Amino Acids and the Primary Structure of Proteins














- Slides: 20

Amino Acids and the Primary Structure of Proteins Important biological functions of proteins 1. Enzymes, the biochemical catalysts 2. Storage and transport of biochemical molecules 3. Physical cell support and shape (tubulin, actin, collagen) 4. Mechanical movement (flagella, mitosis, muscles) (continued)

Amino Acids and the Primary Structure of Proteins 5. Decoding cell information (translation, regulation of gene expression) 6. Hormones or hormone receptors (regulation of cellular processes) 7. Other specialized functions (antibodies, toxins etc)

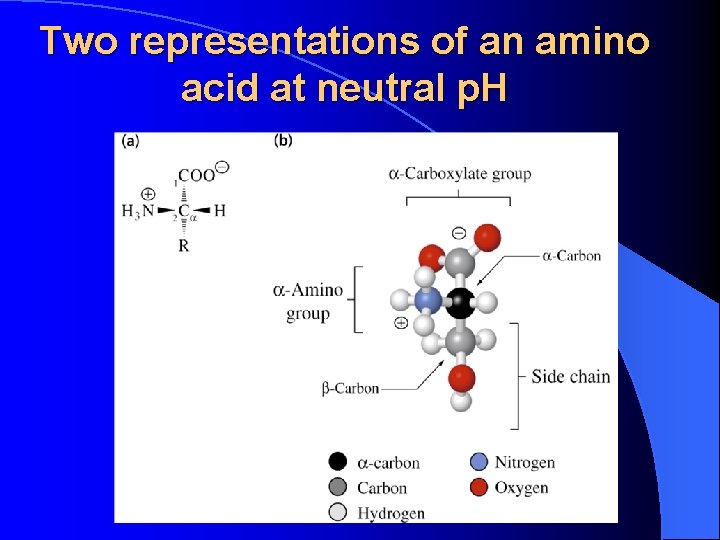
Zwitterionic form of amino acids • Under normal cellular conditions amino acids are zwitterions (dipolar ions): Amino group = Carboxyl group = -NH 3+ -COO-

Two representations of an amino acid at neutral p. H

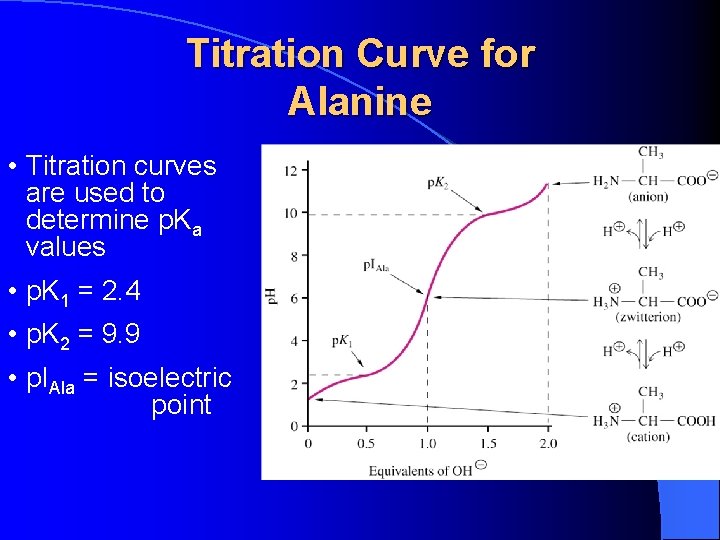
Titration Curve for Alanine • Titration curves are used to determine p. Ka values • p. K 1 = 2. 4 • p. K 2 = 9. 9 • p. IAla = isoelectric point

Aliphatic R Groups • Glycine (Gly, G) - the a-carbon is not chiral since there are two H’s attached (R=H) • Four amino acids have saturated side chains: Alanine (Ala, A) Valine (Val, V) Leucine (Leu, L) Isoleucine (Ile, I) • Proline (Pro, P) 3 -carbon chain connects a-C and N

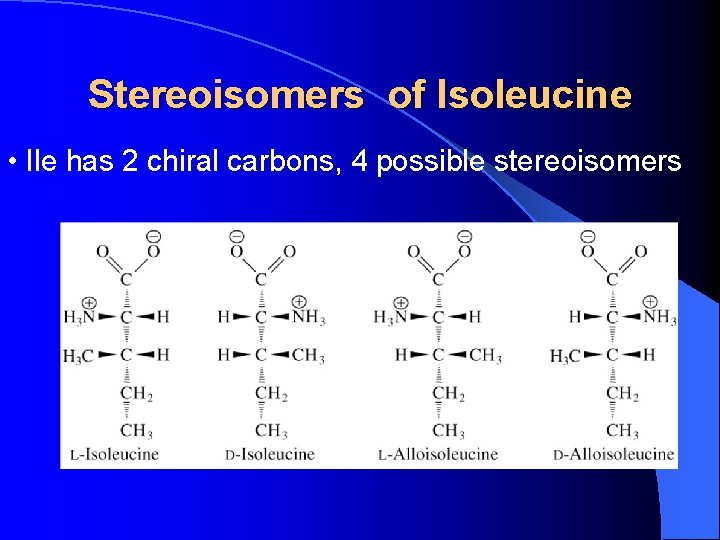
Stereoisomers of Isoleucine • Ile has 2 chiral carbons, 4 possible stereoisomers

Aromatic Amino Acids

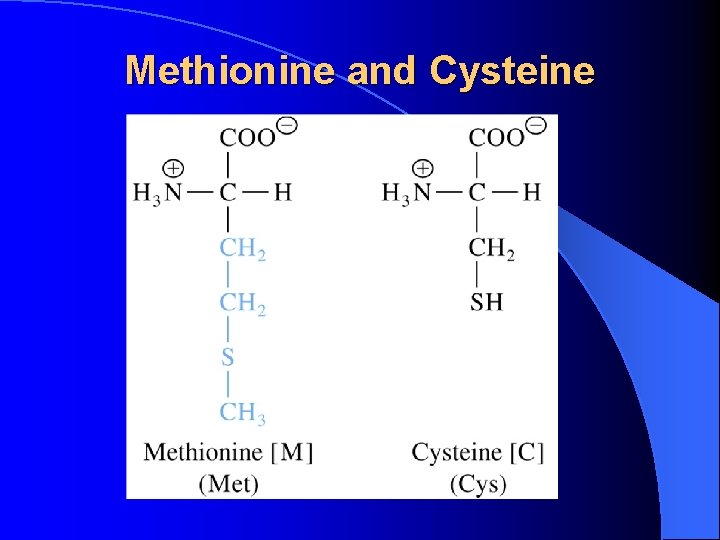
Methionine and Cysteine

Formation of Cystine

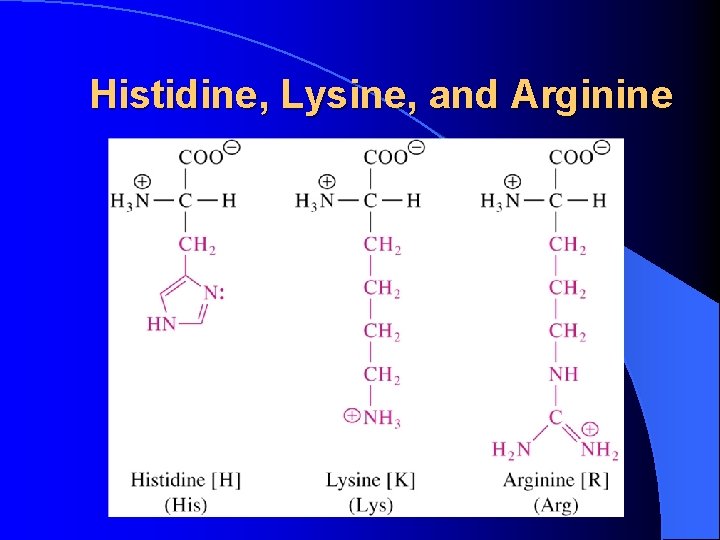
Histidine, Lysine, and Arginine

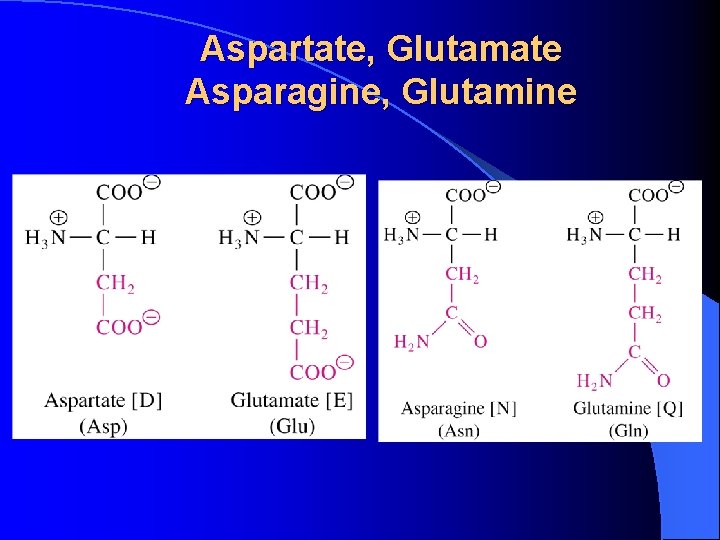
Aspartate, Glutamate Asparagine, Glutamine

Peptide Chain Nomenclature • Amino acid “residues” compose peptide chains • Peptide chains are numbered from the N (amino) terminus to the C (carboxyl) terminus • Example: (N) Gly-Arg-Phe-Ala-Lys (C) (or GRFAK) • Formation of peptide bonds eliminates the ionizable a-carboxyl and a-amino groups of the free amino acids

Peptide Sequencing Edmann Degradation

Edmann Degradation (cont. )

Edmann Degradation (cont. )

Edmann Degradation (cont. )

Cleaving Disulfide bonds and Protecting the thiols formed • Disulfide bonds in proteins must be cleaved: (1) To permit isolation of the PTH-cysteine during the Edman procedure (2) To separate peptide chains • Treatment with thiol compounds reduces the (R-S-S-R) cystine bond to two cysteine (R-SH) residues • Thiols are protected with iodoacetate

Further Protein Sequencing Strategies • Proteins may be too large to be sequenced completely by the Edman method • Proteases (enzymes cleaving peptide bonds) and chemical agents are used to selectively cleave the protein into smaller fragments • Cyanogen bromide (Br. CN) cleaves polypeptides at the C-terminus of Met residues

Protease Enzymes cleave specific peptide bonds • Chymotrypsin - carbonyl side of aromatic or bulky noncharged aliphatic residues (e. g. Phe, Tyr, Trp, Leu) • Trypsin - carbonyl side, basic residues (Lys, Arg). • Staphylococcus aureus V 8 protease - carbonyl side of negatively charged residues (Glu, Asp). NOTE: in 50 m. M ammonium bicarbonate cleaves only at Glu.