Amino Acid Metabolism Metabolism of the 20 common

Amino Acid Metabolism • Metabolism of the 20 common amino acids is considered from the origins and fates of their: (1)Nitrogen atoms (2)Carbon skeletons • For mammals: Essential amino acids must be obtained from diet Nonessential amino acids - can be synthesized

v. Protein catabolism is the breakdown of proteins into amino acids and simple derivative compounds, for transport into the cell through the plasma membrane and ultimately for the polymerization into new proteins via the use of ribonucleic acids (RNA) and ribosomes. v. Protein catabolism, which is the breakdown of macromolecules, is essentially a digestion process. Protein catabolism is most commonly carried out by non-specific endo- and exo-proteases. v. However, specific proteases are used for cleaving of proteins for regulatory and protein trafficking purposes. v. One example is called oligopeptidase. the subclass of proteolytic enzymes

Amino Acid Catabolism • Amino acids from degraded proteins or from diet can be used for the biosynthesis of new proteins • During starvation proteins are degraded to amino acids to support glucose formation • First step is often removal of the α-amino group • Carbon chains are altered for entry into central pathways of carbon metabolism
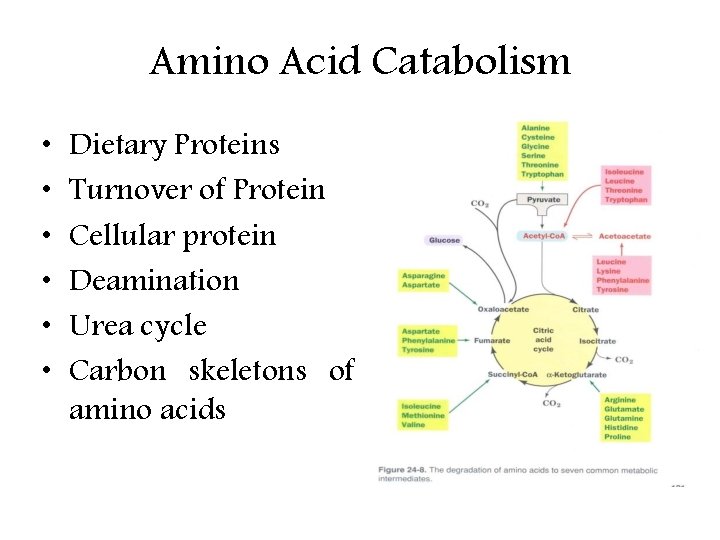
Amino Acid Catabolism • • • Dietary Proteins Turnover of Protein Cellular protein Deamination Urea cycle Carbon skeletons of amino acids

Amino acid catabolism in humans Proteins are broken down in stomach and small intestine to constituent amino acids. Amino acids are either used as building blocks or burned for energy (~10% of our energy needs). Catabolism of amino acids increases for use in gluconeogenesis when glucose is unavailable (e. g. , starvation/diabetes) when protein content of diet exceeds need for building blocks during times of stress

Transamination, a chemical reaction that transfers an amino group to a ketoacid to form new amino acids. This pathway is responsible for the deamination of most amino acids. This is one of the major degradation pathways which convert essential amino acids to nonessential amino acids

v. Transamination in biochemistry is accomplished by enzymes called transaminases or aminotransferases. vα-ketoglutarate acts as the predominant amino-group acceptor and produces glutamate as the new amino acid. v. Aminoacid + α-ketoglutarate ↔ α-keto acid + Glutamate v. Glutamate's amino group, in turn, is transferred to oxaloacetate in a second transamination reaction yielding aspartate. v. Glutamate + oxaloacetate ↔ α-ketoglutarate + aspartate
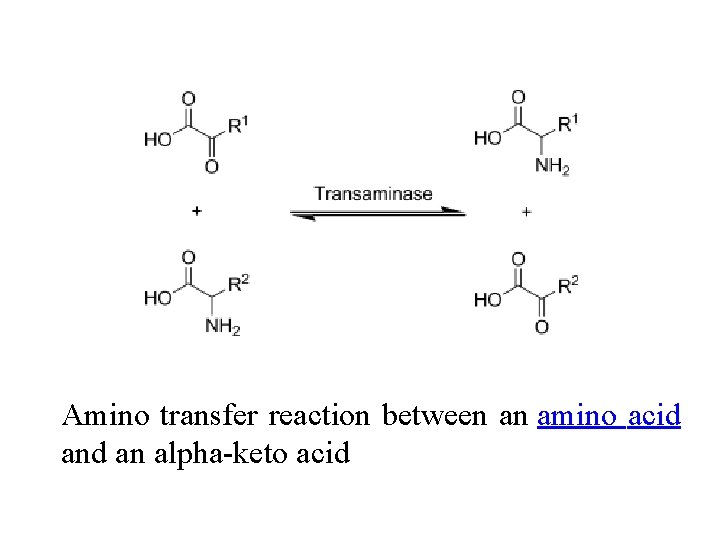
Amino transfer reaction between an amino acid an alpha-keto acid

v. Oxidative deamination is a form of deamination that generates α -keto acids and other oxidized products from amine-containing compounds, and occurs only in the liver. v Oxidative deamination is an important step in the catabolism of amino acids, generating a more metabolizable form of the amino acid, and also generating ammonia as a toxic by-product. v The ammonia generated in this process can then be neutralized into urea via the urea cycle.

What is the Difference Transamination and Deamination Between The main difference between transamination and deamination is that in transamination, the amine group of an amino acid is exchanged with a keto group of another compound whereas, in deamination, an amino acid loses its amine group.

v. Furthermore, in transamination, the amino acid becomes a keto acid while a deamination reaction produced ammonia. v. Moreover, transamination is a biochemical reaction between two molecules responsible for the synthesis of nonessential amino acids. It can occur in all types of cells of the body. In contrast, deamination is responsible for the break down of excess protein in the liver.

v. Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide(CO 2). v. Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain. The reverse process, which is the first chemical step in photosynthesis, is called carboxylation, the addition of CO 2 to a compound. v. Enzymes that catalyze decarboxylations are called decarboxylases or, the more formal term, carboxylyases
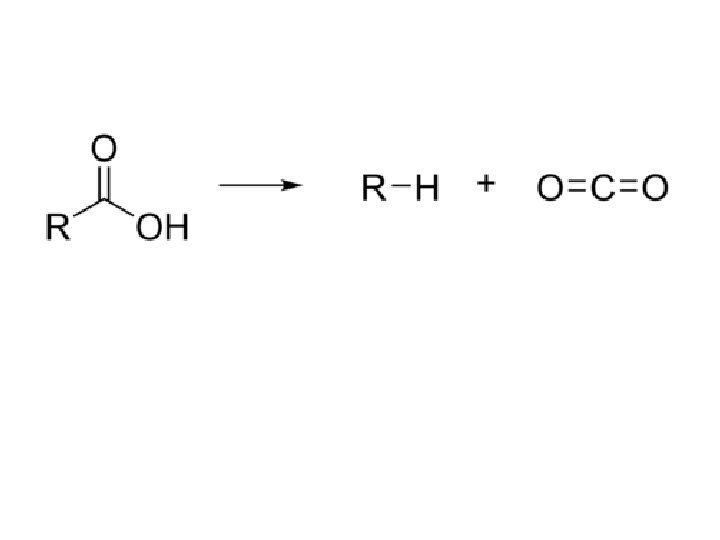

v. Decarboxylations are pervasive in biology. They are often classified according to the cofactors that catalyze the transformations. v. Biotin-coupled processes effect the decarboxylation of malonyl. Co. A to acetyl-Co. A. v. Thiamine (T: ) is the active component for decarboxylation of alphaketoacids, including pyruvate: v. T: + RC(O)CO 2 H → T=C(OH)R + CO 2 v. T=C(OH)R + R'CHO → T: + RC(O)CH(OH)R'

Common biosynthetic oxidative üdecarboxylations of amino acids to amines are: ütryptophan to tryptamine üphenylalanine to phenylethylamine ütyrosine to tyramine ühistidine to histamine üserine to ethanolamine üglutamic acid to GABA ülysine to cadaverine üarginine to agmatine üornithine to putrescine ü 5 -HTP to serotonin üL-DOPA to dopamine
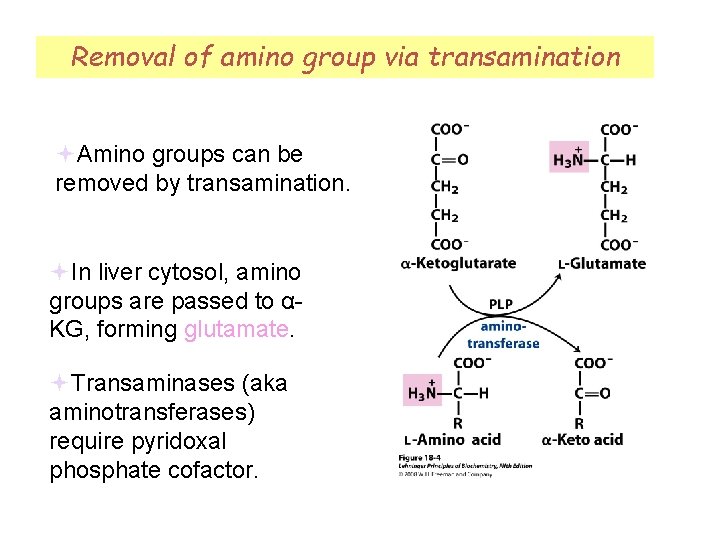
Removal of amino group via transamination Amino groups can be removed by transamination. In liver cytosol, amino groups are passed to αKG, forming glutamate. Transaminases (aka aminotransferases) require pyridoxal phosphate cofactor.
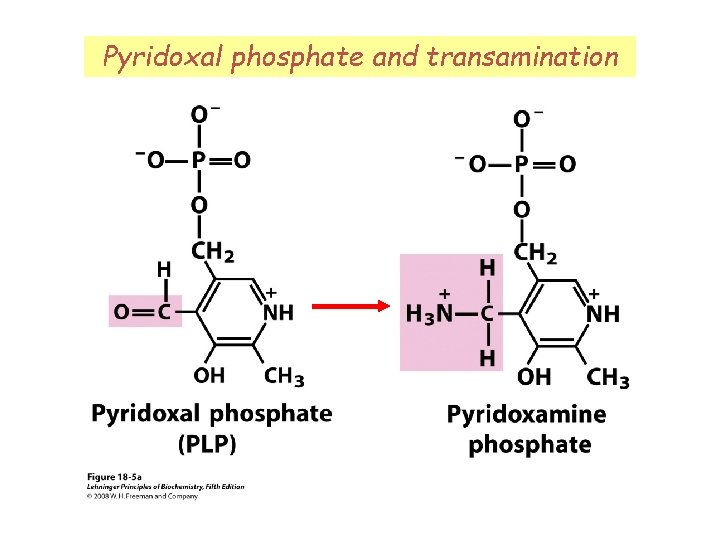
Pyridoxal phosphate and transamination
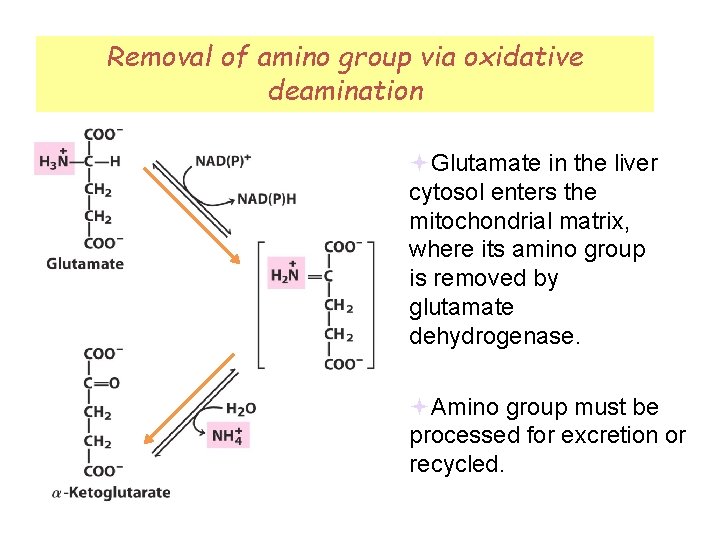
Removal of amino group via oxidative deamination Fig 18 -1 Glutamate in the liver cytosol enters the mitochondrial matrix, where its amino group is removed by glutamate dehydrogenase. Amino group must be processed for excretion or recycled.
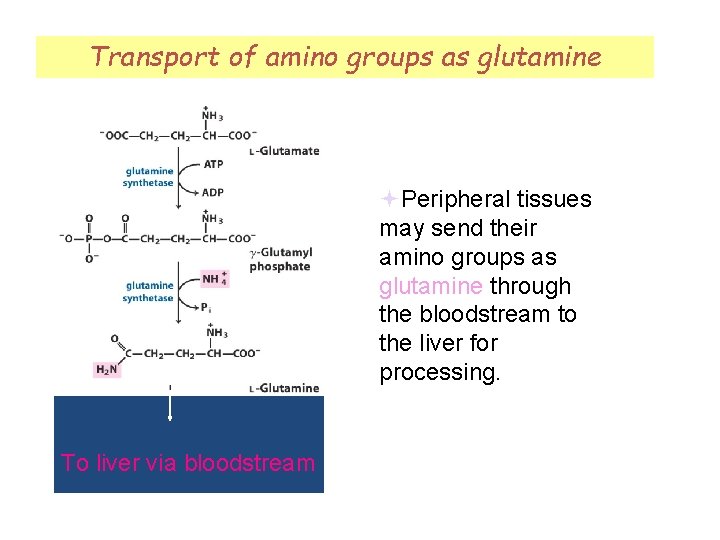
Transport of amino groups as glutamine Peripheral tissues may send their amino groups as glutamine through the bloodstream to the liver for processing. To liver via bloodstream
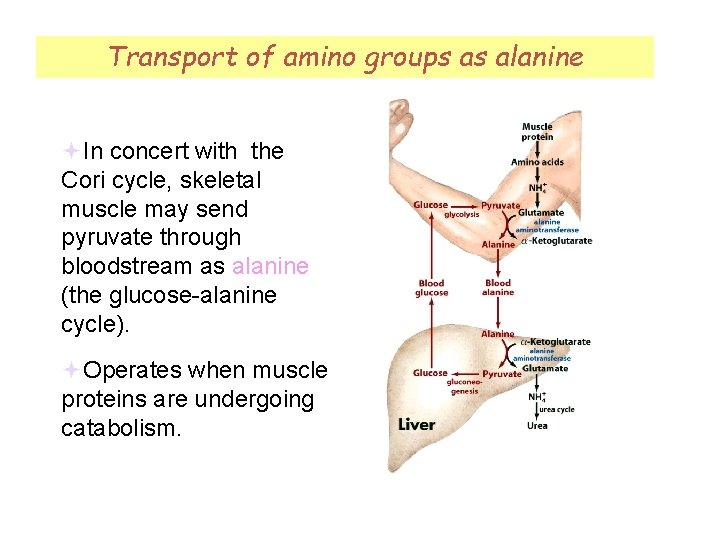
Transport of amino groups as alanine Fig 18 -1 In concert with the Cori cycle, skeletal muscle may send pyruvate through bloodstream as alanine (the glucose-alanine cycle). Operates when muscle proteins are undergoing catabolism.
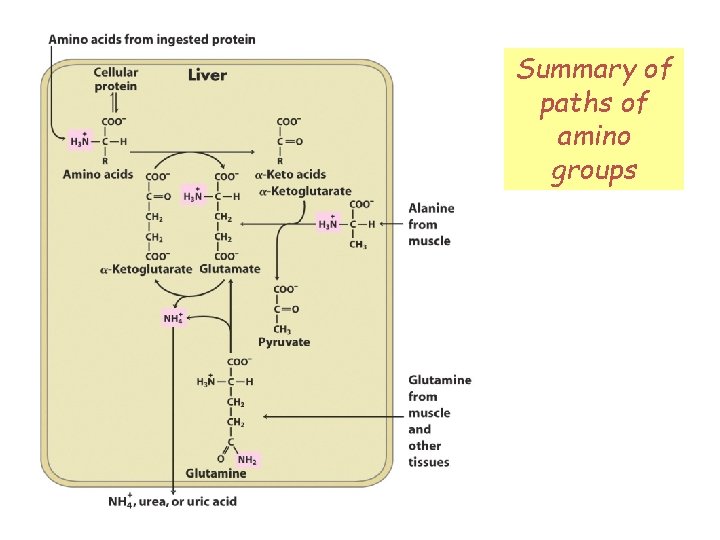
Summary of paths of Fig 18 -1 amino groups

Fate of + NH Fig 18 -1 4 excreted as NH 3 (ammonotelic)

Fate of NH 4+ excreted as uric acid (uricotelic)
Fate of NH 4+
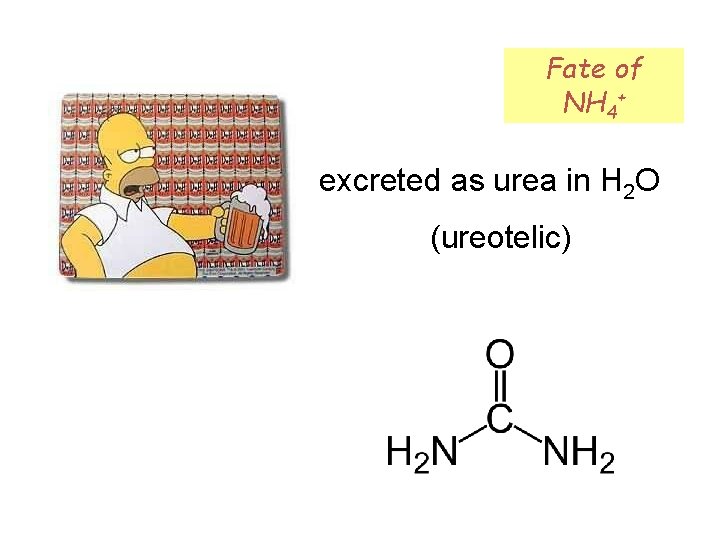
Fate of NH 4+ excreted as urea in H 2 O (ureotelic)
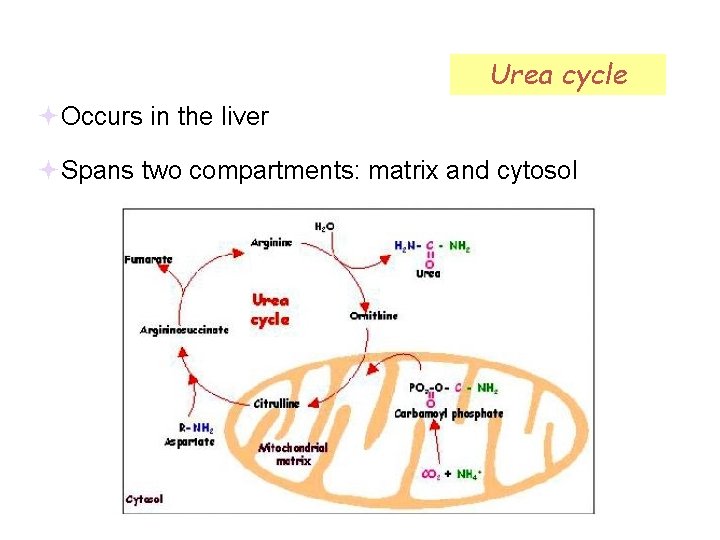
Urea cycle Occurs in the liver Spans two compartments: matrix and cytosol
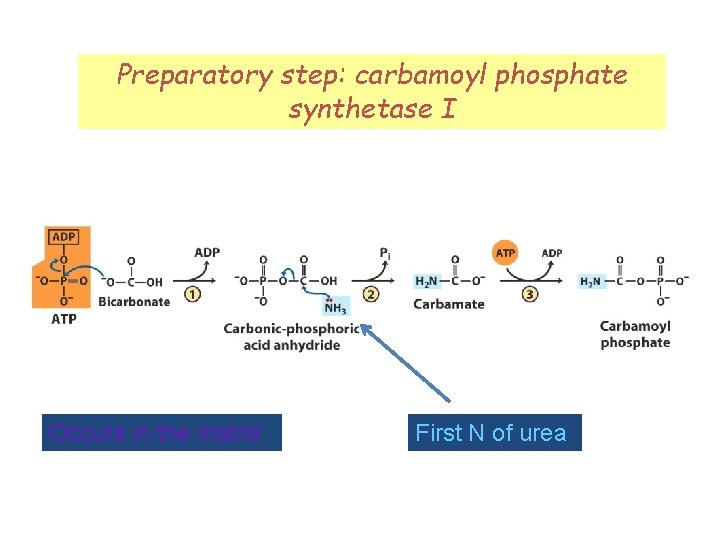
Preparatory step: carbamoyl phosphate synthetase I Occurs in the matrix First N of urea

Step 1: Ornithine transcarbamoylase Ornithine is analogous to OA Also occurs in the matrix, but citrulline is transported to cytosol
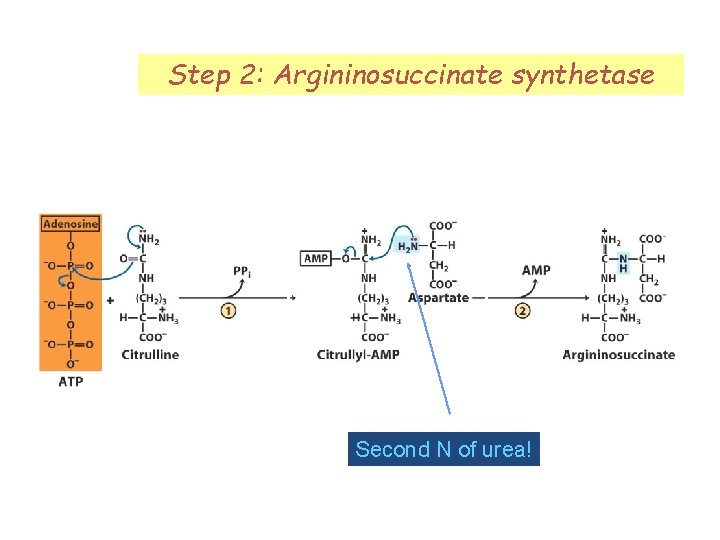
Step 2: Argininosuccinate synthetase Second N of urea!
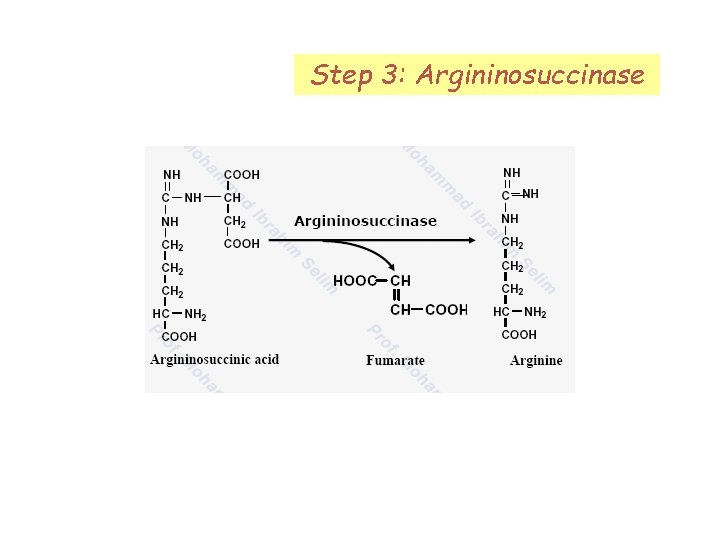
Step 3: Argininosuccinase
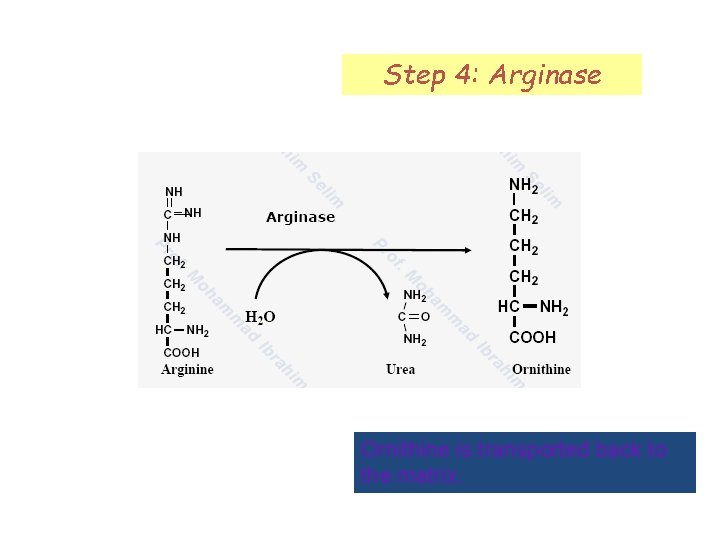
Step 4: Arginase Ornithine is transported back to the matrix.
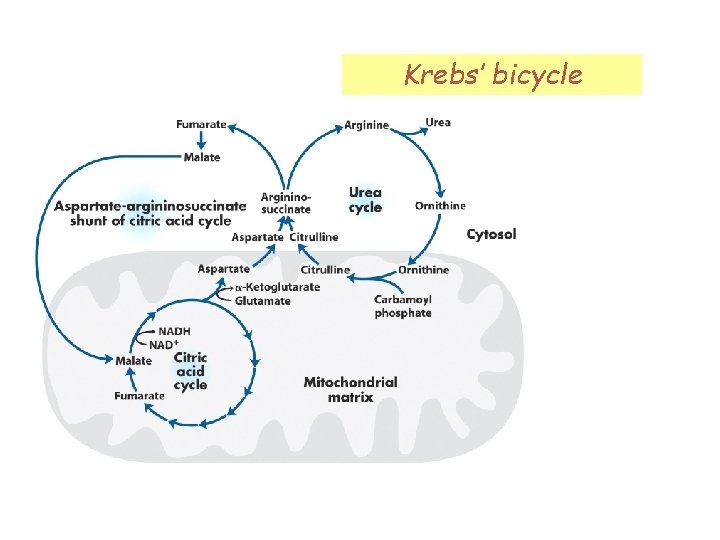
Krebs’ bicycle
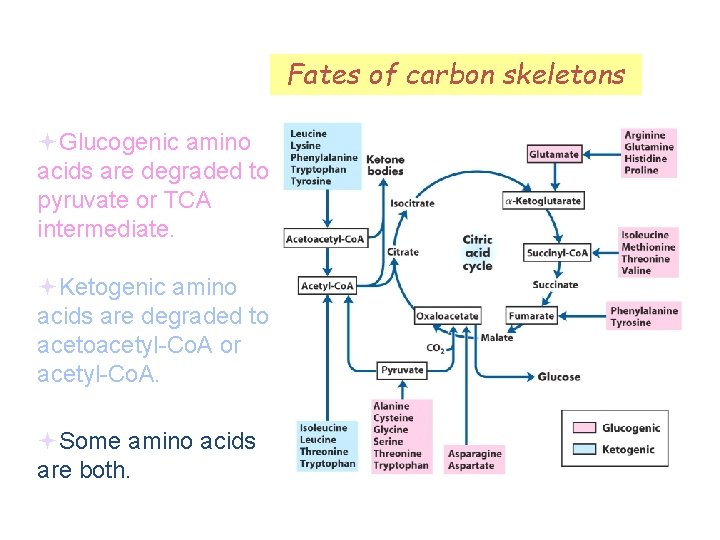
Fates of carbon skeletons Glucogenic amino acids are degraded to pyruvate or TCA intermediate. Ketogenic amino acids are degraded to acetoacetyl-Co. A or acetyl-Co. A. Some amino acids are both.

Thank you
- Slides: 35