Amino acid metabolism III Brake down of amino

Amino acid metabolism III. Brake down of amino acids, glucoplastic and ketoplastic amino acids Figures: Lehninger-4 ed; chapter: 18 Stryer-5 ed; chapter: 23
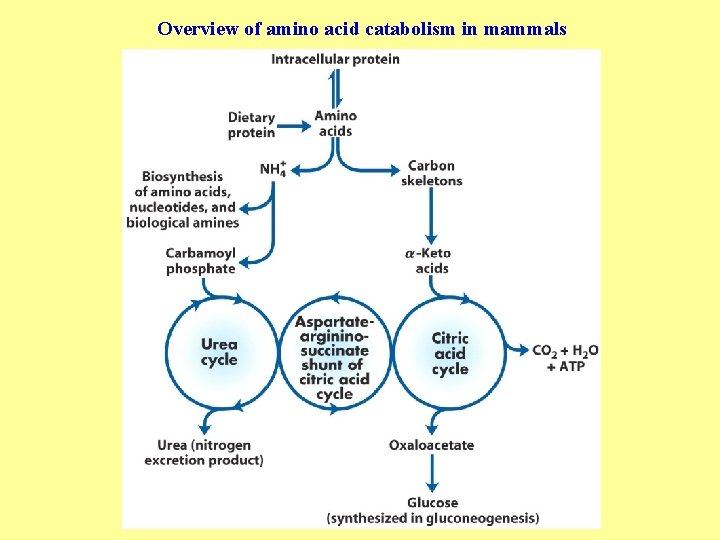
Overview of amino acid catabolism in mammals
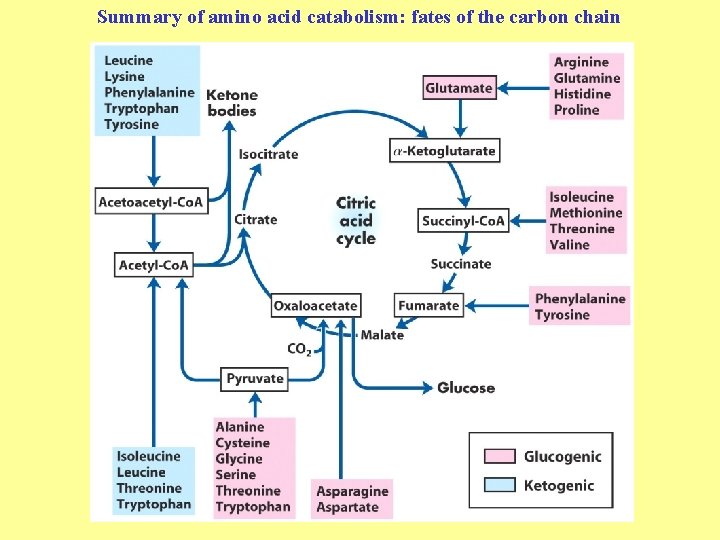
Summary of amino acid catabolism: fates of the carbon chain

Purely ketogenic amino acids: can yield ketone bodies in the liver • leucine (Leu) very common in proteins • lysine (Lys) Glucogenic amino acids: can be converted to glucose and glycogen • alanine (Ala) • cysteine (Cys) • glycine (Gly) • serine (Ser) • asparagine (Asn) • aspartate (Asp) • methionine (Met) • valine (Val) • arginine (Arg) • glutamine (Gln) • glutamate (Glu) • histidine (His) • proline (Pro)

Mixed amino acids (both ketogenic and glucogenic): • tryptophan (Trp) • phenylalanine (Phe) • tyrosine (Tyr) • threonine (Thr) • isoleucine (Ile)
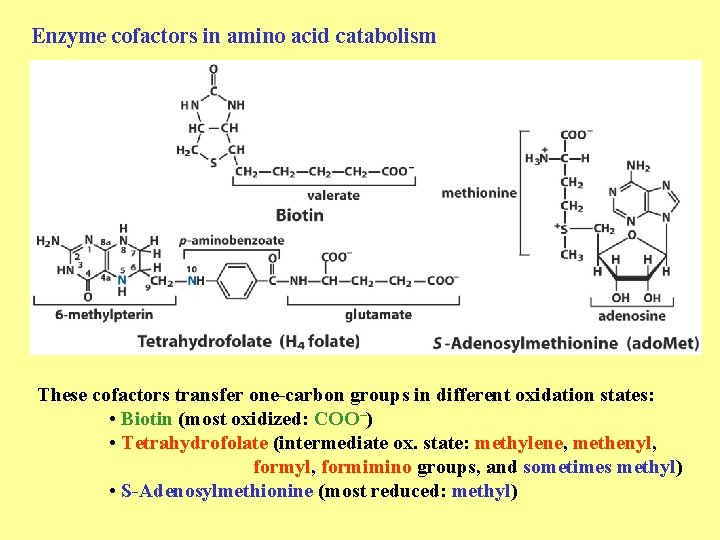
Enzyme cofactors in amino acid catabolism These cofactors transfer one-carbon groups in different oxidation states: • Biotin (most oxidized: COO–) • Tetrahydrofolate (intermediate ox. state: methylene, methenyl, formimino groups, and sometimes methyl) • S-Adenosylmethionine (most reduced: methyl)
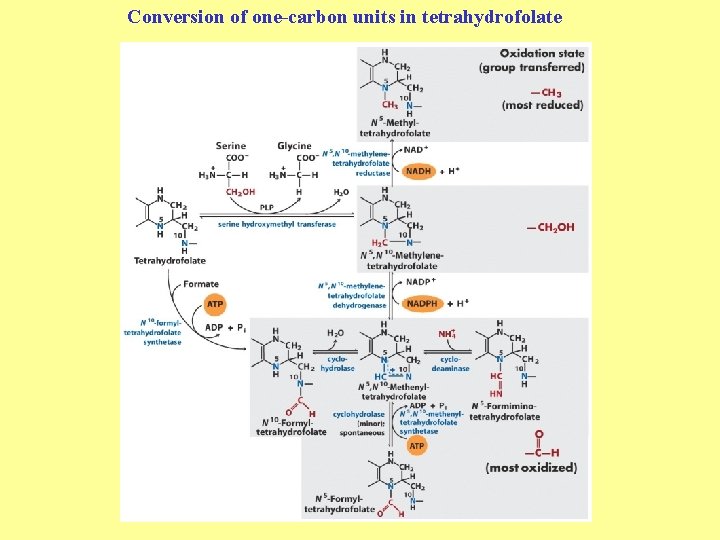
Conversion of one-carbon units in tetrahydrofolate
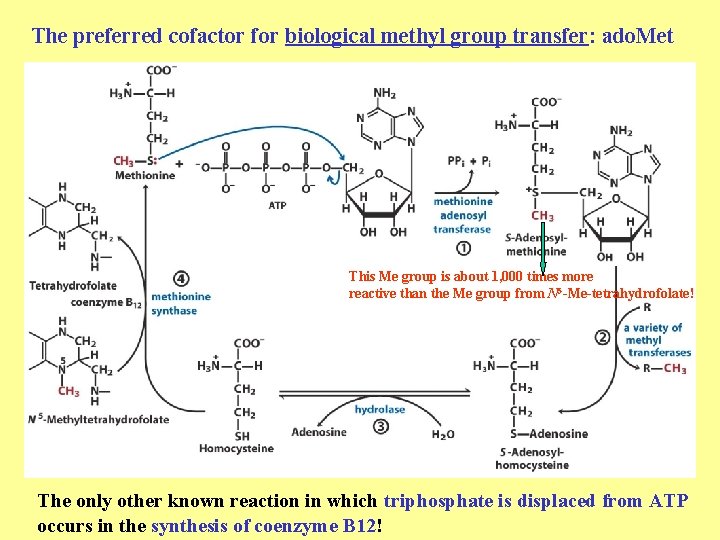
The preferred cofactor for biological methyl group transfer: ado. Met This Me group is about 1, 000 times more reactive than the Me group from N 5 -Me-tetrahydrofolate! The only other known reaction in which triphosphate is displaced from ATP occurs in the synthesis of coenzyme B 12!
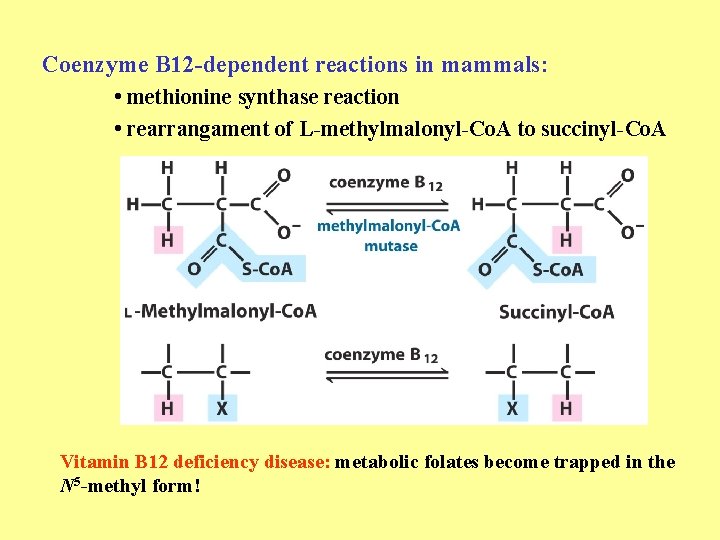
Coenzyme B 12 -dependent reactions in mammals: • methionine synthase reaction • rearrangament of L-methylmalonyl-Co. A to succinyl-Co. A Vitamin B 12 deficiency disease: metabolic folates become trapped in the N 5 -methyl form!
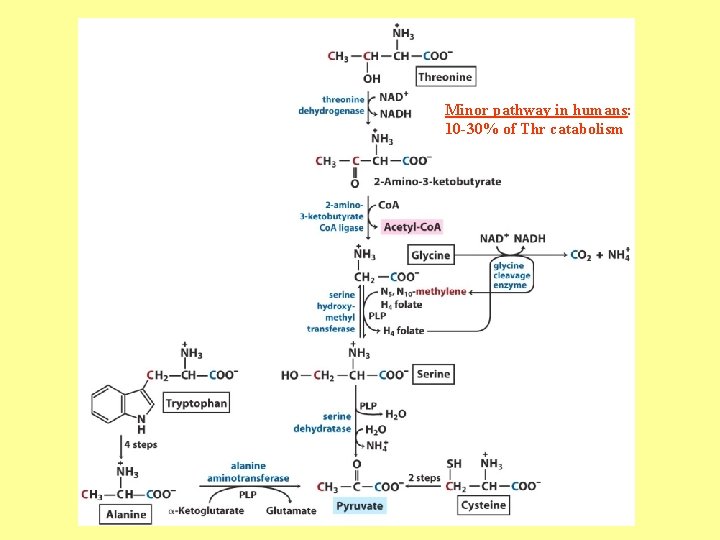
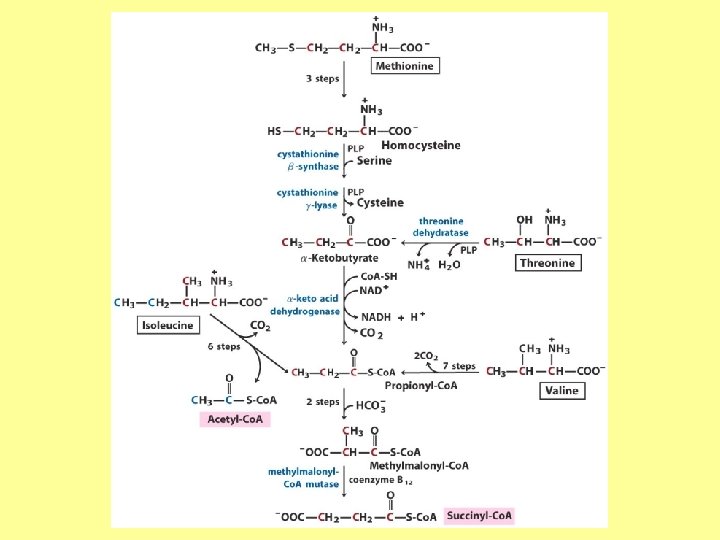
Minor pathway in humans: 10 -30% of Thr catabolism
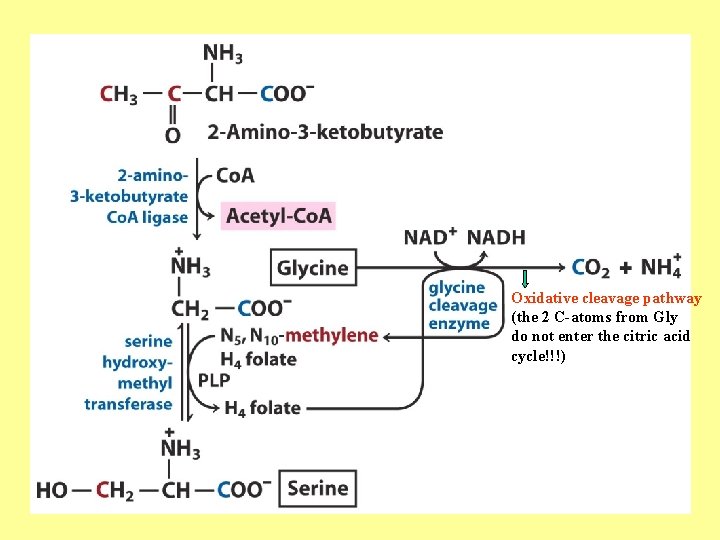
Oxidative cleavage pathway (the 2 C-atoms from Gly do not enter the citric acid cycle!!!)
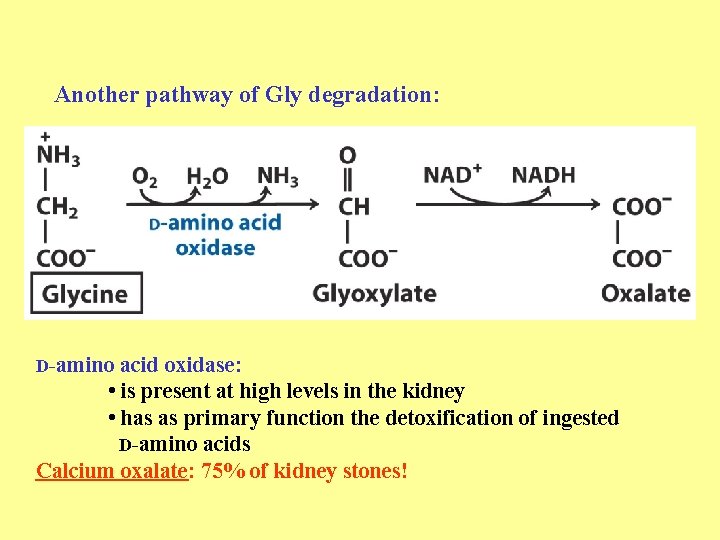
Another pathway of Gly degradation: D-amino acid oxidase: • is present at high levels in the kidney • has as primary function the detoxification of ingested D-amino acids Calcium oxalate: 75% of kidney stones!
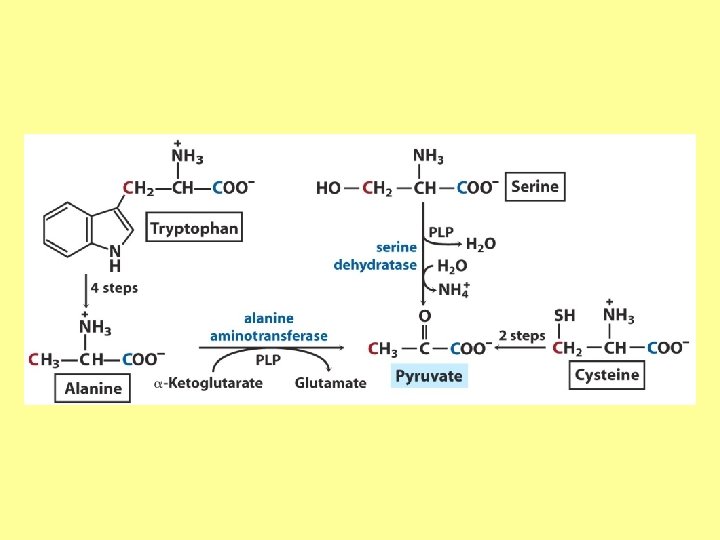
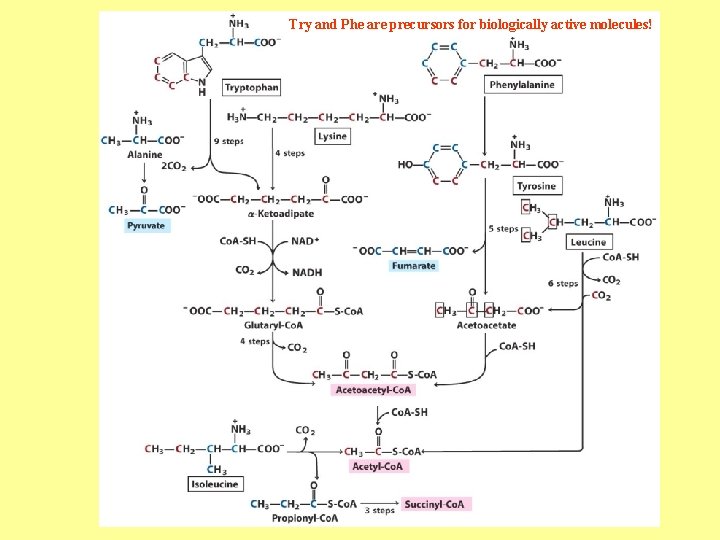
Try and Phe are precursors for biologically active molecules!
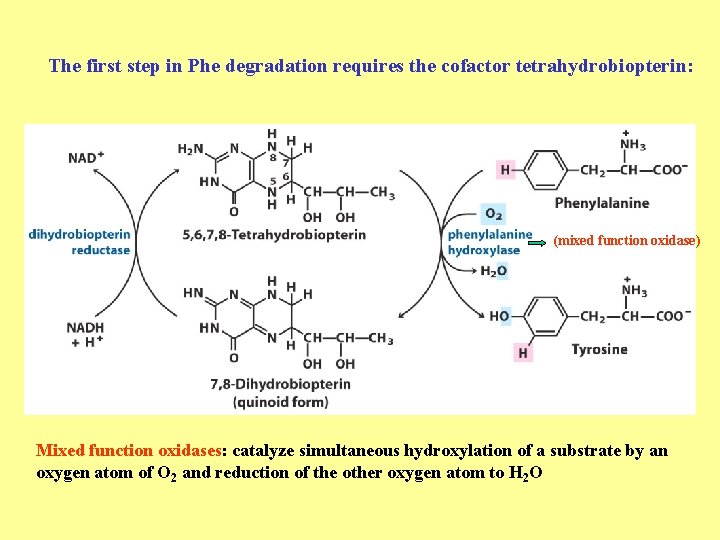
The first step in Phe degradation requires the cofactor tetrahydrobiopterin: (mixed function oxidase) Mixed function oxidases: catalyze simultaneous hydroxylation of a substrate by an oxygen atom of O 2 and reduction of the other oxygen atom to H 2 O
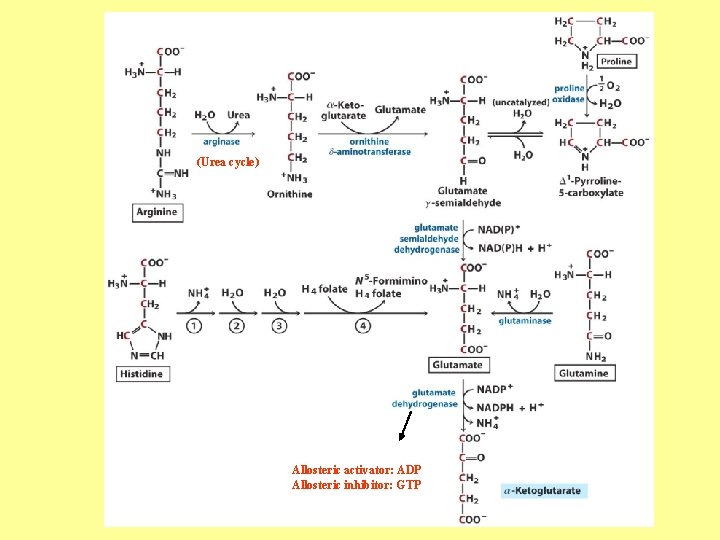
(Urea cycle) Allosteric activator: ADP Allosteric inhibitor: GTP
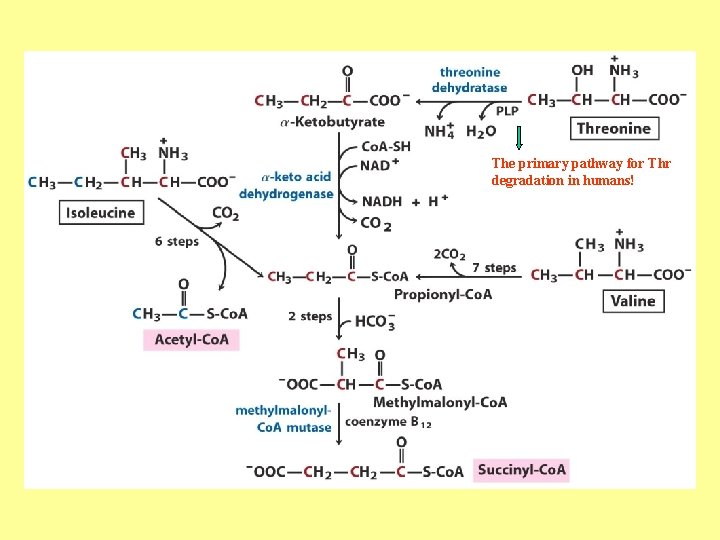
The primary pathway for Thr degradation in humans!
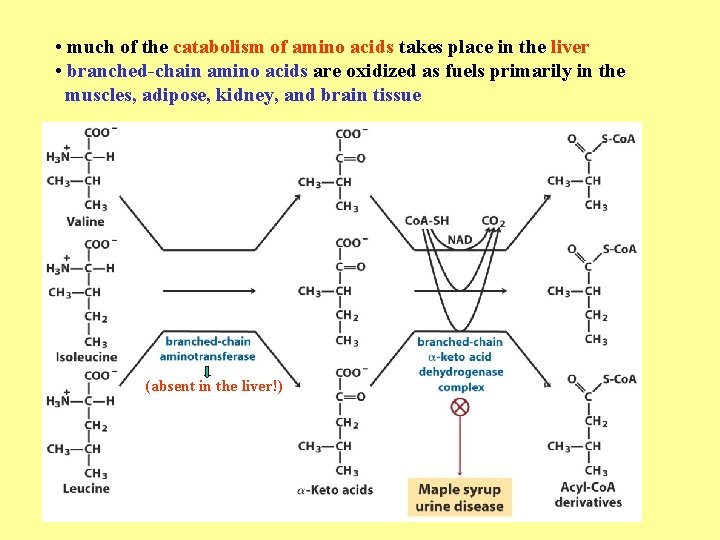
• much of the catabolism of amino acids takes place in the liver • branched-chain amino acids are oxidized as fuels primarily in the muscles, adipose, kidney, and brain tissue (absent in the liver!)

• branched-chain -keto acid dehydrogenase complex • pyruvate dehydrogenase complex • -ketoglutarate dehydrogenase complex similar structure, same reaction mechanism catalyze homologous reactions five cofactors: thiamine pyrophosphate FAD NAD lipoate coenzyme A inactive enzyme complex = phosphorylated form! (when the dietary intake of branched-chain amino acids is low)
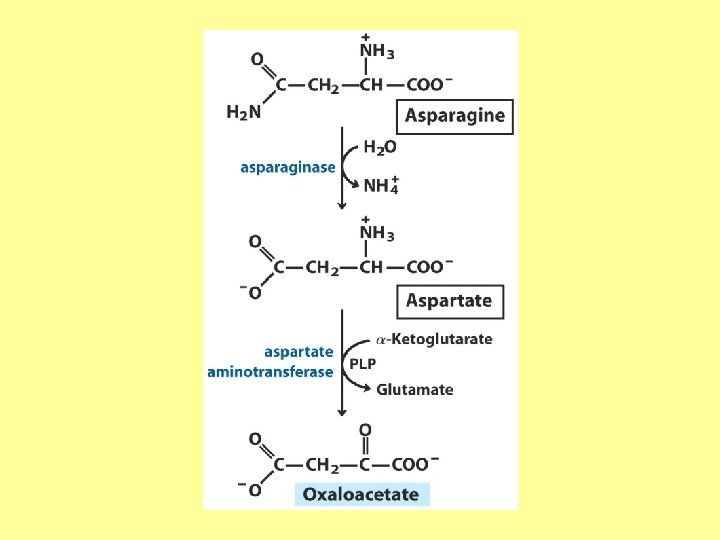
- Slides: 22