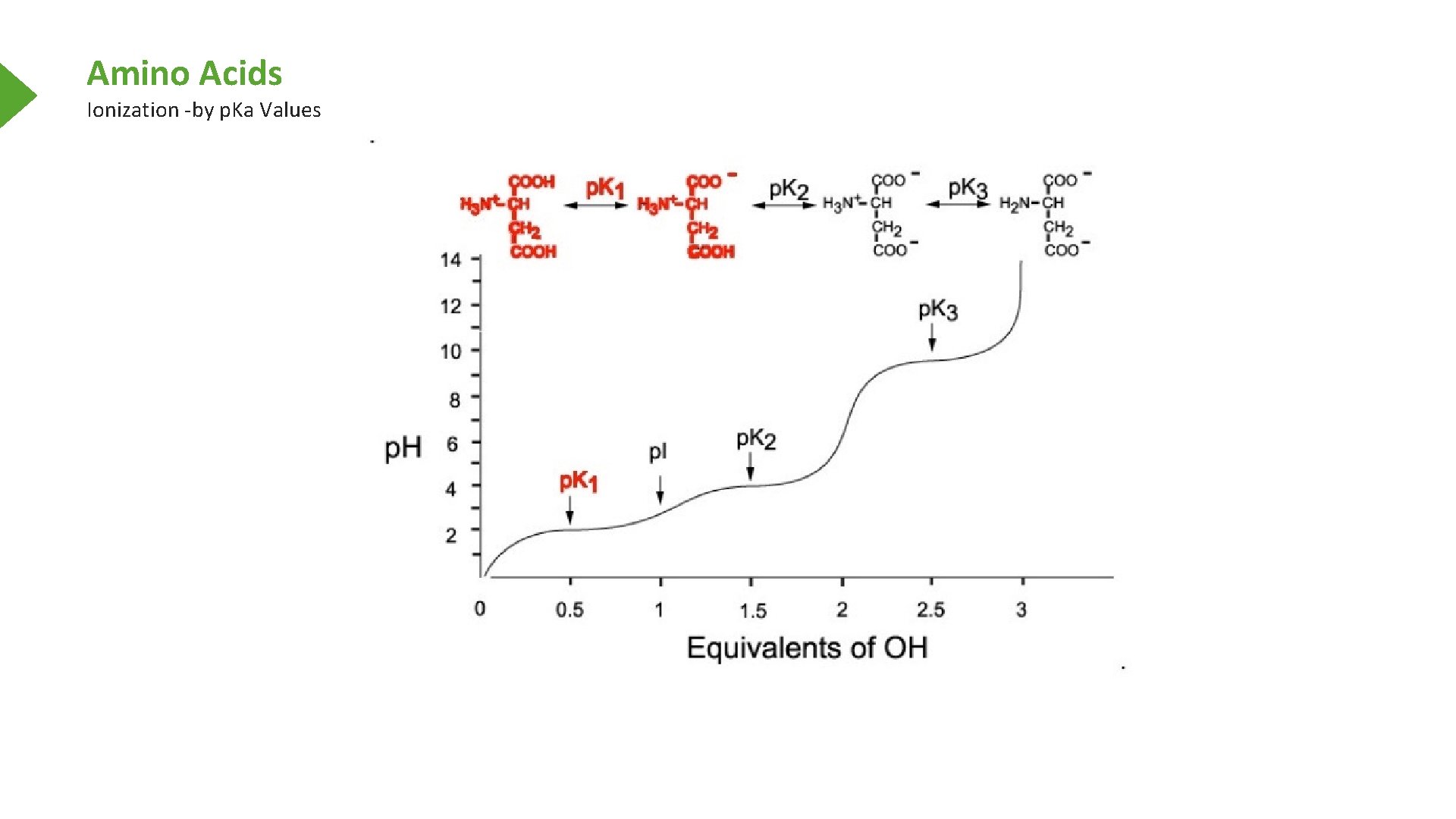
Amino Acids Ionization -by p. Ka Values
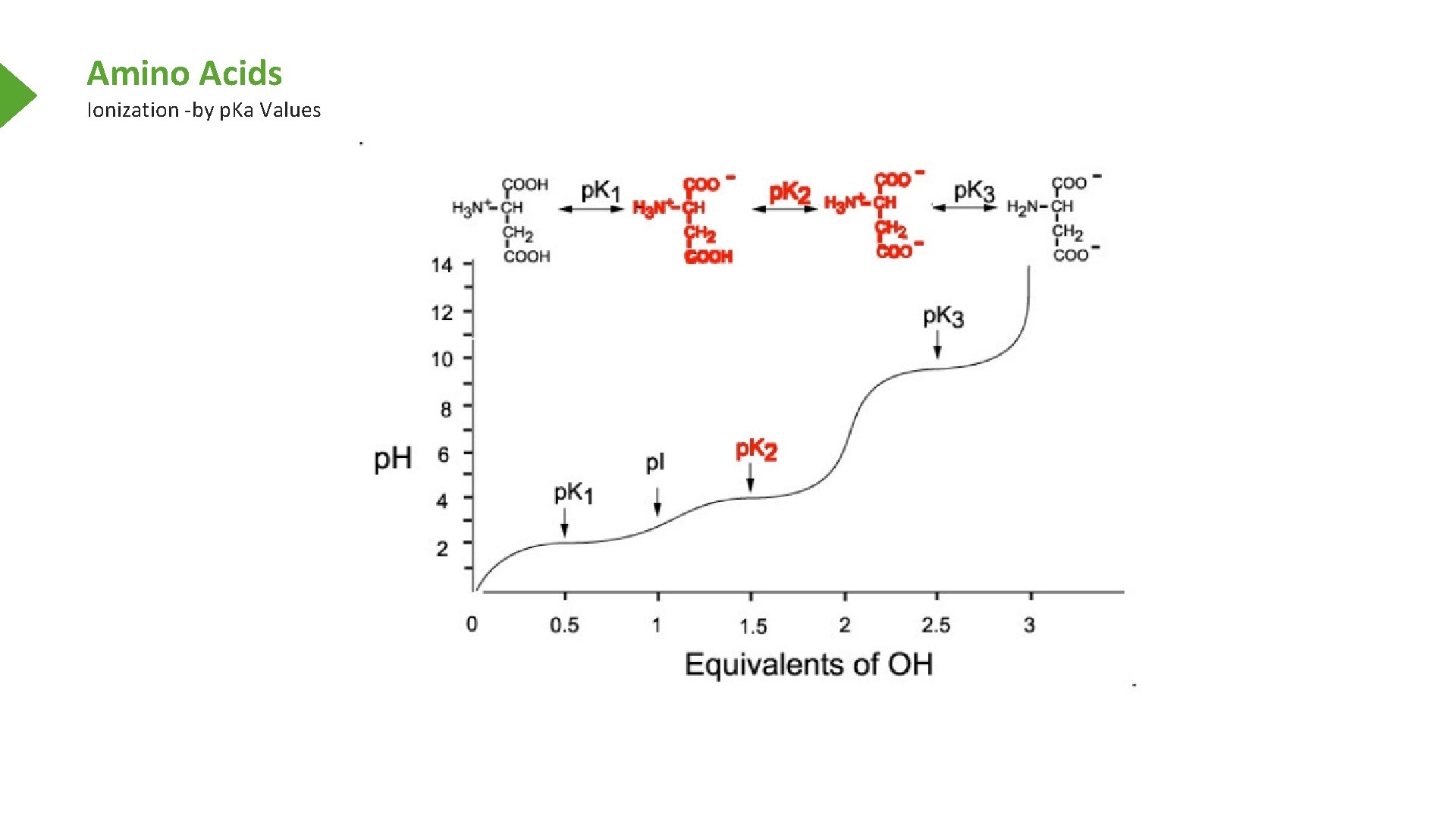
Amino Acids Ionization -by p. Ka Values
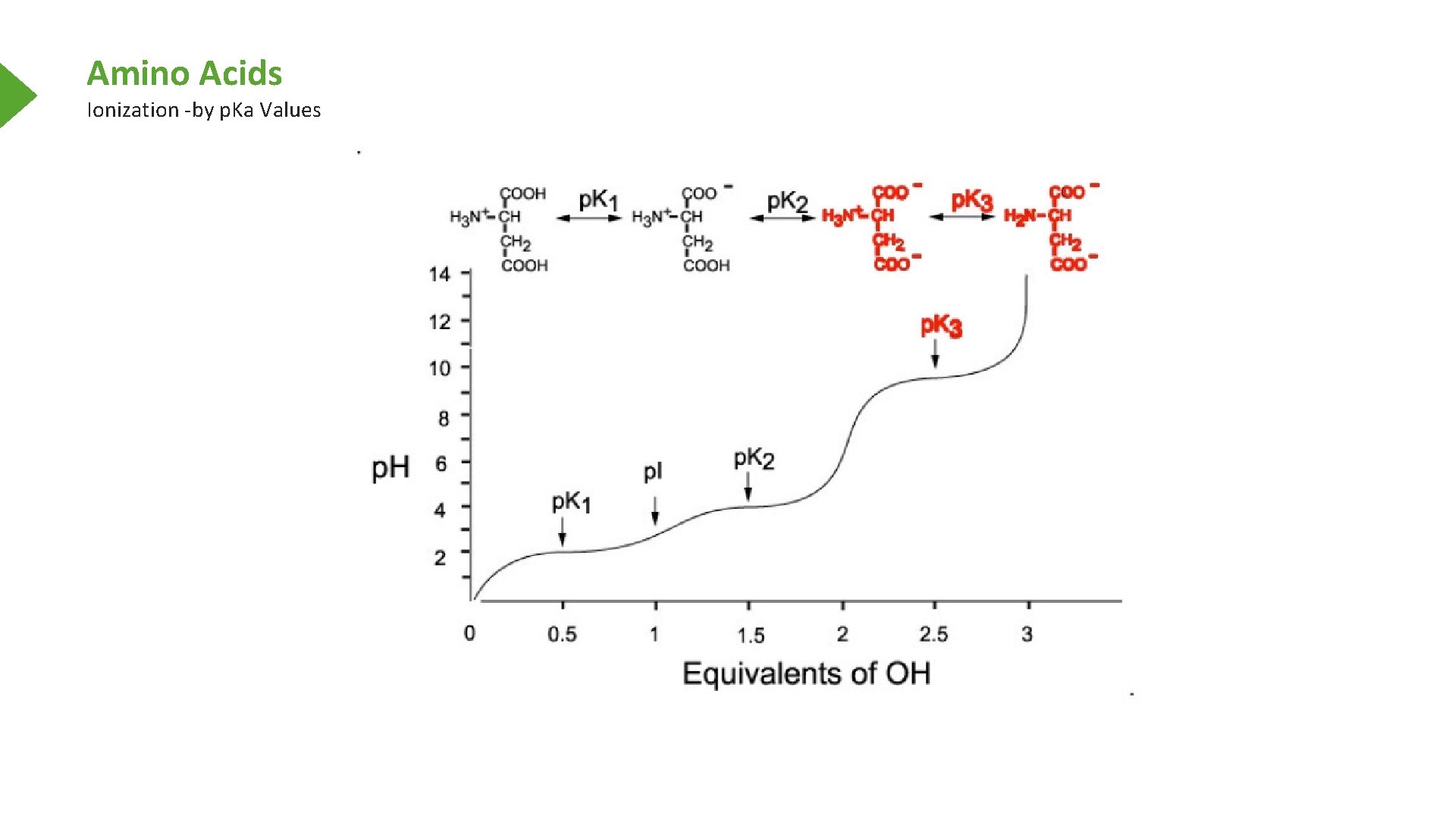
Amino Acids Ionization -by p. Ka Values
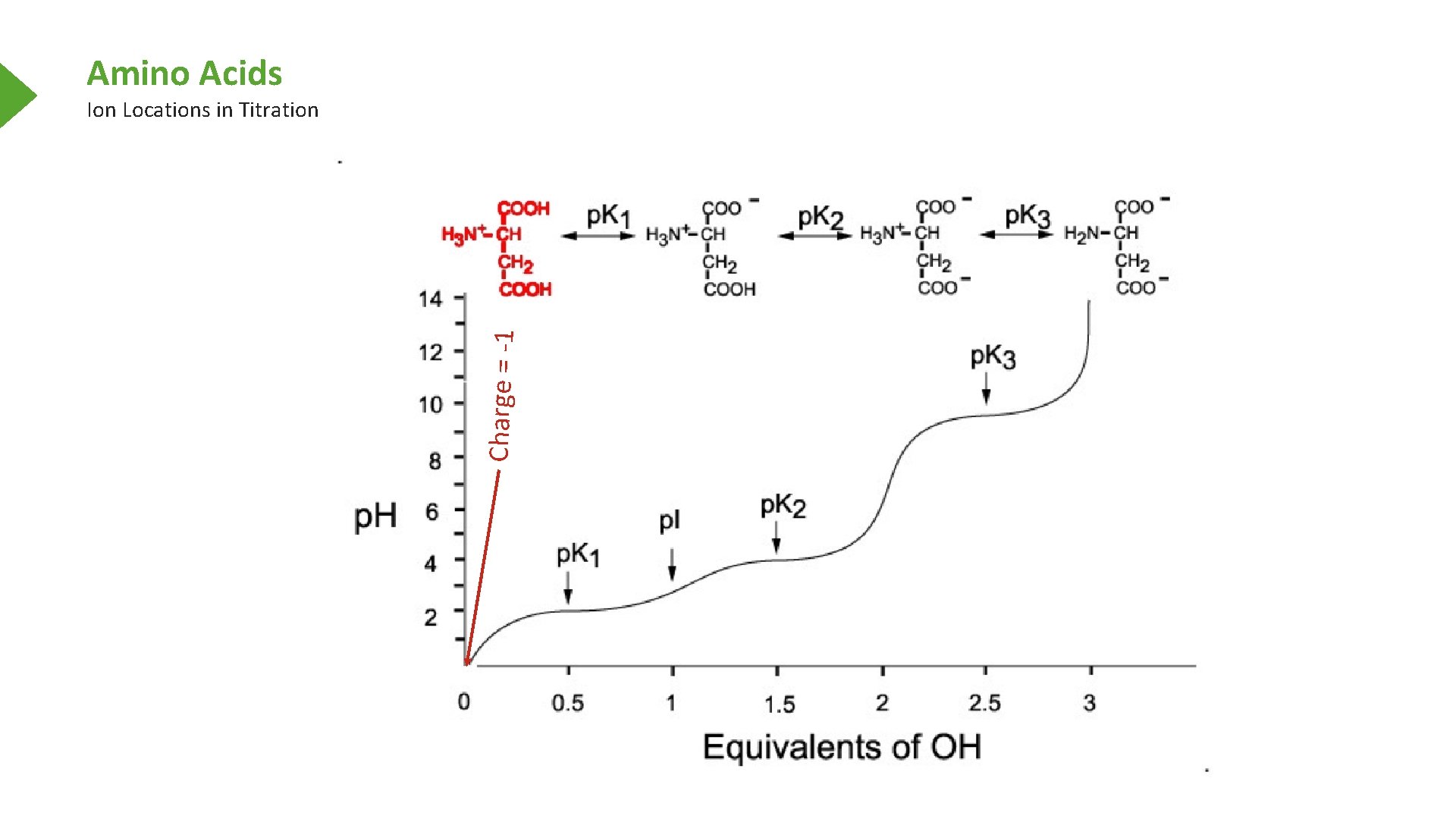
Amino Acids Charge = -1 Ion Locations in Titration
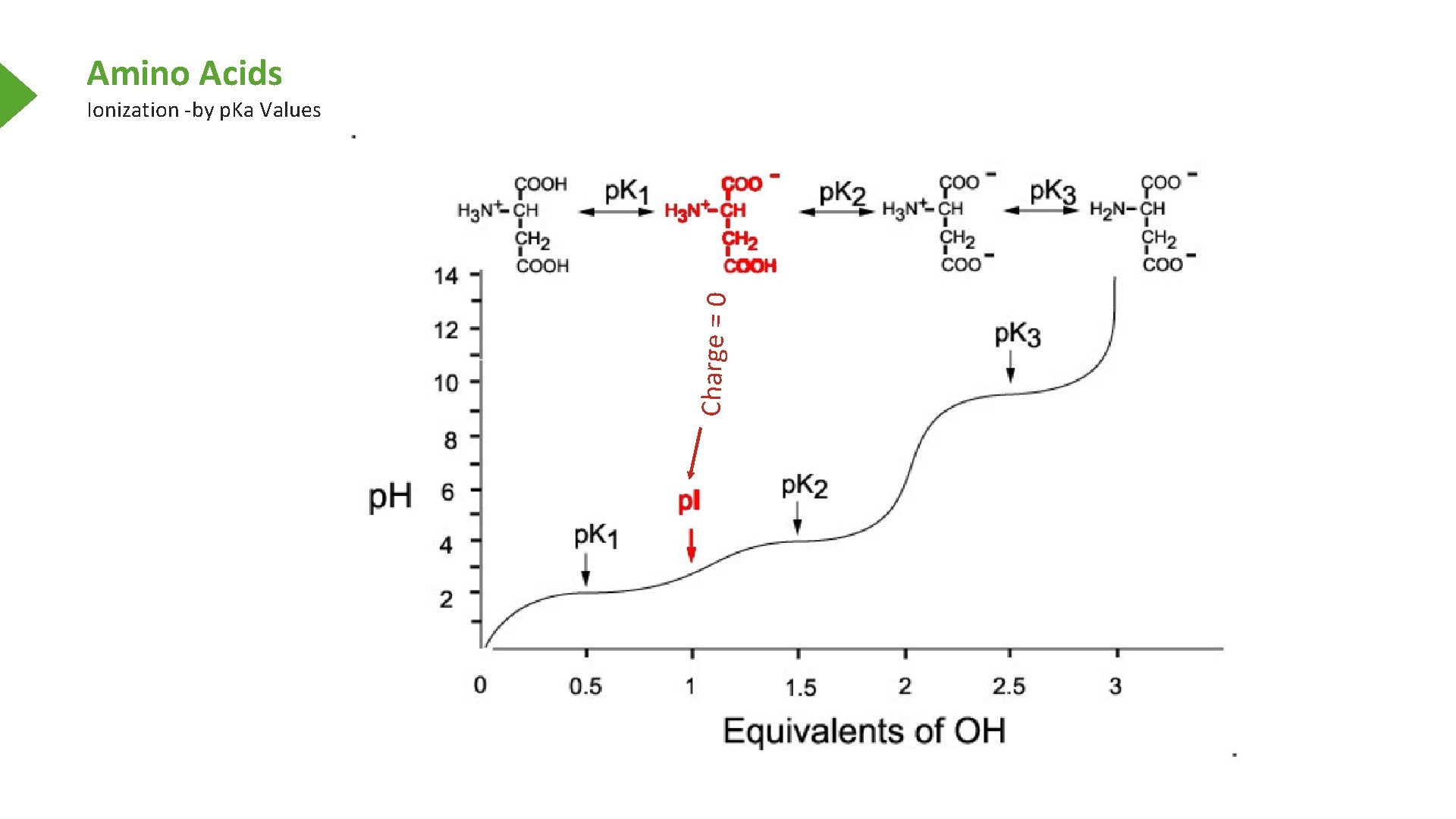
Amino Acids Charge = 0 Ionization -by p. Ka Values
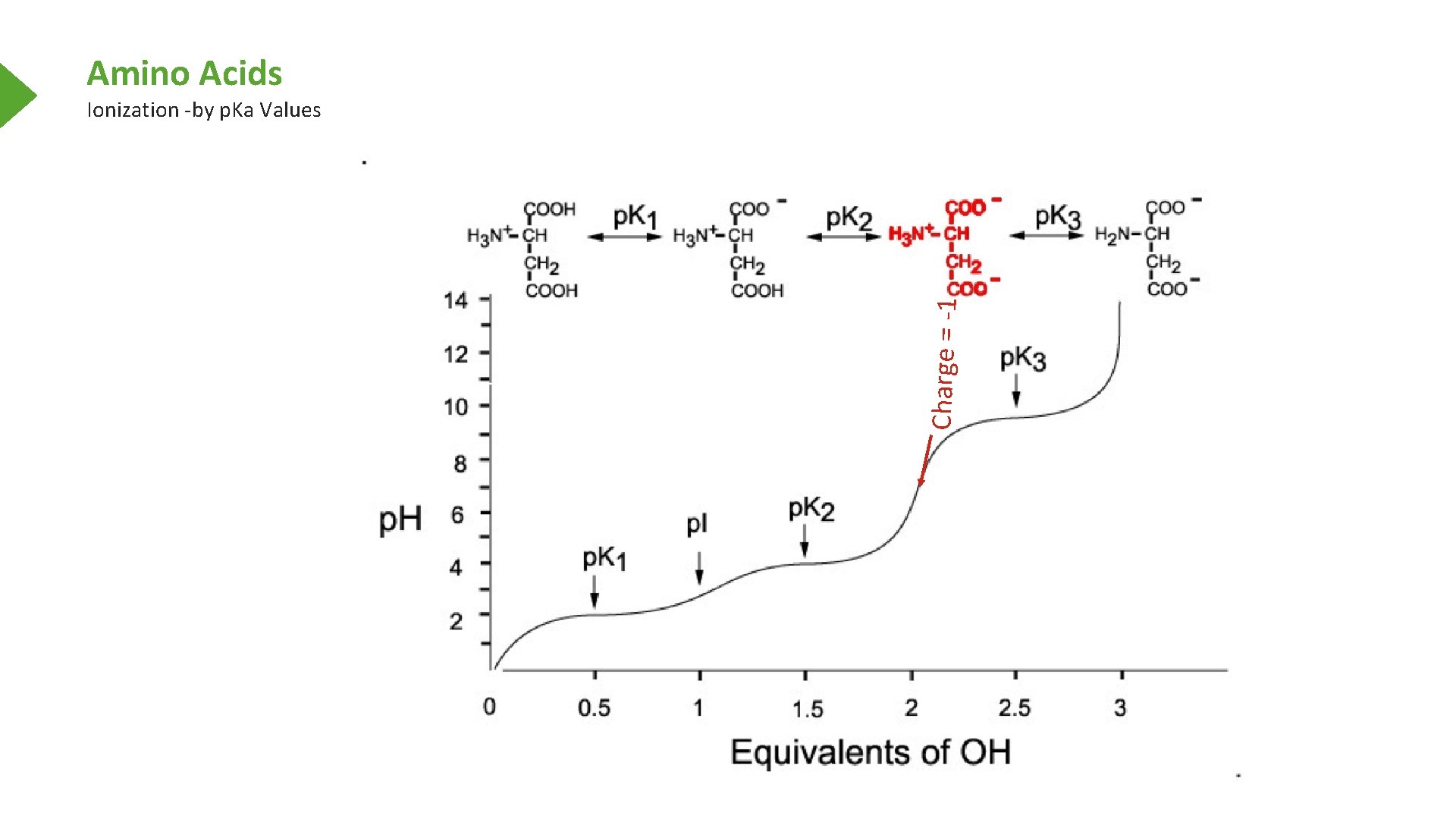
Amino Acids Charge = -1 Ionization -by p. Ka Values
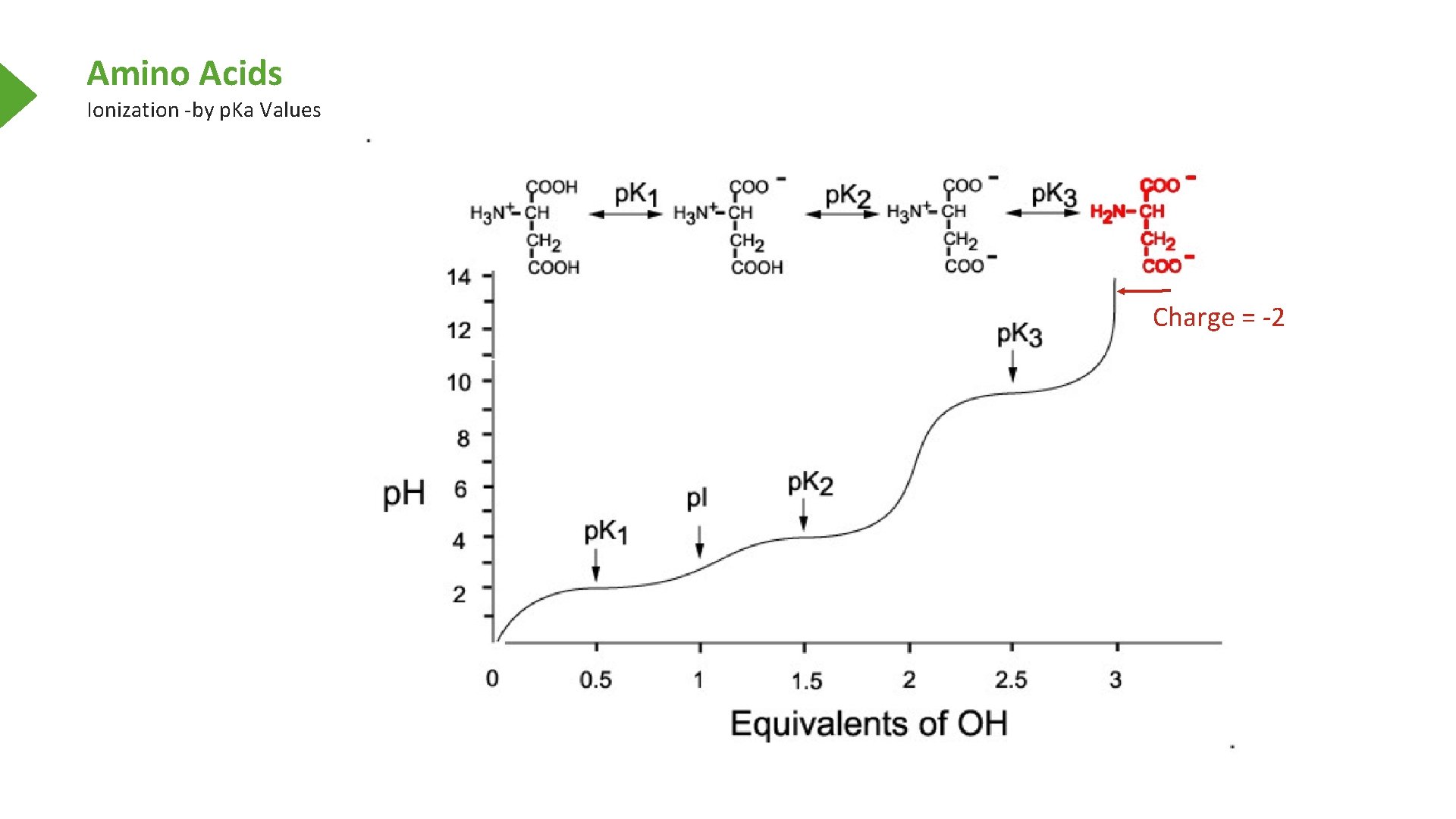
Amino Acids Ionization -by p. Ka Values Charge = -2
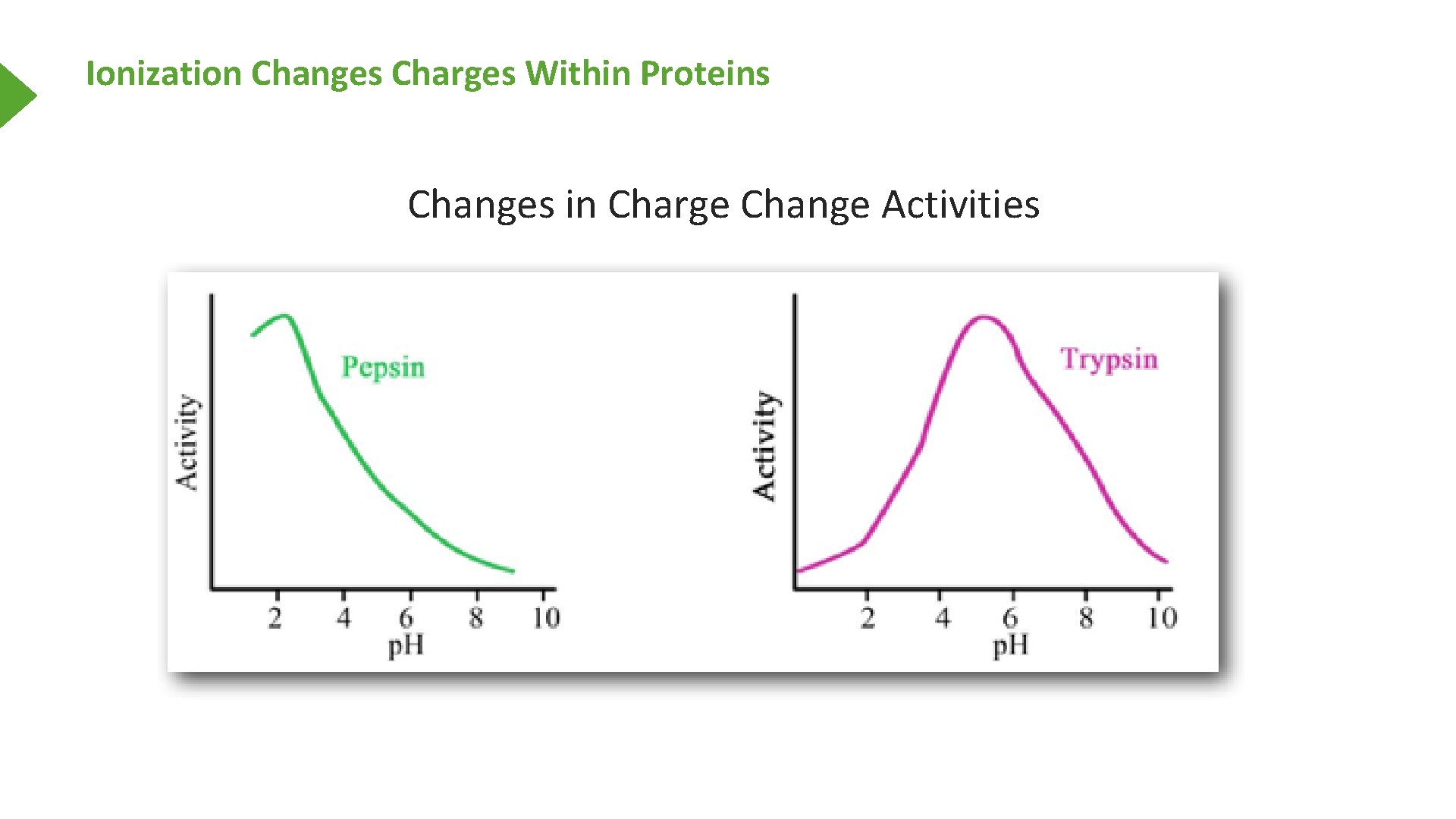
Ionization Changes Charges Within Proteins Changes in Charge Change Activities

Post-translational Modifications
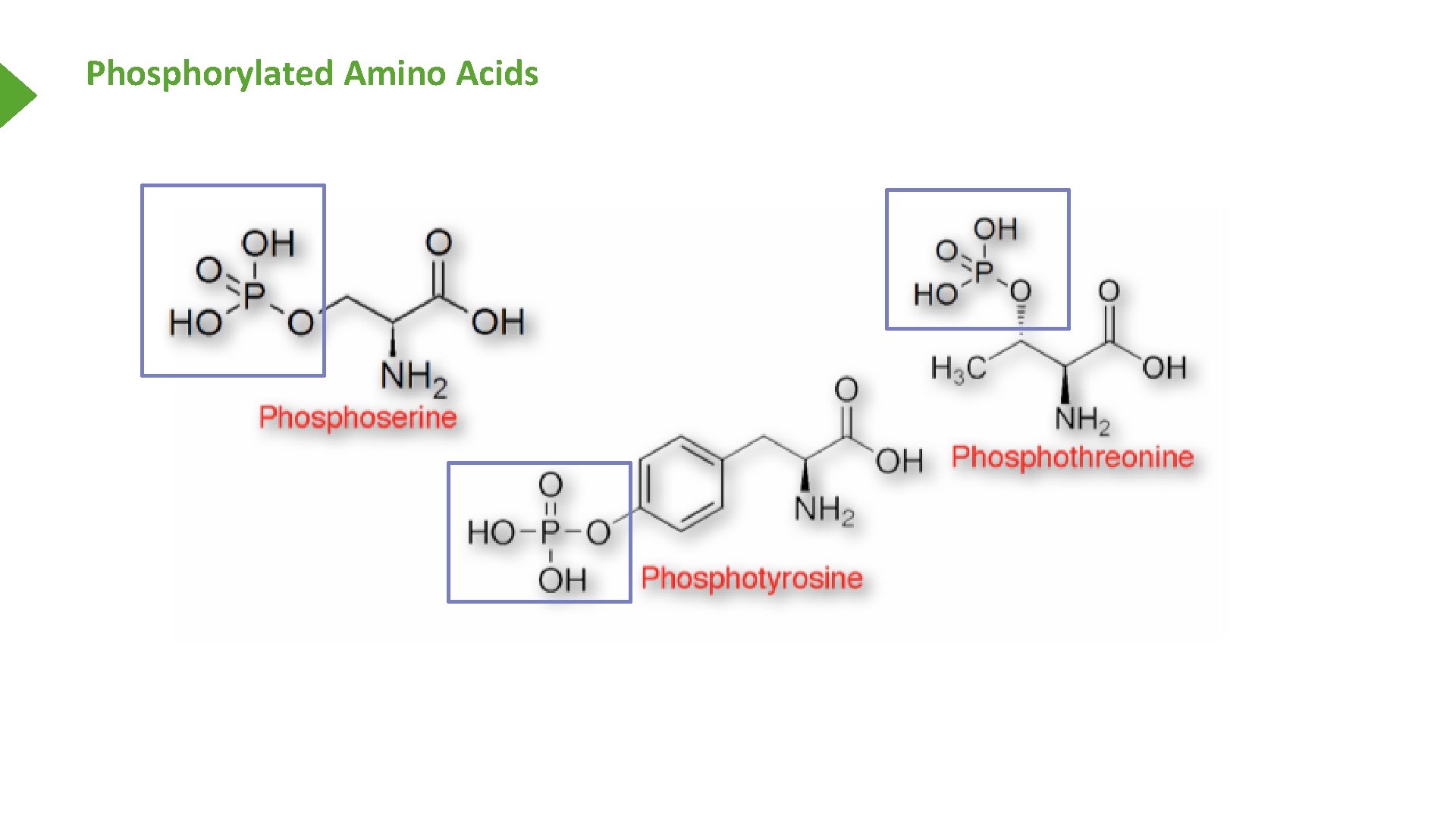
Phosphorylated Amino Acids
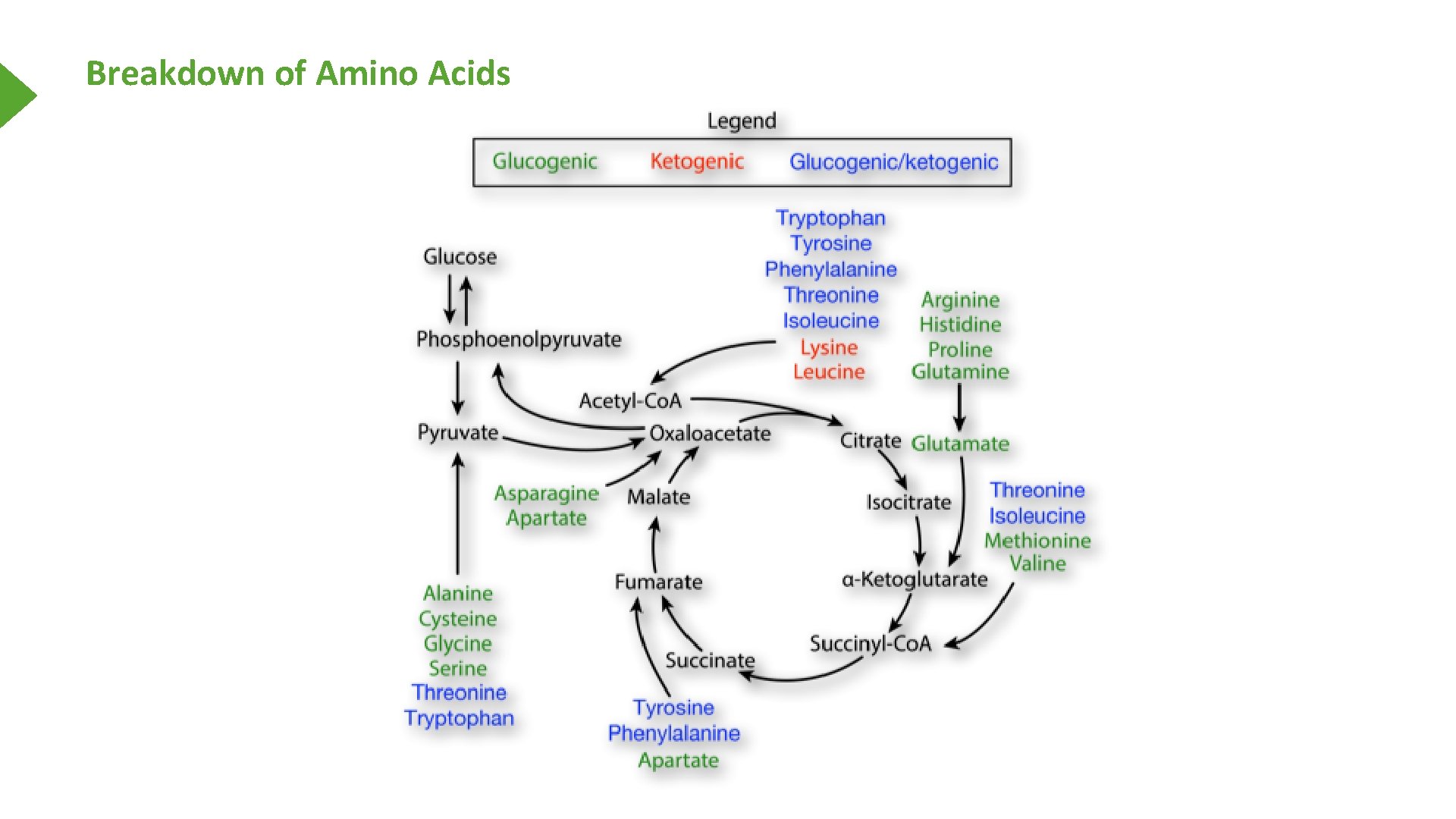
Breakdown of Amino Acids
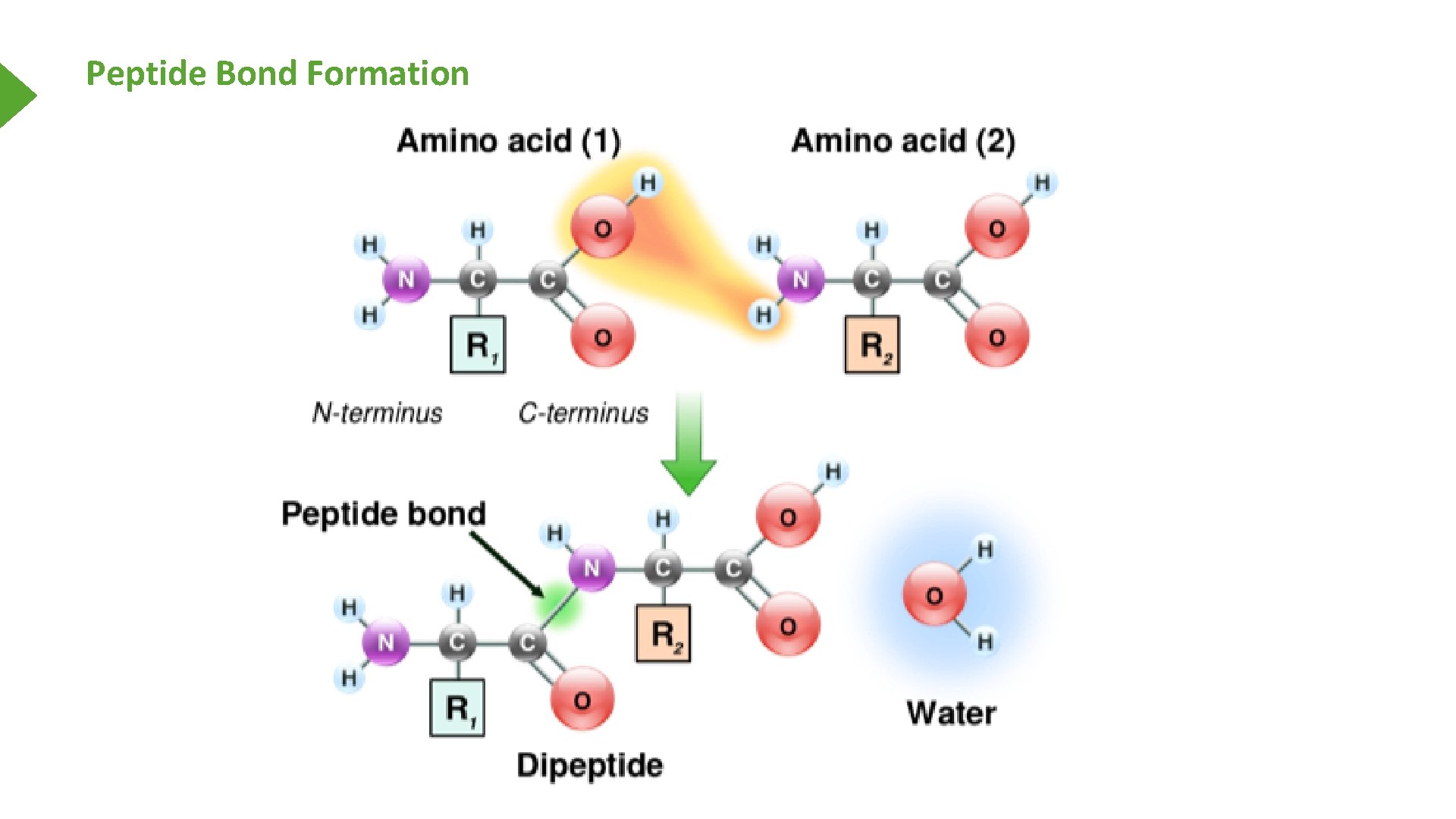
Peptide Bond Formation
- Slides: 45