Amines Draw parahydroxyNmethylbenzoamide Amines Organic derivatives of ammonia

Amines

Draw: para-hydroxy-Nmethylbenzoamide

Amines • Organic derivatives of ammonia

Ammonia H H-N: H

Ammonium H H-N-H H +1
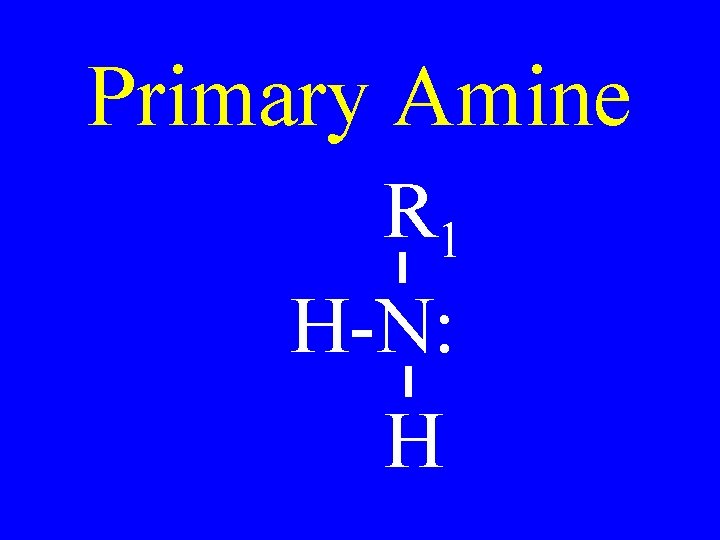
Primary Amine R 1 H-N: H
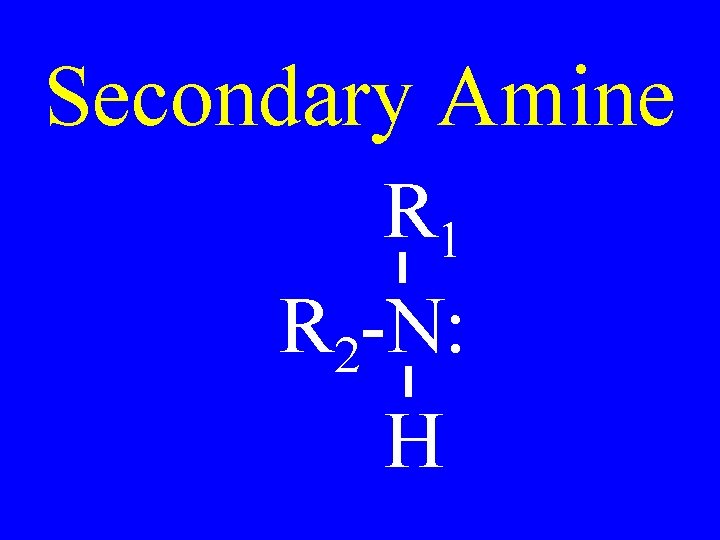
Secondary Amine R 1 R 2 -N: H

Tertiary Amine R 1 R 2 -N: R 3

Quaternary Amine R 1 R 2 -N-R 4 R 3 +1

Naming Amines
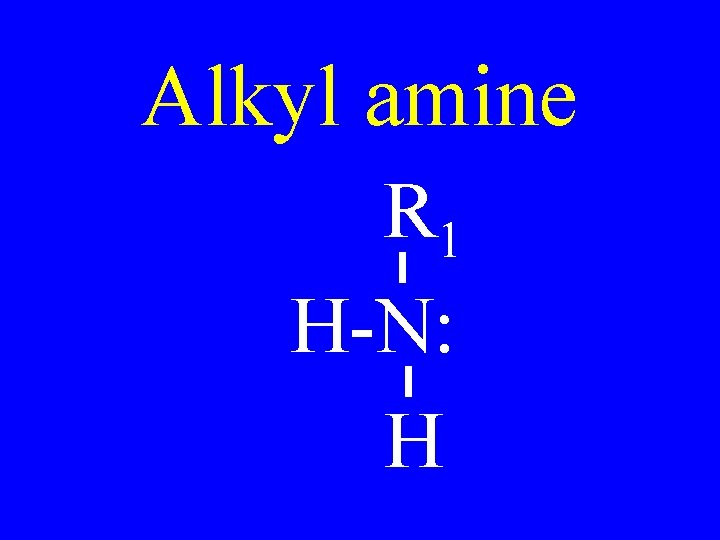
Alkyl amine R 1 H-N: H
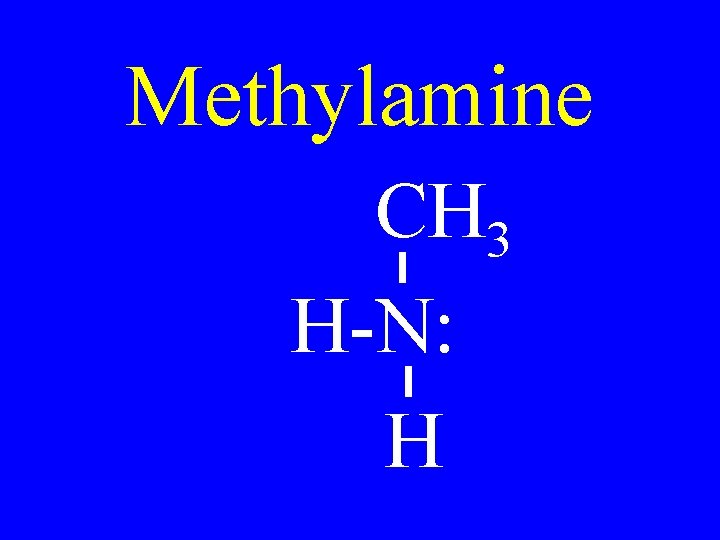
Methylamine CH 3 H-N: H
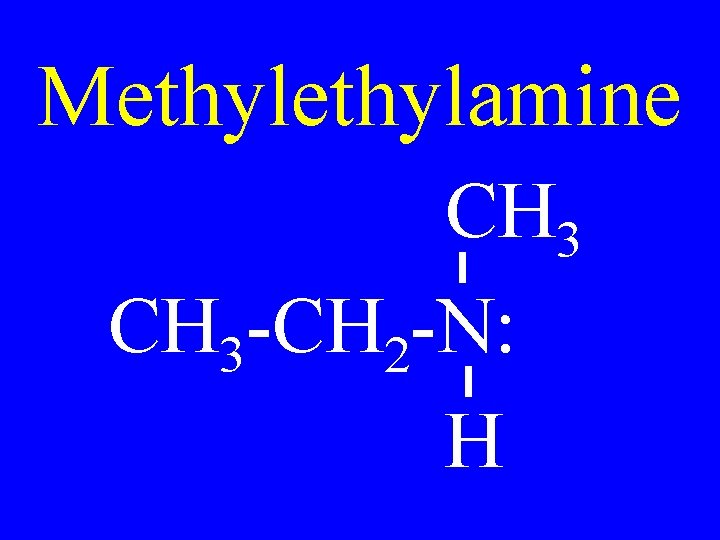
Methylamine CH 3 -CH 2 -N: H
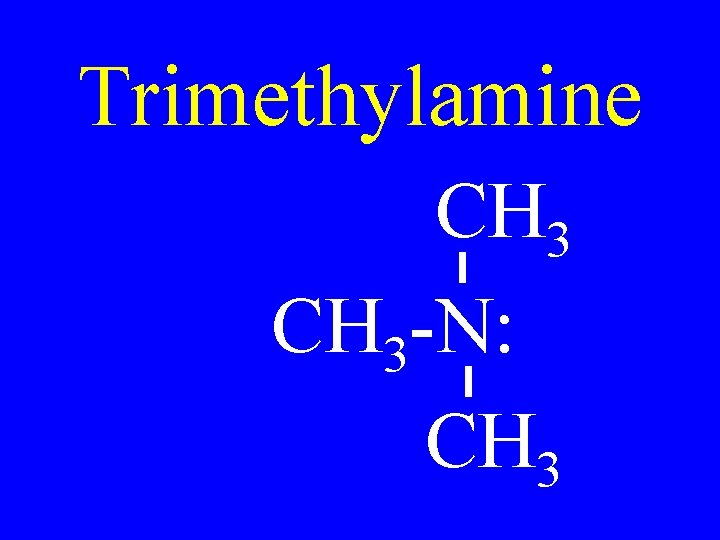
Trimethylamine CH 3 -N: CH 3
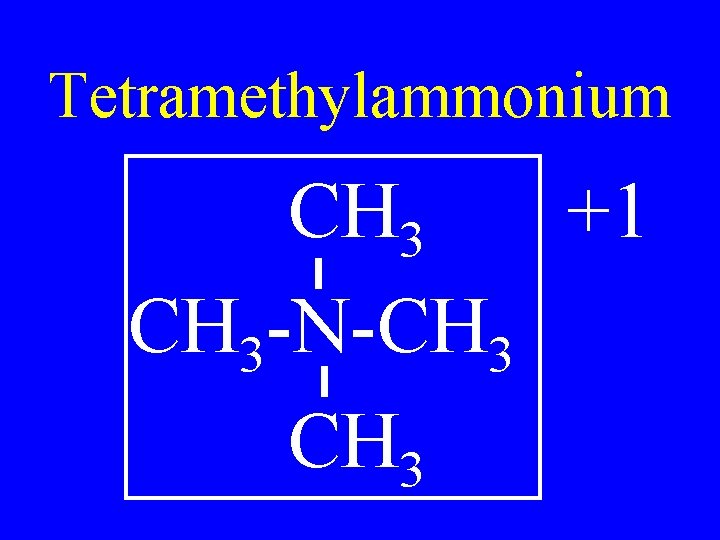
Tetramethylammonium CH 3 +1 CH 3 -N-CH 3
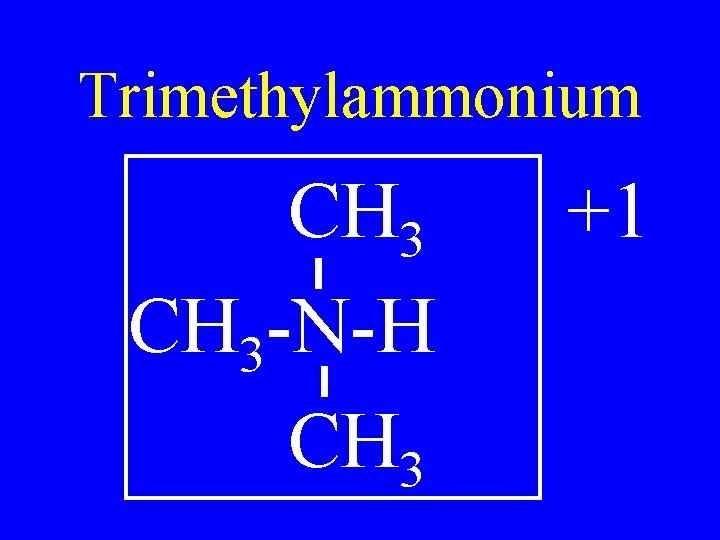
Trimethylammonium CH 3 -N-H CH 3 +1

Common Amines

Phenylamine or Aminobenzene NH 2 Aniline
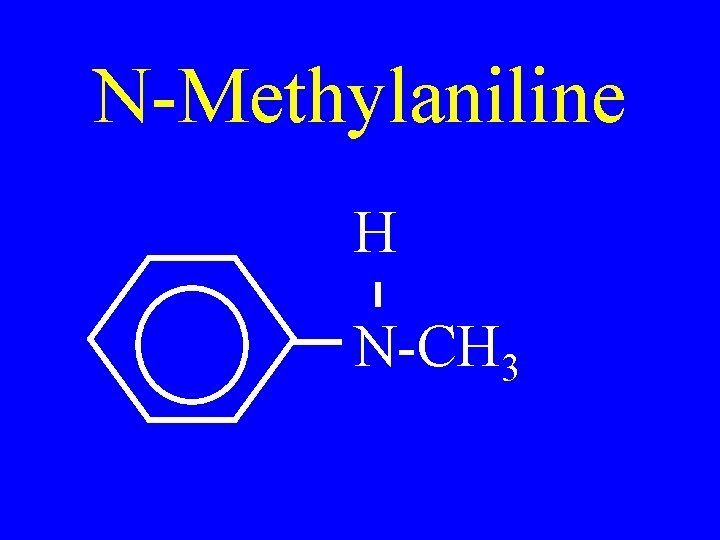
N-Methylaniline H N-CH 3
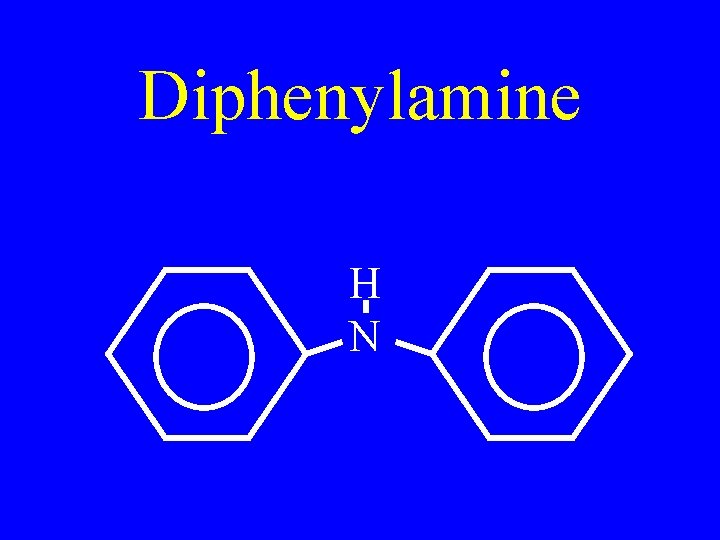
Diphenylamine H N
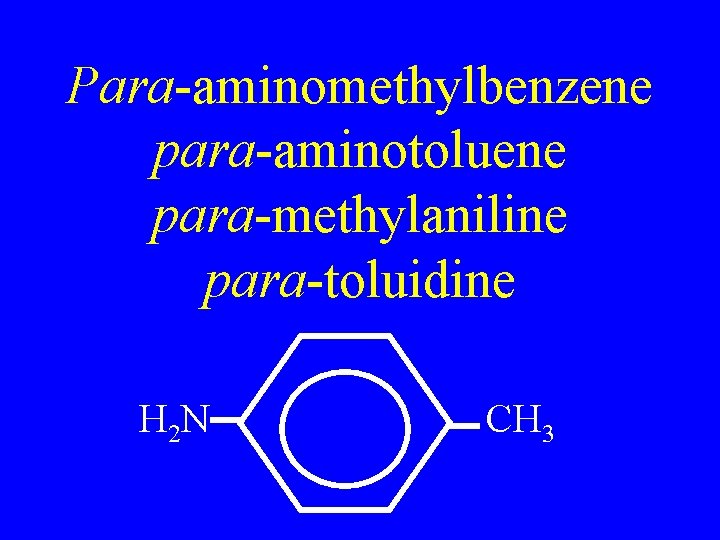
Para-aminomethylbenzene para-aminotoluene para-methylaniline para-toluidine H 2 N CH 3
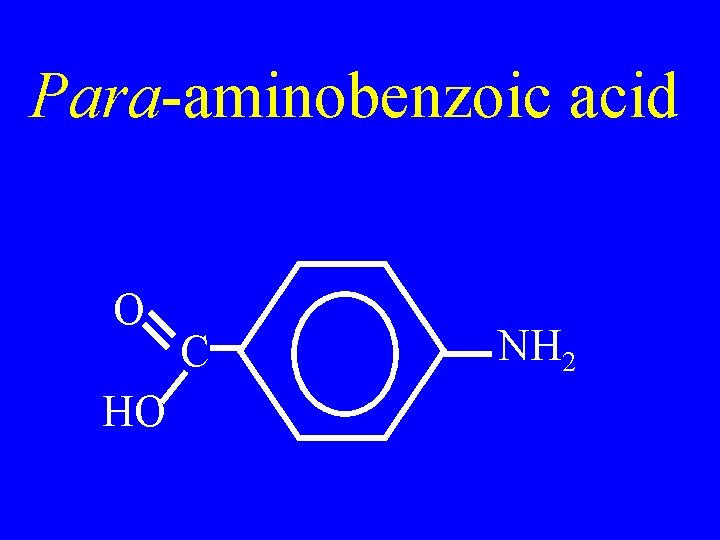
Para-aminobenzoic acid O HO C NH 2
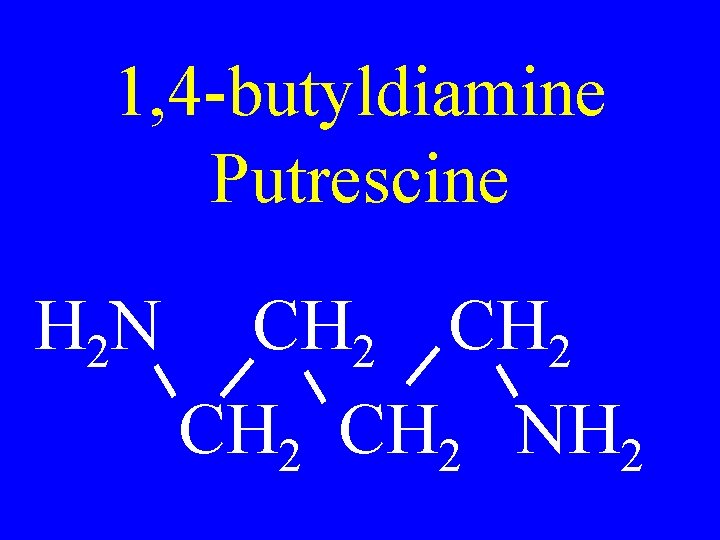
1, 4 -butyldiamine Putrescine H 2 N CH 2 NH 2
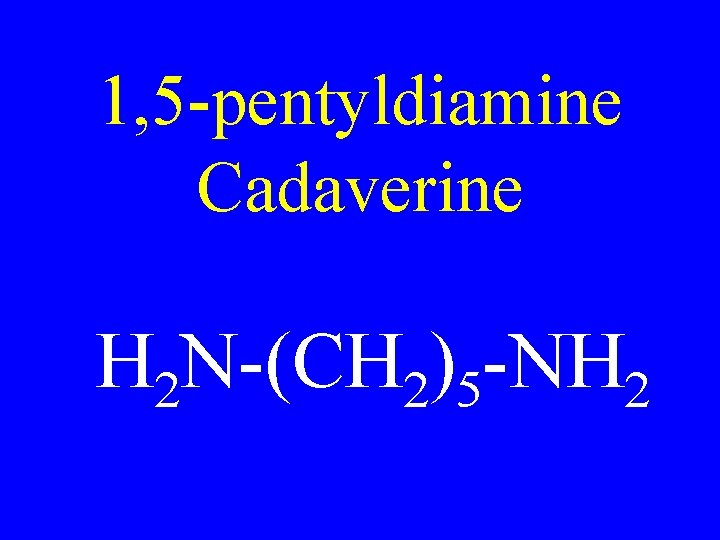
1, 5 -pentyldiamine Cadaverine H 2 N-(CH 2)5 -NH 2

Draw: NCyanoformamide

Heterocyclic Amines
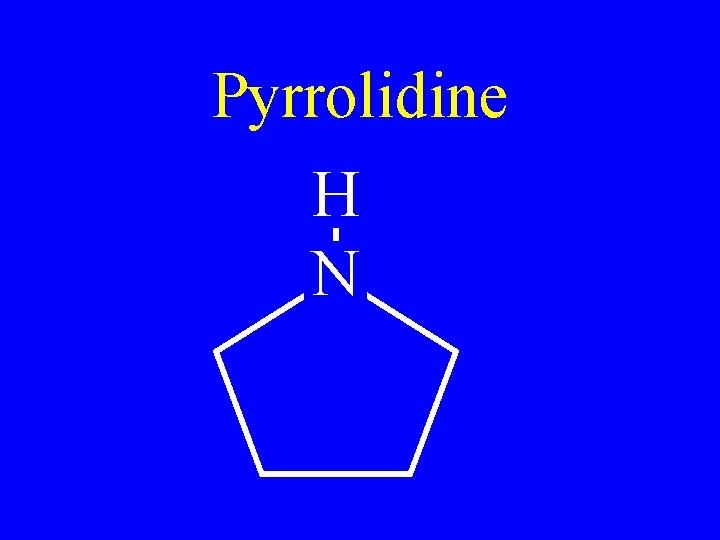
Pyrrolidine H N
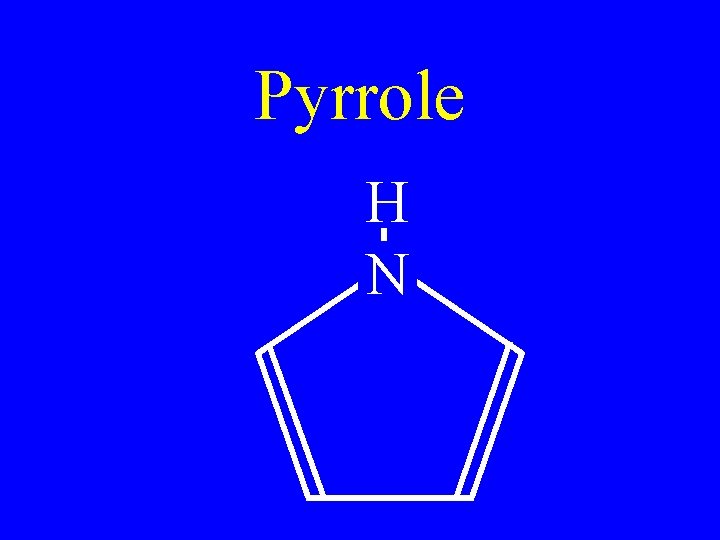
Pyrrole H N
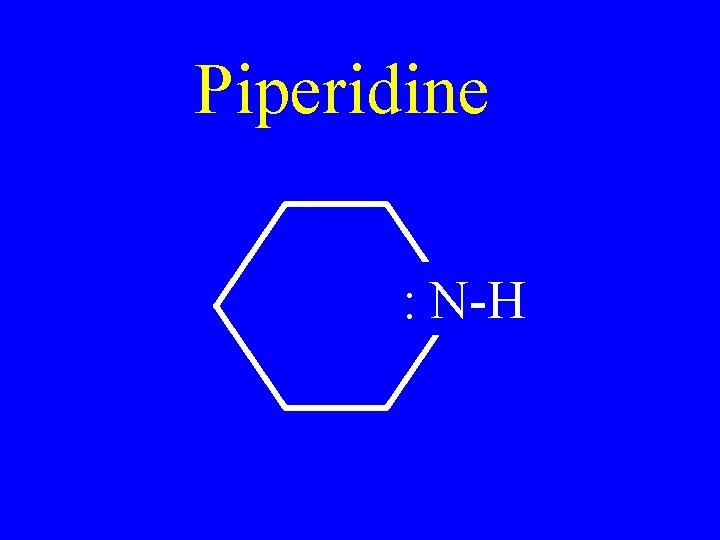
Piperidine : N-H
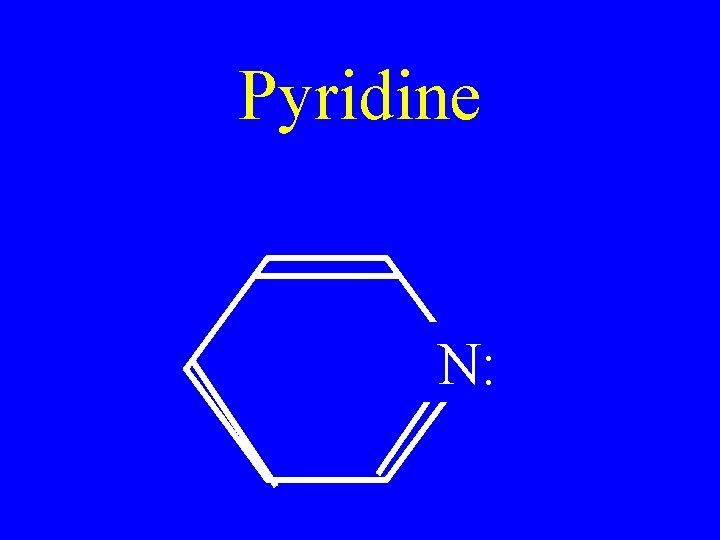
Pyridine N:
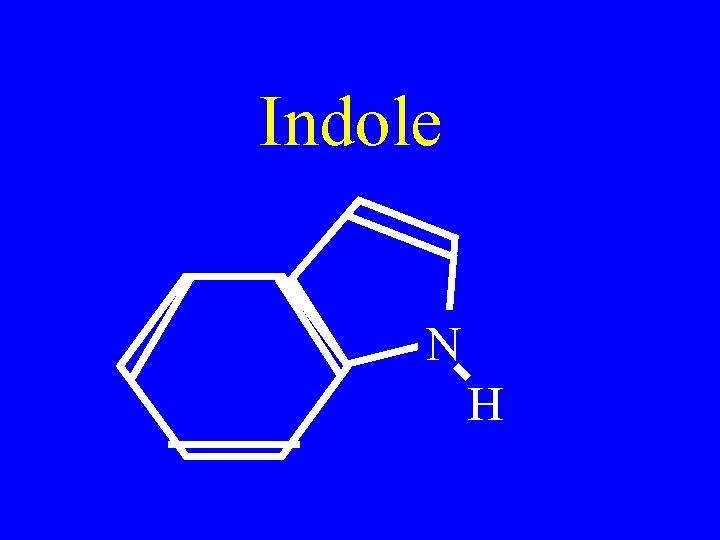
Indole N H
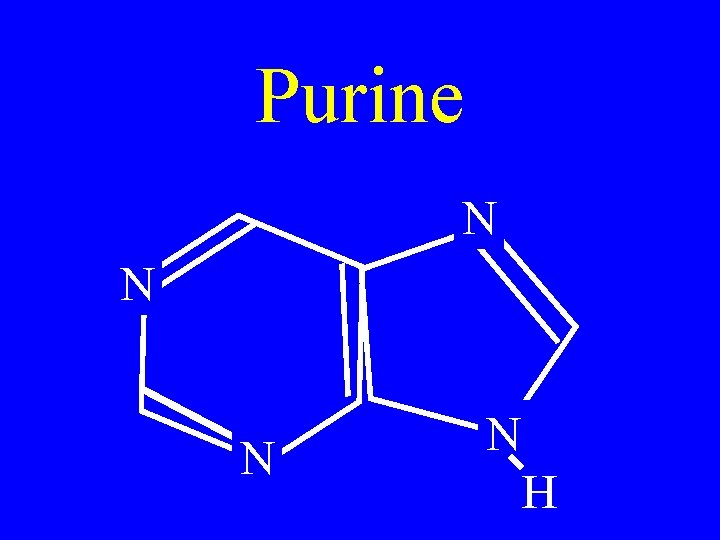
Purine N N H

Pyrimidine N N

Amino Acids

Common Amino Acid H R-C-COOH NH 2
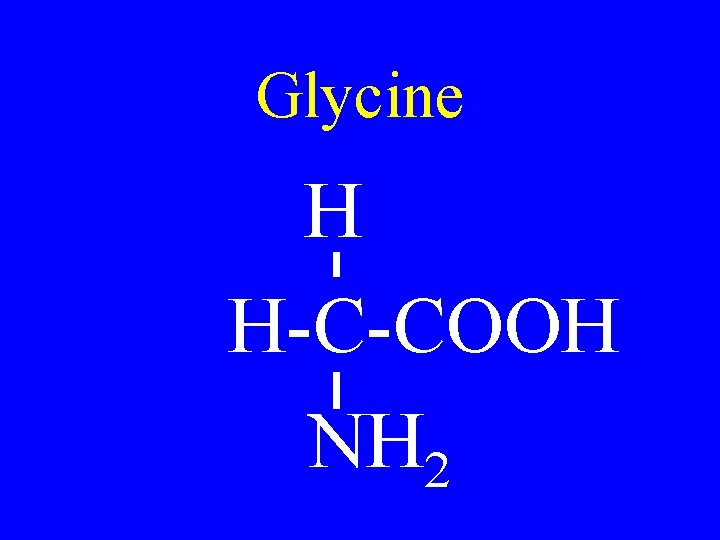
Glycine H H-C-COOH NH 2
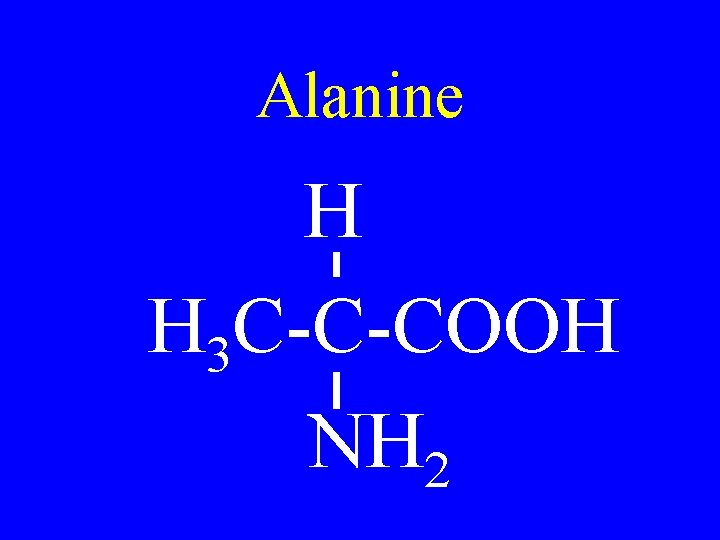
Alanine H H 3 C-C-COOH NH 2
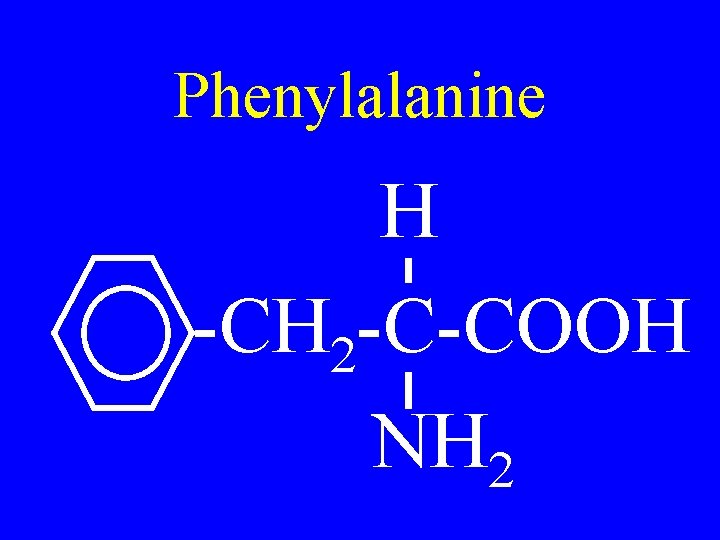
Phenylalanine H -CH 2 -C-COOH NH 2

Making Amines
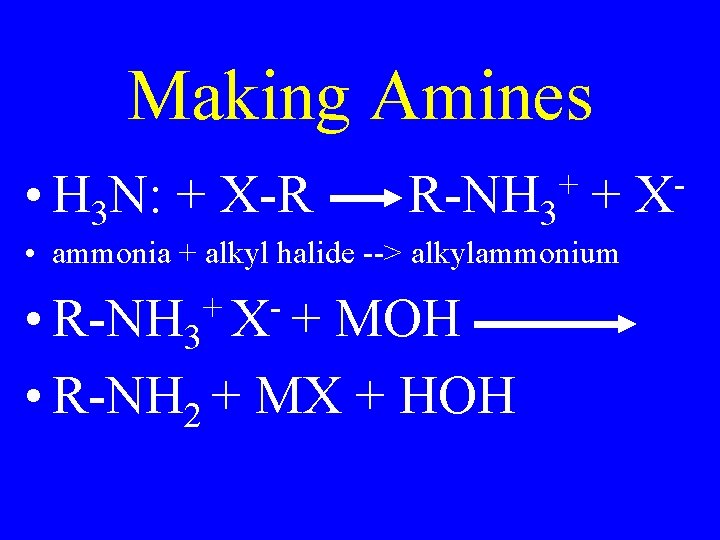
Making Amines • H 3 N: + X-R R-NH 3 + + • ammonia + alkyl halide --> alkylammonium + • R-NH 3 X + MOH • R-NH 2 + MX + HOH X
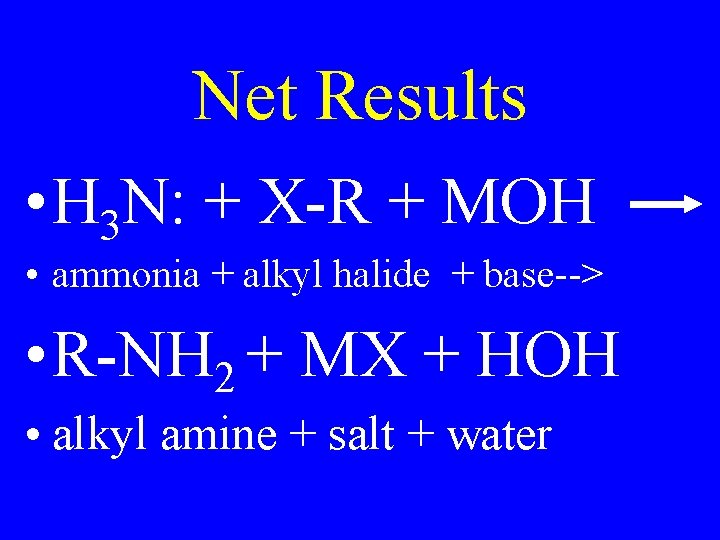
Net Results • H 3 N: + X-R + MOH • ammonia + alkyl halide + base--> • R-NH 2 + MX + HOH • alkyl amine + salt + water
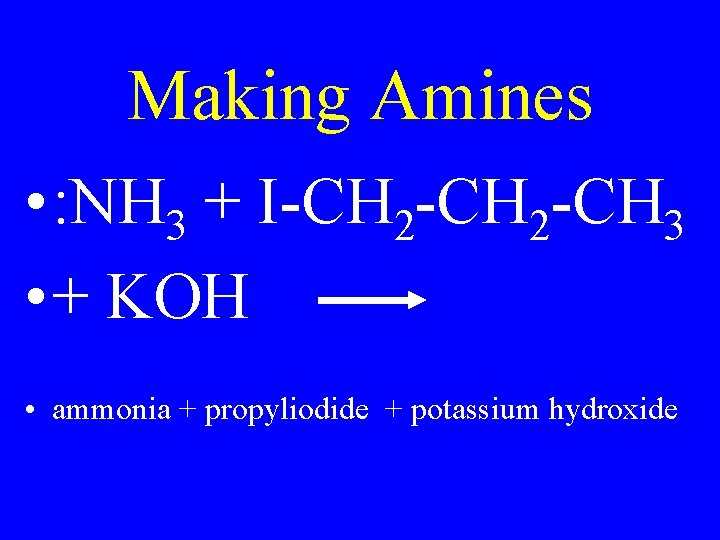
Making Amines • : NH 3 + I-CH 2 -CH 3 • + KOH • ammonia + propyliodide + potassium hydroxide
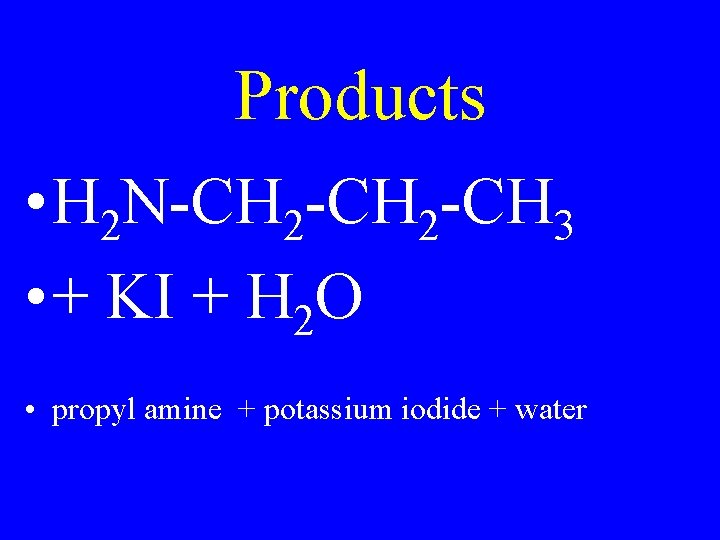
Products • H 2 N-CH 2 -CH 3 • + KI + H 2 O • propyl amine + potassium iodide + water
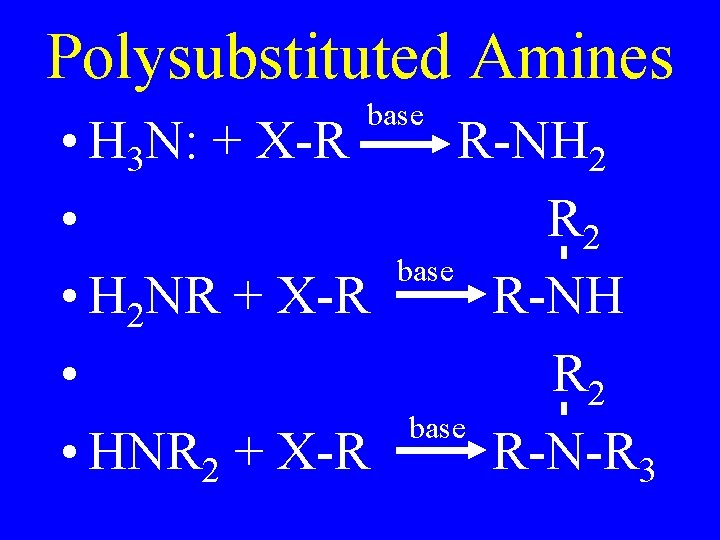
Polysubstituted Amines base • H 3 N: + X-R • • H 2 NR + X-R • • HNR 2 + X-R R-NH 2 R 2 base R-NH R 2 base R-N-R 3
- Slides: 44