Amino acid metabolism Nitrogen balance Dietary protein amino
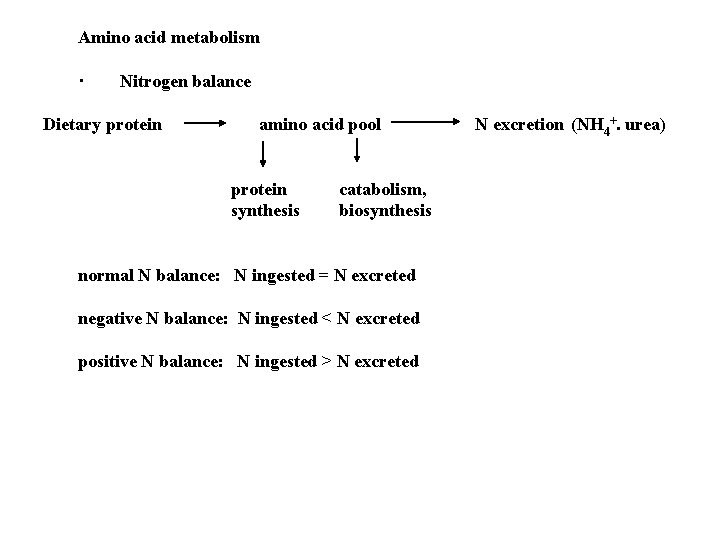
Amino acid metabolism · Nitrogen balance Dietary protein amino acid pool protein synthesis catabolism, biosynthesis normal N balance: N ingested = N excreted negative N balance: N ingested < N excreted positive N balance: N ingested > N excreted N excretion (NH 4+. urea)

Requirement for essential amino acids

Amino acid catabolism · accounts for ~ 10% of energy requirement of adults · When: • · excess protein in diet (amino acids are not stored) • · protein degradation exceeds demand for new protein • · starvation when carbohydrates are not available · (protein storing seeds such as beans, peas, etc. ) ·

Glucogenic vs ketogenic amino acids · ketogenic: yield Ac. Co. A or Ac. Ac as end products of catabolism - leu, lys · glucogenic: are degraded to pyruvate or a member of the TCA cycle (succinyl. Co. A, OAA, a-ketoglutarate, fumarate). In absence of sugars, glucogenic amino acids permit continued oxidation of fatty acids by maintaining TCA cycle intermediates. Also source of carbons for gluconeogenesis in liver - ile, phe, tyr, trp · glucogenic and ketogenic: yield both ketogenic and glucogenic products. - all others

N catabolism General strategy: 1· · removal of N from amino acid by transamination (generally first or second step of amino acid catabolic pathways) and collection of N in glutamic acid 2· deamination of glutamic acid with release of NH 4+ -glutamate dehydrogenase 3. Collection of N in glutamine or alanine for delivery to liver 4· removal of NH 4+ by : i. secretion; or ii. conversion to urea or other less toxic form.
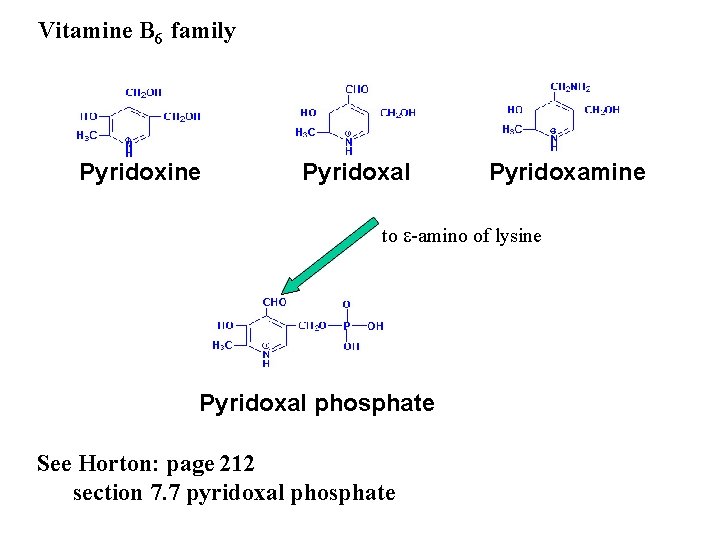
Vitamine B 6 family Pyridoxine Pyridoxal Pyridoxamine to e-amino of lysine Pyridoxal phosphate See Horton: page 212 section 7. 7 pyridoxal phosphate
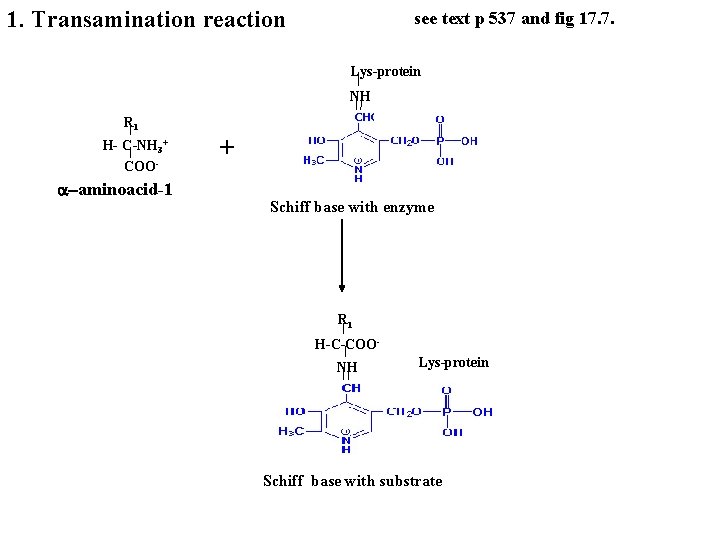
1. Transamination reaction see text p 537 and fig 17. 7. Lys-protein NH R 1 H- C-NH 3+ COO- a-aminoacid-1 + Schiff base with enzyme R 1 H-C-COONH Lys-protein Schiff base with substrate
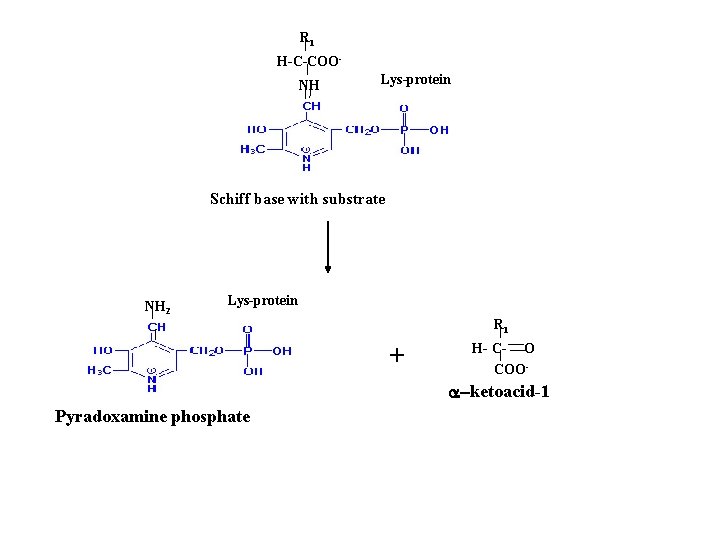
R 1 H-C-COONH Lys-protein Schiff base with substrate NH 2 Lys-protein R 1 + H- C- O COO- a-ketoacid-1 Pyradoxamine phosphate
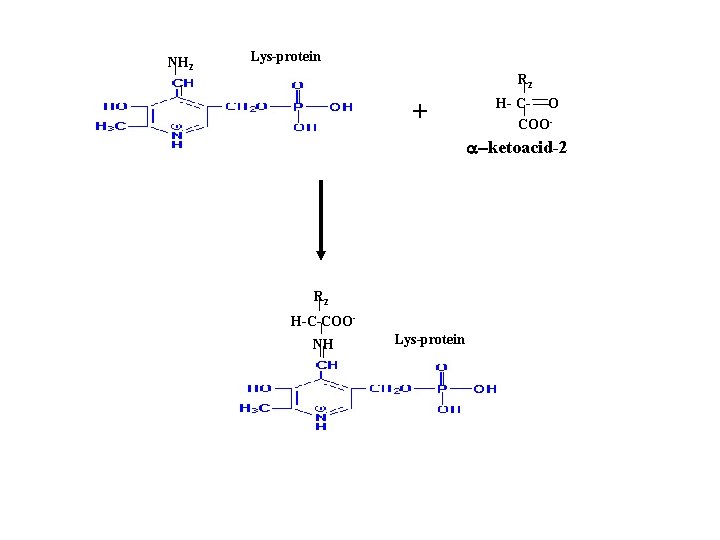
NH 2 Lys-protein R 2 + H- C- O COO- a-ketoacid-2 R 2 H-C-COONH Lys-protein
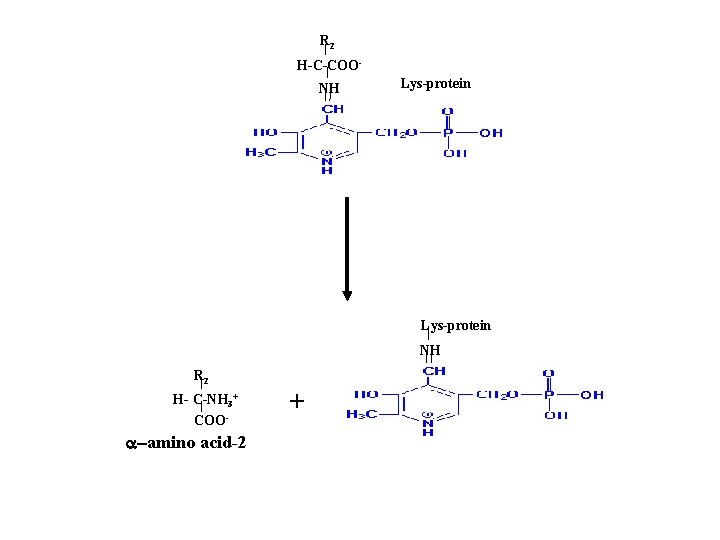
R 2 H-C-COONH Lys-protein NH R 2 H- C-NH 3+ COO- a-amino acid-2 +

Net reaction: a-amino acid-1 + a-ketoacid-2 PLP a-amino acid-2 + a-ketoacid-1 e. g. alanine + a-ketoglutarate pyruvate + glutamate

N catabolism General strategy: 1· · removal of N from amino acid by transamination (generally first or second step of amino acid catabolic pathways) and collection of N in glutamic acid 2· deamination of glutamic acid with release of NH 4+ -glutamate dehydrogenase 3. Collection of N in glutamine or alanine for delivery to liver 4· removal of NH 4+ by : i. secretion; or ii. conversion to urea or other less toxic form.
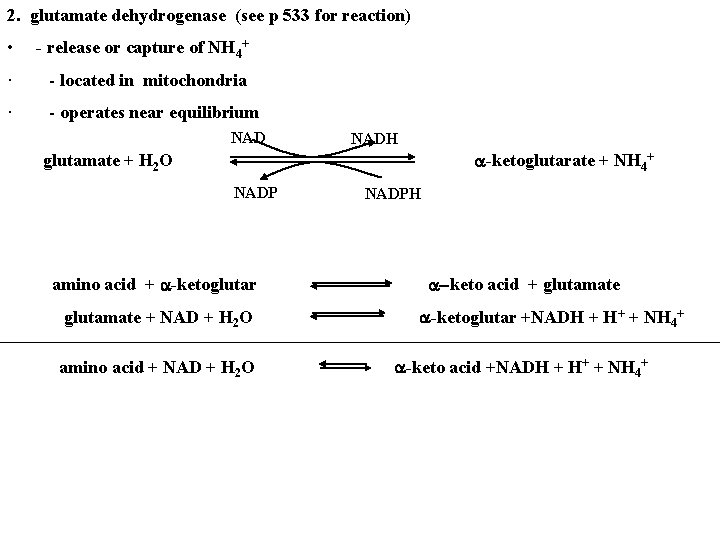
2. glutamate dehydrogenase (see p 533 for reaction) • - release or capture of NH 4+ · - located in mitochondria · - operates near equilibrium NADH a-ketoglutarate + NH 4+ glutamate + H 2 O NADP amino acid + a-ketoglutar glutamate + NAD + H 2 O amino acid + NAD + H 2 O NADPH a-keto acid + glutamate a-ketoglutar +NADH + H+ + NH 4+ a-keto acid +NADH + H+ + NH 4+

3. transport of N to the liver - glutamine synthetase - glutaminase - alanine/glucose cycle 1. Glutamine synthetase ATP glutamate + NH 4 2. Glutaminase glutamine + ADP + Pi glutamine glutamate + NH 4+ Note: glutamate can be used for glucose synthesis. How? 3. Formation of alanine by transamination: alanine/glucose cycle
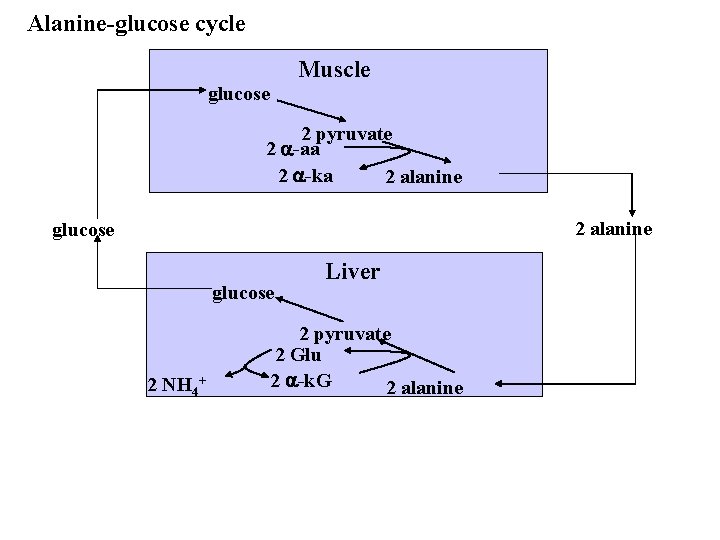
Alanine-glucose cycle Muscle glucose 2 pyruvate 2 a-aa 2 a-ka 2 alanine glucose 2 NH 4+ Liver 2 pyruvate 2 Glu 2 a-k. G 2 alanine
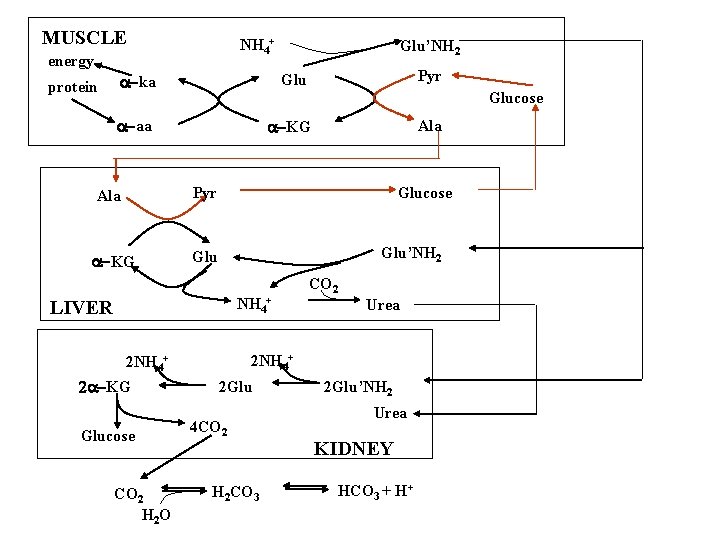
MUSCLE NH 4+ energy protein Glu’NH 2 Pyr a-ka Glu a-aa a-KG Glucose Ala Pyr Ala a-KG Glucose Glu’NH 2 Glu NH 4+ LIVER Glucose CO 2 H 2 O Urea 2 NH 4+ 2 a-KG CO 2 2 Glu 4 CO 2 H 2 CO 3 2 Glu’NH 2 Urea KIDNEY HCO 3 + H+

Urea cycle Where: Liver: mito/cyto Why: disposal of N Immediate source of N: glutamate dehydrogenase glutaminase Fate of urea: liver kidney How much: ~ 30 g urea / day urine
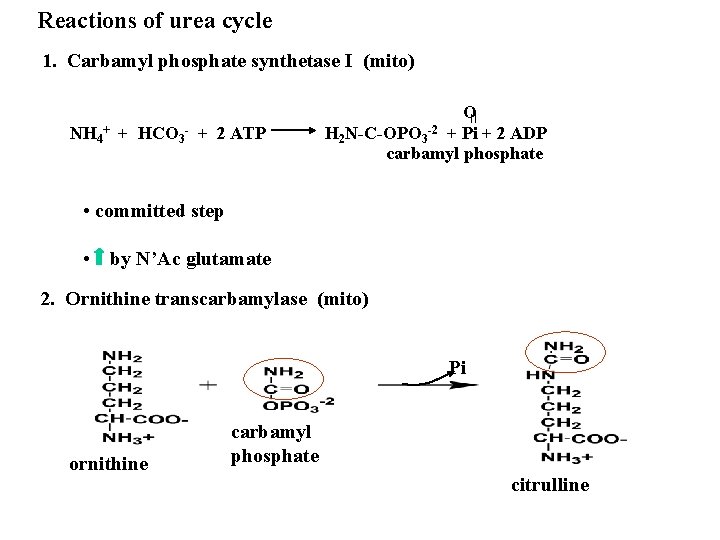
Reactions of urea cycle 1. Carbamyl phosphate synthetase I (mito) NH 4+ + HCO 3 - + 2 ATP O H 2 N-C-OPO 3 -2 + Pi + 2 ADP carbamyl phosphate • committed step • by N’Ac glutamate 2. Ornithine transcarbamylase (mito) Pi ornithine carbamyl phosphate citrulline
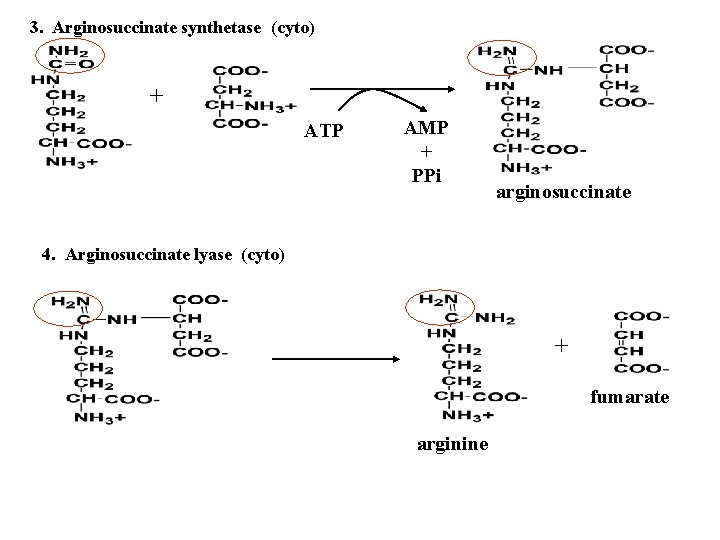
3. Arginosuccinate synthetase (cyto) + ATP AMP + PPi arginosuccinate 4. Arginosuccinate lyase (cyto) + fumarate arginine
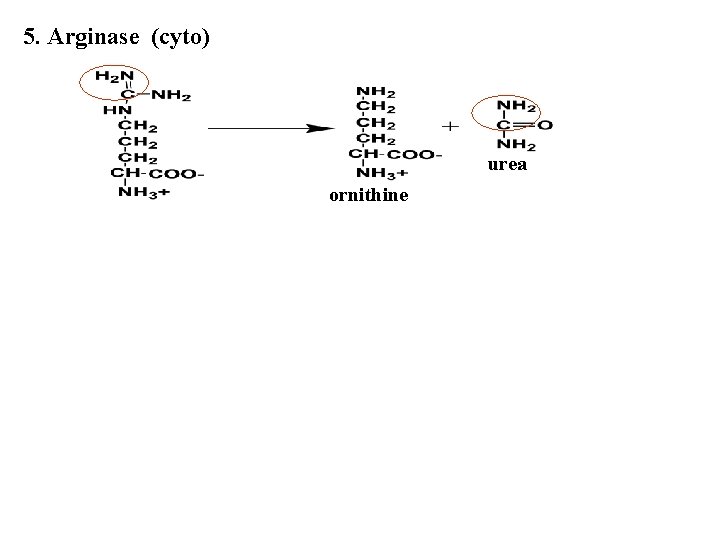
5. Arginase (cyto) urea ornithine
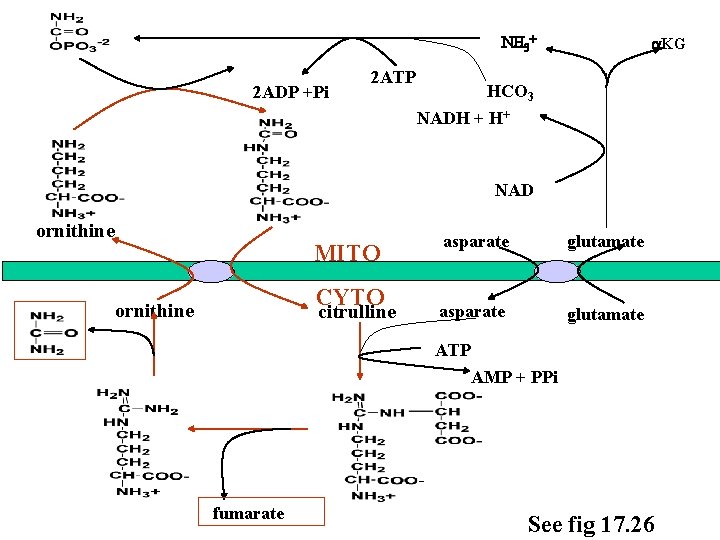
NH 3+ 2 ADP +Pi 2 ATP a. KG HCO 3 NADH + H+ NAD ornithine MITO CYTO ornithine citrulline asparate glutamate ATP AMP + PPi fumarate See fig 17. 26
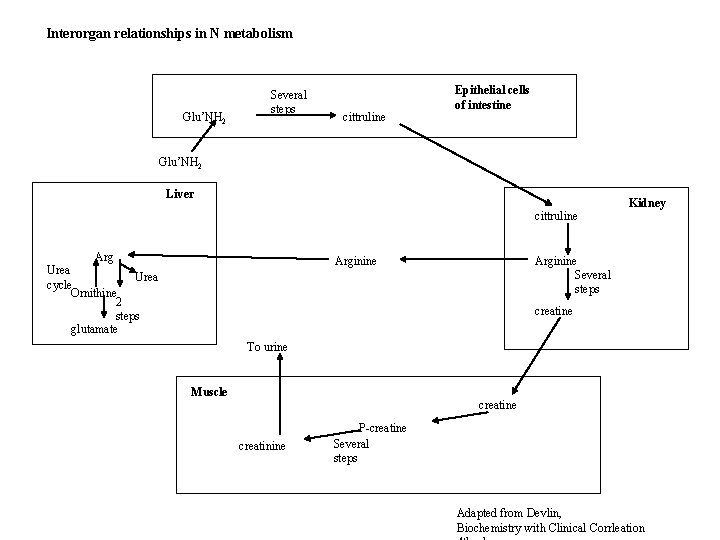
Interorgan relationships in N metabolism Glu’NH 2 Several steps cittruline Epithelial cells of intestine Glu’NH 2 Liver Kidney cittruline Arg Urea cycle Ornithine 2 steps glutamate Arginine Several steps creatine To urine Muscle creatinine P-creatine Several steps Adapted from Devlin, Biochemistry with Clinical Corrleation
- Slides: 22