AMG 420 an AntiBCMA Bispecific Tcell Engager Bi

AMG 420, an Anti-BCMA Bispecific T-cell Engager (Bi. TE®) Molecule, in Patients with R/R Multiple Myeloma: Updated Results of a First-in-Human Phase 1 Dose Escalation Study St Max S Topp, 1 Johannes Duell, 1 Gerhard Zugmaier, 2 Michel Attal, 3 Philippe Moreau, 4 Christian Langer, 5 Jan Krönke, 6 Thierry Facon, 7 Alexey V Salnikov, 8 Robin Lesley, 9 Karl Beutner, 10 James Kalabus, 9 Erik Rasmussen, 10 Kathrin Riemann, 8 Alex C Minella, 10 Gerd Munzert, 8* Hermann Einsele 1* 1 Department of Internal Medicine II, University Hospital Würzburg, Germany, 2 Amgen Research (Munich), Munich, Germany, 3 University of Toulouse, France, 4 Hematology Department Chair, University Hospital Center of Nantes, France, 5 Kempten Clinic, Kempten, Germany, 6 Ulm University, Ulm, Germany, 7 Regional University Hospital of Lille, France, 8 Boehringer Ingelheim, Biberach, Germany, 9 Amgen Inc. , South San Francisco, California, US, 10 Amgen Inc. , Thousand Oaks, California, US, *contributed equally PRESENTED BY: PROF MAX TOPP Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007.
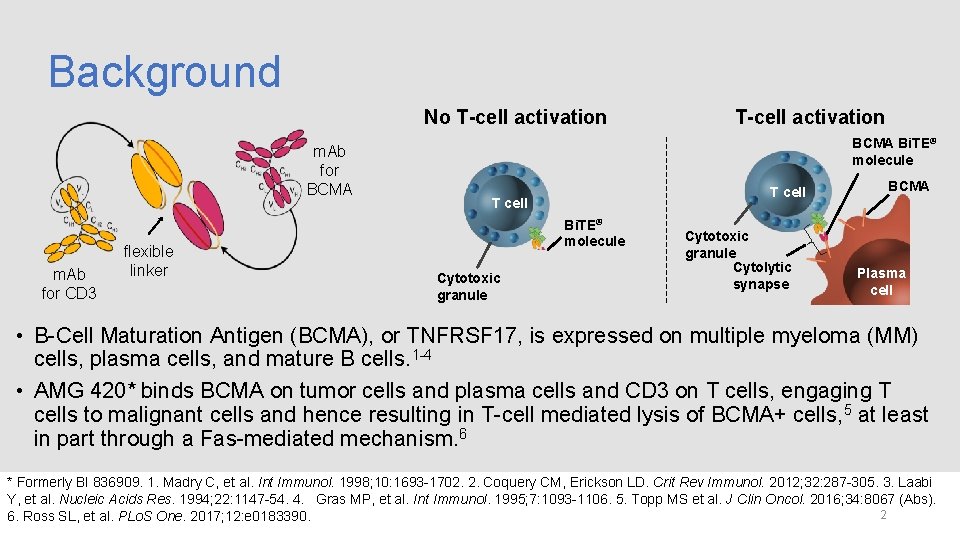
Background No T-cell activation m. Ab for BCMA m. Ab for CD 3 flexible linker T-cell activation BCMA Bi. TE® molecule T cell Bi. TE® molecule Cytotoxic granule Cytolytic synapse BCMA Plasma cell • B-Cell Maturation Antigen (BCMA), or TNFRSF 17, is expressed on multiple myeloma (MM) cells, plasma cells, and mature B cells. 1 -4 • AMG 420* binds BCMA on tumor cells and plasma cells and CD 3 on T cells, engaging T cells to malignant cells and hence resulting in T-cell mediated lysis of BCMA+ cells, 5 at least in part through a Fas-mediated mechanism. 6 * Formerly BI 836909. 1. Madry C, et al. Int Immunol. 1998; 10: 1693 -1702. 2. Coquery CM, Erickson LD. Crit Rev Immunol. 2012; 32: 287 -305. 3. Laabi Y, et al. Nucleic Acids Res. 1994; 22: 1147 -54. 4. Gras MP, et al. Int Immunol. 1995; 7: 1093 -1106. 5. Topp MS et al. J Clin Oncol. 2016; 34: 8067 (Abs). 2 6. Ross SL, et al. PLo. S One. 2017; 12: e 0183390.
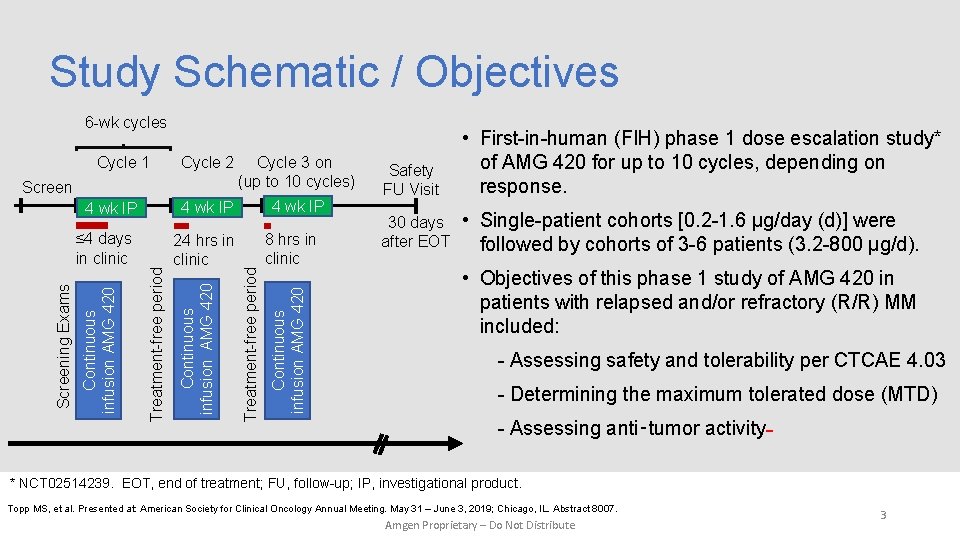
Study Schematic / Objectives 6 -wk cycles Cycle 2 Cycle 3 on (up to 10 cycles) 4 wk IP Treatment-free period Continuous infusion AMG 420 Screening Exams ≤ 4 days in clinic 24 hrs in clinic Continuous infusion AMG 420 4 wk IP 8 hrs in clinic Continuous infusion AMG 420 Screen Treatment-free period Cycle 1 Safety FU Visit • First-in-human (FIH) phase 1 dose escalation study* of AMG 420 for up to 10 cycles, depending on response. 30 days after EOT • Single-patient cohorts [0. 2 -1. 6 µg/day (d)] were followed by cohorts of 3 -6 patients (3. 2 -800 µg/d). • Objectives of this phase 1 study of AMG 420 in patients with relapsed and/or refractory (R/R) MM included: - Assessing safety and tolerability per CTCAE 4. 03 - Determining the maximum tolerated dose (MTD) - Assessing anti‑tumor activity * NCT 02514239. EOT, end of treatment; FU, follow-up; IP, investigational product. Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 3

Eligibility and Endpoints Element Description Key inclusion criteria R/R MM, progression after ≥ 2 prior treatment lines, which included both ≥ 1 proteasome inhibitor and ≥ 1 immunomodulatory imide drug Key exclusion criteria Plasma cell leukemia, extramedullary relapse, known CNS involvement, or prior allogeneic stem cell transplant Treatment For up to 5 cycles or until progressive disease (PD), toxicity, withdrawal of consent, or investigator decision; 5 more cycles could be given per investigator for perceived benefit for up to a possible total of 10 cycles. 1° objectives Determine MTD for this study, dose-limiting toxicities Also assessed Response per IMWG 2006, including minimal residual disease (MRD) negative response, defined for this study as <1 tumor cell / 104 normal cells in the bone marrow per FACS* * Using antibodies to cyt. Igλ, cyt. Igκ, CD 19, CD 56 or CD 138, CD 38, and CD 45 Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 4

Patient Baseline Characteristics Male, n (%) Age, median (range), year ECOG performance status, 0 / 1 / 2, % Disease duration, median (range), year Cytogenetics*, standard / intermediate / high, % Plasma cells at baseline, median (range), % Prior lines of therapy, median (range) Number of prior therapies, median (range)† Prior daratumumab / prior elotuzumab, n (%) Prior auto stem cell transplant, n (%) Refractory to past therapies, number, median (range) Refractory to IMi. D / PI / IMi. D & PI, % Refractory to daratumumab / elotuzumab, % N=42 27 (64%) 65 (39, 79) 57% / 40% / 2% 5. 2 (1. 3 -20) 55% / 31% / 14% 18% (0%-95%) 4 (2 -13) 4 (2 -10) 11 (26%) / 4 (10%) 36 (86%) 1 (0 -6) 55% / 45% / 31% 21% / 10% As of Feb 27, 2019. * Per Rajkumar SV. AJH 2012; 87: 79 -88. †A given therapy could be counted as a line more than once (ie, different dose / schedule after intervening therapies). Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 5

Dose-Limiting Toxicities (DLTs) and MTD DLT 800 µg/d: Cytokine release syndrome (CRS) 800 µg/d: Peripheral polyneuropathy 400 µg/d: Peripheral polyneuropathy* N=42 1 (2%) # Gr 1 - # Gr 2 - # Gr 3 1 1 1 CRS, cytokine release syndrome. * This occurred after the initial 3 patients were dosed at 400 µg/day, ie as de-escalating from 800 to 400 µg/day. • In this study, 800 µg/d was determined to not be tolerable as 2 of 3 patients experienced DLTs, both of which resolved: - Grade 3 CRS within 1 day of initiating treatment, with fever, hypertension, tachycardia, and retrograde amnesia; symptoms resolved after discontinuing AMG 420 - Grade 3 peripheral polyneuropathy • Of doses tested in this study, 400 µg/d was the MTD. As of Feb 27, 2019 Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. 6
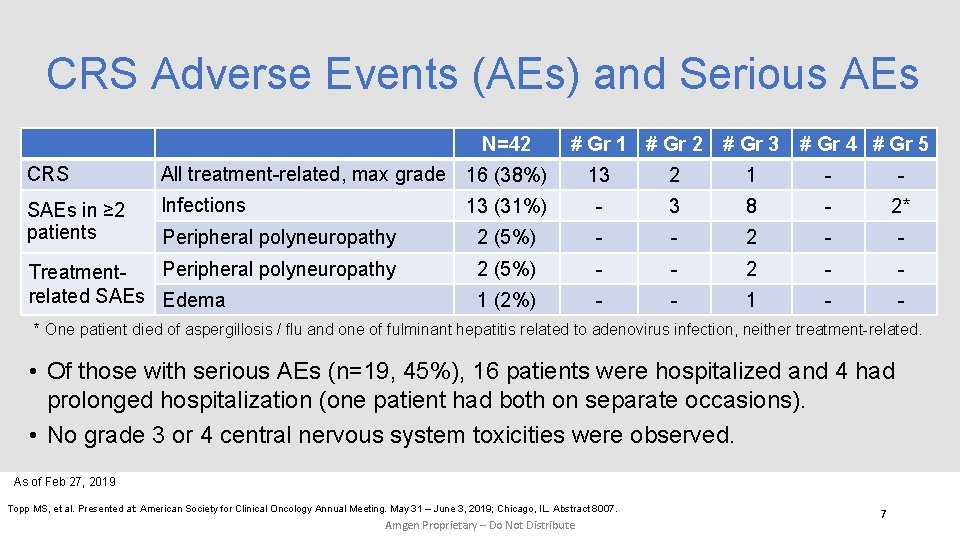
CRS Adverse Events (AEs) and Serious AEs N=42 CRS All treatment-related, max grade 16 (38%) SAEs in ≥ 2 patients Infections # Gr 1 # Gr 2 # Gr 3 # Gr 4 # Gr 5 13 2 1 - - 13 (31%) - 3 8 - 2* Peripheral polyneuropathy 2 (5%) - - 2 - - Peripheral polyneuropathy Treatmentrelated SAEs Edema 2 (5%) - - 2 - - 1 (2%) - - 1 - - * One patient died of aspergillosis / flu and one of fulminant hepatitis related to adenovirus infection, neither treatment-related. • Of those with serious AEs (n=19, 45%), 16 patients were hospitalized and 4 had prolonged hospitalization (one patient had both on separate occasions). • No grade 3 or 4 central nervous system toxicities were observed. As of Feb 27, 2019 Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 7
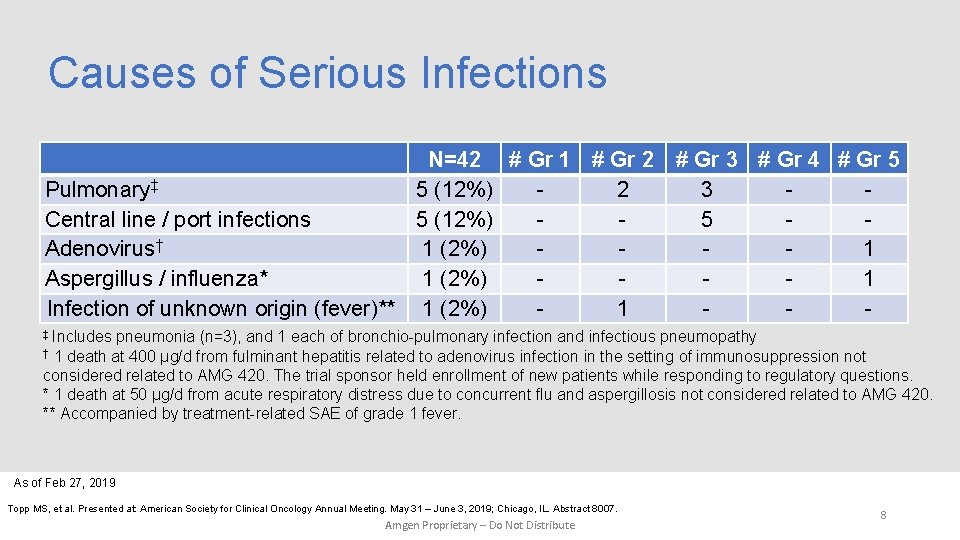
Causes of Serious Infections N=42 # Gr 1 # Gr 2 # Gr 3 # Gr 4 # Gr 5 Pulmonary‡ 5 (12%) 2 3 Central line / port infections 5 (12%) 5 Adenovirus† 1 (2%) 1 Aspergillus / influenza* 1 (2%) 1 Infection of unknown origin (fever)** 1 (2%) 1 ‡ Includes pneumonia (n=3), and 1 each of bronchio-pulmonary infection and infectious pneumopathy † 1 death at 400 µg/d from fulminant hepatitis related to adenovirus infection in the setting of immunosuppression not considered related to AMG 420. The trial sponsor held enrollment of new patients while responding to regulatory questions. * 1 death at 50 µg/d from acute respiratory distress due to concurrent flu and aspergillosis not considered related to AMG 420. ** Accompanied by treatment-related SAE of grade 1 fever. As of Feb 27, 2019 Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 8

Peripheral Polyneuropathy • Peripheral polyneuropathy was observed in two patients, in both cases as a DLT: - At 800 µg/d: Polyneuropathy starting 1 week after initiating treatment, to grade 3 by day 14 and requiring hospitalization. Ø After 15 days of treatment, M protein decreased by 58%. - At 400 µg/d dose de-escalation: Baseline grade 1 polyneuropathy worsened to grade 3 by day 30; after 3 weeks, the polyneuropathy gradually improved and was back to baseline by day 84 (week 12). Ø After 28 days of treatment, M protein decreased by 79%. • Both cases of peripheral polyneuropathy improved with IVIg and corticosteroid treatment; in one case completely resolving in a month and in the other returning to baseline (grade 1) in 2 months. As of Feb 27, 2019 Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 9
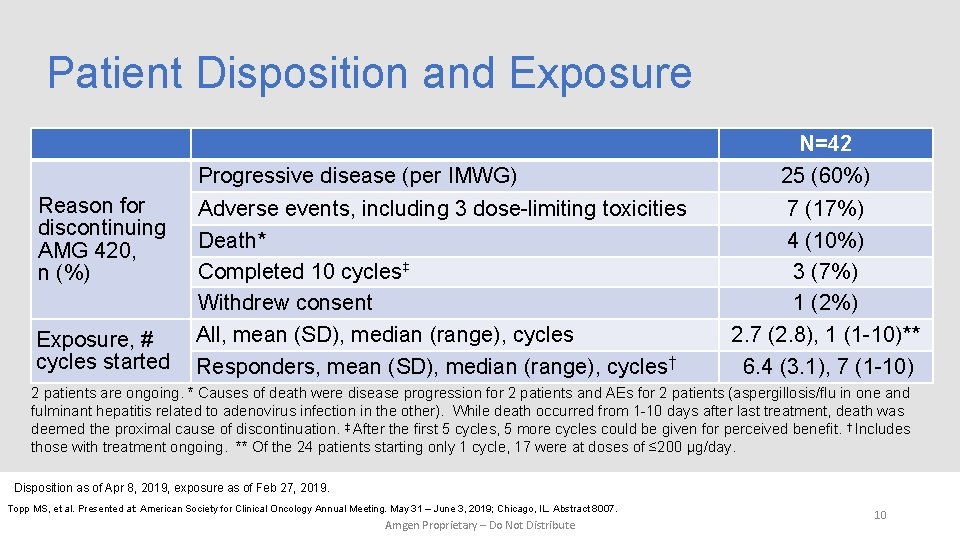
Patient Disposition and Exposure Reason for discontinuing AMG 420, n (%) Exposure, # cycles started Progressive disease (per IMWG) Adverse events, including 3 dose-limiting toxicities Death* Completed 10 cycles‡ Withdrew consent All, mean (SD), median (range), cycles Responders, mean (SD), median (range), cycles† N=42 25 (60%) 7 (17%) 4 (10%) 3 (7%) 1 (2%) 2. 7 (2. 8), 1 (1 -10)** 6. 4 (3. 1), 7 (1 -10) 2 patients are ongoing. * Causes of death were disease progression for 2 patients and AEs for 2 patients (aspergillosis/flu in one and fulminant hepatitis related to adenovirus infection in the other). While death occurred from 1 -10 days after last treatment, death was deemed the proximal cause of discontinuation. ‡ After the first 5 cycles, 5 more cycles could be given for perceived benefit. † Includes those with treatment ongoing. ** Of the 24 patients starting only 1 cycle, 17 were at doses of ≤ 200 µg/day. Disposition as of Apr 8, 2019, exposure as of Feb 27, 2019. Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 10
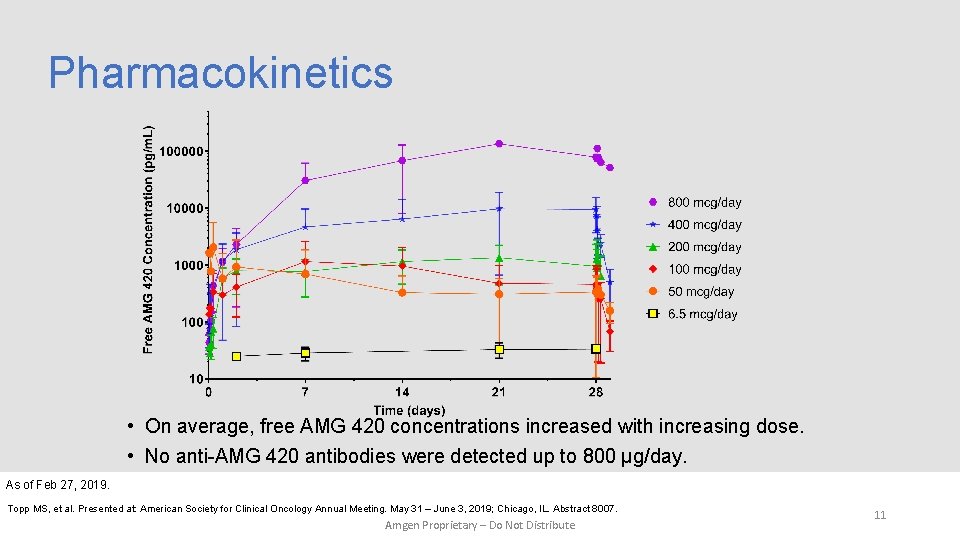
Pharmacokinetics • On average, free AMG 420 concentrations increased with increasing dose. • No anti-AMG 420 antibodies were detected up to 800 μg/day. As of Feb 27, 2019. Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 11
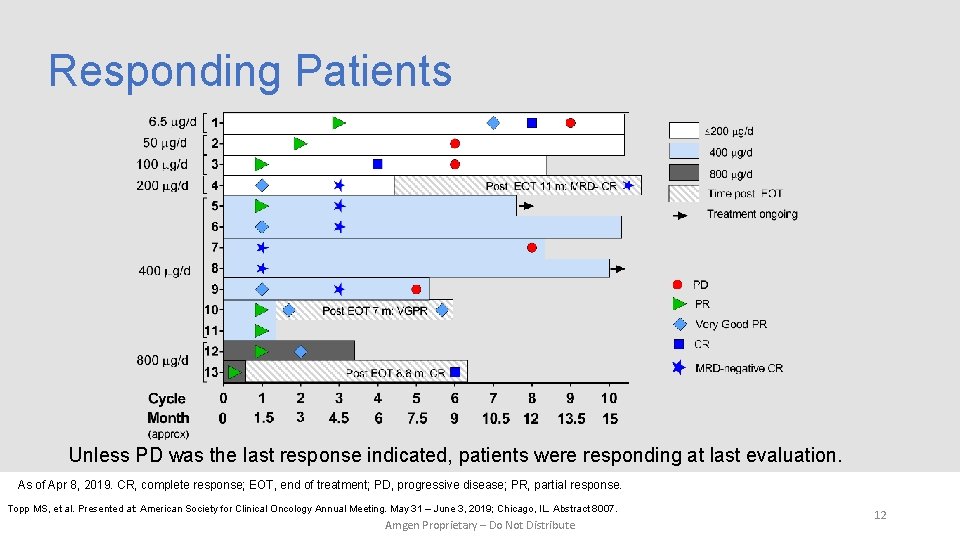
Responding Patients Unless PD was the last response indicated, patients were responding at last evaluation. As of Apr 8, 2019. CR, complete response; EOT, end of treatment; PD, progressive disease; PR, partial response. Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 12
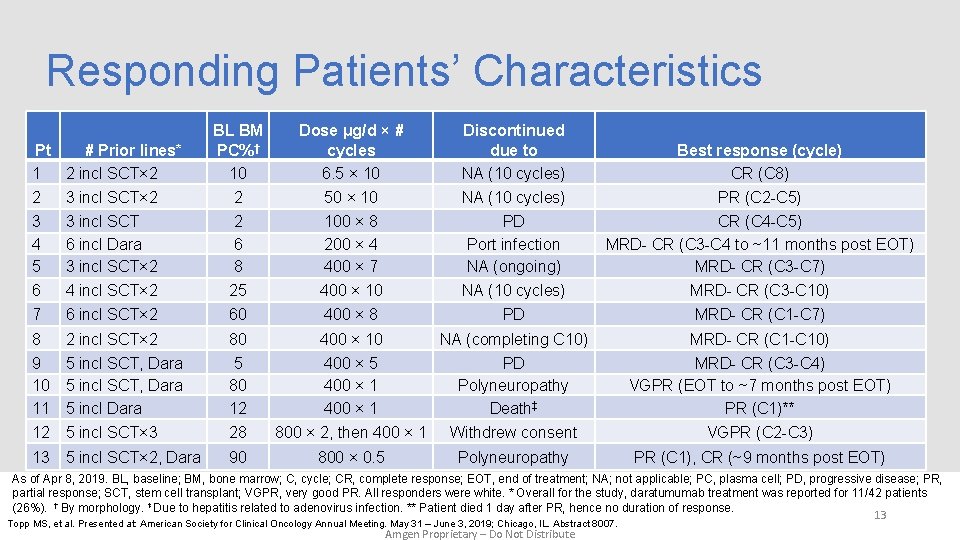
Responding Patients’ Characteristics Pt # Prior lines* 1 2 incl SCT× 2 BL BM PC%† 10 Dose μg/d × # cycles 6. 5 × 10 Discontinued due to NA (10 cycles) Best response (cycle) CR (C 8) 2 3 incl SCT× 2 2 50 × 10 NA (10 cycles) PR (C 2 -C 5) 3 4 5 3 incl SCT 6 incl Dara 3 incl SCT× 2 2 6 8 100 × 8 200 × 4 400 × 7 PD Port infection NA (ongoing) CR (C 4 -C 5) MRD- CR (C 3 -C 4 to ~11 months post EOT) MRD- CR (C 3 -C 7) 6 4 incl SCT× 2 25 400 × 10 NA (10 cycles) MRD- CR (C 3 -C 10) 7 6 incl SCT× 2 60 400 × 8 PD MRD- CR (C 1 -C 7) 8 2 incl SCT× 2 80 400 × 10 NA (completing C 10) MRD- CR (C 1 -C 10) 9 10 11 12 5 incl SCT, Dara 5 incl SCT× 3 5 80 12 28 400 × 5 400 × 1 800 × 2, then 400 × 1 PD Polyneuropathy Death‡ Withdrew consent MRD- CR (C 3 -C 4) VGPR (EOT to ~7 months post EOT) PR (C 1)** VGPR (C 2 -C 3) 90 800 × 0. 5 Polyneuropathy PR (C 1), CR (~9 months post EOT) 13 5 incl SCT× 2, Dara As of Apr 8, 2019. BL, baseline; BM, bone marrow; C, cycle; CR, complete response; EOT, end of treatment; NA; not applicable; PC, plasma cell; PD, progressive disease; PR, partial response; SCT, stem cell transplant; VGPR, very good PR. All responders were white. * Overall for the study, daratumumab treatment was reported for 11/42 patients (26%). † By morphology. ‡ Due to hepatitis related to adenovirus infection. ** Patient died 1 day after PR, hence no duration of response. 13 Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute

Efficacy Summary At 400 µg/d • There were 5 minimal residual disease (MRD)-negative CRs, 1 VGPR, and 1 PR, for a response rate of 7/10 (70%); as of April 8, responses lasted for a median of 9. 0 months (range: 5. 8 -13. 6 months)*, with 2 patients ongoing on treatment. Overall • There were 13/42 responders (6 MRD-negative CRs, 3 CRs, 2 VGPRs, 2 PRs). Median time to any response was 1 month, with 11 of 13 patients responding in the first cycle. • Responses lasted for a median of 8. 4 months (range: 2. 5 -15. 5 months). • MRD-negative CRs lasted for a median of 9. 6 months (range: 2. 8 -12. 8 months); all but one of these 6 patients were in the 400 µg/d cohort. * Excludes patient 11; death 1 day following PR assessment. Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 14

Conclusions In this FIH dose escalation study, AMG 420, a short half-life Bi. TE® molecule targeting BCMA, demonstrated clinical activity in patients with heavily pretreated multiple myeloma: • No major toxicities prior to DLTs at 800 µg/d of CRS and polyneuropathy; a patient in the subsequent 400 µg/d dose group also had a DLT of polyneuropathy, which resolved. • Careful evaluation of infections should be conducted in future clinical trials to enable development of optimal management guidelines. • Of doses tested in this study, 400 µg/d was the MTD; other doses may be tested in the future. § There was a 70% response rate (7/10) with 5 out of 7 responders achieving a MRDnegative CR at 400 µg/d, a recommended dose for further investigation. § All (7/7) responses at this dose started in the first cycle. • These data warrant further clinical investigation of AMG 420; a phase 1 b/2 trial is underway. Topp MS, et al. Presented at: American Society for Clinical Oncology Annual Meeting. May 31 – June 3, 2019; Chicago, IL. Abstract 8007. Amgen Proprietary – Do Not Distribute 15

Disclosures • MT: Honoraria: Amgen, Advisory: Amgen, Roche, Affimed, Regeneron, Jazz, Gilead, Research funding: Affimed, Amgen, Regeneron, Roche, Travel: Amgen, Roche, Affimed, Speaker: Amgen, Expert testimony: Amgen, Boehringer Ingelheim, Affimed, Regeneron, Patents/royalties/other IP: Biomarkers for immunotherapies • JD, MA, CL, and TF: No disclosures. • GZ: Employee and stockholder of Amgen Inc. ; patents, royalties, other intellectual property with Amgen Inc. • PM: Honoraria: Amgen, Celgene, Janssen-Cilag, Novartis, Takeda, Advisory: Amgen, Celgene, Janssen, Novartis, Takeda • JKroenke: Amgen honoraria for speakers’ bureau • AS and KR: Employees of Boehringer Ingelheim • RL, KB, JKalabus, ER, and AM: Employees of and stockholders in Amgen. • GM: Employee of Boehringer Ingelheim; patents, royalties, other intellectual property with Boehringer Ingelheim. • HE: Consulting or Advisory Role - Amgen; Celgene; Janssen; Novartis; Takeda Amgen Proprietary – Do Not Distribute 16

Acknowledgments The authors would like to thank the patients, their families, and study site staff. Others who have contributed to this work include Peter Kufer, Francesco Galimi, and Agnes Ang. Medical writing support was provided by Susanna Mac of Amgen Inc. , and this study and analyses were funded by Amgen Inc. Please send requests for slides to: MEDINFO@AMGEN. COM Amgen Proprietary – Do Not Distribute 17
- Slides: 17